Abstract
Background
This prospective cohort study searched for factors associated with a response to nucleos(t)ide analogue/peg-interferon (NUC/peg-IFN) sequential therapy.
Methods
A total of 95 patients with chronic hepatitis B being treated with NUCs were enrolled. Immediately following NUC cessation, peg-IFN was administered at 180 µg/dose weekly for 48 weeks.
Results
Twenty-six patients (27%) were judged to be responders at 48 weeks after the completion of peg-IFN. Analysis of baseline factors revealed that hepatitis B surface antigen (HBsAg) <3.1 log IU/ml and HB core-related antigen (HBcrAg) <3.9 log U/ml were significant indicators of a treatment response. The levels of the markers decreased in both responders and non-responders during peg-IFN therapy but continued falling in responders only after halting peg-IFN. Lower HBsAg (<2.0 log IU/ml) and HBcrAg (<3.8 log U/ml) levels at the time of response judgment were also significantly associated with a favorable response. While lower HBcrAg at baseline was the sole predictor of decreased HBcrAg levels at judgment, lower HBsAg, lower HBcrAg, and the use of adefovir dipivoxil at baseline predicted decreased HBsAg levels at the study endpoint. The use of adefovir dipivoxil was also associated with higher serum IFN-λ3, which might have contributed to the reduction in patient HBsAg levels.
Conclusions
The combinational use of HBsAg and HBcrAg levels at baseline and their changes throughout sequential therapy may be useful for predicting a response to NUC/peg-IFN sequential therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With an estimated 350 million carriers worldwide, hepatitis B virus (HBV) infection is a widespread health problem that frequently causes chronic hepatitis and eventual liver cirrhosis and hepatocellular carcinoma (HCC) [1,2,3]. The majority of HBV carriers in Japan have genotype C and become infected perinatally or during early childhood. Response rates to antiviral therapies are lower in the Japanese as compared with Caucasian patients because of suspected differences in the main mode of transmission and genotype [4].
Currently available antiviral agents for hepatitis B include nucleos(t)ide analogues (NUCs) and interferon-alpha (IFN). NUCs are orally administered and associated with low rates of adverse effects. Although treatment with such NUCs as lamivudine (LAM), adefovir dipivoxil (ADV), entecavir (ETV), and tenofovir disoproxil fumarate (TDF) induces virological and biochemical responses in most patients, NUC therapy also carries the risk of drug resistance. Furthermore, patients with hepatitis B are required to undergo extended treatment with NUCs since early discontinuance often leads to relapse. In contrast, the remission of chronic hepatitis B by IFN is prolonged but achieved only in a small percentage of patients. Combination therapy with NUCs and IFN may therefore confer additive or synergistic antiviral effects. Serfaty et al. [5] conducted a pilot study on sequential therapy using LAM and IFN and suggested a cooperative effect of the antiviral agents. However, ensuing reports [6,7,8,9,10] have been unable to confirm this.
We previously evaluated 50 patients with chronic hepatitis B who underwent 24 weeks of sequential IFN therapy for the cessation of NUCs and concluded that the combinational use of hepatitis B surface antigen (HBsAg) and hepatitis B core-related antigen (HBcrAg) levels [11, 12] might be useful in predicting the 24-month outcome. Since the study was retrospective and the number of patients was small, the present prospective cohort investigation of sequential therapy, in which 48 weeks of peg-IFN alpha 2a (peg-IFN) was administered subsequently to NUCs, was undertaken to clarify the factors associated with the effects of NUC/peg-IFN sequential therapy.
Patients and methods
Study design
A prospective cohort study entitled “Study on usefulness of PEG-IFN alpha-2a administration to discontinue nucleos(t)ide analogue treatment in patients with chronic hepatitis B” was registered at the University Hospital Medical Information Network Center of Japan on January 10, 2012 (UMIN 000007045). The investigation originally consisted of three non-randomized arms with different procedures aiming at the discontinuation of NUCs. In the present study, we analyzed one of the three arms in which patients with NUC/peg-IFN sequential therapy were enrolled. We consider that such selection causes minimum bias because each arm proceeds independently.
Patients with chronic hepatitis B who had been receiving NUCs for at least 1 year were recruited. Immediately after stopping NUCs, peg-IFN (Chugai Pharmaceutical Co., Ltd., Tokyo, Japan) was administered at 180 µg/dose once weekly for 48 weeks. The dose of peg-IFN could be reduced to 90 µg during exceptional circumstances, such as side effects. Patients were followed for 48 weeks after completing peg-IFN administration, at which time the treatment response was judged.
Patients who met the following conditions were classified as responders according to the guidelines of the Japan Society of Hepatology: alanine transaminase (ALT) <31 IU/l, negative result for HBeAg, HBV DNA <4.0 log copies/ml, and no recommencement of NUCs [13]. Patients who did not meet all of these criteria were recorded as non-responders.
