Abstract
Objective
The aim of this sub-analysis was to highlight the MASCC/ISOO clinical practice guidelines for the management of oral mucositis (OM) in pediatric patients and to present unique considerations in this patient population.
Methods
This sub-analysis of the pediatric patient population is based on the systematic review conducted by the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISSO) published in 2019/2020. Studies were scored and assigned a level of evidence based on previously published criteria. Data regarding adverse effects and compliance was collected from the original publications.
Results
A total of 45 papers were included and assessed in this sub-analysis, including 21 randomized controlled trials (RCTs). Chewing gum was demonstrated to be not effective in preventing OM in pediatric cancer patients in 2 RCTs. The efficacy of all other interventions could not be determined based on the available literature.
Conclusion
There is limited or conflicting evidence about interventions for the management of OM in pediatric cancer patients, except for chewing gum which was ineffective for prevention. Therefore, currently, data from adult studies may need to be extrapolated for the management of pediatric patients. Honey and photobiomodulation therapy in this patient population had encouraging potential. Implementation of a basic oral care protocol is advised amid lack of high level of evidence studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Oral mucositis (OM) is an important complication related to anti-neoplastic therapy, such as chemotherapy (CT), radiotherapy (RT), radio-chemotherapy (RT-CT) and hematopoietic stem cell transplantation (HSCT) [1, 2]. Pain and difficulty eating have been associated with a negative impact on the patient’s quality of life [3, 4]. In addition, OM is associated with an increased risk for systemic infections [5, 6] and high financial costs [1, 7].
The Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) has published clinical practice guidelines for the management of OM since 2003, with updates published in 2007, 2014 and 2019–20 [8,9,10,11,12,13,14,15,16]. The guidelines are presented according to the following categories of interventions: (1) anti-inflammatory; (2) antimicrobials, mucosal coating agents, anesthetics, and analgesics; (3) basic oral care; (4) cryotherapy; (5) growth factors and cytokines; (6) photobiomodulation (PBM); and (7) natural and miscellaneous agents. Studies on the interventions for gastrointestinal mucositis were reviewed too [9].
Although high-quality studies investigating OM in pediatric patients are still scarce, there has been some increase in the literature pertaining to this population. Therefore, it is timely to synthesize the available evidence on the management of mucositis in this sub-population.
As part of a better understanding of the MASCC/ISOO clinical practice guidelines for the management of cancer therapy-induced OM, the aim of this sub-analysis is to highlight the evidence related to OM management in the pediatric patient population and to focus on unique considerations.
Methods
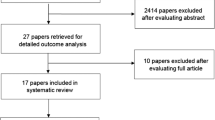
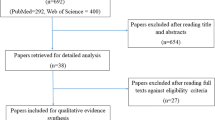
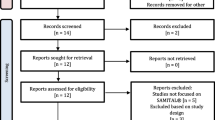
The methods related to this sub-analysis are described in detail in Ranna et al. [17]. Briefly, a literature search for relevant papers indexed from January 1, 2011, to June 2016 was conducted using PubMed and Web Science, with papers selected for review based on defined inclusion and exclusion criteria [17]. Additionally, the literature review identified manually randomized controlled trials (RCT) published until July 2019. The list of keywords used by all sections is detailed in Ranna et al. and in the respective section’s articles [8,9,10,11,12,13,14,15,16]. Studies were scored for their level of evidence (LoE) based on Somerfield criteria [18], and flaws were listed according to Hadorn criteria [19]. A well-designed study was defined as a study with no major flaws per the Hadorn criteria. Findings from the reviewed studies were merged with the evidence examined in the previous MASCC/ISOO guideline update, culminating in a systematic review of all literature until March 2020. Then, data was integrated into the guidelines based on the LoE for each intervention. Studies were grouped based on (1) the aim of the intervention (prevention or treatment of mucositis), (2) the cancer treatment modality (RT, CT, RT-CT or HSCT with or without total body irradiation (TBI), (3) the route of administration of the intervention and (4) the agent. Additionally, publications on the pediatric patient population were reviewed from each section and data about patient compliance with the treatment and adverse effects (AEs) were collected using a standard electronic form. Guidelines of this sub-analysis are presented based on the category of intervention.
Results
The literature search identified a total of 10,195 papers from all sections, which were triaged. Following the merge with the papers included in the previous MASCC/ISOO guidelines, 340 (175 RCT) papers were included in the systematic review. For the pediatric sub-analysis, 45 papers were included, 20 RCTs, 6 comparator RCTs (randomizing to either another intervention or to an active control), 4 non-randomized comparative (comparing an intervention to a placebo-control or no treatment), 1 cross-over, 5 before-and-after, 4 case-control, and 5 cohort studies.
Efficacy studies on basic oral care
Multi-agent combination oral care protocols
The preventive efficacy of combined multi-agent oral care protocols for pediatric patients with hematologic malignancies treated with CT regimens were assessed in 5 studies (1 RCT, 3 comparative studies, and 1 before-and-after study) [20,21,22,23,24]. The RCT [20] found no significant difference in OM incidence between the experimental group that received a combination of enhanced oral physiotherapy, use of 0.05% non-alcoholic fluoride mouthwashes daily and 20% miconazole oral gel applications (n = 5) and the control group with no intervention (n = 7). The findings from the 4 remaining non-RCTs [21,22,23,24] consistently indicated that the implementation of a multi-agent combination of oral care protocols is beneficial for the prevention of OM or pain during CT for pediatric cancer patients (Tables 1 and 2).
Patient education
A single before-and-after study including hematologic and non-hematologic pediatric cancer patients (n = 16) concluded that both the degree of OM and OM-related pain decreased (p < 0.05) when patients were given mouth care education before CT and when they regularly performed mouth care (Tables 1 and 2) [25].
Chlorhexidine
Three studies (2 RCTs and 1 comparative study) assessed OM outcomes in pediatric cancer patients (Tables 1, 2, 3, 4 and 5) [26,27,28]. The RCT, which compared 23 patients in the intervention group with 24 in a placebo group, concluded that 0.12% chlorhexidine mouthwash did not significantly reduce the incidence of OM in pediatric HSCT recipients [28]. The comparative study analyzed 14 pediatric patients (7 patients each for both the experimental and control group) treated with intensive CT regimens for hematologic malignancies and found positive results on the effectiveness of chlorhexidine 0.12% in reducing incidence of OM in the experimental group (p < 0.05) [27]. The comparator RCT compared chlorhexidine to benzydamine and the details are given in the next paragraph under benzydamine (Tables 4 and 5) [26].
