Abstract
Purpose
To explore the best schedule of oral cryotherapy for the prevention of oral mucositis in recipients of myeloablative hematopoietic stem cell transplantation (HSCT).
Methods
A prospective randomized study was conducted to recruit allogeneic HSCT recipients, who were then randomly allocated into four arms to accept the following: oral cryotherapy during the whole course (arm A) or second half of the course (arm B) of cytotoxic agents administration, regular oral cryotherapy twice a day (arm C), or conventional oral care without cryotherapy (arm D). Status of oral mucositis was daily assessed from the first day of conditioning to the 15th day post-HSCT. A myeloablative conditioning regimen was used which was composed of busulfan, cyclophosphamide, and cytarabine.
Results
Totally 160 cases were consecutively enrolled in this study, and 145 cases were eligible for oral mucositis assessment. Both arm A and arm B were associated with a lower incidence and short duration of severe mucositis (≥ grade 3), although no statistical difference was found between these two groups (p = 0.463, p = 0.678). The highest incidence of severe mucositis was observed in arm C. Recovery of mucositis also had a significant diversity among the 4 arms (F = 4.133, p = 0.008).
Conclusions
Risk and outcome of severe oral mucositis could be ameliorated by oral cryotherapy during the administration of cytotoxic agents for allogeneic HSCT patients receiving non-radiation myeloablative conditioning regimen, and a half-course schedule could acquire a comparable efficacy compared with the whole-course schedule.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Oral mucositis (OM) is defined as an ulcerative and inflammatory disease of the oral mucosa [1, 2]. OM is the most common complication in patients treated with high-dose chemotherapy and/or total body irradiation before hematopoietic stem cell transplantation (HSCT) [3]. Compared with other types of myeloablative conditioning, patients who receive busulfan-cyclophosphamide (BUCY) regimen exhibit a higher incidence of OM [4]. OM is also closely associated with morbidity, treatment compliance, and treatment results, and negatively affects the feeding and quality of life of the patients [3, 5,6,7,8].
Cryotherapy, through placing ice cubes in the mouth during chemotherapy administration, causes blood vessel constriction, thereby reducing the penetration of chemotherapy agents into the mucous membrane of the mouth. Cryotherapy has been shown to effectively reduce the incidence and severity of oral mucositis in patients receiving high-dose chemotherapy followed by stem cell transplantation [3, 9,10,11,12,13]. In a recent Cochrane review, oral cryotherapy was found to reduce severe OM in adults receiving high-dose melphalan-based chemotherapy before hematopoietic stem cell transplantation [14]. However, some patients complained that a long period of cryotherapy was uncomfortable [10]. It has been reported that cryotherapy during the distribution phase is as effective as cryotherapy of longer duration [11, 15]. Oral cryotherapy is characterized by high safety, few side effects, easy availability, and low cost, which makes its wide application in clinical settings worthwhile [16]. However, it is not clear how this simple method should be applied in HSCT patients receiving different myeloablative conditioning regimes and what timing or duration is optimal [1]. Furthermore, whether cryotherapy could be a part of daily routine nursing practice has received little attention [17]. The aim of the present study was to evaluate the effect of cryotherapy in preventing OM following HSCT with BUCY as a myeloablative conditioning regime and to try to identify the optimal timing and duration.
Patients and methods
Patients and protocol design
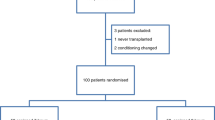
This study was conducted in the Bone Marrow Transplantation Unit of the First Affiliated Hospital of Soochow University. Patients with an age ≥ 18 years with confirmed literacy, who were undergoing BUCY regimens as myeloablative conditioning before transplantation, were recruited and randomly assigned to four arms. Patients with clinical evidence of mucositis at enrollment were excluded. Details are shown in Fig. 1.