Patients
A total of 95 patients with chronic hepatitis B who had been receiving NUC therapy were prospectively enrolled in the present study between February 2012 and December 2013. The cohort included 68 men and 27 women of a median age of 45 years (range, 27–87 years) at peg-IFN commencement. HBe antigen was positive in 33 (35%) patients. HBV genotype was A in seven (7%) patients, B in four (4%) patients, C in 77 (82%) patients, other in three (3%) patients, and undetermined in four (4%) patients. Genotype C was predominant as previously reported in Japan [14]. The NUCs at cessation were ETV in 58 (61%) patients, ETV + ADV in 12 (13%) patients, LAM in two (2%) patients, and LAM + ADV in 23 (24%) patients. The median duration of NUC administration was 4.9 years (range, 1.0–12.3 years).
HBsAg was confirmed to be positive on a minimum of two occasions at least 6 months apart in all patients before NUC treatment. Tests for hepatitis C and human immunodeficiency virus antibodies were negative. Patients complicated with HCC or signs of hepatic failure at the cessation of NUC administration were excluded from the study, as were patients with contraindications for peg-IFN therapy. HBsAg level, HBcrAg level, hepatitis B e antigen (HBeAg) status, NUCs used, and HBV genotype were not considered during patient recruitment. With few exceptions, subjects were seen at least once a month during peg-IFN therapy and at least once every 3 months afterwards. No patient developed HCC or hepatic failure during the follow-up period. Stored serum samples were kept frozen at −20 °C or below until assayed.
This study was approved by the Ethics Committees of all participating institutions (approval references 1906 and 353 for Shinshu University, 1172 and 92 for Hyogo College of Medicine, 184 and 125 for Musashino Red Cross Hospital, 45-11-0028 and 67 for Nagoya City University Graduate School of Medical Sciences, 2272 for Osaka City University Graduate School of Medicine, 639 and 379 for Chiba University, 2089 and 91 for St. Marianna University School of Medicine, 2485 and 23020 for National Hospital Organization Nagasaki Medical Center, 121-27 and 2086 for Tokai University School of Medicine, 1170 for Kawasaki Medical School, 1326 for Okayama University Graduate School of Medicine, B120705027 for Yokohama City University Graduate School of Medicine, 411 for Kumamoto Shinto General Hospital, and “Study on usefulness of Peg-IFN alpha 2a administration to discontinue NUC treatment in patients with chronic hepatitis B” for Teine Keijinkai Hospital and Kagawa Prefectural Central Hospital).
Hepatitis B viral markers
Serological markers for HBV, including HBsAg, HBeAg, and antibody to HBeAg, were tested using commercially available enzyme immunoassay kits (Abbott Japan Co., Ltd., Tokyo, Japan). HBsAg was quantitatively measured using a chemiluminescence enzyme immunoassay (CLEIA)-based ARCHITECT HBsAg QT assay manufactured by Abbott Japan Co., Ltd. [15] with a quantitative range of 0.05–250 IU/ml (−1.3 to 2.4 log IU/ml). End titer was determined by diluting samples according to the manufacturer’s instructions when initial results exceeded the upper limit of the assay range.
Serum HBcrAg levels were measured using a CLEIA HBcrAg assay kit with a fully automated Lumipulse System analyzer (Fujirebio Inc., Tokyo, Japan) as described previously [16]. The HBcrAg assay measures all antigens transcribed and translated from the pre-core and core genes of the HBV genome, which include hepatitis B e, core, and p22cr antigens [12, 16]. HBcrAg concentration was calculated based on a standard curve generated using recombinant pro-HBeAg. The immunoreactivity of pro-HBeAg at 10 fg/ml was defined as 1 U/ml. We expressed HBcrAg in terms of log U/ml, with a quantitative range set at 3.0–6.8 log U/ml.
Serum HBV DNA was determined using a COBAS TaqMan HBV kit (Roche, Tokyo, Japan) with a quantitative range of 2.1–9.0 log copies/ml [17]. According to the manufacturer’s instructions, detection of a positive signal below the quantitative range was described as <2.1 log copies/ml and no signal detection was regarded as negative. Six HBV genotypes (A–F) were evaluated according to the restriction patterns of DNA fragments from the method reported by Mizokami et al. [18].
Intrahepatic HBV covalently closed circular DNA (cccDNA) was measured based on the method reported by Suzuki et al. [19]. Briefly, liver biopsy specimens were obtained and stored at −80 °C until DNA extraction, after which the concentration of extracted HBV DNA was determined according to absorbance at 260 nm. Two oligonucleotide primers, cccF2 (nucleotides 1424–1444) and cccR4 (nucleotides 1755–1737), and a cccP2 probe (nucleotides 1672–1655) were designed to flank the direct repeat region between the hepatitis B core and the polymerase gene. The use of cccF2 and cccR4 oligonucleotide primers spanning the direct repeat region of the HBV genome enabled the polymerase chain reaction of native viral DNA in the Dane particle to block the amplification of products since the partially double-stranded HBV DNA was disrupted in the direct repeat region [20]. Twenty-five microliters of the extracted DNA (0.5 µg) was analyzed with a sequence detector system (SRL Inc., Tokyo, Japan). The concentration of HBV cccDNA was expressed as the log copy number per 1 µg of total DNA extracted from the liver tissue at a lower quantitative limit of 1.7 log copies/µg. Detection of a positive signal below the quantitative threshold was described as <1.7 log copies/µg and no signal detection was regarded as negative. Measurement of intrahepatic HBV cccDNA was not considered essential since the study protocol permitted liver biopsy histology samples obtained within 1 year before starting peg-IFN therapy.