Efficacy studies about anti-inflammatory agents
Benzydamine
There were no RCTs comparing benzydamine to a placebo. A single RCT comparing benzydamine to chlorhexidine reported their findings in 3 publications (Tables 4 and 5) [26, 29, 30]. The aim of this RCT was the prevention of OM in pediatric patients undergoing CT for hematologic and solid cancer. The study was designed as a non-blinded cross-over trial, where each patient behaved as their own control. The results of this RCT reported benzydamine was not superior to chlorhexidine. Another small cross-over study of 4 patients assessing benzydamine for the treatment of OM in pediatric patients with hematologic malignancies and solid tumors treated with CT reported benzydamine was not effective in reducing pain (Tables 1, 2 and 3) [31]. In this study, benzydamine mouthwash was being alternated with Hospital for Sick Children (HSC) mouthwash (nystatin 7000 U/mL, lidocaine viscous 0.58 mL/mL in NaCl 0.9%) over 2 cycles.
Efficacy studies about antimicrobials, mucosal coating agents, anesthetics, and analgesics
Sucralfate
A single double-blind RCT assessed the effectiveness of swish and swallow suspension with sucralfate in hematological cancer pediatric patients experimental (n = 24) and control (n = 24) groups who underwent CT (Tables 1, 2 and 3) [32]. Patients receiving sucralfate reported less oral discomfort (p = 0.06) but there was no statistically significant difference in pain severity between the groups.
Polymyxin-tobramycin-amphotericin B lozenges
A single comparator study evaluated the use of topical polymyxin-tobramycin-amphotericin B lozenges (n = 12) to a mouthwash containing diphenhydramine, and topical anesthetic in an oral suspension of aluminum hydroxide and magnesium hydroxide (n = 14) in the treatment of OM in hematological and solid cancer pediatric patients that underwent HSCT with or without TBI (Tables 4 and 5) [33]. The OM score in the group taking polymyxin-tobramycin-amphotericin-B was significantly lower (p < 0.05). The 2014 mucositis guidelines paper of the MASCC/ISOO reported that the clinical difference was small [34].
Morphine
A double-blind cross-over RCT evaluating the use of morphine in pediatric patients that underwent autologous or allogenic HSCT found no statistical difference in daily pain scores between the compared groups (Tables 4 and 5) [35]. There were two groups that differed in the orders of the interventions. The first group was composed of pediatric patients that received morphine followed by hydromorphone and then morphine (n = 5). The second group initiated the protocol with hydromorphone followed by morphine and then hydromorphone (n = 5), both delivered as patient-controlled analgesia (PCA) protocol. A second RCT evaluated pediatric patients that underwent HSCT with or without TBI and compared OM-related pain outcomes between a PCA protocol and a continuous infusion regimen (Tables 4 and 5) [36]. It concluded that there was a significantly lower morphine intake in the PCA group (p < 0.01), but there was no difference in pain intensity between the two groups.
Ketamine
A single before-and-after study [37] analyzed the addition of ketamine to morphine PCA or nurse-controlled analgesia (NCA) protocols in regard to OM-related pain relief. This study showed that ketamine added either to a PCA or NCA protocol improved analgesic efficacy in pediatric patients with hematologic and solid malignancies that underwent variable CT regimens (p = 0.01) (Tables 1, 2 and 3).
Efficacy studies about growth factors and cytokines
Keratinocyte growth factors
A single RCT, published in 2 parts, about intravenous (IV) keratinocyte growth factors (KGF-1) to prevent OM in pediatric patients undergoing HSCT reported the results of 27 patients treated with either IV KGF-1 or placebo (Tables 1, 2 and 3) [38, 39]. This RCT showed effectiveness in reducing the severity (p = 0.03) and duration of OM (p < 0.001). Three other studies (1 cohort [40] and 3 case-control studies [41,42,43]) reported that KGF-1 was effective in reducing mucositis severity or duration, and that patients complied well.
Granulocyte colony-stimulating factor
A single RCT studied the effect of subcutaneous (SC) granulocyte colony-stimulating factor (G-CSF) included 148 pediatric patients with hematologic malignancies treated with high-dose CT. The results indicated that the use of G-CSF was ineffective to prevent severe OM (Tables 1, 2 and 3) [44].
Granulocyte-macrophage colony-stimulating factor
There is limited evidence for the use of IV granulocyte-macrophage colony-stimulating factor (GM-CSF) in pediatric patients undergoing HSCT for the prevention of OM (Tables 1, 2 and 3) [45]. This cohort reported of non-significant reduction in duration of OM.
Transforming growth factor-β—nutrition or mouthwash
A single RCT on the use of transforming growth factor-β (TGF-β) for the prevention of OM in 25 pediatric patients treated with CT (Tables 1, 2 and 3) [46] concluded that TGF-β is not effective for the prevention of CT induced OM.
Efficacy studies about natural and miscellaneous agents
Supersaturated calcium phosphate rinse
A single RCT of 29 pediatric patients undergoing HSCT or CT reported the efficacy of supersaturated calcium phosphate rinse (SCPR) (Caphosol®, EUSA Pharma, Hemel Hempstead, UK) for the treatment of OM. The study reported that SCPR is ineffective for the treatment of OM in alleviating severity, duration, and pain severity (Tables 1, 2 and 3) [47].
Glutamine
A single RCT including 118 patients [48] and a case-control study [49] that assessed the efficacy of parenteral glutamine in pediatric patients reported no beneficial effect of glutamine (parenteral) for prevention of OM in HSCT (Tables 1, 2 and 3). Contrary to this a single RCT on oral glutamine based on the [50] same patient population (n = 120) showed glutamine to be effective in reducing pain duration (p = 0.01) and to have a tendency to reduce OM severity. Further, some studies with lower quality designs reported that parenteral glutamine was ineffective in the prevention of OM in patients with hematologic cancer treated with CT [51, 52].
Vitamin E
A single RCT compared topical vitamin E to a placebo for the prevention of OM in hematologic malignancies and solid cancer patients treated with CT failed to demonstrate a reduction in OM severity (Tables 1, 2 and 3) [53]. The sample size was 16 patients; however, it is unclear how many patients were in each arm since data was reported per treatment cycle.
