Abstract
Purpose
Although physical activity has been demonstrated to increase cancer survival in epidemiological studies, breast cancer patients tend toward inactivity after treatment.
Methods
Breast cancer patients were quasi-randomly allocated to two different groups, intervention (IG) and control (CG) groups. The intervention group (n = 111) received an individual 3-week exercise program with two additional 1-week inpatient stays after 4 and 8 months. At the end of the rehabilitation, a home-based exercise program was designed. The control group (n = 83) received a 3-week rehabilitation program and did not obtain any follow-up care. Patients from both groups were measured using questionnaires on physical activity, fatigue, and quality of life (QoL) at five time points, 4 months (t1), 8 months (t2), 12 months (t3), 18 months (t4), and 24 months (t5) after the beginning of the rehabilitation.
Results
After 2 years, the level of physical activity (total metabolic rate) increased significantly from 2733.16 ± 2547.95 (t0) to 4169.71 ± 3492.27 (t5) metabolic equivalent (MET)-min/week in the intervention group, but just slightly changed from 2858.38 ± 2393.79 (t0) to 2875.74 ± 2590.15 (t5) MET-min/week in the control group (means ± standard deviation). Furthermore, the internal group comparison showed significant differences after 2 years as well. These results came along with a significantly reduced fatigue syndrome and an increased health-related quality of life.
Conclusions
The data indicate that an individual, according to their preferences, and physical-resource-adapted exercise program has a more sustainable impact on the physical activity level in breast cancer patients than the usual care. It is suggested that the rehabilitation program should be personalized for all breast cancer patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Purpose
Breast cancer is the most commonly diagnosed type of cancer in women both in the developed and less-developed world. It is estimated that in 2013, over 1.8 million women worldwide were diagnosed with breast cancer [1]. One in eight women is confronted with breast cancer diagnosis during her lifetime, which is in Germany alone 69.550 women each year [2]. Breast cancer survival rates in low- and middle-income countries vary between 40 and 60%. Due to early screening, detection, better awareness, and treatment options, which keep getting better constantly, the curing prospects in high-income countries are over 80% [1]. Despite continuously improving treatment options, the medical treatment for breast cancer has a lot of side effects on the physical [3, 4], mental [5, 6], and social levels [7], which have a negative influence on the quality of life [8, 9]. Over the past few years, a number of studies have shown that physical activity and exercise programs have significantly positive influence over those very common side effects such as lymphedema and fatigue [10, 11]. Several epidemiological studies suggest that physical activity can even significantly decrease the mortality risk in patients with breast cancer [12, 13]. However, studies indicate that a lot of patients lose their activity level after completing the rehabilitation phase and exercise programs and fall back to their base level [14, 15]. This of course involves the risk that diseases due to physical inactivity and medical treatment-related side effects may reappear. To change their physical activity and exercise habits, it is assumed that patients seem to prefer an individual and according to their preferences and physical resource-adapted exercise program, instructed by specialized staff [16]. Therefore, we initiated a quasi-randomized, intervention-controlled trial with breast cancer patients in the rehabilitation phase in two different rehabilitation clinics over 3 weeks. We hypothesized that an individual-supervised exercise program shows a more positive and more sustainable impact on physical activity level, fatigue syndrome, and health-related quality of life than usual care.
Methods
Study design and measure points
The Kissinger Individualization in Rehabilitation and Activity (KIRA) study is a quasi-randomized, controlled intervention trial that analyzes the sustainable impact of an individualized exercise program on physical activity level (primary endpoint) and fatigue syndrome on breast cancer patients in a rehabilitation center, and it took place between 2010 and 2011 with a follow-up examination in 2013. A randomization of patients into two different groups in one rehabilitation clinic was not possible, which is why we conducted a two-center study with an intervention group located in the “Klinik Am Kurpark” in Bad Kissingen (Germany) and a control group located in the “Klinik Ob der Tauber” in Bad Mergentheim (Germany). The patients from the first clinic received an individualized exercise program; the patients from the second clinic received usual care. The control group received a 3-week rehabilitation program according to the German rehabilitation guidelines and did not obtain any follow-up care. They were measured at the time points of 4 months (t1), 8 months (t2), 12 months (t3), 18 months (t4), and 24 months (t5) after the beginning of the rehabilitation using postal questionnaires on physical activity, fatigue syndrome, and health-related quality of life. An exact sample size calculation was not conducted, but we calculated the number of 200 recruited breast cancer patients considering the inpatient number in the hospital per year.