Interleukin-28B (IL28B) genotyping and serum IFN-λ3 level
Patients were evaluated for single-nucleotide polymorphisms near IL28B (rs8099917) using the Invader Plus assay (Invader Chemistry, Madison, WI, USA), as previously reported [21]. We did not investigate the polymorphism of rs12979860 because rs8099917 and rs12979860 have shown strong linkage disequilibrium in Japanese patients. Written informed consent was obtained from each patient.
Serum levels of IFN-λ3 were evaluated by a recently introduced CLEIA [22,23,24] that enabled the quantification of serum IFN-λ3 without any overlap of IFN-λ1 or IFN-λ2. The measurement range for this assay was 0.1–10,000 pg/ml.
Liver histological examination
Histological examination of liver samples was performed in 85 (89%) of the 95 patients within 1 year before starting peg-IFN. Histological findings were assessed according to the new Inuyama classification system [25]. Briefly, staging of liver fibrosis was classified as F0 (no fibrosis), F1 (mild fibrosis), F2 (moderate fibrosis), F3 (severe fibrosis), or F4 (liver cirrhosis). Grading of liver inflammation was classified as A0 (no activity), A1 (mild activity), A2 (moderate activity), or A3 (severe activity).
Statistical analyses
The Fisher’s exact and Pearson’s Chi-square tests were adopted to test for differences between patient subgroups. The Mann–Whitney U test was employed to compare continuous data. The Friedman test with Bonferroni correction was used to assess changes in HBsAg and HBcrAg levels. Multivariate analysis was performed with a logistic model for the 48-week response to NUC/peg-IFN sequential therapy using factors whose p value was less than 0.2 in univariate analysis. All continuous data values were translated into categorical data and divided by the cut-off value decided by Youden’s index using receiver operating characteristic (ROC) curve analysis. A value of 0.5 was substituted for any missing values. Correlations between the levels of HBsAg, HBcrAg, and cccDNA were calculated using the Spearman’s rank correction correlation coefficient test.
All tests were performed using the IBM SPSS Statistics Desktop for Japan ver. 23.0 (IBM Japan Inc., Tokyo, Japan). p values of less than 0.05 were considered to be statistically significant.
Results
Of the 95 patients enrolled, 75 (79%) completed peg-IFN therapy without dose reduction, 15 (16%) completed it with a necessary dose reduction, and the remaining five (5%) discontinued treatment due to side effects of epilepsy (15 weeks after starting peg-IFN), ALT elevation up to 937 IU/ml (16 weeks), interstitial pneumonia (17 weeks), retinal hemorrhage (42 weeks), or chronic thyroiditis (43 weeks). Five (5%) patients were lost to follow-up during the 48 weeks subsequent to peg-IFN completion.
All patients requiring re-administration of NUCs after stopping peg-IFN had ALT >80 IU/l and/or HBV DNA >5.8 log copies/ml at or just before the point of NUC resumption that fulfilled the established requirements for restarting NUCs [11, 12, 26]. Under these conditions, NUCs were recommenced in 37 (39%) of the 95 patients prior to the study endpoint.
Twenty-six (27%) of the 95 patients were judged as responders to NUC/peg-IFN sequential therapy and the remaining 69 (73%) were determined to be non-responders. The reasons for a non-response were lost to follow-up in five (7%) patients, high ALT in five (7%) patients, high HBV DNA in nine (13%) patients, high ALT and HBV DNA in 13 (19%) patients, and recommencement of NUCs in 37 (54%) patients.
Clinical background, laboratory data, and viral markers are compared between the 26 responders and 69 non-responders in Table 1. Age distribution tended to be higher in responders, while gender and history were similar between the groups. Genotype C was over 80% in both groups. Approximately 60% of patients were treated with ETV alone and 35% were treated with regimens that included ADV just prior to starting peg-IFN in both groups. Histological liver staging and grading were comparable between the groups, as was the frequency of peg-IFN administration completion. The distribution of the major and minor IL28B genotypes was similar among the 82 (86%) of 95 patients tested. Laboratory data, including blood cell count and transaminase enzyme levels, were similar between the groups and HBV DNA levels were comparably low, possibly reflecting the effects of NUCs. HBeAg positivity tended to be lower in responders than in non-responders, while levels of HBsAg and HBcrAg were significantly lower in this group. Median HBV cccDNA level was lower in responders (n = 6, median 2.5 log copies/µg, range: negative result −4.0 log copies/µg) than in non-responders (n = 22, median 3.6 log copies/µg, range: negative result −8.1 log copies/µg), but this difference was not statistically significant (p = 0.158) likely due to the small number of patients analyzed.