Another RCT that evaluated the efficacy of swish and swallow vitamin E compared with pycnogenol or glycerin (vehicle-control) in 72 pediatric patients treated with CT (Tables 1, 2 and 3) [54] reported no significant difference between both active arms. However, the two active arms were effective compared with the control arm for the treatment of OM (p < 0.001).
Calcitriol
A single RCT described the use of calcitriol for the prevention of OM in 28 patients undergoing HSCT due to Fanconi anemia (Tables 1, 2, and 3) [55]. The study did not report considerable benefits for calcitriol in preventing OM.
Honey
A single placebo-controlled RCT of topical honey for the treatment of OM in 90 pediatric patients with hematologic malignancies undergoing CT (Tables 1, 2 and 3) [56] reported the effectiveness of the Trifolium alexandrinum-based honey in reducing the healing time of ulcerative OM. (p = 0.0005). Additionally, a mixture of honey, olive oil-propolis extract, and beeswax (HOPE) was significantly more effective than in the control (p = 0.0056).
Propolis
A single RCT reported the ineffectiveness of topical propolis for the treatment of OM in pediatric patients (n = 40) treated with CT for hematological and solid cancers (Tables 1 and 2) [57].
Traumeel
Two RCTs investigated the use of Traumeel for the prevention of OM in pediatric patients undergoing HSCT (Tables 1, 2 and 3) [58, 59]. The pediatric patients were instructed to swish and swallow in both studies. In the first RCT with hematologic patients (n = 30), Traumeel illustrated a significant reduction in the severity and duration of OM (p < 0.01) [58]. On the other hand, in the second RCT, Traumeel was ineffective for the prevention of OM in pediatric patients with hematologic and solid cancers (n = 181) [59].
Chewing gum
Two RCTs described the use of chewing gum for the prevention of OM in pediatric patients treated with CT for hematologic malignancies and solid cancers (Tables 1, 2 and 3) [60, 61]. In one RCT with 145 pediatric patients, oral care alone was compared with oral care combined with a chewing gum routine [61]. In the other RCT with 130 pediatric patients, the use of chewing gum and “magic mouthwash” (nystatin, diphenhydramine, and aluminum) was compared with “magic mouthwash” alone [60]. Both studies reported chewing gum was not effective to prevent OM.
Pycnogenol
A single RCT reported on pycnogenol compared with vitamin E or a control group and results are presented under the “Vitamin E” section above [54].
Efficacy studies on cryotherapy
One study evaluated the effectiveness of cryotherapy together with the vasoconstrictor propantheline (n = 12), compared to a single intervention (cryotherapy or propantheline; n = 5) and to a control group receiving no treatment (n = 7) for the prevention of OM in pediatric patients undergoing HSCT with or without TBI (Tables 1, 2 and 3) [62]. Cryotherapy was applied 5–10 min before and at least 30 min following the CT infusion. Propantheline was administered at 1.5 mg/kg once daily from the day before CT infusion until one day before the HSCT. The results showed that the combination protocol was significantly more effective in reducing the incidence of severe OM (grades 3–4) compared to the other two groups (p = 0.0069).
Efficacy studies on PBM
The management of OM with intra-oral PBM in a mixed pediatric patient population, including hematologic malignancies and solid cancers, treated with either high-dose CT or HSCT has been described by two studies (Tables 1, 2, 3 and 6) [63, 64]. The RCT with 123 pediatric patients observed the effectiveness of intra-oral PBM in pain reduction (p < 0.005) [63]. A cohort study (n = 18) with intra-oral PBM therapy reported a significant decrease in the severity of OM (p < 0.05) and pain reduction (p < 0.001) [64]
.
Another cohort study on the effectiveness of extra-oral PBM for the prevention of OM in a mixed pediatric patient population (n = 32), including hematologic malignancies and solid cancers treated with HSCT was reported (Tables 1, 2 and 3) [65]. The results demonstrated a significant reduction in OM-associated pain.
AEs and tolerability
The active agents used in this sub-analysis were generally well tolerated by pediatric patients (Table 3). Interventions for which AEs were reported are detailed below.
Chlorhexidine and benzydamine
Chlorhexidine (0.12–0.2%) and benzydamine (0.15%) have been related to stinging or burning sensations [26, 27, 29, 30]. In an RCT, the dilution of the chlorhexidine and benzydamine mouthwashes with saline or water was required in 6% and 3% of patients, respectively [30]. In another RCT, once OM developed, there was an increase in patients who required dilution of chlorhexidine and benzydamine due to stinging and burning sensations in the oral cavity to 33% and 20% of patients, respectively [29]. Nevertheless, no patient discontinued the use of mouthwashes with both active agents. On the other hand, a small study reported severe oral stinging when benzydamine (0.15%) mouthwash was administered, and 3 out of 4 patients dropped out of the study due to this adverse effect [31].
In addition, a minor taste alteration has been reported by pediatric patients when using chlorhexidine (0.2%) and benzydamine (0.15%); in 6% and 3–9% of patients, respectively [29, 30].
Propolis
A transient burning sensation has been reported by 26.7% of pediatric patients immediately after application of a HOPE mixture [56].
Photobiomodulation
A burning sensation was reported following PBM therapy in 50% of pediatric patients (9 out 18) [64]. Other studies did not report any AEs.
KGF-1
AEs related to KGF-1 were skin rash (22–44% of patients) [38,39,40,41,42], altered taste (10–20%) [38, 39, 42], gingival hyperplasia (11%) [40], a sensation of increased tongue thickness (12.5–16%) [38, 39], lip swelling (11%) [40], pharyngeal plaque (11%) [40], and face edema (11%) [40].
Sucralfate
Eight (33.3%) patients that received a placebo suspension and 4 (16.6%) patients that received a swish and swallow sucralfate suspension experienced cutaneous rash [32].
Morphine and hydromorphone
Patients that underwent a cross-over of morphine and hydromorphone reported mild sedation, nausea, vomiting, and pruritus during the administration of the protocol. Also, 1 patient presented with urinary retention during hydromorphone use, which required urinary catheterization [35]. The single administration of morphine by a PCA or continuous-infusion was associated with mild nausea, concentration issues, and sleepiness during both administered protocols [36].
Ketamine
There was an overall incidence of nausea/vomiting and pruritus related to ketamine in 58% and 34% of patients, respectively. One patient had the ketamine withdrawn after becoming acutely confused during a septic episode and required intensive care [37].
