Abstract
Key message
Photosynthetic capacity of yew needles depends primarily on light environment, with little evidence that developmental stage is a determinant photosynthetic performance. However, needle morphology is mostly determined by plant size.
Abstract
Populations of yew Taxus baccata are often characterized by a lack of natural regeneration and stands of this species have been afforded priority conservation status within the European Union. Light availability is known to be a key factor influencing recruitment dynamics in yew. To explore potential size-related responses to light, we investigated the distribution, photosynthetic efficiency and needle morphological traits in yew across a gradient of light availability at Atlantic oak woodland in southwest Ireland. Young plants were more common in shaded areas whereas saplings and juvenile yews were distributed in areas of higher light availability. Photosynthetic capacity of yew needles was found to depend primarily on light environment, with little evidence that the developmental stage is a determinant of photosynthetic performance. Photoinhibition was evident in needles sampled from high light environments across all demographic size classes. However, larger plants also showed particularly strong needle morphological responses (needle length, width, thickness, and specific leaf area) to light levels, which were mostly lacking in seedlings. We conclude that there is a dual control of yew light responses; the efficiency of the photosynthetic light reactions is largely linked to the light environment in which the plants grew, while needle morphology is mostly determined by the size of the plants. The findings of this study highlight the importance of considering all life-history stages and multiple traits when evaluating species response to light availability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Light controls plant growth and development by facilitating photosynthesis and through a range of photoreceptor-mediated photomorphogenic processes. The quantity and quality of light are key factors in determining growth and survival in plant communities (Canham et al. 1990; Messier et al. 1999) and interspecific differences in shade tolerance are recognized as fundamental drivers of succession (Kobe et al. 1995; Lusk 2004; Lusk and Jorgensen 2013). While a shortage of light can compromise growth and survival, excessive irradiance can also negatively affect plant performance by causing photoinhibitory stress (Jansen et al. 1999). As a result, in forested landscapes an increase in canopy openness may cause severe stress to seedlings and saplings due to exposure to excessive radiation (Wayne and Bazzaz 1993; Mitamura et al. 2009).
Much of the research on growth and survival of plants subject to varying degrees of insolation has focused on shade tolerance of young seedlings (Niinemets and Valladares 2006). Some authors have, however, highlighted the need to adopt a life cycle view of the responses of plants to their light environments (Valladares and Niinemets 2008; Kunstler et al. 2009). Tree species may be subject to major changes in light quantity and quality during their lifetime, ranging from low light understory or gap conditions as seedlings, to fully exposed canopy conditions as adults. Changes in physiological and morphological traits of the photosynthetic apparatus, leaf and canopy structure of tree species will occur during development (Augspurger and Bartlett 2003). Furthermore, as plant size increases, a decreasing ratio of photosynthetic to non-photosynthetic biomass, and associated higher maintenance costs, may lead to a reduction in shade tolerance (Givnish 1988; Kunstler et al. 2009).
Yew Taxus baccata L. is a dioecious gymnosperm, occurring throughout Europe with scattered populations extending to North Africa and the Middle East (Thomas and Polwart 2003). Yew has declined over recent millennia and populations are now isolated or locally extinct in many areas (Dubreuil et al. 2008; Myking et al. 2009). Due to concern for the conservation status of natural populations of this species (Dhar et al. 2006; Iszkulo et al. 2009), several conservation and restoration programmes have been established (Dhar et al. 2007; Piovesan et al. 2009). The absence of natural regeneration has been attributed to a range of factors, including overgrazing by ungulates, seed predation, genetic isolation and intraspecific competition (Hilfiker et al. 2004; Perrin et al. 2006; Farris and Filigheddu 2008; Piovesan et al. 2009; Devaney et al. 2013). Numerous authors have also reported that light availability is a key factor influencing recruitment dynamics in yew (Svenning and Magård 1999; Thomas and Polwart 2003; Iszkulo et al. 2007).