Multivariate analysis of factors associated with a response to NUC/peg-IFN sequential therapy at peg-IFN initiation revealed that lower levels of HBsAg and HBcrAg were significantly associated with responders (Table 2). The area under the curve (AUC) to predict responders was 0.748 (p < 0.001) for HBsAg and 0.692 (p = 0.005) for HBcrAg according to ROC analysis. The calculated threshold to predict responders was 3.1 log IU/ml for HBsAg [sensitivity: 64.0%, specificity: 67.1%, positive predictive value (PPV): 41.0%, negative predictive value (NPV): 83.9%, accuracy: 66.3%] and 3.9 log U/ml for HBcrAg (sensitivity: 62.5%, specificity: 73.5%, PPV: 45.5%, NPV: 84.7%, accuracy: 70.8%). Figure 1a shows the distribution of responders and non-responders according to HBsAg and HBcrAg level at the start of peg-IFN. The response rate was highest (56%) when both HBsAg and HBcrAg levels were low and was lowest (6%) when both levels were high (p = 0.003). The maximum response rate (56%) was 10–15% higher than those predicted using low levels of HBsAg (41.0%) or HBcrAg (45.5%) separately. The minimum response rate (6%) was approximately 10% lower than those predicted using high levels of HBsAg (16.1%) or HBcrAg (15.3%) separately.
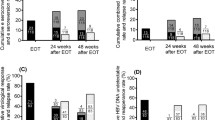
Distribution of responders and non-responders according to serum levels of HBsAg and HBcrAg. a Antigen levels at the start of peg-IFN therapy. b Antigen levels at 48 weeks after stopping peg-IFN therapy. Figures are divided into four areas according to quantitative thresholds. The vertical dashed lines delineate the upper and lower detection limits of HBcrAg. The horizontal dashed line indicates the lower detection limit of HBsAg. The percentage in each area represents the responder rate. Differences in responder rates among areas were analyzed by the Mantel–Haenszel test
The changes in HBsAg and HBcrAg levels during peg-IFN therapy and the ensuing follow-up period are compared between responders and non-responders in Fig. 2. Median antigen levels were significantly lower in responders at all three points examined for both HBsAg and HBcrAg. Median HBsAg levels decreased during peg-IFN administration in responders (from 2.9 to 2.1 IU/ml) and non-responders (from 3.4 to 3.1 IU/ml). However, antigen levels subsequently decreased in responders (from 2.1 to 2.0 IU/ml) but increased in non-responders (from 3.1 to 3.2 IU/ml) during follow-up after peg-IFN therapy. Similarly, median HBcrAg levels decreased during peg-IFN administration in responders (from 3.7 to 3.4 U/ml) and non-responders (from 4.6 to 4.0 U/ml) but decreased in responders (from 3.4 to <3.0 U/ml) and increased in non-responders (from 4.0 to 4.4 U/ml) during post-peg-IFN therapy follow-up.
Comparison of changes in HBsAg and HBcrAg levels during peg-IFN administration and follow-up of 48 weeks afterwards. Data are expressed as the median and 25–75% range. Open circles indicate responders and closed circles indicate non-responders. The horizontal dashed lines in the right figure delineate the upper and lower quantitative ranges of HBcrAg. Changes in the antigen levels of responders and non-responders were analyzed by the Friedman test with Bonferroni correction
Lower levels of HBsAg [odds ratio 3.1, 95% confidence interval (CI) 1.0–9.2, p = 0.041] and HBcrAg (odds ratio 8.8, 95% CI 2.3–33.3, p = 0.001) at 48 weeks after completing peg-IFN administration were also significantly associated with a favorable response to sequential therapy. The AUC to predict responders was 0.748 (p < 0.001) for HBsAg and 0.692 (p = 0.005) for HBcrAg at 48 weeks after completing peg-IFN administration according to ROC analysis. The threshold to predict responders was 2.0 log IU/ml for HBsAg (sensitivity: 64.0%, specificity: 67.1%, PPV: 41.0%, NPV: 83.9%, accuracy: 66.3%) and 3.8 log U/ml for HBcrAg (sensitivity: 62.5%, specificity: 73.5%, PPV: 45.5%, NPV: 84.7%, accuracy: 70.8%). Figure 1b presents the distribution of responders and non-responders according to HBsAg and HBcrAg levels at 48 weeks after completing peg-IFN. The response rate was high (60%) only when both levels were low (p < 0.001).
Multivariate analysis revealed that lower levels of HBsAg and HBcrAg at the initiation of peg-IFN and the use of ADV before peg-IFN therapy were significant factors associated with lower HBsAg levels (<2.0 log IU/ml) at 48 weeks after peg-IFN therapy (Table 3). Thresholds to predict lower HBsAg were 3.0 log IU/ml for HBsAg (AUC 0.872, p < 0.001), 4.0 U/ml for HBcrAg (AUC 0.703, p = 0.008), and ADV use (AUC 0.643, p < 0.061). The only factor associated with decreased HBcrAg (<3.8 log U/ml) levels at the study endpoint was itself at 4.8 log IU/ml (AUC 0.852, p < 0.001) (Table 3).