Inclusion and exclusion criteria
In order for the patients to participate in the study, they had to be between 18 and 75 years old and diagnosed with non-metastatic, histologically proven breast cancer not longer than 5 years. In addition, fluent German language skills were required for the complete comprehension of the questionnaires. Exclusion criteria were second malignancies, metastatic diseases, major medical or psychiatric comorbidities, or other chronic diseases excluding regular physical activity, further alcohol and drug abuse, and non-compliance with therapy.
Exercise intervention
The intervention group received a 3-week rehabilitation program with an individual and according to their preferences and physical resource-adapted exercise program. Therefore, face-to-face meetings and a detailed diagnosis of a patient’s movement and exercise habits, circumstances of life, and exercise possibilities in their home place were conducted. In addition, a physical performance test (IPN test) by physiotherapists was performed. The individualized exercise program of 15 metabolic equivalents (METs) and the focus of the therapy units were based on the results of the previous tests and diagnostics. If one patient could not name any preferred sport activity, the first week of the rehabilitation was used to find their most suitable type of movement (walking, treadmill, ergometer, bicycle, machine training, etc.) for the following exercise focus. Furthermore, the patients from the intervention group received an individual, home-based exercise program. The aftercare involved a 1-week inpatient stay at the clinic, 4 and 8 months after the first 3-week stay. An additional personal phone call took place 1 month after the first discharge, and the patients were questioned again at the time points of 12, 18, and 24 months after the beginning of the rehabilitation with postal questionnaires on physical activity, fatigue syndrome, and health-related quality of life.
The main purpose of the individual and detailed history of physical activity (habits, preference, possibilities at home), the telephone aftercare, and the home-based training schedule was to generate a best possible individual and precise therapy and exercise program in order to sustainably improve the physical activity level and achieve the recommended activity level (sport metabolic rate) of 15 MET-h/week [12, 13].
Assessment
Patients of both groups filled in an anthropometric questionnaire, which comprised clinical and sociodemographic data. Information about tumor stage, chemotherapy, and radiation were used from the patient records.
The registration of the primary endpoint (level of physical activity) was measured with the validated “Freiburger Fragebogen zur körperlichen Aktivität” (Freiburg questionnaire on physical activity; FFkA, German version) involving 12 questions for physical activity in daily life, leisure time, and exercise activity in hours per week [17]. Activity is divided into the following subgroups: basic activity (e.g., shopping), leisure activity (e.g., taking a walk), and sports activity.
The health-related quality of life (QoL) was determined with the European Organization for Research and Treatment of Cancer (EORTC) “Quality-of-Life questionnaire (QLQ-C30)” [18].
To survey fatigue syndrome, the “Multidimensional Fatigue Inventory” (MFI) was used. The 20-itemself-report questionnaire is subdivided in the dimensions general, physical, and mental fatigue; reduced activity; and reduced motivation, with four questions in each dimension and scoring from 1 to 5 points with higher results indicating higher grades of fatigue syndrome [19, 20].
Statistical analysis
Following the intention-to-treat (ITT) approach, data from all participants that completed t 0 assessment were included in the following analyses (N = 194). Missing values due to dropouts from the study were replaced using the last-observation-carried-forward (LOCF) method.
Potential baseline differences between treatment groups in age, height, weight, BMI, time since diagnosis, duration of radiotherapy, duration of chemotherapy, EORTC-QLQ-C30, MFI-20, and FKKA subscales were investigated using separate independent t tests. Due to the central limit theorem, we assumed the approximate normality of sampling distribution [21, 22]. Independent t test assumption of homogenous variances between groups was tested using Levene’s test. Potential baseline differences between treatment groups regarding distribution of radiotherapy, chemotherapy, and postmenopausal patients were investigated using separate Fisher’s exact tests.