Prior to HSCT, all patients underwent an oral examination and during their hospital stay; the patients were instructed to wash the oral cavity with chlorhexidine mouthwash half an hour before and after eating or half an hour before sleeping, for 3 min each time. Dentists were consulted if any oral problems arose, and the use of toothpicks or dental floss was prohibited. Since mucositis symptoms develop between 5 and 14 days following chemotherapy and no subsequent alleviation has been observed in our unit; the stages and grades of oral mucositis were evaluated by a nurse every day from the first day of myeloablative conditioning until the 15th day after HSCT.
Cryotherapy administration
Ice cubes whose corners had been rounded to avoid irritation, with a size (3.2 cm × 3.3 cm × 1 cm) suitable for being easily moved in the mouth, were used. While receiving cryotherapy, the patients were informed of the importance of keeping the oral cavity constantly cool. As the ice cubes melted, patients were advised to spit out the cold liquid and take another ice cube. During cryotherapy, the patients were asked to take as many ice cubes as possible but only one piece at a time, and patients could take a 60–180s break if they felt discomfort.
Transplant procedures
Before transplantation, all the patients received a BUCY regimen (including cytarabine, busulfan, and cyclophosphamide) for myeloablative conditioning for 7 days. Cytarabine (8 g/msq) was used on day 1 to day 2 and was infused for 3 h/day; busulfan (9.6 mg/kg) was used from day 3 to day 5 and was infused for 8 h/day (divided into four infusion periods a day of 2 h each); and cyclophosphamide (3.6 g/msq) was used from day 6 to day 7 and was infused for 3 h/day. Infusion speeds were controlled by infusing pumps (B.Braun Melsungen, Germany).
Evaluations
General data collection form
The data collection form included the following general information about the patient that might influence the development of oral mucositis after conditioning regimen: age, sex, residence, diagnosis, graft type, systemic diseases, oral hygiene habits, oral disease history, dietary habits (fondness of taste), OM history, the frequency of chemotherapy treatment.
Nurse-judged mucositis grading
The nurses utilized the National Cancer Institute Common Toxicity Criteria (NCI-CTC version 2) form to assess oral mucositis [18] according to the following scale: grade 0, none; grade 1, painless ulcers, erythema, and/or mild soreness in the absence of lesions; grade 2, painful erythema, edema, or ulcers, but able to swallow; grade 3, painful erythema, edema, or ulcers preventing swallowing or requiring hydration or parenteral (or enteral) nutritional support; and grade 4, severe ulceration requiring prophylactic intubation or resulting in documented aspiration pneumonia.
Patient-judged mucositis questionnaire
This questionnaire was developed from a Chinese version of Oral Mucositis Daily Questionnaire (OMDQ), which has 0.959 criterion-related validity compared with the WHO Oral Mucositis Assessment Scale [19]. The form was amended and pretested by the clinical specialists of our center. It contains the grades of symptoms of oral mucositis (such as pain, swallow sense, taste sense), information on the use of ice cubes, and routine oral care.
Statistical analysis
The data were recorded and analyzed using the Statistical Program for Social Sciences version 19.0 for Windows (SPSS 19.0). For purposes of analysis, a maximal score of oral mucositis was attributed to each patient. Incidence of oral mucositis was compared among four groups by χ2 test. The chi-square statistic or Fisher’s exact test was applied for categorical variables, and variance analysis and the Kruskal-Wallis test were applied for continuous variables. Multivariate analyses were conducted by logistic regression and linear regression. All p values are two-sided with the type I error rate fixed at 0.05.
Results
Patient characteristics
There were 160 transplant patients enrolled in this study from January 2017 to April 2018. Among these individuals, fifteen patients were excluded due to protocol deviation, including the two patients in arm A who could not endure a long duration of cryotherapy and suffered severe diarrhea; four patients in arm B who had not get cryotherapy accordingly; three patients in arm C and five patients in the arm D because of disease progression; and one patient in arm C quitted because of severe vomiting during myeloablative conditioning. Thus, 145 patients were finally evaluated. Among these individuals, 110 patients were in the experimental groups (38 patients in arm A, 36 patients in arm B and 36 patients in arm C), and 35 patients were in the control group (arm D). Demographic and transplant-related factors were compared among the patients of four arms, and no statistical significance was found (details are shown in Table 1).
