Abstract
Key message
Winter and spring wet and cool conditions often linked to El Niño enhance growth in five Mexican pine species which showed different responses to drought.
Abstract
Climate warming could intensify drought stress in NW México mountains as the Sierra Madre Occidental which are not only centers of pine diversification but also subject to a high hydroclimatic variability. In this region, rainfall and thus forest growth depend on changes in sea-surface temperatures (SST) across the equatorial Pacific related to the El Niño Southern Oscillation (ENSO). Winter moist-cool conditions usually correspond to El Niño episodes characterized by warm SSTs in the equatorial Pacific, whilst dry winters correspond to cold SSTs (La Niña). Consequently, growth responsiveness to dry spells could be used as a proxy of forest vulnerability to increased drought stress. Here, we characterize the growth responsiveness of five coexisting Mexican pine species to hydroclimate, drought, and the ENSO using dendrochronology. Pinus lumholtzii and P. durangensis showed a low responsiveness to maximum temperatures and drought, whereas others as P. arizonica, P. engelmannii, and P. leiophylla were very responsive. Among these drought-sensitive pine species, we found responses to ca. half- (e.g. P. engelmannii) and 1-year long (e.g. P. arizonica) droughts. The most responsive species to drought, P. arizonica and P. leiophylla, were also those showing a higher association between ENSO-related indices and growth. Wet and cool winter and spring conditions, linked to warm SSTs over the equatorial Pacific, were the main hydroclimate factors enhancing growth. Pine species presented different growth responsiveness to drought which suggests that they present diverse tolerances to water shortage. This implies that coexisting pine species will differently face the more arid conditions forecasted for many drought-prone areas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate warming is expected to increase drought stress reducing both forest productivity and growth, and increasing tree mortality, particularly in semi-arid areas as the SW USA (Williams et al. 2013). In the nearby NW México, climate models also predict increased aridification and more frequent drought spells reducing long-term water availability (Cook and Seager 2013). A higher evaporative demand as a consequence of a higher vapour pressure deficit could lead to a growth decline in this drought-prone region, and such reduction in productivity could portend forest dieback (McDowell et al. 2008; Camarero et al. 2015). Nevertheless, such predictions should explicitly consider the different responsiveness to drought of coexisting tree species, particularly in diversity hotspots where drought tolerance in front of the forecasted warmer and drier climate can vary among co-occurring species.
To assess how drier and warmer climate conditions affect the tolerance to drought of forests and tree species across NW México, it must be considered that El Niño–Southern Oscillation (ENSO) events and related moist-cool conditions enhance forest productivity and tree radial growth in this region (Stahle and Cleaveland 1993; Stahle et al. 1998). Hydroclimatic conditions in this region are, in fact, tightly associated with ENSO-related atmospheric patterns (Andrade and Sellers 1988). The precipitation variability over this area is controlled by sea-surface temperatures (SST) of the tropical Pacific and Atlantic Oceans (Seager et al. 2009). Warm SSTs in the equatorial Pacific and El Niño events characterize wet winters, whilst cold SSTs and La Niña events correspond to dry winters (Andrade and Sellers 1988; Kiladis and Díaz 1993; Magaña et al. 1999). For instance, mountain pine forests from NW México experienced severe winter droughts in the mid 1990s (e.g. 1996) due to a displacement of the storm track caused by a more La Niña-like state of the tropical Pacific (Seager et al. 2009). These droughts negatively impacted water-intensive crops grown in NW México, mostly cultivated for exportation (e.g., alfalfa, fruits), reduced corn and grain harvests, decreased reservoir capacity to less than 20%, and initiated conflicts between farmers, cattle ranchers, and indigenous communities causing migration of rural population to the USA (Liverman 1999).
It is uncertain if climate warming could extend the drying of the semi-arid NW México to the central part of the country and the wet south by expanding the subtropical dry zone, weakening the Mexican summer monsoon or delaying its onset date in winter (Grantz et al. 2007; Seager et al. 2009). For example, Cook and Seager (2013) argue that shifts in the seasonal distribution of precipitation within the North American Monsoon over NW México would have major climatic and ecological consequences. In any case, the projected warming will further menace water resources negatively impacting forest ecosystems (Martínez and Fernández 2004). This potential threat justifies investigating how drought-prone and diverse NW Mexican pine forests respond to recent drought stress in such vulnerable areas.
The forest growth of coexisting pines showing different responsiveness to drought in such semi-arid areas can be regarded as a reliable proxy of rangeland productivity (Cleaveland et al. 2003). Coexistence of many pine species in México occurs, because this country has more pine species than any other country of similar size in the world, and it is one of the top three areas worldwide for pine speciation (Perry 1991; Farjon and Styles 1997; Farjon 2010). Of the 111 world pine species, 47 (31 diploxylon + 16 haploxylon species) occur in México representing 43% of the world pine species, and at least 35 of these species (e.g. pinyon pines) are endemic to the country (Carvajal and McVaugh 1992; Sánchez-González 2008). Other numbers reveal the importance of pine forests to México where ca. 33 million ha are occupied by these species which accounted for a third of the total forested area in the 1980s, and represented about 80% of the total annual logged timber volume in NW México (Ruelas Monjardín and Dávalos Sotelo 1999).
The greatest diversity of pine species concentrates in highland forests of the Sierra Madre Occidental, the largest mountain range (extending 1200 km) in México, where there are reports on 24 pine species (González-Elizondo et al. 2012; Gernandt and Pérez de la Rosa 2014). In this area, pine forests are ecologically and economically very valuable, since they provide both ecosystem services and direct economic resources (timber, lumber, cones, plant collection, wildlife hunting, cattle raising, etc) to native people as the Tarahumara Indians (Fulé et al. 2011).
Pine-dominated upland Mexican forests seem to be the result of drought cycles mediated by climate conditions (Figueroa-Rangel et al. 2008). Mexican pines forests are subjected to marked climatic gradients forming diverse formations either as pure stands (e.g., Pinus durangensis) or as assemblages of up to seven pine species often, including other genera of conifers (firs and junipers) and hardwood (oaks) species (Rzedowski 2006). These climatic gradients usually lead to the replacement of pine species along altitudinal gradients (Yeaton 1982; Earle 2007).