Effects of time point (t 0 vs. t 1 vs. t 2 vs. t 3 vs. t 4 vs. t 5), group (CG vs. IG), and time point × group interaction on the FFKA, EORTC, and MFI subscales were assessed using separate 2 × 6 mixed analyses of variance (mixed ANOVA). Again, the normality of sampling distribution was assumed due to the central limit theorem. ANOVA assumption of sphericity for variances of differences between categories of within-subject factor time point was tested using Mauchly’s test. If Mauchly’s test had reached significance, the Greenhouse-Geisser rectification method was applied. ANOVA assumption of homogeneous variances in between factor categories was tested using Levene’s test. Interaction effects were further investigated through simple effect analyses. Alpha error accumulation at simple effect analyses was controlled using Bonferroni adjustment. For significant effects, partial eta-squared (ƞ 2 p) values were reported as effect size estimates.
For all inferential statistical analyses, significance was defined as the p value less than 0.05. All descriptive and inferential statistical analyses were conducted using SPSS 22® (IBM®, Armonk, NY, USA). Two-tailed probability tests were used throughout all inferential statistical testing.
Results
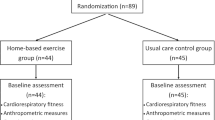
A total of 194 patients were recruited between May 15, 2010, and April 15, 2011, of which 111 patients were assigned to the intervention group and 83 patients to the control group.
Both groups were comparable with respect to the most relevant clinical and sociodemographic data. However, patients in the intervention group were significantly younger (p = 0.001). All patients had finished medical treatment at the beginning of the study. For the intervention group, the mean durations after the end of the medical treatment were 1.7 ± 1.9 months (after chemotherapy) and 1.4 ± 0.7 months (after radiation), and for the control group, 1.8 ± 2.0 months (after chemotherapy) and 1.3 ± 0.8 months (after radiation). The beginning of the rehabilitation took place 11.4 ± 7.6 and 12.8 ± 8.5 months after breast cancer diagnosis in the intervention and the control groups, respectively (Table 1). During the study, some patients from both intervention and control groups ended their participation due to personal and medical reasons. Detailed information concerning the dropouts is enclosed as an attachment.
Level of physical activity
We have observed that 74.1% of the patients in the intervention group achieved the recommended activity level (sport metabolic rate) of 15 MET-h/week after 2 years (t5). In the control group, 48.7% reached that level. The internal group comparison showed significant differences after 2 years. There were differences at t1 (p = 0.002), t2 (p = 0.020), t3 (p = 0.005), and t5 (p = 0.005) in the intervention group in the internal group comparison with respect to the total metabolic rate (Fig. 1). After 2 years, the level of physical activity (total metabolic rate) significantly increased from 2733.16 ± 2547.95 (t0) to 4169.71 ± 3492.27 (t5) MET-min/week in the intervention group, but just slightly from 2858.38 ± 2393.79 (t0) to 2875.74 ± 2590.15 (t5) MET-min/week in the control group (means ± standard deviation) (Table 2). Baseline (p = 0.813) and t4 (p = 0.196) showed no significant differences. Additionally, the intragroup comparison revealed six significant differences in the intervention group at eight parameters related to an increase in the physical activity level. There were no significant differences regarding the control group. The intervention group increased in respect to all parameters from t0 to t5, while the control group decreased in respect to basic metabolic rate, total activity, and basic activity.
In relation to the parameter “activity,” we observed a significant increase in the total activity (p = 0.001), sports activity (p = 0.001), and leisure activity (p = 0.003) between t0 and t5 in the intervention group. No significant changes in basic activity were observed (p = 0.265). Within this context, no significant changes were noted in the control group. The total activity of the intervention group increased from 10.02 ± 8.1 h/week (t0) to 15.36 ± 11.17 h/week (t3), decreased to 12.36 ± 9.36 h/week (t4), and increased to 14.15 ± 11.47 h/week (t5). In the CG, an increase was observed in the total activity of 10.52 ± 9.54 h/week (t0) to 11.68 ± 9.56 h/week (t3) with a subsequent decrease to 10.43 ± 9.08 h/week, which was below the baseline value.