IFN-λ3 levels at peg-IFN commencement were measured in 80 (84%) of the 95 patients. Median IFN-λ3 was roughly ten times higher (p < 0.001) in the 29 patients who received ADV (median 18.4, range 1.2–118.6 pg/ml) than in the 51 patients who did not (median 1.4, range 0.3–23.0 pg/ml). On the other hand, IFN-λ3 level did not differ (p = 0.706) between patients with the IL28B-SNP major type (n = 16, median 19.4 pg/ml, range 1.2–55.6 pg/ml) and those with the minor type (n = 10, median 24.2 pg/ml, range 2.9–118.6 pg/ml) in subjects receiving ADV. Similarly, IFN-λ3 level did not differ (p = 0.708) between patients with the IL28B-SNP major type (n = 39, median 1.4 pg/ml, range 0.3–23.0 pg/ml) and those with the minor type (n = 6, median 1.8 pg/ml, range 0.3–7.5 pg/ml) in subjects not receiving ADV. These findings were in agreement with the results described by Murata et al. [24].
Figure 3 assessed for correlations among serum HBsAg, serum HBcrAg, and intrahepatic HBV cccDNA in 28 patients receiving NUC therapy. Serum HBV DNA was suppressed to the level of signal-positive or negative in 26 (93%) patients. HBsAg and HBcrAg were both significantly correlated with HBV cccDNA, but only weakly with each other.
Discussion
NUCs and IFN are commonly used anti-HBV agents worldwide. NUCs induce a rapid reduction in serum HBV DNA and hepatitis remission in many cases. They also produce histological improvement resulting in a diminished occurrence of HCC [27]. However, it is difficult to eradicate the virus completely, and NUCs face such complications as the emergence of resistant strains and hepatitis relapse following treatment discontinuation. One reason for these problems is that while NUCs can efficiently reduce serum HBV DNA, they have virtually no effect on HBV cccDNA levels in hepatocyte nuclei, which are the origin of HBV replication. Based on this fact, the simple discontinuation of NUCs is discouraged. On the other hand, the duration of peg-IFN therapy is usually limited to 48 weeks [28, 29] and a sustained off-treatment response can be expected in some patients. Thus, the sequential administration of peg-IFN after NUCs may be useful to successfully halt NUC treatment [30]. In this context, we analyzed the efficacy of NUC/peg-IFN sequential therapy in patients with chronic hepatitis B who were treated with NUCs and searched for factors associated with a favorable response.
Responders were defined as patients who fulfilled the conditions of inactive HBV carriers with a generally benign clinical course [1, 26]. A considerable fraction of non-responders required re-administration of NUCs during follow-up after completing peg-IFN therapy. NUC resumption was decided by physicians only when the patient satisfied the established requirements [11, 12, 26], which indicated that individuals with ALT >80 IU/l and/or HBV DNA >5.8 log copies/ml after stopping peg-IFN were likely to be non-responders. The percentage of responders was 27.4% by intention-to-treat analysis in the present study. Although this figure seems relatively low, it is noteworthy that over 80% of patients with genotype C are reported to be mostly resistant to interferon therapy [31].
Among various factors at the start of peg-IFN, lower levels of HBcrAg and HBsAg were determined as associated with a response to NUC/peg-IFN sequential therapy. HBsAg has been reported to closely relate to peg-IFN therapy outcome [32,33,34,35]. Moucari et al. analyzed HBeAg-negative hepatitis B patients who had been treated with peg-IFN for 48 weeks and concluded that an early serum HBsAg drop was strongly predictive of a sustained virological response [34]. Sonneveld et al. assessed HBeAg-positive hepatitis B patients who had received peg-IFN with or without LAM for 52 weeks and observed that patients experiencing no decline in HBsAg level from baseline at week 12 had little chance of achieving a sustained response and no possibility of an HBsAg loss [32]. HBcrAg includes antigens that are transcribed and translated from pre-core and core genes of the HBV genome, and HBeAg is a primary component of these antigens [16]. HBcrAg levels have been reported to predict the risk of hepatitis relapse after NUC discontinuation [12, 36] as well as the likelihood of developing HCC in patients under NUC therapy [37, 38]. We previously described that a combinational use of HBsAg and HBcrAg levels was useful to predict the 24-month outcome of NUC/IFN sequential therapy [39]. Martinot-Peignoux et al. also reported that HBsAg and HBcrAg levels either separately or in combination could estimate the response to peg-IFN-based precision therapy [40]. However, the above studies suffered from a retrospective design and relatively small number of patients. It was therefore meaningful that the present prospective cohort study confirmed the ability of combined HBsAg and HBcrAg levels for predicting a response to NUC/peg-IFN sequential therapy.