The diverse pine forests of NW México subjected to a marked variability in precipitation offer a unique opportunity to study how tree growth of coexisting tree species responds to drought stress. In fact, several dendroclimatic studies have been performed in this area, including some of the first American tree-ring chronologies (Schulman 1944). These studies showed that growth of most tree species was enhanced in northern Mexico forests as spring and summer rainfall increased, which often occurs during El Niño events (Stahle and Cleaveland 1993; Barton et al. 2001; Therrell et al. 2002; Villanueva-Díaz et al. 2000, 2006, 2007). However, these studies did not evaluate climate–growth relationships among different coexisting pine species subjected to similar local climate conditions, which is a reliable approach to understand the growth responsiveness to drought at species level. Here, we address this issue by following a dendroecological approach. Our first objective is to analyze how radial growth of five pine species coexisting in the Sierra Madre Occidental responds to climate conditions and drought stress. The second objective involves a better understanding of the role played by ENSO-related atmospheric patterns driving hydroclimate conditions in NW México, thus affecting drought stress and tree growth. We argue that a better understanding of climate–growth relationships would improve our ability to determine the vulnerability of Mexican pine forests to the forecasted warmer and drier climate conditions. This argument could be applied to other mid-latitude centers of pine diversity (e.g., Mediterranean forests) which are also subjected to seasonal drought.
Materials and methods
Study site
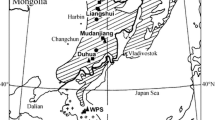
The study site (Cuchíwachi) is located in the Sierra Madre Occidental, Chihuahua state, NW México (27° 04′ 38″–27° 05′ 31″N, 107° 08′ 11″–107° 08′ 22″W). The site has a mean elevation of 1390 m a.s.l. and the average slope is 3%. The mean crown cover is 60% and the mean standing volume is 70 m3 ha−1. The dominant vegetation is formed by mixed pine and oak–pine forests with the presence of other species of the Juniperus and Arbutus genera (González-Elizondo et al. 2012). The climate in the study area is temperate with dry winters and hot summers (Cwa according to the Köppen–Geiger climate classification). The mean temperature is 16.2 °C and annual precipitation is about 779 mm with 46% of this precipitation being recorded from July to August (INEGI 2003; data from nearby Guachochi station, 107° 04′ 48″W, 26° 49′ 48″N, 2420 m a.s.l.). The warmest and coldest months are June (mean maximum temperature 31.4 °C) and January (mean minimum temperature −1.0 °C), respectively. Soils are mainly Leptosols and Regosols of volcanic origin.
Pine species
The five studied pine species are: Pinus arizonica Engelm., Pinus durangensis Martínez, Pinus engelmannii var. blancoi (Martínez) Martínez, Pinus leiophylla Schiede ex Schltdl. & Cham., and Pinus lumholtzii B.L. Rob. & Fernald. They belong to the Martínez (1948) “Ponderosa” group (mostly three-needled species) which occurs in the Sierra Madre Occidental. Indigenous local people use the trunks and branches for making tools, but logging has reduced the range of some species as P. durangensis and P. arizonica which have been exploited for timber and construction lumber (González-Elizondo et al. 2012).
According to the Flora of North America (http://www.efloras.org/), the study pine species are native to NW Mexico, Arizona, New Mexico, and Texas, where they can reach maximum heights of 22 (P. engelmannii) to 35 m (P. arizonica). P. arizonica coexists with oaks, junipers, and other pine species at elevations of 2000–2700 m in the Sierra Madre Occidental, mainly on deep soils (Perry 1991; González-Elizondo et al. 2012). P. durangensis is mainly distributed at an altitudinal range of 1400–3000 m and at its upper limit can coexist with fir species in wet sites (about 1000 m of annual precipitation). P. engelmannii inhabits mountains, valleys, and plateaus at 1200–3000 m, often forming post-fire pure stands (Barton 2002). P. leiophylla appears at 1300–2600 m under warm-temperate climate conditions forming mixed pine–oak–fir stands on deep and well-drained soils (Perry 1991). P. lumholtzii occurs from 1600 to 2900 m, often on sites with shallow and acid soils (González-Elizondo et al. 2012). In the study area, P. engelmannii and P. arizonica usually inhabit drier sites, often characterized by rocky and shallow soils with a low water-holding capacity, than P. durangensis, whereas P. lumholtzii and P. leiophylla are also associated to semi-arid climate conditions (González-Elizondo et al. 2012).
Four of the studied species have been sampled in México for dendrochronological investigations, mainly focused on describing forest dynamics and reconstructing hydroclimatic conditions (P. durangensis in Barton et al. 2001; P. durangensis in Villanueva-Díaz et al. 2000, 2006, 2007; P. engelmannii and P. lumholtzii in Villanueva-Díaz et al. 2006 and Bickford et al. 2011).
Field sampling and dendrochronological methods
Sampled individuals were randomly selected and sampled in a plot of 1 ha. We collected wood samples (discs) at 0.3 m of 10 living individuals for 4 of the study species and from 20 individuals for P. durangensis felled for timber in late 2015. In addition, we collected increment cores from five trees per species using a Pressler increment borer.
In the laboratory, the wood discs and cores were air-dried and sanded using successively finer grained sandpapers until the tree rings were clearly visible. Tree-ring series were visually cross-dated by comparing and detecting characteristic narrow rings among radii of the same tree and then by matching the series from different trees of the same species. We measured the ring widths from the most recent ring up to the pith along two radii per sample (separated by 90° in the case of discs to account for within-tree-growth variability). Tree rings were measured with a 0.01-mm resolution under a binocular scope using a LINTAB measuring device (Rinntech, Heidelberg, Germany). Then, the previous visual cross-dating was checked using the COFECHA program which compares all tree series with the master-dating chronology of each species (Holmes 1983). Samples failing to pass this check were excluded to develop the species ring-width chronologies (five trees per species were excluded excepting P. durangensis, for which six trees were excluded).