Vitamin E
Vitamin E was associated with an uncomfortable “oily texture” by 12 out of 16 patients in a single RCT [53].
Compliance with treatment
The compliance was reported in 31 out of 45 of the papers (Tables 3 and 5). The compliance of pediatric patients was based on patient self-reports or parental reports. Studies reported compliance as a dichotomic categorical variable (good/poor) [21, 31, 38,39,40,41,42,43, 46, 47, 51,52,53,54,55,56,57,58,59, 62, 64, 65] or as a quantitative analysis [22, 23, 26, 28,29,30, 50, 59, 61].
Compliance was adequate in most of the studied patients, except for some cases. For example, there was a limitation specific to younger children to stay awake during oral cryotherapy application when it was performed in the evening [62].
Discussion
This sub-analysis paper of the MASCC/ISOO clinical practice guidelines for the management of mucositis presented the available evidence regarding interventions for OM in pediatric cancer patients. Although over a dozen studies were large RCTs, the type and quality of the study design, as well as the volume of the studies are not as robust as the evidence in the adult patient population, except for chewing gum. Furthermore, the diversity of the pediatric cancer patients population in these studies, and the combination of experimental agents in a single study protocol, compromise the conclusion about an effective intervention for oral mucositis. Therefore, interventional protocols in pediatric patients need to rely on extrapolation from the currently available evidence for adult cancer patients.
The literature included in this sub-analysis reported of either oral or systemic drug-related AEs. Benzydamine or chlorhexidine necessitates dilution or dose reduction [29, 30]. Drug withdrawal was infrequently needed for benzydamine or ketamine [31, 37]. Notwithstanding, parenteral glutamine was reported to be associated with increased relapse and mortality in adult HSCT patients [66].
In the pediatric oncology setting, specific instruments may be helpful to assess OM and OM-related pain. Depending on the age of the patient, language development, emotional difficulty in expressing themselves, comprehensive skills, and attention capacity, it can be difficult to apply the conventional scales used for adult cancer patient assessment [67]. Some examples such as the Children’s International Mucositis Evaluation Scale [25, 54], a self-reporting scale designed for children with cancer, as well as visual “smiley face” scales, are validated and reliable tools for this population [22, 23, 31, 37, 54, 63, 65]. Among the selected studies in the current sub-analysis, several types of assessment tools were used, and only some of them were adapted for children.
The effectiveness of an experimental intervention relies on its capacity to prevent or mitigate the signs and symptoms of OM, as well as on its acceptance. The acceptance is driven by ease of use and lack of AEs, psycho-emotional state, and general health condition of the patient [29, 62]. In order to overcome the challenges related to pediatric patient acceptance, unique treatment strategies should be employed. For example, the use of popsicles instead of ice or ice water for younger patients undergoing oral cryotherapy may increase compliance during the application [62]. Likewise, extra-oral PBM may be tolerated better than the intra-oral approach [65, 68, 69]. Studies in cancer and non-cancer pediatric patients suggested strategies utilizing cognitive-behavioral approaches may help to handle stressful situations and consequently enhance engagement in children. For example, the use of storyboards, gaming, virtual-reality experiences, medical clowning, animal-assisted therapy, or a parent-guided therapy approach [70,71,72,73,74,75].
Following the timeframe of the literature search of the MASCC/ISOO guidelines update, 15 RCTs were published addressing interventions for the management of OM in pediatric cancer patients. The new RCTs investigated the use of natural & miscellaneous agents [76,77,78,79,80,81,82], PBM therapy [68, 83,84,85,86], oral cryotherapy [87], G-CSF [88], and KGF-1 vs. chlorhexidine mouthwashes [89]. These newer RCTs suggest efficacy for honey and inefficacy for SCPR. The PBM studies had conflicting results and, due to the diverse PBM protocols, it is impossible to conclude about a preferred PBM setting. The long-term effects of these interventions are unknown. Although the speculative benefit of honey on OM prevention, some concerns about an increased risk of dental caries still remains [14].
In summary, this is the first focused view of MASCC/ISOO on guidelines for the management of OM for pediatric patients. It is a detailed sub-analysis of the guidelines that were developed by this group [90]. There is evidence that chewing gum does not prevent OM. In addition, there is intriguing evidence regarding the efficacy of PBM and honey. Despite the absence of high evidence studies, the implementation of basic oral care protocol is very appropriate.
References
Sonis ST, Oster G, Fuchs H, Bellm L, Bradford WZ, Edelsberg J, Hayden V, Eilers J, Epstein JB, LeVeque FG, Miller C, Peterson DE, Schubert MM, Spijkervet FKL, Horowitz M (2001) Oral mucositis and the clinical and economic outcomes of hematopoietic stem-cell transplantation. J Clin Oncol. 19(8):2201–2205
He M, Zhang B, Shen N, Wu N, Sun J (2018) A systematic review and meta-analysis of the effect of low-level laser therapy (LLLT) on chemotherapy-induced oral mucositis in pediatric and young patients. Eur J Pediatr. 177(1):7–17
Mazhari F, Shirazi AS, Shabzendehdar M (2019) Management of oral mucositis in pediatric patients receiving cancer therapy: A systematic review and meta-analysis. Pediatr Blood Cancer. 66(3):e27403