Incidence of oral mucositis
According to the nurse-judged mucositis grading, 67.58% of the patients exhibited grade 2–3 mucositis, and no grade 4 mucositis was observed in any group. The mucositis rate was similar between arm A and arm B and between arm C and arm D (Fig. 2). The patients in both arm A and arm B patients presented a lower incidence of mild mucositis compared with those in the arm D over the observation period, while the patients in arm C exhibited more severe mucositis than the control group after chemotherapy (Fig. 2).
The incidence rates of severe oral mucositis (grades 3 and 4) during the observation period were 13.2% in arm A, 19.4% in arm B, 41.7% in arm C, and 39.4% in arm D. There was no significant difference between arm A and arm B (p = 0.463). However, compared with arm D, arm A and arm B showed a significant difference (p = 0.011, 0.068, respectively); arm C showed no significant difference (p = 0.848). In addition, there was a significant difference between arm A and arm C as well as between arm B and arm C (p = 0.006 and 0.041, respectively), as shown in Table 2.
Duration and recovery time of severe oral mucositis
The duration of mucositis presented no significant difference in patients with grade 1–2 mucositis (Table 2). The median duration of mucositis of grades 3–4 was 2 days in both arm A (range 1–4) and arm B (range 1–6), which was much shorter than the duration in arm D (7, range 3–20). A significant difference was also found between arm A and arm D and between arm B and arm D (p = 0.003, p = 0.01). The median duration of mucositis of grades 3–4 was 7 days in arm C, although no significant difference was found compared with arm D (p = 0.072). However, when arm C was compared with arm A and arm B, significant differences were found (p = 0.001, 0.004).
In regard to the time of recovery, the patients in arm A showed an average time of 6. 39± 3.41 days; patients in arm B showed an average time of 6.76 ± 3.17 days; patients in arm C showed an average time of 8.97 ± 4.42 days; and the patients in arm D showed an average time of 9.51 ± 6.96 days. Comparison of the four groups revealed a significant difference (F = 4.133, p = 0.006), as shown in Table 2.
Manifestation of oral mucositis
According to patient-judge mucositis questionnaire, 63.45% patients had taste change; 18.62% patients had a limitation of mouth opening; 49.66% patients had a limitation of swallowing; 33.79% patients had a limitation of speaking, and 60% patients had a limitation of sleeping. Compared with four groups, the incidence of taste change had a significant difference (p = 0.015), as shown in Table 3.
Multivariate analysis
In multivariate logistic regression analysis, arm A was a dependent affect factor with the incidence of severe oral mucositis (OR = 0.022, 0.080–0.821), and there was no statistical significance between arm A and arm B (p = 0.530).
The correlation between the time of recovery and certain variables was analyzed through multiple linear regression (Table 4). The analysis showed that the time of recovery was significantly associated with patient’s education level (p = 0.048), oral mucositis history (p = 0.041), and dietary habits (fondness for hot food) (p = 0.032). No significant correlation was found between the time of recovery and other variables, including sex, age, diagnosis, graft types, the frequency of chemotherapy treatment, and underlying disease.
Discussion
This study explored the role of cryotherapy in the prevention of oral mucositis in patients receiving a BUCY regimen (containing cytarabine, busulfan, and cyclophosphamide) as their myeloablative conditioning regimen. All these chemotherapeutic agents have a short plasma half-life, produce active metabolites, and have negative effects on the oral mucosa [4, 20]. We found that cryotherapy with a certain timing and duration (arm A and arm B) could significantly decrease the incidence of severe oral mucositis and reduce the recovery time of oral mucositis. However, it is not desirable to use cryotherapy as daily oral care (arm C).