To built species chronologies, we first removed the long-term growth trends potentially related to non-climatic fluctuations (tree ontogeny, stand dynamics) by fitting negative exponential curves to each series of ring-width measurements, and then obtaining ring-width indices. Second, an autoregressive model was fitted to each of these detrended series to remove most of the temporal autocorrelation related to the previous year growth. Finally, a biweight robust mean was used to obtain species chronologies of residual indices (mean = 1). This procedure was performed using the ARSTAN program (Cook and Holmes 1986).
The resulting species chronologies were characterized by calculating several statistics: the first-order autocorrelation (AC) which quantifies the year-to-year persistence in tree-ring width; the mean sensitivity (MS), a measure of the relative change in ring-width index between consecutive years; and the mean correlation between trees (Rbt) for the common interval 1970–2015 (Fritts 2001). We also calculated the expressed population signal (EPS) for this period to determine how suitable are the species chronologies for capturing the hypothetical population signal, with a minimum threshold value of 0.85 being usually considered to define well-replicated chronologies (Wigley et al. 1984).
Climate data, drought index, and ENSO-related indices
In NW México highland pine forests, a few climate records extend back into the early 20th century and most climate stations are sparsely distributed and do not have complete records (Stahle et al. 2000). Consequently, we obtained monthly climate data (mean maximum and minimum temperatures, and total precipitation) for the 0.5°-grid centered on the study site (27.0–27.5°N, 107.0–107.5°W) from the Climate Research Unit data set, version 3.22 (Harris et al. 2014), using the KNMI-Climate explorer webpage http://climexp.knmi.nl/ (Van Oldenborgh and Burgers 2005). These data indicated that mean maximum and minimum temperatures have warmed significantly since 1970 in the study area, particularly during spring and summer but not in winter.
To quantify drought stress, we used monthly values of the Standardised Precipitation-Evapotranspiration Index (SPEI; see Vicente-Serrano et al. 2010). The SPEI considers the effect of temperatures on the evapotranspiration rate with negative and positive SPEI values reflecting dry and wet conditions, respectively. The SPEI was calculated for the same 0.5° grid, including the study site, and it was obtained from the Webpage http://sac.csic.es/spei/index.html.
To determine how the ENSO affects hydroclimate and tree growth in NW México, we obtained the following indices: the Southern Oscillation Index (SOI), a standardised index based on the observed sea level pressure and temperature differences between the western and eastern tropical Pacific (Ropelewski and Jones 1987); El Niño 3.4 index based on the SST anomalies in the “El Niño 3 + 4” region (5°N–5°S, 120°–170°W) of the equatorial Pacific (Rayner et al. 2003); and the Pacific Decadal Oscillation (PDO) which is a long-term El Niño-like pattern of climate variability characterized by warm and cool SST anomalies in the NE and tropical Pacific Ocean (Zhang et al. 1997). ENSO years were defined by 5-month running means of SSTs anomalies in the Niño 3.4 region exceeding +0.4 °C for at least 6 consecutive months (Trenberth 1997). The SSTs were obtained from the NOAA 0.25° daily Optimum Interpolation data (period 1981–2013; Reynolds et al. 2007). In general, prolonged periods of negative (positive) SOI values coincide with very warm (cold) SSTs across the eastern tropical Pacific (e.g., El Niño 3.4 region) typical of El Niño (La Niña) episodes.
Statistical analyses
We calculated Pearson correlations between monthly climate data (mean maximum and minimum temperatures), SPEI, atmospheric indices (SOI, El Niño 3.4, PDO), and residual chronologies of each species for the common period 1969–2014. In the case of precipitation, we used the Spearman coefficient to account for its non-normal distribution. We considered the temporal window from the previous June up to July of the growth year based on the previous dendroclimatic analyses carried out in NW México (Villanueva-Díaz et al. 2006, 2007). We also used the Seascorr program (Meko et al. 2011) to calculate partial correlations for the secondary climate variable (mean maximum temperatures) to identify tree-growth response related specifically to that variable, independent of variance related to the primary variable (precipitation). Subsequently, we identified periods when climate–growth correlations reached maximum values by analyzing 3-, 6-, 9- and 12-months long scales. To evaluate how drought stress affected year-to-year growth variability, the monthly SPEI indices calculated from 1 to 28-months long scales (representing cumulative water deficit for those time scales) were correlated to the residual ring-width indices considering a time window from the previous October to current December. Finally, to illustrate and to summarize the geographical climate–growth patterns, we calculated field correlations based on Pearson coefficients by relating the grand mean of the five species chronologies and climate data (CRU data set: temperature, precipitation; NOAA SST data set) using the KNMI-Climate explorer webpage. Climate and SST monthly or seasonal data used to calculate the field correlations were previously selected based on the climate- or SST-growth associations.
Results
Pine growth patterns
The pine species with the widest rings were P. engelmannii (2.96 mm) followed by P. arizonica (2.77 mm), whilst the species with the narrowest rings were P. durangensis (1.35 mm) and P. lumholtzii (1.47 mm), albeit both of these species also presented the oldest trees (Table 1). The species with the lowest first-order autocorrelation and the highest mean sensitivity were P. leiophylla and P. engelmannii, whereas the lowest mean sensitivity was observed in P. lumholtzii. The highest coherence in growth between conspecific trees was observed in P. engelmannii, whilst the lowest was found in P. durangensis, the species which presented the most difficult cross-dating (Table 1). Accordingly, all pine species chronologies presented EPS values above the 0.85 threshold excepting P. durangensis (EPS = 0.77).
Considering the common 1970–2015 period, the two ring-width chronologies most tightly related were P. arizonica and P. leiophylla (r = 0.897), whereas the pair P. durangensis–P. leiophylla showed the lowest correlation value (r = 0.673). Overall, P. durangensis was the species with the lowest correlation values among the pine species (Table 2).