We witnessed that levels of both HBsAg and HBcrAg were significantly associated with that of HBV cccDNA in the liver, even under the sufficient suppression of HBV replication by NUCs. Intrahepatic HBV cccDNA is a key molecule in HBV replication whose amount is closely related to viral activity. Several reports have shown that HBV cccDNA is associated with the response to antiviral therapies using peg-IFN and NUCs [35, 41, 42]. Sung et al. analyzed HBeAg-positive hepatitis B patients who had been treated with either LAM monotherapy or a combination of peg-IFN and LAM and concluded that intrahepatic HBV cccDNA level at the end of therapy was superior to serum HBV DNA in predicting a sustained virological response [41]. Serum HBV DNA is correlated with intrahepatic HBV cccDNA and is widely used as an indicator of HBV replication activity. However, such associations may be incompatible with antiviral therapies, and especially NUC treatment, since the drugs directly hamper production of the HBV virion by inhibiting reverse transcription of pre-genomic RNA without affecting HBV cccDNA directly. As levels of serum HBsAg and HBcrAg are easier to measure than that of intrahepatic HBV cccDNA, these assays may be more suitable as surrogate markers of HBV replication activity in patients undergoing antiviral therapy. It is possible that HBsAg and HBcrAg have complimentary roles in monitoring antiviral effects because their production is regulated by alternative enhancer-promoter systems in the HBV genome. Accordingly, the levels of the two antigens showed a weak, non-significant correlation in the present study.
HBsAg and HBcrAg levels decreased in both responders and non-responders during peg-IFN administration but continued falling in responders only after stopping peg-IFN. Consequently, we presumed that the levels of HBsAg and HBcrAg at the treatment response judgment point of 48 weeks after peg-IFN cessation were closely associated with therapy response. Analysis of baseline parameters associated with low HBcrAg levels (<3.8 log U/ml) at response judgment revealed lower HBcrAg to be the sole significant factor. On the other hand, lower HBsAg, lower HBcrAg, and the use of ADV were significant baseline factors predicting decreased HBsAg levels (<2.0 log IU/ml) at response judgment. Murata et al. reported that serum IFN-λ3 level increased during ADV and TDF administration but did not with LAM and ETV in patients with chronic hepatitis B. They also showed that ADV and TDF exerted the additional pharmacological effect of IFN-λ3 upregulation, which further induced IFN-stimulated genes and resulted in a reduction of HBsAg production in a cell culture system [24]. IFN-λ3 levels at baseline were approximately ten times higher in patients with ADV treatment than in those without in our cohort. It is therefore plausible that the administration of ADV is related to HBsAg level reduction during sequential therapy. Further studies are required to clarify this notion.
In conclusion, the combinational use of HBsAg and HBcrAg levels was able to predict the response to NUC/peg-IFN sequential therapy. Decreases in these antigens during and after peg-IFN administration were closely associated with a favorable response. The use of ADV was related to higher serum IFN-λ3 and suspected to be linked to a rapid decrease in HBsAg level during sequential therapy.
Abbreviations
- HBV:
-
Hepatitis B virus
- HCC:
-
Hepatocellular carcinoma
- NUCs:
-
Nucleos(t)ide analogues
- IFN:
-
Interferon-alpha
- LAM:
-
Lamivudine
- ADV:
-
Adefovir dipivoxil
- ETV:
-
Entecavir
- TDF:
-
Tenofovir disoproxil fumarate
- HBsAg:
-
Hepatitis B surface antigen
- HBcrAg:
-
Hepatitis B core-related antigen
- peg-IFN:
-
Peg-IFN-alpha 2a
- ALT:
-
Alanine transaminase
- HBeAg:
-
Hepatitis B e antigen
- CLEIA:
-
Chemiluminescence enzyme immunoassay
- cccDNA:
-
Covalently closed circular DNA
- IFN-λ:
-
Interferon-lambda
- ROC:
-
Receiver operating characteristic curve
- AUC:
-
Area under the curve
- PPV:
-
Positive predictive value
- NPV:
-
Negative predictive value
- CI:
-
Confidence interval
References
Hoofnagle JH, Doo E, Liang TJ, et al. Management of hepatitis B: summary of a clinical research workshop. Hepatology. 2007;45:1056–75.
Lee WM. Hepatitis B virus infection. N Engl J Med. 1997;337:1733–45.
Lok AS, McMahon BJ. Chronic hepatitis B. Hepatology. 2007;45:507–39.
Okanoue T, Minami M. Update of research and management of hepatitis B. J Gastroenterol. 2006;41:107–18.
Serfaty L, Thabut D, Zoulim F, et al. Sequential treatment with lamivudine and interferon monotherapies in patients with chronic hepatitis B not responding to interferon alone: results of a pilot study. Hepatology. 2001;34:573–7.
Shi M, Wang RS, Zhang H, et al. Sequential treatment with lamivudine and interferon-alpha monotherapies in hepatitis B e antigen-negative Chinese patients and its suppression of lamivudine-resistant mutations. J Antimicrob Chemother. 2006;58:1031–5.
Manesis EK, Papatheodoridis GV, Hadziyannis SJ. A partially overlapping treatment course with lamivudine and interferon in hepatitis B e antigen-negative chronic hepatitis B. Aliment Pharmacol Ther. 2006;23:99–106.