The mechanisms by which oral cryotherapy alleviates oral mucositis are thought to be related to local vasoconstriction, causing decreased blood flow to the oral mucosa, thus reducing the distribution of the drugs among the cells located in the mucous membranes and decreasing the risk of oral mucositis development [2, 21]. Although cryotherapy is generally regarded as a safe and well-tolerated method, it can sometimes be painful and constitute a negative stimulus to mucosa if excessively applied [2, 22]. Pharmacologic studies have shown that the concentration of drugs delivered to a patient is higher during the distribution phase than during the elimination phase and therefore plays a more crucial role in causing oral mucositis [13, 18]. We hypothesized that shorter administration of cryotherapy, only during the drug distribution phase, might be effective and easily tolerated. However, in clinical practice, nurses could not always apply cryotherapy at the correct time, and they complained that these interventions require much time in routine care. Therefore, the present study was designed to assess the efficacy of cryotherapy in stem cell transplantation following the administration of a highly toxic myeloablative regimen and to compare the effect of a short or long period of application of cryotherapy as well as whether cryotherapy could be included as a part of daily oral care. Our study showed a significant reduction in the incidence and/or severity of oral mucositis by nurse assessment, according to a lower incidence of score 3–4 oral mucositis as well as a shorter duration of grade 3–4 mucositis noted in arm B. We also found that cryotherapy could not be used casually, as it could increase the incidence or duration of OM, as noted in arm C.
Oral mucositis is an important, painful complication of myeloablative conditioning that requires intervention. Mucositis related to direct stomatoxicity usually develops within 3 to 7 days after chemotherapy and improves within approximately 2 to 3 weeks, in parallel with neutrophil recovery [20, 23]. Previous studies showed that the severity and duration of oral mucositis in patients undergoing myeloablative conditioning with high-dose melphalan regimes exceeded those associated with busulfan, and cryotherapy could prevent severe oral mucositis under melphalan treatment [4, 22, 24]. Our study showed that the median recovery duration for oral mucositis under BUCY myeloablative conditioning was less than 10 days. A shortened duration of cryotherapy could significantly reduce the duration of recovery from oral mucositis (the median recovery time for oral mucositis in arm B patients was 2 days).
OM has been complained by patients as the most debilitating complication of HSCT, while they reported it could affect their oral functions [25]. In our study, we found 63.45% patients had taste change, which was the most common manifestation patients reported. Cryotherapy might be a good intervention to alleviate taste change [26]. As we found, patients received certain timing and duration cryotherapy (arm A and arm B) reported less incidence of taste change than patients with no cryotherapy (arm D).
The factors associated with the duration of recovery from oral mucositis were of less concern. For nurses, it is important to have knowledge of effective intervention measures and provide patients with good advice when they suffer from OM. In our research, we found that patients with a lower educational level exhibited a longer recovery time from OM (OR = − .728, − .082~1.538). The reason for this finding is probably that patients with poorer education often do not understand the importance of oral hygiene well or pay little attention to oral hygiene while undergoing hematopoietic stem cell transplantation. We also found that patients with a history of OM usually performed oral care carefully and maintained better oral hygiene conditions when they suffered OM during HSCT. Chinese people are fond of hot food due to the belief that consuming hot food is a healthful dietary habit and keeps their body healthy. However, when patients have oral mucositis, hot food stimulates the local mucosa and worsens their lesions. Accordingly, we found that patients who prefer hot food exhibited a longer recovery time from oral mucositis (OR = 1.368, − 2.964~0.227).
Conclusion
In conclusion, we found that cryotherapy during the administration of cytotoxic agents for allogeneic HSCT patients receiving non-radiation myeloablative conditioning regimen could decrease the incidence and duration of severe oral mucositis and a half-course schedule could acquire a comparable efficacy compared with the whole-course schedule. However, cryotherapy should not be applied as a part of daily routine care because this could damage the oral mucosa. During mucositis, patients with poor education or who are fond of hot food may suffer a long duration of recovery. Nurses should convey more concern and advise these patients to maintain their oral hygiene during the period of HSCT.