The five pine species showed similar growth patterns characterized by pointer years that were common among all species (Fig. 1). This was particularly evident regarding inter-annual growth variability and considering some dry years corresponding to narrow rings (e.g., 1970, 1974, 1978, 1980, 1982, 1989–1990, 1996, 1999–2000, 2002–2003, and 2011–2012). Conversely, wet winters often linked to ENSO events corresponded to wide rings (e.g., 1983, 1987, 1991–1993, 1997–1998, 2010, and 2015).
Growth patterns (a tree-ring width; b ring-width index) obtained for the five pine species coexisting in NW Mexico. Growth is related to accumulated precipitation from the previous August to current May (a bars and right y axis) and sea surface temperature anomalies over the equatorial Pacific (b bars and right y axis). In the lowermost plot, ENSO events are indicated by filled bars
Climate–growth associations
Overall, wet and cool winters were associated with a higher growth rate, but each pine species showed different sensitivity to these climatic conditions. Warm conditions in the previous summer, fall, and winter (October–January) and also in the current spring and early summer (March–June) were negatively and significantly related to growth of all pine species excepting P. durangensis which only responded to warm conditions from April to June of the year of tree-ring formation (Fig. 2). Contrastingly, we only found negative responses to high minimum temperatures in the previous August and the current April and June in the case of P. arizonica, P. engelmannii and P. leiophylla. Warmer minimum temperatures in the previous November and December were positively associated to growth, particularly in the case of P. lumholtzii and P. leiophylla, albeit not significantly. High January–February precipitation was related to enhanced growth of all species, being P. arizonica and P. durangensis the most and least responsive species to changes in winter rainfall. Wet conditions during the previous August and the current spring (April–May–June) also favored growth, being P. durangensis the most sensitive species to changes in spring rainfall.
Climate–growth associations (Pearson correlation coefficients) calculated for the five Mexican pine species by relating climate variables (mean maximum and minimum temperatures, total precipitation) to the residual ring-width chronologies. Correlations were calculated from the previous August to the current July. The previous- and current-year months are abbreviated by lowercase and uppercase letters, respectively. The dashed and dotted horizontal lines indicate the 0.05 and 0.001 significance levels
The analyses of the seasonalized precipitation identifies highest correlations with growth of most pine species for periods of lengths 3, 6, 9, and 12 months ending in January, March, June, and July, respectively (Fig. 3). Again, the pine species whose growth was most responsive to precipitation variability were P. arizonica, P. engelmannii, and P. leiophylla, whereas P. lumholtzii and P. durangensis were the least responsive. In addition, P. durangensis presented highest precipitation-growth correlations for 3- and 6-month long periods in the previous November and January, respectively. This species was the least responsive to maximum temperatures, whilst P. lumholtzii was the most responsive.
Correlations obtained by relating precipitation (P) as primary climate variable and mean maximum temperature (T) as secondary climate variable to the residual ring-width chronologies of the five Mexican pine species. Precipitation and temperature were summed or averaged for periods of 3, 6, 9, and 12 months whose ending months are shown in the lowermost x axis. Correlations for the secondary climate variable (T) were calculated independently of variance related to the primary variable (P). Significant correlations (P < 0.05) are shown by dark filled bars. Note the different scales of the correlation coefficient (y axes)
Drought–growth relationships
In general, dry conditions (negative SPEI values) were associated to low ring-width indices, but again, we observed different species-specific responses to drought (Fig. 4). Three species were very sensitive to changes in drought severity (P. arizonica, P. leiophylla and P. engelmannii) and two were less responsive (P. durangensis and P. lumholtzii). The three most sensitive species reacted to dry conditions as reflected by low SPEI values from January to July at scales from 2 to 16 months with P. engelmannii and P. arizonica particularly responding to shorter (2–10 months) and longer (14–16 months) time scales of cumulative water deficit. P. durangensis presented the most delayed, albeit weak, response to growth from August to September at long time scales (12–14 months).
Drought–growth associations calculated for the five Mexican pine species by relating the SPEI drought index to the residual ring-width chronologies. The SPEI was calculated at 1- to 28-month long scales (x axes) from the previous October to current December (y axes). The color scale shows Pearson correlation coefficients with significant (P < 0.05) values above the threshold r = |0.30|
ENSO and pine growth
All pine species excepting P. durangensis presented negative growth associations with high January and March–April SOI values in agreement with wet winter conditions during El Niño episodes which are characterized by negative SOI values (Fig. 5). Similarly, these four species responded positively to warm SSTs across the El Niño 3.4 region across the eastern tropical Pacific with the strongest responses being observed from January to March and particularly in three species (P. arizonica, P. leiophylla, and P. engelmannii). Regarding the PDO, P. arizonica and P. leiophylla presented the strongest positive associations to growth during the previous October and the current June, respectively.
Associations observed between the residual ring-width chronologies of the five Mexican species and selected indices related to the El Niño–Southern Oscillation: Southern Oscillation Index (SOI), sea-surface temperature anomalies in the equatorial Pacific (El Niño 3.4 region), and the Pacific Decadal Oscillation (PDO). The rest of explanations are as in Fig. 2
Considering the field correlations based on the grand mean chronology of the five pine species, the positive growth responses to cool and wet January conditions were mainly concentrated in NE and NW México, respectively (Fig. 6a, b). This corresponded to warm SSTs across the Mexican Pacific coast (e.g., Baja California, central México; Fig. 6c). Pine growth was positively associated with spring and summer (April to June) warm SSTs in the Californian Pacific waters (Fig. 6d).