The leisure activity and the leisure metabolic rate principally remained unchanged in all measure points (t0 to t5) and tended to be higher in the control group. Basic activity and basic metabolic rate increased between t0 and t3 in the intervention group, while they decreased between t3 and t4. At the last time point (between t4 and t5), both parameters increased. There was no change observed in the control group between t0 and t3, but there was a decrease regarding the basic activity between t3 and t5 lower than the t0 baseline level (Table 2).
Fatigue
Comparing both groups in respect to the general fatigue score, we have observed significantly lower fatigue scores in the intervention group at t1 (p = 0.034), t2 (p = 0.011), and t4 (p = 0.025) (Fig. 2). In the intervention group, fatigue syndrome remained in all scales and at all measure points (t1 to t5) on a lower level compared to the control group. In the intervention group, a decrease was observed regarding fatigue syndrome in all dimensions from t0 to t2, while the control group showed an increase in four dimensions (mental fatigue, reduced motivation, reduced activity, and physical fatigue) and a decrease in general fatigue. Between t2 and t5, the control group showed hardly any differences while the intervention group showed slightly reduced effects on fatigue syndrome. The intervention group demonstrated, from baseline to t5 fatigue, reductions in four dimensions (general fatigue, mental fatigue, reduced activity, and physical fatigue) while the control group revealed fatigue reductions only in two dimensions (general fatigue and physical fatigue).
Health-related quality of life
Both groups slightly proved an increase in the general health-related quality of life from t0 to t5. The data of the intervention group were higher at all time points between t1 and t5 compared to the control group but not significant (Fig. 3).
Discussion
This quasi-randomized, controlled study hypothesized that a personalized, supervised exercise program, including an additional 1-week inpatient stay after 4 and 8 months, has a more positive and more sustainable impact on physical activity level in breast cancer patients compared to the usual care in the rehabilitation clinic. In addition, we expected a reduction regarding the fatigue syndrome and an improvement on the health-related quality of life.
After 2 years, the patients in the intervention group showed a sustainable and significant increase in the level of physical activity (total metabolic rate) (t0 vs. t5) by 50.36% while the control group increased just by 0.61%. The intervention group showed an increase in all parameters from t0 to t5 by up to 105.24% (sport activity) while the control group showed a decrease in respect to basic metabolic rate (−25.49%), total activity (−0.86%), and basic activity (−26.07%). The fatigue syndrome revealed a significant decrease as well, but had no effects on the health-related quality of life. To our knowledge, no studies have investigated the sustainable impact of an individual exercise program on the physical activity level in breast cancer patients proving significant effects after 2 years.
The results revealed a significant and sustainable improvement of the physical activity level in the intervention group between t0 and t5. This is mainly observed due to the individual exercise control, regarding the preferred method of exercise, and the professional instruction of skilled therapists. Furthermore, two additional 1-week inpatient stays took place after 4 and 8 months, which provided a high motivation and supported a change of lifestyle. The patients have experienced positive effects of physical activity as fatigue syndrome showed a decrease after physical exercise [10]. To obtain a sustainable improvement, it is also important to include the conditions of life in the particular home setting in the individual exercise program.
The unexpected decrease in both total activity level and total metabolic rate between t3 and t4 in the intervention group and the following increase between t4 and t5 are possibly explained due to the season of the year as the decrease in these parameters took place during the winter, when people tend to be less active [23].
Several studies already proved that physical activity is safe and effective in both medical treatment and follow-up care of breast cancer therapy [24, 25]. Furthermore, the increase in physical activity in the intervention group was accompanied by a substantial improvement in fatigue and a significantly higher health-related quality of life. This is already documented in other studies [10, 26–33], but was not observed in our study. The parameter of QoL is a very complex concept, dependent upon multiple variables. In the rehabilitation clinic, the patients received various other therapies, such as psycho-oncology, and we assume that this fact is a possible reason that in this study, no significant effects between the groups regarding QoL were observed.
One possible limitation in this study can be the fact that this study is not a typical randomized, controlled (RCT) study. There were also differences at baseline between both groups in the physical activity level. Additionally, some patients ended their participation in the study due to personal and medical reasons. Nevertheless, the dropouts were higher in the control group (t0 vs. t5) with a mean of 33.73–35.28% compared to the dropouts in the intervention group (30.63–31.53%).