Epstein JB, Thariat J, Bensadoun RJ, Barasch A, Murphy BA, Kolnick L, Popplewell L, Maghami E (2012) Oral complications of cancer and cancer therapy: from cancer treatment to survivorship. CA Cancer J Clin. 62(6):400–422
Cohen J, Donnelly JP, Worsley AM, Catovsky D, Goldman JM, Galton DA (1983) Septicaemia caused by viridans streptococci in neutropenic patients with leukaemia. Lancet. 2(8365-66):1452–1454
Elting LS, Bodey GP, Keefe BH (1992) Septicemia and shock syndrome due to viridans streptococci: a case-control study of predisposing factors. Clin Infect Dis. 14(6):1201–1207
Bezinelli LM, de Paula EF, da Graca Lopes RM, Biazevic MG, de Paula EC, Correa L et al (2014) Cost-effectiveness of the introduction of specialized oral care with laser therapy in hematopoietic stem cell transplantation. Hematol Oncol. 32(1):31–39
Ariyawardana A, Cheng KKF, Kandwal A, Tilly V, Al-Azri AR, Galiti D et al (2019) Systematic review of anti-inflammatory agents for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer. 27(10):3985–3995
Bowen JM, Gibson RJ, Coller JK, Blijlevens N, Bossi P, Al-Dasooqi N et al (2019) Systematic review of agents for the management of cancer treatment-related gastrointestinal mucositis and clinical practice guidelines. Support Care Cancer. 27(10):4011–4022
Correa MEP, Cheng KKF, Chiang K, Kandwal A, Loprinzi CL, Mori T, Potting C, Rouleau T, Toro JJ, Ranna V, Vaddi A, Peterson DE, Bossi P, Lalla RV, Elad S (2020) Systematic review of oral cryotherapy for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer. 28(5):2449–2456
Hong CHL, Gueiros LA, Fulton JS, Cheng KKF, Kandwal A, Galiti D et al (2019) Systematic review of basic oral care for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer. 27(10):3949–3967
Logan RM, Al-Azri AR, Bossi P, Stringer AM, Joy JK, Soga Y et al (2020) Systematic review of growth factors and cytokines for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer. 28(5):2485–2498
Saunders DP, Rouleau T, Cheng K, Yarom N, Kandwal A, Joy J et al (2020) Systematic review of antimicrobials, mucosal coating agents, anesthetics, and analgesics for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer. 28(5):2473–2484
Yarom N, Hovan A, Bossi P, Ariyawardana A, Jensen SB, Gobbo M et al (2020) Systematic review of natural and miscellaneous agents, for the management of oral mucositis in cancer patients and clinical practice guidelines - part 2: honey, herbal compounds, saliva stimulants, probiotics, and miscellaneous agents. Support Care Cancer. 28(5):2457–2472
Yarom N, Hovan A, Bossi P, Ariyawardana A, Jensen SB, Gobbo M et al (2019) Systematic review of natural and miscellaneous agents for the management of oral mucositis in cancer patients and clinical practice guidelines-part 1: vitamins, minerals, and nutritional supplements. Support Care Cancer. 27(10):3997–4010
Zadik Y, Arany PR, Fregnani ER, Bossi P, Antunes HS, Bensadoun RJ et al (2019) Systematic review of photobiomodulation for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer. 27(10):3969–3983
Ranna V, Cheng KKF, Castillo DA, Porcello L, Vaddi A, Lalla RV et al (2019) Development of the MASCC/ISOO clinical practice guidelines for mucositis: an overview of the methods. Support Care Cancer. 27(10):3933–3948
Somerfield MR, McCrae RR (2000) Stress and coping research. Methodological challenges, theoretical advances, and clinical applications. Am Psychol. 55(6):620–625
Hadorn DC, Baker D, Hodges JS, Hicks N (1996) Rating the quality of evidence for clinical practice guidelines. J Clin Epidemiol. 49(7):749–754
Rojas de Morales T, Zambrano O, Rivera L, Navas R, Chaparro N, Bernardonni C et al (2001) Oral-disease prevention in children with cancer: testing preventive protocol effectiveness. Med Oral. 6(5):326–334
Chen CF, Wang RH, Cheng SN, Chang YC (2004) Assessment of chemotherapy-induced oral complications in children with cancer. J Pediatr Oncol Nurs. 21(1):33–39
Cheng KK, Molassiotis A, Chang AM (2002) An oral care protocol intervention to prevent chemotherapy-induced oral mucositis in paediatric cancer patients: a pilot study. Eur J Oncol Nurs. 6(2):66–73
Cheng KK, Molassiotis A, Chang AM, Wai WC, Cheung SS (2001) Evaluation of an oral care protocol intervention in the prevention of chemotherapy-induced oral mucositis in paediatric cancer patients. Eur J Cancer. 37(16):2056–2063
Levy-Polack MP, Sebelli P, Polack NL (1998) Incidence of oral complications and application of a preventive protocol in children with acute leukemia. Spec Care Dentist. 18(5):189–193
Yavuz B, Bal YH (2015) Investigation of the effects of planned mouth care education on the degree of oral mucositis in pediatric oncology patients. J Pediatr Oncol Nurs. 32(1):47–56
Cheng KK, Chang AM, Yuen MP (2004) Prevention of oral mucositis in paediatric patients treated with chemotherapy; a randomised crossover trial comparing two protocols of oral care. Eur J Cancer. 40(8):1208–1216
Costa EM, Fernandes MZ, Quinder LB, de Souza LB, Pinto LP (2003) Evaluation of an oral preventive protocol in children with acute lymphoblastic leukemia. Pesqui Odontol Bras. 17(2):147–150
Raether D, Walker PO, Bostrum B, Weisdorf D (1989) Effectiveness of oral chlorhexidine for reducing stomatitis in a pediatric bone marrow transplant population. Pediatr Dent. 11(1):37–42
Cheng KK (2004) Children's acceptance and tolerance of chlorhexidine and benzydamine oral rinses in the treatment of chemotherapy-induced oropharyngeal mucositis. Eur J Oncol Nurs. 8(4):341–349
Cheng KK, Chang AM (2003) Palliation of oral mucositis symptoms in pediatric patients treated with cancer chemotherapy. Cancer Nurs. 26(6):476–484
Lever SA, Dupuis LL, Chan HS (1987) Comparative evaluation of benzydamine oral rinse in children with antineoplastic-induced stomatitis. Drug Intell Clin Pharm. 21(4):359–361
Shenep JL, Kalwinsky DK, Hutson PR, George SL, Dodge RK, Blankenship KR, Thornton D (1988) Efficacy of oral sucralfate suspension in prevention and treatment of chemotherapy-induced mucositis. J Pediatr. 113(4):758–763