Field correlations calculated by relating the mean of the five residual tree-ring width chronologies of pine species sampled in NW Mexico and January a mean maximum temperature, b precipitation, and c sea-surface temperatures (SST). The plot d shows the correlation with the average April–June SSTs across the Equatorial Pacific and Atlantic oceans. In all the cases, the field significance is lower than 10%. The asterisks indicate the approximate location of the study site. The last climatograph illustrates the distribution of monthly precipitation P (e) and temperature T (f) for the study area
Discussion
Here, we show how drought was related to decrease forest growth in five pine species inhabiting the Sierra Madre Occidental, NW México. Specifically, high maximum temperatures and low rainfall in January, hydroclimate conditions which are often associated in the study area (Bickford et al. 2011), were linked to narrow tree rings. As expected, cool and wet winters related to ENSO events (anomalous warm SSTs across the equatorial Pacific) usually coincided with enhanced growth of most species. However, we detected species-specific growth responses to climate and drought which greatly modulated how pines responded to climate and drought. These responses could be associated with diverse drought tolerances of coexisting pine species, but it should be also considered that temperatures are becoming warmer in the study area which could increase evapotranspiration rates and magnify drought stress.
Climatic drivers of pine growth
As expected growth of Mexican pine species was enhanced by cool and wet conditions during winter (December to February) and spring (April) (Figs. 2, 3). However, we found clear differences in growth sensitivity to climate and drought across pine species at different temporal scales (Figs. 2, 3). Cooler winter temperatures and elevated precipitation allow moisture to penetrate deep soil layers that are important for wood formation in deep-rooted tree species from dry areas (Fritts 2001). This is also observed in other conifers from northern México as Douglas fir (Pseudotsuga menziesii) whose growth was enhanced by wet conditions during the previous fall and winter and the current spring (González-Elizondo et al. 2005; Villanueva-Díaz et al. 2006). High winter rainfall combined with cool conditions ensures moisture availability in early spring, when NW Mexican pines typically start to grow (Villanueva-Díaz et al. 2006). A similar response has also been observed in drought-prone Mexican forests (Bickford et al. 2011; Pompa-García and Camarero 2015), and also in semi-arid Mediterranean Aleppo pine (Pinus halepensis) forests where soil moisture recharge was postulated as an explanation of these climate–growth associations (Pasho et al. 2012). Interestingly, Aleppo pine forests are characterized by showing high photosynthesis rates during late winter (Maseyk et al. 2008), when cool and wet conditions lead to low leaf vapour pressure deficit, whereas wood and needle growth mostly occur in spring (Camarero et al. 2010). The temporal decoupling between photosynthesis, wood formation, and needle growth could lead to different predictions on the effects of the forecasted warming and drying on productivity of drought-prone woodlands as Mexican pine forests. This decoupling and a better knowledge of xylogenesis could also explain why some species showing a low responsiveness to drought as P. durangensis responded more to spring than to the previous-winter precipitation (Fig. 2).
During the previous late fall and winter, minimum temperatures could enhance growth through warmer night conditions, i.e., higher minimum temperatures (Way and Oren 2010). The different temperature responses of trees imply that the stem contracts during the day, due to transpiration, and it expands and grows during the night when overnight hydration of woody tissues occurs (Vieira et al. 2013). Thus, warmer nights could encourage growth, but warmer maximum temperatures would lead to growth reduction during the day by increasing water loss through stomata, reducing sapwood water content, triggering stem shrinkage, or enhancing respiration (Fig. 2). Warmer winter conditions may also favor radial growth by triggering an earlier budburst (Huang et al. 2010). Contrastingly, during current spring and early summer (April, June), warm night and day conditions were associated with reduced growth due to increased evapotranspiration rates, and possibly depleted soil moisture reserves, affecting both more (P. arizonica, P. lumholtzii) and less drought- and temperature-sensitive species (P. durangensis). It is also notable that the least responsive P. durangensis also showed maximum responses to precipitation at short time scales (3–6 months) in a different month than the rest of pine species (Fig. 3) suggesting a different phenology of growth formation in this species. Finally, P. lumholtzii seems to be the species most sensitive to temperatures (Fig. 3), so it could be hypothesized that it would be the most negatively affected by warmer and drier conditions.
Drought influences on pine growth
The three most drought-responsive pine species (P. arizonica, P. leiophylla, and P. engelmannii) could be considered those most drought tolerant based also on their current distribution (Sánchez-González 2008). On the contrary, the two least drought-responsive pine species (P. durangensis and P. lumholtzii) could be considered those showing a lower drought tolerance. The three responsive species showed positive growth-SPEI associations from January to July at scales from 2 to 16 months (Fig. 4), but P. engelmannii responded more to short-term droughts (2–10 months), whilst P. arizonica responded more to long-term droughts (14–16 months). It could be hypothesized that species responding to longer droughts are more dependent on deeper soil moisture reserves than species responding to shorter droughts, but this should be tested in the field using, for instance, hydrogen isotopes of soil and xylem water and carbon isotopes of wood (Sarris et al. 2013). The higher growth sensitivity to climate variability of some species (e.g., P. engelmannii) as compared with others (e.g. P. lumholtzii) (Figs. 2, 3) was also observed in another study carried out in NW México (Bickford et al. 2011) and attributed to different drought tolerance of the evaluated pine species (Perry 1991). The most drought-responsive pine species usually inhabit drier habitats than those less responsive species (Martínez 1948). The lower responsiveness of some species (P. durangensis and P. lumholtzii) could also be explained by a decoupling of regional climate variability due to their presence in microhabitats characterized by wet and cool conditions, although this was not observed in the study site. However, we still lack adequate field and empirical data on adult trees (for instance regarding: phenology on needle and wood formation, photosynthesis, and water-use efficiency) to properly explain these contrasting growth responses to drought. Studies based on seedlings support our results and also emphasize the great intraspecific variability in the responses to drought of some of these species as P. leiophylla and P. engelmannii (Martínez et al. 2002; Prieto-Ruiz et al. 2004). Carbon and oxygen isotopes of tree-ring wood and needles could be used to infer long-term changes in water-use by these species (Gómez-Guerrero et al. 2013). Finally, it could be stated that warmer and drier conditions have a less negative effect on growth of species showing a low responsiveness to maximum temperatures and drought as P. lumholtzii or P. durangensis than on sensitive species as P. arizonica and P. engelmannii. Further studies should evaluate if this species-specific effect of drought on growth also affects other aspects of the species’ dynamics, including regeneration, mortality rates, and potential shifts, along the altitudinal gradients of the Sierra Madre Occidental.