In conclusion, due to the fact that diseases and therapy can vary from case to case, every patient needs to get an individualized and suitable treatment and professional therapeutic support. To achieve the best possible therapy effects, physical exercise and rehabilitation programs should be individual and holistic and include all side effects of the medical treatment.
Further studies are required to confirm these results, and based on the present study, there are a lot of new challenges and complications that have to be approached. The obtained data need a translation into the patient-centered care. Furthermore, we recommend new rehabilitation concepts. The usual 3-week rehabilitation is not personalized enough to obtain sustainable improvements, which is why patients need further follow-up inpatient stays with individual support. Therefore, a specialized education of physiotherapists and sport therapists according to the state of the art is required. To ensure this progress, the government, the health insurance funds, and the medical care system need to place higher financial investments in the cancer aftercare and rehabilitation programs.
References
Global Burden of Disease Cancer Collaboration (2015) The global burden of cancer 2013. JAMA Oncol 1:505–527. doi:10.1001/jamaoncol.2015.0735
Robert-Koch-Institut und die Gesellschaft der epidemiologischen Krebsregister in Deutschland e. V. (Hrsg.) (2015) Krebs in Deutschland 2011/2012. Berlin, Germany. doi:10.17886/rkipubl-2015-004
Skarin A (2000) Diagnosis in oncology: side effects of chemotherapy. J Clin Oncol 18:693–698
Hidding JT, Beurskens CH, van der Wees PJ, van Laarhoven HW, Nijhuis-van der Sanden MW (2014) Treatment related impairments in arm and shoulder in patients with breast cancer: a systematic review. PLoS One 9:e96748. doi:10.1371/journal.pone.0096748
Burish TG, Tope DM (1992) Psychological techniques for controlling the adverse side effects of cancer chemotherapy: findings from a decade of research. J Pain Symptom Manag 7:287–301. doi:10.1016/0885-3924(92)90062-M
Asher A (2011) Cognitive dysfunction among cancer survivors. American Journal of Physical Medicine & Rehabilitation/Association of Academic Physiatrists 90:16–26. doi:10.1097/PHM.0b013e31820be463
Kwok C, White K (2014) Perceived information needs and social support of Chinese-Australian breast cancer survivors. Support Care Cancer 22:2651–2659. doi:10.1007/s00520-014-2252-x
Kayl AE, Meyers CA (2006) Side-effects of chemotherapy and quality of life in ovarian and breast cancer patients. Current Opinion in Obstetrics & Gynecology 18:24–28. doi:10.1097/01.gco.0000192996.20040.24
Palmer BV, Walsh GA, McKinna JA, Greening WP (1980) Adjuvant chemotherapy for breast cancer: side effects and quality of life. BMJ 281:1594–1597. doi:10.1136/bmj.281.6255.1594
Velthuis MJ, Agasi-Idenburg SC, Aufdemkampe G, Wittink HM (2010) The effect of physical exercise on cancer related fatigue during cancer treatment: a meta-analysis of randomised controlled trials. ClinOncol 22:208–221. doi:10.1016/j.clon.2009.12.005
Robert JT, Holm M, Al-Adhami A (2014) Physical activity after cancer: an evidence review of the international literature. British Journal of Medical Practitioners 7(1):a708
Holmes MD, Chen WY, Feskanich, Kroenke CA, Colditz GA (2005) Physical activity and survival after breast cancer diagnosis. JAMA 293:2479–2486. doi:10.1001/jama.293.20.2479
Irwin ML, Wilder Smith A, McTiernan A et al (2008) Influence of pre- and postdiagnosis physical activity on mortality in breast cancer survivors: the health, eating, activity, and lifestyle study. J ClinOncol 26:3958–3964. doi:10.1200/JCO.2007.15.9822
Pinto BM, Rabin C, Papandomatos GD, Frierson GM, Trunzo JJ, Marcus BH (2008) Maintenance of effects of a home-based physical activity program among breast cancer survivors. Support Care Cancer 16(11):1279–1289. doi:10.1007/s00520-008-0434-0