Enomoto M, Nishiguchi S, Tamori A, et al. Entecavir and interferon-alpha sequential therapy in Japanese patients with hepatitis B e antigen-positive chronic hepatitis B. J Gastroenterol. 2013;48:397–404.
Enomoto M, Tamori A, Nishiguchi S, et al. Combination therapy with a nucleos(t)ide analogue and interferon for chronic hepatitis B: simultaneous or sequential. J Gastroenterol. 2013;48:999–1005.
Minami M, Okanoue T. Management of HBV infection in Japan. Hepatol Res. 2007;37:S79–82.
Matsumoto A, Tanaka E, Suzuki Y, et al. Combination of hepatitis B viral antigens and DNA for prediction of relapse after discontinuation of nucleos(t)ide analogs in patients with chronic hepatitis B. Hepatol Res. 2012;42:139–49.
Tanaka E, Matsumoto A. Guidelines for avoiding risks resulting from discontinuation of nucleoside/nucleotide analogs in patients with chronic hepatitis B. Hepatol Res. 2014;44:1–8.
Kumada H, Okanoue T, Onji M, et al. Guidelines for the treatment of chronic hepatitis and cirrhosis due to hepatitis B virus infection for the fiscal year 2008 in Japan. Hepatol Res. 2010;40:1–7.
Orito E, Ichida T, Sakugawa H, et al. Geographic distribution of hepatitis B virus (HBV) genotype in patients with chronic HBV infection in Japan. Hepatology. 2001;34:590–4.
Schuttler CG, Wend UC, Faupel FM, et al. Antigenic and physicochemical characterization of the 2nd International Standard for hepatitis B virus surface antigen (HBsAg). J Clin Virol. 2010;47:238–42.
Kimura T, Rokuhara A, Sakamoto Y, et al. Sensitive enzyme immunoassay for hepatitis B virus core-related antigens and their correlation to virus load. J Clin Microbiol. 2002;40:439–45.
Ronsin C, Pillet A, Bali C, et al. Evaluation of the COBAS AmpliPrep-total nucleic acid isolation-COBAS TaqMan hepatitis B virus (HBV) quantitative test and comparison to the VERSANT HBV DNA 3.0 assay. J Clin Microbiol. 2006;44:1390–9.
Mizokami M, Nakano T, Orito E, et al. Hepatitis B virus genotype assignment using restriction fragment length polymorphism patterns. FEBS Lett. 1999;450:66–71.
Suzuki F, Miyakoshi H, Kobayashi M, et al. Correlation between serum hepatitis B virus core-related antigen and intrahepatic covalently closed circular DNA in chronic hepatitis B patients. J Med Virol. 2009;81:27–33.
Mason AL, Xu L, Guo L, et al. Molecular basis for persistent hepatitis B virus infection in the liver after clearance of serum hepatitis B surface antigen. Hepatology. 1998;27:1736–42.
Ito K, Higami K, Masaki N, et al. The rs8099917 polymorphism, when determined by a suitable genotyping method, is a better predictor for response to pegylated alpha interferon/ribavirin therapy in Japanese patients than other single nucleotide polymorphisms associated with interleukin-28B. J Clin Microbiol. 2011;49:1853–60.
Aoki Y, Sugiyama M, Murata K, et al. Association of serum IFN-lambda3 with inflammatory and fibrosis markers in patients with chronic hepatitis C virus infection. J Gastroenterol. 2015;50:894–902.
Sugiyama M, Kimura T, Naito S, et al. Development of specific and quantitative real-time detection PCR and immunoassays for lambda3-interferon. Hepatol Res. 2012;42:1089–99.
Murata K, Asano M, Matsumoto A, et al. Induction of IFN-lambda3 as an additional effect of nucleotide, not nucleoside, analogues: a new potential target for HBV infection. Gut. 2017 (in press).
Ichida F, Tsuji T, Omata M, et al. New Inuyama classification; new criteria for histological assessment of chronic hepatitis. Int Hepatol Commun. 1996;6:112–9.
Drafting Committee for Hepatitis Management Guidelines and the Japan Society of Hepatology. JSH guidelines for the management of hepatitis B virus infection. Hepatol Res. 2014;44(Suppl S1):1–58.
Papatheodoridis GV, Lampertico P, Manolakopoulos S, et al. Incidence of hepatocellular carcinoma in chronic hepatitis B patients receiving nucleos(t)ide therapy: a systematic review. J Hepatol. 2010;53:348–56.
Marcellin P, Lau GK, Bonino F, et al. Peginterferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HBeAg-negative chronic hepatitis B. N Engl J Med. 2004;351:1206–17.
Lau GK, Piratvisuth T, Luo KX, et al. Peginterferon alfa-2a, lamivudine, and the combination for HBeAg-positive chronic hepatitis B. N Engl J Med. 2005;352:2682–95.
Kim V, Abreu RM, Nakagawa DM, et al. Pegylated interferon alfa for chronic hepatitis B: systematic review and meta-analysis. J Viral Hepat. 2016;23:154–69.