Winter–spring temperature and precipitation were the main hydroclimatic factors controlling growth of the studied Mexican pine species, excepting the least drought-sensitive P. durangensis. We assume that this was not due to cross-dating difficulties in this species and suggest that the low EPS value of P. durangensis indicates that this species is less prone to climate variability (Table 2).
ENSO influences on climate variables affecting growth
The correlations found between growth and ENSO indices (Fig. 5) confirm that El Niño events associated to cool and wet conditions enhanced growth, whereas the La Niña events and dry conditions reduced growth (Fig. 1). This was also reported for other tree species in NW México (Therrell et al. 2002; Pompa-García and Némiga 2014) and for Pinus lagunae in the Baja California (see also the SST pattern of the Pacific Mexican coast in Fig. 6c) where tree growth was enhanced by very wet conditions linked to El Niño events as 1983, whereas La Niña events corresponded to below-normal precipitation years, such as those recorded in the late 1980s (Díaz et al. 2001). We also observed a growth increase after the 1983 El Niño which lasted several years, while wet conditions were wet even if the SSTs over the tropical Pacific dropped (Fig. 1). It is remarkable that wet conditions lasting for several years linked to warm SSTs in the “El Niño 3 + 4” region (e.g. 1983, 1991–1993) enhanced and stabilized growth, whilst dry conditions linked to cold SSTs reduced growth (e.g., 1970s and 1999–2001) but also increased year-to-year growth variability (Fig. 1). In other words, successive droughts could compromise the resilience capacity of the most sensitive pine species (e.g., P. arizonica). This species and P. leiophylla were the species whose ring-width chronologies were most tightly related to some of the ENSO indices (January and March–April SOI, January to April El Niño 3.4, October, and June PDO; Fig. 5), confirming the links between this ocean–atmosphere circulation pattern, winter–spring hydroclimate, and productivity in the most drought-sensitive species. Geographically, pine growth was negatively associated to January precipitation across an ample region, including N México and the SW USA (Fig. 6a), but positively associated with January precipitation mainly in the focal area of NW México (Fig. 6b) and California (Fig. 6d). We could not find relationships of pine growth with SSTs over the Equatorial pacific implying that the local climate variables driving growth of the studied species did not respond to such large-scale ENSO patterns. It is also possible that tree growth mainly responds to very wet (El Niño events) or very dry (La Niña events) climate conditions corresponding to extreme ENSO values (Fig. 1). Overall, these patterns suggest similar growth to responses to hydroclimate across these North American forests subjected to frequently dry springs which lead to abundant rainfall in July caused by the North American monsoon (Adams and Comrie 1997).
It could be hypothesized that current climate warming would amplify drought stress or alter the ENSO-hydroclimate connections intensifying aridification and reducing forest productivity making some tree species more vulnerable to other biotic stressors as bark beetles (McDowell et al. 2008). Growth decline could portend dieback events and rises in mortality rates similar to reports from other drought-prone pine forests in the SW USA (Breshears et al. 2005) and the Mediterranean Basin (Camarero et al. 2015). Here, we show that a characterization of climate–growth associations aids to infer the tolerance to drought in coexisting tree species and it could be used as a proxy of growth sensitivity to water shortage.
To conclude, this study is among the few ones characterizing the climatic drivers of pine growth in a hotspot of pine diversity, the Mexican Sierra Madre Occidental. We found different growth responsiveness to drought among coexisting pine species, and we suggest that these diverse responses could correspond to different tolerances to dry conditions. We found that wet and cool winter and spring conditions, linked to warm SSTs over the equatorial Pacific, were the main hydroclimate factors enhancing growth of pine species. However, among the five studied species, some presented a high responsiveness to drought (P. arizonica, P. leiophylla, and P. engelmannii), whilst others were less responsive (P. lumholtzii and P. durangensis). The presented findings could be used by managers and conservation agencies as it provides information on the potential impacts of climate warming and drought on this understudied and very diverse region of NW México.
Author contribution statement
MGC and MPG sampled the trees. MGC measured the samples. JJC lead the data analyses. All authors contributed to data analyses and writing.