References
Harris DJ, Eilers J, Harriman A, Cashavelly BJ, Maxwell C (2008) Putting evidence into practice: evidence-based interventions for the management of oral mucositis. Clin J Oncol Nurs 12(1):141–152. https://doi.org/10.1188/08.CJON.141-152
Karagözoglu S, Filiz Ulusoy M (2005) Chemotherapy: the effect of oral cryotherapy on the development of mucositis. J Clin Nurs 14(6):754–765
Svanberg A, Ohrrn K, Birgegard G (2010) Oral cryotherapy reduces mucositis and improves nutrition-a randomised controlled trial. J Clin Nurs 19(15–16):2146–2151. https://doi.org/10.1111/j.1365-2702.2010.03255.x
Wardly AM, Jayson GC, Swindell R, Morgenstern GR, Chang J, Bloor R et al (2000) Prospective evaluation of oral mucositis in patients receiving myeloablative conditioning regimes and haemopoietic progenitor rescue. Br J Haematol 110(2):292–299
Svanberg A, Öhrn K, Birgegård G (2015) Caphosol(®) mouthwash gives no additional protection against oral mucositis compared to cryotherapy alone in stem cell transplantation. A pilot study. Eur J Oncol Nurs 19(1):50–53. https://doi.org/10.1016/j.ejon.2014.07.011
Demarosi F, Bez C, Carrassi A (2002) Prevention and treatment of chemo-and radiotherapy-induced oral mucositis. Minerva Stomatol 51(5):173–186
Gori E, Arpinati M, Bonifazi F, Errico A, Mega A, Alberani F et al (2007) Cryotherapy in the prevention of oral mucositis in patients receiving low-dose methotrexate following myeloablative allogeneic stem cell transplantation: a prospective randomized study of the Gruppo Italiano Trapianto di Midollo Osseo nurses group. Bone Marrow Transplant 39(6):347–352
Katranci N, Ovayolu N, Ovayolu O, Sevinc A (2012) Evaluation of the effect of cryotherapy in preventing oral mucositis associated with chemotherapy - a randomized controlled trial. Eur J Oncol Nurs 16(4):339–344. https://doi.org/10.1016/j.ejon.2011.07.008
Tartarone A, Matera R, Romano G, Vigliotti ML, Di Renzo N (2005) Prevention of high-dose melphalan-induced mucositis by cryotherapy. Leuk Lymphoma 46(4):633–634
Lilleby K, Garcia P, Gooley T, McDonnell P, Taber R, Holmberg L et al (2006) A prospective, randomized study of cryotherapy during administration of highdose melphalan to decrease the severity and duration of oral mucositis in patients with multiple myeloma undergoing autologous peripheral blood stem cell transplantation. Bone Marrow Transplant 37(11):1031–1035
Mori T, Aisa Y, Yamazaki R, Mihara A, Ikeda Y, Okamoto S (2006) Cryotherapy for the prevention of high-dose melphalan-induced oral mucositis. Bone Marrow Transplant 38(9):637–638
Svanberg A, Birgegård G, Öhrn K (2007) Oral cryotherapy reduces mucositis and opioid use after myeloablative therapy – a randomized controlled trial. Support Care Cancer 15(10):1155–1161
Riley P, Glenny AM, Worthington HV, Littlewood A, Clarkson JE, McCabe MG (2015) Interventions for preventing oral mucositis in patients with cancer receiving treatment: Oral cryotherapy. Cochrane Database Syst Rev 23(12):CD011552. https://doi.org/cncr.28592/14651858.CD011552.pub2 Review
Wang L, Gu Z, Zhai R, Zhao S, Luo L, Li D et al (2015) Efficacy of oral cryotherapy on oral mucositis prevention in patients with hematological malignancies undergoing hematopoietic stem cell transplantation: a meta-analysis of randomized controlled trials. PLoS One 10(5):e0128763. https://doi.org/10.1371/journal.pone.0128763 eCollection 2015