Bondi E, Baroni C, Prete A, Gatti M, Carrassi A, Lodi G, Porters SR (1997) Local antimicrobial therapy of oral mucositis in paediatric patients undergoing bone marrow transplantation. Oral Oncol. 33(5):322–326
Saunders DP, Epstein JB, Elad S, Allemano J, Bossi P, van de Wetering MD et al (2013) Systematic review of antimicrobials, mucosal coating agents, anesthetics, and analgesics for the management of oral mucositis in cancer patients. Support Care Cancer. 21(11):3191–3207
Collins JJ, Geake J, Grier HE, Houck CS, Thaler HT, Weinstein HJ, Twum-Danso NY, Berde CB (1996) Patient-controlled analgesia for mucositis pain in children: a three-period crossover study comparing morphine and hydromorphone. J Pediatr. 129(5):722–728
Mackie AM, Coda BC, Hill HF (1991) Adolescents use patient-controlled analgesia effectively for relief from prolonged oropharyngeal mucositis pain. Pain. 46(3):265–269
James PJ, Howard RF, Williams DG (2010) The addition of ketamine to a morphine nurse- or patient-controlled analgesia infusion (PCA/NCA) increases analgesic efficacy in children with mucositis pain. Paediatr Anaesth. 20(9):805–811
Lucchese A, Matarese G, Ghislanzoni LH, Gastaldi G, Manuelli M, Gherlone E (2016) Efficacy and effects of palifermin for the treatment of oral mucositis in patients affected by acute lymphoblastic leukemia. Leuk Lymphoma. 57(4):820–827
Lucchese A, Matarese G, Manuelli M, Ciuffreda C, Bassani L, Isola G, Cordasco G, Gherlone E (2016) Reliability and efficacy of palifermin in prevention and management of oral mucositis in patients with acute lymphoblastic leukemia: a randomized, double-blind controlled clinical trial. Minerva Stomatol. 65(1):43–50
Morris J, Rudebeck M, Neudorf S, Moore T, Duerst R, Shah AJ, Graham M, Aquino V, Morris C, Olsson B (2016) Safety, pharmacokinetics, and efficacy of palifermin in children and adolescents with acute leukemias undergoing myeloablative therapy and allogeneic hematopoietic stem cell transplantation: a pediatric blood and marrow transplant consortium trial. Biol Blood Marrow Transplant. 22(7):1247–1256
Czyzewski K, Debski R, Krenska A, Wysocki M, Styczynski J (2014) Palifermin in children undergoing autologous stem cell transplantation: a matched-pair analysis. Anticancer Res. 34(12):7379–7382
Lauritano D, Petruzzi M, Di Stasio D, Lucchese A (2014) Clinical effectiveness of palifermin in prevention and treatment of oral mucositis in children with acute lymphoblastic leukaemia: a case-control study. Int J Oral Sci. 6(1):27–30
Vitale KM, Violago L, Cofnas P, Bishop J, Jin Z, Bhatia M, Kung AL, George D, Garvin J, Satwani P (2014) Impact of palifermin on incidence of oral mucositis and healthcare utilization in children undergoing autologous hematopoietic stem cell transplantation for malignant diseases. Pediatr Transplant. 18(2):211–216
Patte C, Laplanche A, Bertozzi AI, Baruchel A, Frappaz D, Schmitt C, Mechinaud F, Nelken B, Boutard P, Michon J (2002) Granulocyte colony-stimulating factor in induction treatment of children with non-Hodgkin's lymphoma: a randomized study of the French Society of Pediatric Oncology. J Clin Oncol. 20(2):441–448
Gordon B, Spadinger A, Hodges E, Ruby E, Stanley R, Coccia P (1994) Effect of granulocyte-macrophage colony-stimulating factor on oral mucositis after hematopoietic stem-cell transplantation. J Clin Oncol. 12(9):1917–1922
de Koning BA, Philipsen-Geerling B, Hoijer M, Hahlen K, Buller HA, Pieters R (2007) Protection against chemotherapy induced mucositis by TGF-beta(2) in childhood cancer patients: results from a randomized cross-over study. Pediatr Blood Cancer. 48(5):532–539
Raphael MF, den Boer AM, Kollen WJ, Mekelenkamp H, Abbink FC, Kaspers GJ et al (2014) Caphosol, a therapeutic option in case of cancer therapy-induced oral mucositis in children? : Results from a prospective multicenter double blind randomized controlled trial. Support Care Cancer. 22(1):3–6
Uderzo C, Rebora P, Marrocco E, Varotto S, Cichello F, Bonetti M, Maximova N, Zanon D, Fagioli F, Nesi F, Masetti R, Rovelli A, Rondelli R, Valsecchi MG, Cesaro S (2011) Glutamine-enriched nutrition does not reduce mucosal morbidity or complications after stem-cell transplantation for childhood malignancies: a prospective randomized study. Transplantation. 91(12):1321–1325
Kuskonmaz B, Yalcin S, Kucukbayrak O, Cetin N, Cetin M, Tezcan I, Uckan D (2008) The effect of glutamine supplementation on hematopoietic stem cell transplant outcome in children: a case-control study. Pediatr Transplant. 12(1):47–51
Aquino VM, Harvey AR, Garvin JH, Godder KT, Nieder ML, Adams RH et al (2005) A double-blind randomized placebo-controlled study of oral glutamine in the prevention of mucositis in children undergoing hematopoietic stem cell transplantation: a pediatric blood and marrow transplant consortium study. Bone Marrow Transplant. 36(7):611–616
Ward E, Smith M, Henderson M, Reid U, Lewis I, Kinsey S, Allgar V, Bowers D, Picton SV (2009) The effect of high-dose enteral glutamine on the incidence and severity of mucositis in paediatric oncology patients. Eur J Clin Nutr. 63(1):134–140
Yildirim ZK, Bidev D, Buyukavci M (2013) Parenteral glutamine supplementation has no effect on chemotherapy-induced toxicity in children with non-Hodgkin lymphoma. J Pediatr Hematol Oncol. 35(5):371–376
Sung L, Tomlinson GA, Greenberg ML, Koren G, Judd P, Ota S, Feldman BM (2007) Serial controlled N-of-1 trials of topical vitamin E as prophylaxis for chemotherapy-induced oral mucositis in paediatric patients. Eur J Cancer. 43(8):1269–1275
Khurana H, Pandey RK, Saksena AK, Kumar A (2013) An evaluation of vitamin E and pycnogenol in children suffering from oral mucositis during cancer chemotherapy. Oral Dis. 19(5):456–464