References
Adams D, Comrie A (1997) The North American Monsoon. Am Meteorol Soc 78:2197–2213
Andrade ER, Sellers WD (1988) El Niño and its effect on precipitation in Arizona and western New Mexico. J Climatol 8:403–410
Barton AM (2002) Intense wildfire in southeastern Arizona: transformation of a Madrean oak–pine forest to oak woodland. For Ecol Manag 165:205–212
Barton AM, Swetnam TW, Baisan Ch (2001) Arizona pine (Pinus arizonica) stand dynamics: local and regional factors in a fire-prone madrean gallery forest of Southeast Arizona, USA. Landsc Ecol 16:351–369
Bickford IN, Fulé PZ, Kolb TE (2011) Growth sensitivity to drought of co-occurring Pinus spp. along an elevation gradient in northern Mexico. West N Am Nat 71:338–348
Breshears DD, Cobb NS, Rich PM, Price KP, Allen CD, Balice RG, Romme WH, Kastens JH, Floyd ML, Belnap J, Anderson JJ, Myers OB, Meyer CW (2005) Regional vegetation die-off in response to global-change-type drought. PNAS USA 102:15144–15148
Camarero JJ, Olano JM, Parras A (2010) Plastic bimodal xylogenesis in conifers from continental Mediterranean climates. New Phytol 185:471–480
Camarero JJ, Gazol A, Sangüesa-Barreda G, Oliva J, Vicente-Serrano SM (2015) To die or not to die: early-warning signals of dieback in response to a severe drought. J Ecol 103:44–57
Carvajal S, McVaugh R (1992) Pinus L., In: Flora Novo-Galiciana 17 (ed) R. McVaugh. Univ. Michigan Herbarium, Ann Arbor, pp 32–100
Cleaveland MK, Stahle DW, Therrell MD, Villanueva-Díaz J, Burns BT (2003) Tree-ring reconstructed winter precipitation and tropical teleconnections in Durango, Mexico. Clim Change 59:369–388
Cook ER, Holmes RL (1986) Users manual for program ARSTAN. In: Holmes RL, Adams RK, Fritts HC (eds) Tree-ring chronologies of western North America: California, eastern Oregon and northern Great Basin. Laboratory of Tree-Ring Research, Tucso, pp 50–65
Cook BI, Seager R (2013) The response of the North American Monsoon to increased greenhouse gas forcing. J Geophys Res Atmos 118:1690–1699
Díaz SC, Touchan R, Swetnam TW (2001) A tree-ring reconstruction of past precipitation for Baja California Sur, Mexico. Int J Climatol 21:1007–1019
Earle JC (2007) Gymnosperm database. http://www.conifers.org/. Accessed 17 May 2016
Farjon A (2010) A Handbook of the World’s Conifers. Brill, The Netherlands, p 1112
Farjon A, Styles BT (1997) Pinus (Pinaceae). Flora Neotropica Monograph 75. The New York Botanical Garden, USA, p 291
Figueroa-Rangel BL, Willis KJ, Olvera Vargas M (2008) 4200 years of pine-dominated upland forest dynamics in west-central Mexico: human or natural legacy? Ecology 89:1893–1907
Fritts HC (2001) Tree rings and climate. Blackburn Press, USA, p 567
Fulé PZ, Ramos-Gómez MA, Cortés Montaño C, Miller AM (2011) Fire regime in a Mexican forest under indigenous resource management. Ecol Appl 21:764–775
Gernandt DS, Pérez de la Rosa JA (2014) Biodiversity of Pinophyta (conifers) in México. Rev Mex Biodivers 85:126–133
Gómez-Guerrero A, Silva LCR, Barrera-Reyes M, Kishchuk B, Velázquez-Martínez A, Martínez-Trinidad T, Plascencia-Escalante FO, Horwath WR (2013) Growth decline and divergent tree ring isotopic composition (δ13C and δ18O) contradict predictions of CO2 stimulation in high altitudinal forests. Global Change Biol 19:1748–1758
González-Elizondo M, Jurado E, Navar J, Gonzalez-Elizondo MS, Villanueva J, Aguirre O, Jiménez J (2005) Tree-rings and climate relationships for Douglas-fir chronologies from the Sierra Madre Occidental, Mexico: a 1681–2001 rain reconstruction. Forest Ecol Manag 213:39–53
González-Elizondo MS, González-Elizondo M, Tena-Flores JA, Ruacho-González L, López-Enríquez IL (2012) Vegetación de la Sierra Madre Occidental, México: Una síntesis. Act Bot Mexico 100:351–403
Grantz K, Rajagopalan B, Clark M, Zagona E (2007) Seasonal shifts in the North American monsoon. J Clim 20:1923–1935
Harris I, Jones PD, Osborn TJ, Lister DH (2014) Updated high-resolution grids of monthly climatic observations—the CRU TS3.10 Dataset. Int J Climatol 34:623–642
Holmes RL (1983) Computer-assisted quality control in treering dating and measurement. Tree Ring Bull 43:69–78
Huang J, Tardif JC, Bergeron Y, Denneler B, Berninger F, Girardin MP (2010) Radial growth response of four dominant boreal tree species to climate along a latitudinal gradient in the eastern Canadian boreal forest. Global Change Biol 16:711–731
Instituto Nacional de Estadística y Geografía-INEGI (2003) Conjunto de datos vectoriales de la carta de climas escala 1:1000000. http://www3.inegi.org.mx/sistemas/biblioteca/ficha.aspx?upc=702825267568. Accessed 25 March 2016
Kiladis GN, Díaz HF (1993) ENSO and precipitation variability over Mexico during the last 90 years. In: Redmon KT, Tharp VL (eds) Proceedings of the north annual Paclim climate (PACLIM) workshop. Asilomar, USA, California Department of Water Resources, pp 63–70
Liverman DM (1999) Vulnerability and adaptation to drought in Mexico. Nat Res J 29:99–115
Magaña V, Perez JL, Vazquez JL, Carrizosa E, Perez J (1999) El Niño y el clima. In: Magaña V (ed) Los impactos de El Niño en México. CONACYT, México, pp 23–68
Martínez M (1948) Los Pinos Mexicanos, 2ª edn. Universidad Autónoma de México, México
Martínez J, Fernández A (eds) (2004) Cambio climático: una visión desde México. Instituto Nacional de Ecología, México D.F.