Keefe DM, Schubert MM, Elting LS, Sonis ST, Epstein JB, Raber-Durlacher JE et al (2007) Updated clinical practice guidelines for the prevention and treatment of mucositis. Cancer 109(5):820–831
Peterson DE, Ohrn K, Bowen J, Fliedner M, Lees J, Loprinzi C et al (2013) Systematic review of oral cryotherapy for management of oral mucositis caused by cancer therapy. Support Care Cancer 21(1):327–332. https://doi.org/10.1007/s00520-012-1562-0
Sharifi H, Heydari A, Salek R, Emami, Zeydi A (2017) Oral cryotherapy for preventing chemotherapy-induced oral mucositis: an effective but yet neglected strategy. J Can Res Ther 13(2):386–387. https://doi.org/10.4103/0973-1482.188301
Mori T, Yamazaki R, Aisa Y, Nakazato T, Kudo M, Yashima T et al (2006) Brief oral cryotherapy for the prevention of high-dose melphalan-induced stomatitis in allogeneic hematopoietic stem cell transplant recipients. Support Care Cancer 14(4):392–395
Gu YH, Gong LL, Hu Y (2014) Reliability and validity of Chinese version of Oral Mucositis Daily Questionnaire. Clin J Nurs 49(10):109–112
Wu ZZ, Zhou HJ, Xu SW (2000) General clinical pharmacology [M]. Science of China
Hejna M, Brodowicz T, Zielinski CC (1999) Local use of GM-CSF for severe mucositis. Eur J Cancer 35(Suppl.3):S14–S17
Aisa Y, Mori T, Kudo M, Yashima T, Kondo S, Yokoyama A et al (2005) Oral cryotherapy for the prevention of high-dose melphalan-induced stomatitis in allogeneic hematopoietic stem cell transplant recipients. Support Care Cancer 13(4):266–269
Wojtaszek C (2000) Management of chemotherapy-induced stomatitis. Clin J Oncol Nurs 4(6):263–270
Lalla RV, Bowen J, Barasch A, Elting L, Epstein J, Keefe DM et al (2014) MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer 120(10):1453–1461. https://doi.org/cncr.28592 Review. Erratum in: Cancer. 2015 Apr 15; 121(8):1339
Epstein JB, Phillips N, Parry J, Epstein MS, Nevill T et al (2002) Quality of life, taste, olfactory and oral function following high-dose chemotherapy and allogeneic hematopoietic cell transplantation. Bone Marrow Transplant 30(11):785–792
Okada N, Hanafusa T, Abe S, Sato C, Nakamura T et al (2016) Evaluation of the risk factors associated with high-dose chemotherapy-induced dysgeusia in patients undergoing autologous hematopoietic stem cell transplantation: possible usefulness of cryotherapy in dysgeusia prevention. Support Care Cancer 24(9):3979–3985. https://doi.org/10.1007/s00520-016-3244-9
Acknowledgments
We sincerely acknowledge Dr. Jing Yang for her assistance with the statistical consultant in this study. We also express our gratitude to the clinical staff in the Stem Cell Transplantation Unit of the First Affiliated Hospital of Suzhou University.
Funding
This study was supported by a grant from Suzhou People’s Livelihood Science and Technology project (SYSD2016083).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All participants provided informed consent, and the study was approved by the ethical institution of the First Affiliated Hospital of Soochow University (no. 2017021).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lu, Y., Zhu, X., Ma, Q. et al. Oral cryotherapy for oral mucositis management in patients receiving allogeneic hematopoietic stem cell transplantation: a prospective randomized study. Support Care Cancer 28, 1747–1754 (2020). https://doi.org/10.1007/s00520-019-04966-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-019-04966-z