Hamidieh AA, Sherafatmand M, Mansouri A, Hadjibabaie M, Ashouri A, Jahangard-Rafsanjani Z, Gholami K, Javadi MR, Ghavamzadeh A, Radfar M (2016) Calcitriol for oral mucositis prevention in patients with Fanconi anemia undergoing hematopoietic SCT: a double-blind, randomized, placebo-controlled trial. Am J Ther. 23(6):e1700–e17e8
Abdulrhman M, Elbarbary NS, Ahmed Amin D, Saeid ER (2012) Honey and a mixture of honey, beeswax, and olive oil-propolis extract in treatment of chemotherapy-induced oral mucositis: a randomized controlled pilot study. Pediatr Hematol Oncol. 29(3):285–292
Tomaževič T, Jazbec J (2013) A double blind randomised placebo controlled study of propolis (bee glue) effectiveness in the treatment of severe oral mucositis in chemotherapy treated children. Complement Ther Med. 21(4):306–312
Oberbaum M, Yaniv I, Ben-Gal Y, Stein J, Ben-Zvi N, Freedman LS, Branski D (2001) A randomized, controlled clinical trial of the homeopathic medication TRAUMEEL S in the treatment of chemotherapy-induced stomatitis in children undergoing stem cell transplantation. Cancer. 92(3):684–690
Sencer SF, Zhou T, Freedman LS, Ives JA, Chen Z, Wall D, Nieder ML, Grupp SA, Yu LC, Sahdev I, Jonas WB, Wallace JD, Oberbaum M (2012) Traumeel S in preventing and treating mucositis in young patients undergoing SCT: a report of the Children's Oncology Group. Bone Marrow Transplant. 47(11):1409–1414
Eghbali A, Taherkhanchi B, Bagheri B, Sadeghi SB (2016) Effect of chewing gum on oral mucositis in children undergoing chemotherapy: a randomized controlled study. Iran J Ped Hematol Oncol. 6(1):9–14
Gandemer V, Le Deley MC, Dollfus C, Auvrignon A, Bonnaure-Mallet M, Duval M et al (2007) Multicenter randomized trial of chewing gum for preventing oral mucositis in children receiving chemotherapy. J Pediatr Hematol Oncol. 29(2):86–94
Sato A, Saisho-Hattori T, Koizumi Y, Minegishi M, Iinuma K, Imaizumi M (2006) Prophylaxis of mucosal toxicity by oral propantheline and cryotherapy in children with malignancies undergoing myeloablative chemo-radiotherapy. Tohoku J Exp Med. 210(4):315–320
Amadori F, Bardellini E, Conti G, Pedrini N, Schumacher RF, Majorana A (2016) Low-level laser therapy for treatment of chemotherapy-induced oral mucositis in childhood: a randomized double-blind controlled study. Lasers Med Sci. 31(6):1231–1236
Chermetz M, Gobbo M, Ronfani L, Ottaviani G, Zanazzo GA, Verzegnassi F, Treister NS, di Lenarda R, Biasotto M, Zacchigna S (2014) Class IV laser therapy as treatment for chemotherapy-induced oral mucositis in onco-haematological paediatric patients: a prospective study. Int J Paediatr Dent. 24(6):441–449
Whelan HT, Connelly JF, Hodgson BD, Barbeau L, Post AC, Bullard G, Buchmann EV, Kane M, Whelan NT, Warwick A, Margolis D (2002) NASA light-emitting diodes for the prevention of oral mucositis in pediatric bone marrow transplant patients. J Clin Laser Med Surg. 20(6):319–324
Pytlík R, Benes P, Patorková M, Chocenská E, Gregora E, Procházka B, Kozák T (2002) Standardized parenteral alanyl-glutamine dipeptide supplementation is not beneficial in autologous transplant patients: a randomized, double-blind, placebo controlled study. Bone Marrow Transplant. 30(12):953–961
Tomlinson D, Gibson F, Treister N, Baggott C, Judd P, Hendershot E, Maloney AM, Doyle J, Feldman B, Kwong K, Sung L (2009) Understandability, content validity, and overall acceptability of the Children's International Mucositis Evaluation Scale (ChIMES): child and parent reporting. J Pediatr Hematol Oncol. 31(6):416–423
Noirrit-Esclassan E, Valera MC, Vignes E, Munzer C, Bonal S, Daries M, Vaysse F, Puiseux C, Castex MP, Boulanger C, Pasquet M (2019) Photobiomodulation with a combination of two wavelengths in the treatment of oral mucositis in children: The PEDIALASE feasibility study. Arch Pediatr. 26(5):268–274
Treister NS, London WB, Guo D, Malsch M, Verrill K, Brewer J, Margossian S, Duncan C (2016) A feasibility study evaluating extraoral photobiomodulation therapy for prevention of mucositis in pediatric hematopoietic cell transplantation. Photomed Laser Surg. 34(4):178–184
Ljungman L, Cernvall M, Ghaderi A, Ljungman G, von Essen L, Ljótsson B (2018) An open trial of individualized face-to-face cognitive behavior therapy for psychological distress in parents of children after end of treatment for childhood cancer including a cognitive behavioral conceptualization. PeerJ. 6:e4570
Barros IM, Alcântara TS, Mesquita AR, Santos AC, Paixão FP, Lyra DP Jr (2014) The use of pictograms in the health care: a literature review. Res Social Adm Pharm. 10(5):704–719
Li WH, Chung JO, Ho EK (2011) The effectiveness of therapeutic play, using virtual reality computer games, in promoting the psychological well-being of children hospitalised with cancer. J Clin Nurs. 20(15-16):2135–2143
Lopes-Junior LC, Silveira DSC, Olson K, Bomfim EO, Veronez LC, Santos JC et al (2019) Clown intervention on psychological stress and fatigue in pediatric patients with cancer undergoing chemotherapy. Cancer Nurs
Silva NB, Osório FL (2018) Impact of an animal-assisted therapy programme on physiological and psychosocial variables of paediatric oncology patients. PLoS One. 13(4):e0194731
Lopez-Rodriguez MM, Fernández-Millan A, Ruiz-Fernández MD, Dobarrio-Sanz I, Fernández-Medina IM (2020) New technologies to improve pain, anxiety and depression in children and adolescents with cancer: a systematic review. Int J Environ Res Public Health 17(10)
Konuk Sener D, Aydin M, Cangur S, Guven E (2019) The effect of oral care with Chlorhexidine, vitamin E and honey on mucositis in pediatric intensive care patients: a randomized controlled trial. J Pediatr Nurs. 45:e95–e101
Alkhouli M, Laflouf M, Alhaddad M (2020) Efficacy of aloe-vera use for prevention of chemotherapy-induced oral mucositis in children with acute lymphoblastic leukemia: a randomized controlled clinical trial. Compr Child Adolesc Nurs.:1–14