Martínez TT, Vargas HJJ, Muñoz OA, López UJ (2002) Respuesta al déficit hídrico de Pinus leiophylla: Consumo de agua y crecimiento en plántulas de diferentes poblaciones. Agrociencia 36:365–376
Maseyk KS, Lin T, Rotenberg E, Grünzweig JM, Schwartz A, Yakir D (2008) Physiology–phenology interactions in a productive semi-arid pine forest. New Phytol 178:603–616
McDowell N, Pockman WT, Allen CD, Breshears DD, Cobb N, Kolb T, Plaut J, Sperry J, West A, Williams DG, Yepez EA (2008) Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? New Phytol 178:719–739
Meko DM, Touchan R, Anchukaitis KJ (2011) Seascorr: a MATLAB program for identifying the seasonal climate signal in an annual tree-ring time series. Comput Geosci 34:1234–1241
Pasho E, Camarero JJ, Vicente-Serrano SM (2012) Climatic impacts and drought control of radial growth and seasonal wood formation in Pinus halepensis. Trees 26:1875–1886
Perry JP (1991) The pines of Mexico and Central America. Timber Press, Portland
Pompa-García M, Camarero JJ (2015) Reconstructing evaporation from pine tree rings in northern Mexico. Tree Ring Res 71:95–105
Pompa-García M, Némiga XA (2014) ENSO index teleconnection with seasonal precipitation in a temperate ecosystem of northern Mexico. Atmósfera 28:43–50
Prieto-Ruiz JA, Cornejo Oviedo EH, Domínguez Calleros PA, Návar Chaidez JJ, Marmolejo Moncivais JG, Jiménez Pérez J (2004) Estrés hídrico en Pinus engelmannii Carr., producido en vivero. Inv Agr Sist Rec For 13:443–451
Rayner NA, Parker DE, Horton EB, Folland CK, Alexander LV, Rowell DP, Kent EC, Kaplan A (2003) Global analyses of sea surface temperature, sea ice, and night marine air temperature since the late nineteenth century. J Geophys Res 108:4407
Reynolds RW, Smith TM, Liu C, Chelton DB, Casey KS, Schlax MG (2007) Daily high-resolution blended analyses for sea surface temperature. J Clim 20:5473–5496
Ropelewski CF, Jones PD (1987) An extension of the Tahiti-Darwin Southern Oscillation Index. Mon Weather Rev 115:2161–2165
Ruelas Monjardín LC, Dávalos Sotelo R (1999) La industria forestal en el estado de Chihuahua. Madera y Bosques 5:79–91
Rzedowski J (2006) Vegetación de México. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México
Sánchez-González A (2008) Una visión actual de la diversidad y distribución de los pinos de México. Madera y Bosques 14:107–120
Sarris D, Siegwolf R, Körner Ch (2013) Inter- and intra-annual stable carbon and oxygen isotope signals in response to drought in Mediterranean pines. Agric Forest Meteorol 168:59–68
Schulman E (1944) Dendrochronology in Mexico 1. Tree Ring Bull 10:18–24
Seager R, Ting M, Davis M, Cane M, Naik N, Nakamura J, Li C, Cook ER, Stahle D (2009) Mexican drought: an observational modeling and tree ring study of variability and climate change. Atmósfera 22:1–31
Stahle DW, Cleaveland MK (1993) Southern Oscillation extremes reconstructed from tree rings of the Sierra Madre Occidental and southern great plains. J Clim 6:129–140
Stahle DW, D’Arrigo RD, Krusic PJ, Cleaveland MK, Cook ER, Allan RJ, Cole JE, Dunbar RB, Therrell MD, Gay DA, Moore MD, Stokes MA, Burns BT, Villanueva-Diaz J, Thompson LG (1998) Experimental dendroclimatic reconstruction of the Southern Oscillation. Bull Am Meteorol Soc 79:2137–2152
Stahle DW, Cook ER, Cleaveland MK, Therrell MD, Meko DM, Grissino Mayer HD, Watson E, Luckman BH (2000) Tree-ring data document 16th century megadrought over North America. Eos 81:121
Therrell M, Stahle D, Cleaveland M, Villanueva J (2002) Warm season tree growth and precipitation over Mexico. J Geophys Res 107:14–24
Trenberth KE (1997) The definition of el Niño. Bull Am Meteorol Soc 78:2771–2777
Van Oldenborgh GJ, Burgers G (2005) Searching for decadal variations in ENSO precipitation teleconnections. Geophys Res Lett 32:L15701
Vicente-Serrano SM, Beguería S, López-Moreno JI (2010) A multiscalar drought index sensitive to global warming: the Standardized Precipitation Evapotranspiration Index. J Clim 23:1696–1718
Vieira J, Rossi S, Campelo F, Freitas H, Nabais C (2013) Seasonal and daily cycles of stem radial variation of Pinus pinaster in a drought-prone environment. Agric Forest Meteorol 180:173–181
Villanueva-Díaz D, Stahle DW, Cleaveland MK, Therrell MD (2000) Estado actual de la dendrocronología en México. Ciencia Forestal 25:5–36
Villanueva-Díaz J, Cerano-Paredes J, Stahle DW, Luckman BH, Therrell MD, Cleaveland MK (2006) The climatic response of tree-ring chronologies in the Sierra Madre Occidental, Mexico. In: Symposium on climate change: organizing the science in the American Cordillera (Mendoza, Argentina), pp 91–92
Villanueva-Díaz J, Stahle DW, Luckman B, Cerano-Paredes J, Therrell MD, Cleaveland MK, Cornejo-Oviedo E (2007) Winter-spring precipitation reconstructions from tree rings for northeast México. Clim Change 83:117–131
Way DA, Oren R (2010) Differential responses to changes in growth temperature between trees from different functional groups and biomes: a review and synthesis of data. Tree Physiol 30:669–688
Wigley TML, Briffa KR, Jones PD (1984) On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J Appl Meteorol 23:201–213
Williams AP, Allen CD, Macalady AK, Griffin D, Woodhouse CA, Meko DM, Swetnam TW, Rauscher SA, Seager R, Grissino-Mayer HD, Dean JS, Cook ER, Gangodagamage C, Cai M, McDowell NG (2013) Temperature as a potent driver of regional forest drought stress and tree mortality. Nat Clim Change 3:292–297
Yeaton RI (1982) The altitudinal distribution of the genus Pinus in the western United States and Mexico. Bol Soc Bot México 42:55–71
Zhang Y, Wallace JM, Battisti DS (1997) ENSO-like interdecadal variability: 1900–93. J Clim 10:1004–1020
Acknowledgements
We acknowledge the financial support given by Consejo Nacional de Ciencia y Tecnología (CONACYT) through project 222522. We thank local forestry landowners for facilitating gathering field data. We also thank R. Sánchez-Salguero for his help with data analyses and the editors and two anonymous reviewers for their valuable comments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by E. Liang.
Rights and permissions
About this article
Cite this article
González-Cásares, M., Pompa-García, M. & Camarero, J.J. Differences in climate–growth relationship indicate diverse drought tolerances among five pine species coexisting in Northwestern Mexico. Trees 31, 531–544 (2017). https://doi.org/10.1007/s00468-016-1488-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-016-1488-0