Shi YH. Correlation between hepatitis B virus genotypes and clinical outcomes. Jpn J Infect Dis. 2012;65:476–82.
Sonneveld MJ, Rijckborst V, Boucher CA, et al. Prediction of sustained response to peginterferon alfa-2b for hepatitis B e antigen-positive chronic hepatitis B using on-treatment hepatitis B surface antigen decline. Hepatology. 2010;52:1251–7.
Brunetto MR, Moriconi F, Bonino F, et al. Hepatitis B virus surface antigen levels: a guide to sustained response to peginterferon alfa-2a in HBeAg-negative chronic hepatitis B. Hepatology. 2009;49:1141–50.
Moucari R, Mackiewicz V, Lada O, et al. Early serum HBsAg drop: a strong predictor of sustained virological response to pegylated interferon alfa-2a in HBeAg-negative patients. Hepatology. 2009;49:1151–7.
Chan HL, Wong VW, Tse AM, et al. Serum hepatitis B surface antigen quantitation can reflect hepatitis B virus in the liver and predict treatment response. Clin Gastroenterol Hepatol. 2007;5:1462–8.
Matsumoto A, Tanaka E, Minami M, et al. Low serum level of hepatitis B core-related antigen indicates unlikely reactivation of hepatitis after cessation of lamivudine therapy. Hepatol Res. 2007;37:661–6.
Hosaka T, Suzuki F, Kobayashi M, et al. HBcrAg is a predictor of post-treatment recurrence of hepatocellular carcinoma during antiviral therapy. Liver Int. 2010;30:1461–70.
Kumada T, Toyoda H, Tada T, et al. Effect of nucleos(t)ide analogue therapy on hepatocarcinogenesis in chronic hepatitis B patients: a propensity score analysis. J Hepatol. 2013;58:427–33.
Matsumoto A, Yatsuhashi H, Nagaoka S, et al. Factors associated with the effect of interferon-alpha sequential therapy in order to discontinue nucleoside/nucleotide analog treatment in patients with chronic hepatitis B. Hepatol Res. 2015;45:1195–202.
Martinot-Peignoux M, Lapalus M, Maylin S, et al. Baseline HBsAg and HBcrAg titres allow peginterferon-based ‘precision medicine’ in HBeAg-negative chronic hepatitis B patients. J Viral Hepat. 2016;23:905–11.
Sung JJ, Wong ML, Bowden S, et al. Intrahepatic hepatitis B virus covalently closed circular DNA can be a predictor of sustained response to therapy. Gastroenterology. 2005;128:1890–7.
Wursthorn K, Lutgehetmann M, Dandri M, et al. Peginterferon alpha-2b plus adefovir induce strong cccDNA decline and HBsAg reduction in patients with chronic hepatitis B. Hepatology. 2006;44:675–84.
Acknowledgements
We thank Ms. Hiroe Banno for her secretarial assistance and Mr. Trevor Ralph for his English editorial assistance. This research is partially supported by the Research Program on Hepatitis and the Program for Basic and Clinical Research on Hepatitis from the Ministry of Health, Labor, and Welfare of Japan as well as by the Japan Agency for Medical Research and Development (AMED).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Nishiguchi S. received lecture fees from Gilead Sciences and MSD; Dr. Tanaka Y. received honoraria from Bristol-Myers Squibb, FUJIREBIO, Chugai, and Glaxo Smith Kline; Dr. Kanda T. received a research Grant from MSD, AbbVie and Chugai; Dr. Okuse C. received honoraria from AbbVie, and Otsuka; Dr. Takaguchi K. received lecture fees from Bristol Meyer Squib, AbbVie, and Astra Zeneca; Dr. Hino K. received lecture fees from Bristol Meyers Squibb, Otsuka, MSD, Dainippon Sumitomo, and Gilead Sciences. Dr. Hino K. also received a research grant from Bristol Meyers Squibb; Dr. Tsuge M. received Grant/Research Support from Bristol-Myers Squibb; Dr. Chayama K. received honoraria from Dainippon Sumitomo, Otsuka, Gilead, Bristol-Myers Squibb, AbbVie, MSD, Ajinomoto, Eisai, Toray, Tanabe Mitsubishi, Chugai, Daiichi Sankyo, Takeda, and Nihon Kayaku. Dr. Chayama K. also received commercial research funding from Bristol-Myers Squibb, AbbVie, Toray, and Dainippon Sumitomo; Dr. Suzuki Y. received lecture fees from AbbVie, GK, and Bristol-Myers Squibb; Dr. Tanaka E. received honoraria from Chugai, Bristol-Myers Squibb, Glaxo Smith Kline, and Gilead Sciences.
Rights and permissions
About this article
Cite this article
Matsumoto, A., Nishiguchi, S., Enomoto, H. et al. Combinational use of hepatitis B viral antigens predicts responses to nucleos(t)ide analogue/peg-interferon sequential therapy. J Gastroenterol 53, 247–257 (2018). https://doi.org/10.1007/s00535-017-1360-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-017-1360-z