Al Jaouni SK, Al Muhayawi MS, Hussein A, Elfiki I, Al-Raddadi R, Al Muhayawi SM et al (2017) Effects of Honey on oral mucositis among pediatric cancer patients undergoing chemo/radiotherapy treatment at King Abdulaziz University Hospital in Jeddah, Kingdom of Saudi Arabia. Evid Based Complement Alternat Med 2017:5861024
Eslami H, Pouralibaba F, Falsafi P, Bohluli S, Najati B, Negahdari R, Ghanizadeh M (2016) Efficacy of Hypozalix spray and propolis mouthwash for prevention of chemotherapy-induced oral mucositis in leukemic patients: A double-blind randomized clinical trial. J Dent Res Dent Clin Dent Prospects. 10(4):226–233
Tavakoli Ardakani M, Ghassemi S, Mehdizadeh M, Mojab F, Salamzadeh J, Ghassemi S, Hajifathali A (2016) Evaluating the effect of Matricaria recutita and Mentha piperita herbal mouthwash on management of oral mucositis in patients undergoing hematopoietic stem cell transplantation: A randomized, double blind, placebo controlled clinical trial. Complement Ther Med. 29:29–34
Treister N, Nieder M, Baggott C, Olson E, Chen L, Dang H, Krailo M, August A, Sung L (2017) Caphosol for prevention of oral mucositis in pediatric myeloablative haematopoietic cell transplantation. Br J Cancer. 116(1):21–27
Bardellini E, Amadori F, Schumacher RF, D'Ippolito C, Porta F, Majorana A (2016) Efficacy of a solution composed by verbascoside, polyvinylpyrrolidone (PVP) and sodium hyaluronate in the treatment of chemotherapy-induced oral mucositis in children with acute lymphoblastic leukemia. J Pediatr Hematol Oncol. 38(7):559–562
Gobbo M, Verzegnassi F, Ronfani L, Zanon D, Melchionda F, Bagattoni S, Majorana A, Bardellini E, Mura R, Piras A, Petris MG, Mariuzzi ML, Barone A, Merigo E, Decembrino N, Vitale MC, Berger M, Defabianis P, Biasotto M, Ottaviani G, Zanazzo GA (2018) Multicenter randomized, double-blind controlled trial to evaluate the efficacy of laser therapy for the treatment of severe oral mucositis induced by chemotherapy in children: laMPO RCT. Pediatr Blood Cancer. 65(8):e27098
Medeiros-Filho JB, Maia Filho EM, Ferreira MC (2017) Laser and photochemotherapy for the treatment of oral mucositis in young patients: Randomized clinical trial. Photodiagnosis Photodyn Ther. 18:39–45
Ribeiro da Silva VC, da Motta Silveira FM, Barbosa Monteiro MG, da Cruz MMD, Caldas Junior AF, Pina Godoy G. Photodynamic therapy for treatment of oral mucositis: Pilot study with pediatric patients undergoing chemotherapy. Photodiagnosis Photodyn Ther. 2018;21:115-20.
Vitale MC, Modaffari C, Decembrino N, Zhou FX, Zecca M, Defabianis P (2017) Preliminary study in a new protocol for the treatment of oral mucositis in pediatric patients undergoing hematopoietic stem cell transplantation (HSCT) and chemotherapy (CT). Lasers Med Sci. 32(6):1423–1428
Kamsvåg T, Svanberg A, Legert KG, Arvidson J, von Essen L, Mellgren K et al (2020) Prevention of oral mucositis with cryotherapy in children undergoing hematopoietic stem cell transplantations-a feasibility study and randomized controlled trial. Support Care Cancer
Tsurusawa M, Watanabe T, Gosho M, Mori T, Mitsui T, Sunami S, Kobayashi R, Fukano R, Tanaka F, Fujita N, Inada H, Sekimizu M, Koh K, Kosaka Y, Komada Y, Saito AM, Nakazawa A, Horibe K, for the lymphoma committee of the Japanese Pediatric Leukemia/lymphoma Study Group (2016) Randomized study of granulocyte colony stimulating factor for childhood B-cell non-Hodgkin lymphoma: a report from the Japanese pediatric leukemia/lymphoma study group B-NHL03 study. Leuk Lymphoma 57(7):1657–1664
Gholizadeh N, Mehdipoor M, Sajadi H, Moosavi MS (2016) Palifermin and chlorhexidine mouthwashes in prevention of chemotherapy-induced mucositis in children with acute lymphocytic leukemia: a randomized controlled trial. J Dent (Shiraz). 17(4):343–347
Elad S, Cheng KKF, Lalla RV, Yarom N, Hong C, Logan RM, Bowen J, Gibson R, Saunders DP, Zadik Y, Ariyawardana A, Correa ME, Ranna V, Bossi P; Mucositis Guidelines Leadership Group of the Multinational Association of Supportive Care in Cancer and International Society of Oral Oncology (MASCC/ISOO) (2020). MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer. https://doi.org/10.1002/cncr.33100
Acknowledgments
The authors would like to thank the members of the Mucositis Study Group of MASC/ISOO who contributed to the guidelines development. The authors thank Rachel Lubart, PhD, and Lilach Gavish, PhD, for advising on laser physics.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
PRA has received grants and personal fees from PhotomedTech, personal fees from Kerber Applied Research, personal fees from Seaborough, personal fees from Shepherd University, non-financial support from National Institute of Aging, NIH, personal fees from Curalaser, personal fees and non-financial support from Vielight, personal fees from Zoovv, non-financial support from Thor Photomedicine, non-financial support from Weber Medical, non-financial support from K laser (Summa), non-financial support from Biolase, non-financial support from Irradia, non-financial support from Quasar Biotech, and non-financial support from Light scalpel, outside the submitted work; In addition, PRA has a patent light-based dental systems issued.
PB has served an advisory role for Merck Serono, Sanofi, Merck Sharp & Dohme, Sun Pharma, Angelini, Astra Zeneca, Bristol-Myers-Squibb, Helsinn, and GSK and received grants from Merck, Kyowa Hakko Kirin, and Roche. WMS, WGS, YZ, NY, ARAA, CHLH, AA, DPS, MEC, JB, KKFC, WJET, and SE declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Miranda-Silva, W., Gomes-Silva, W., Zadik, Y. et al. MASCC/ISOO clinical practice guidelines for the management of mucositis: sub-analysis of current interventions for the management of oral mucositis in pediatric cancer patients. Support Care Cancer 29, 3539–3562 (2021). https://doi.org/10.1007/s00520-020-05803-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-020-05803-4