Huy C, Schmidt ME, Vrieling A, Chang-Claude J, Steindorf K (2012) Physical activity in a German breast cancer patient cohort: one-year trend and characteristics associated with change in activity level. Eur J Cancer 48:297–304. doi:10.1016/j.ejca.2011.08.005
Rogers LQ, Hopkins-Price P, Vicari S et al (2009) A randomized trial to increase physical activity in breast cancer. Med Sci Sports Exerc 41:935–946. doi:10.1249/MSS.0b013e31818e0e1b
Frey I, Berg A, Gratwohl D, Keul J (1999) Freiburger Fragebogen zur körperlichen Aktivität—entwicklung, prüfung und anwendung. Soz Praventivmed 44:55–64. doi:10.1007/BF01667127
Aaronson NK, Ahmedzai S, Bergman B et al (1993) The European Organisation for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst 85:365–376. doi:10.1093/jnci/85.5.365
Smets EM, Garssen B, Bonke B, De Haes JC (1995) The Multidimensional Fatigue Inventory (MFI) psychometric qualities to assess fatigue. J Psychosom Res 39:315–325. doi:10.1016/0022-3999(94)00125-O
Smets EM, Garssen B, Cull A, De Haes JC (1996) Application of the multidimensional fatigue inventory (MFI-20) in cancer patients receiving radiotherapy. Br J Cancer 73:241–245
Rosner B (2000) Fundamentals of biostatistics, 5th edn. Duxbury, Pacific Grove, p. 308
Hays WL (1994) Statistics, 5th edn. Holt, Rinehart and Winston, New York
Uitenbroek DG (1993) Seasonal variation in leisure time physical activity. Med Sci Sports Exerc 25:755–760
Fong SSM, Ng SSM, Luk WS et al (2014) Effects of qigong exercise on upper limb lymphedema and blood flow in survivors of breast cancer: a pilot study. Integr Cancer Ther 13:54–61. doi:10.1177/1534735413490797
McKenzie DC, Kalda AL (2003) Effect of upper extremity exercise on secondary lymphedema in breast cancer patients: a pilot study. J Clin Oncol 21:463–466. doi:10.1200/JCO.2003.04.069
Mishra SI, Scherer RW, Snyder C et al (2012) Exercise interventions on health-related quality of life for cancer survivors. Cochrane Database Syst Rev 15:CD007566. doi:10.1002/14651858.CD007566.pub2
Markes M, Brockow T, Resch KL (2006) Exercise for women receiving adjuvant therapy for breast cancer. Cochrane Database Syst Rev 18:CD005001. doi:10.1002/14651858.CD005001.pub2
Galvao DA, Newton RU (2005) Review of exercise intervention studies in cancer patients. J Clin Oncol 23:899–909. doi:10.1200/JCO.2005.06.085
Conn VS, Hafdahl AR, Porock DC et al (2007) A meta-analysis of exercise interventions among people treated for cancer. Support Care Cancer 14:699–712. doi:10.1007/s00520-005-0905-5
McNeely ML, Campbell KL, Rowe BH et al (2006) Effects of exercise on breast cancer patients and survivors: a systematic review and meta-analysis. CMAJ 175:34–41. doi:10.1503/cmaj.051073
Schmitz KH, Holtzman J, Courneya KS et al (2005) Controlled physical activity trials in cancer survivors: a systematic review and meta-analysis. Cancer Epidemiol Biomark Prev 14:1588–1595. doi:10.1158/1055-9965.EPI-04-0703
Mehnert A, Veers S, Howaldt D et al (2011) Effects of a physical exercise rehabilitation group program an anxiety, depression, body image, and health related quality of life among breast cancer patients. Onkologie 34:248–253. doi:10.1159/000327813
Dimeo FC (2001) Effects of exercise on cancer-related fatigue. Am Cancer Soc 92:1689–1693
Acknowledgements
We would like to thank the “Deutsche Rentenversicherung Baden Württemberg” for the financial support to conduct this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was in accordance with the Declaration of Helsinki and approved by the ethics committee of the German Sports University of Cologne. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Baumann, F.T., Bieck, O., Oberste, M. et al. Sustainable impact of an individualized exercise program on physical activity level and fatigue syndrome on breast cancer patients in two German rehabilitation centers. Support Care Cancer 25, 1047–1054 (2017). https://doi.org/10.1007/s00520-016-3490-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-016-3490-x