Yew is known to be one of the most shade-tolerant tree species of the European flora (Thomas and Polwart 2003; Iszkulo and Boratynski 2006). However, this perceived tolerance to low light conditions does not necessarily infer a requirement for shade and indeed, conflicting reports exist on the role of light in recruitment in yew. For example, Perrin and Mitchell (2013) demonstrated that under ex situ experimental light conditions, no mortality of yew saplings was attributable to heavy shade (<3 % daylight), yet Iszkulo (2010) reported high mortality of young yew seedlings under similar low light conditions (<2 % daylight). The absence of young seedlings from many areas is often credited to the dense shade cast by mature Fagus spp. and yew canopies (Watt 1926; Krol 1978; Rodwell 1991). Furthermore, increased canopy closure by shade-causing species such as Fagus sylvatica may lead to a reduction in strobilus production and recruitment to the sapling stage in yew (Svenning and Magård 1999). Conversely, recent evidence suggests that seedlings of yew are susceptible to photoinhibition when exposed to full sunlight (Robakowski and Wyka 2009). Nevertheless, older yew saplings and adult trees are often located in areas of high light availability such as limestone pavement and cliff faces (Jackson and Sheldon 1949; Thomas and Polwart 2003; Myking et al. 2009). These findings suggest that the response of yew to light depends on the age and/or developmental stage. A number of studies have identified similar size-related light requirement transitions in other tree species during their development (Poorter et al. 2005; Coopman et al. 2008). With few notable exceptions (Iszkulo and Boratynski 2006) research to date has yet to assess possible size-related changes in the response of yew to light availability.
The objective of this study was to investigate potential size-related responses to light availability in yew. Specifically, we tested the following hypotheses: (1) the natural distribution of yew in consecutive developmental stages is differentially related to light availability (2) the relationship between activity of photosystem II in yew and light availability differs between developmental stages and (3) the relationship between needle morphological traits and light availability differs between developmental stages.
Materials and methods
Study site
The study was carried out at the Glengarriff Wood Nature Reserve (GWNR; 51° 45′ N, 9° 34′ W) in southwest Ireland. The GWNR is a 301 ha area situated in primarily forested hilly terrain with many rocky outcrops. In most areas of the reserve the underlying sandstone is covered by acidic peaty soil with some podsolization (Little 1994). Due to its coastal Atlantic location, the climate is mild and oceanic. The predominant habitat at the GWNR is semi-natural sessile oak woodland, of the association Blechno-Quercetum petraeae, an association listed in Annex I of the EU Habitats Directive (Council Directive 92/43/EEC). The principal canopy tree is Quercus petraea with Ilex aquifolium, Betula pendula, Sorbus aucuparia, Salix spp. and Arbutus unedo forming well-developed understory and shrubs layers. Frequent plants in the field layer include Calluna vulgaris, Vaccinium myrtillus and Pteridium aquilinum. Studies by (O’Neill 2003) and McKenna (2003) have noted a high incidence of seedlings and saplings of yew, generally located in the vicinity of mature adult conspecifics, which occur as infrequent understory trees.
The relationship between yew size and light availability
As part of a wider study (Devaney 2013) the spatial distribution and height of all yew individuals within a 1 ha study area in the GWNR were recorded. For the current study, a stratified random sample (n = 158) of individuals were selected across four demographic size classes based on plant height: seedlings 0–0.20 m; young saplings 0.21–0.50 m; saplings 0.51–2 m; and juveniles >2 –6 m. To examine the relationship between the occurrence of yew individuals within these size classes and light availability, the percentage of full daylight available at the top of each plant was recorded. Light availability was determined by measuring photosynthetic photon flux density (PPFD; µmol m−2 s−1) above each plant, on a uniformly overcast day using a SKYE 200 Photosynthetically Active Radiation meter, whilst obtaining a simultaneous measurement in an open area. PPFD values for open areas ranged from 94–420 µmol m−2 s−1 during sampling. Relative full daylight availability was determined by calculating the ratio of light intensity above each plant to the light intensity recorded in the open area.
Chlorophyll-a fluorescence of yew needles in response to light availability
The same stratified random sample of plants was used to investigate the activity of photosystem II of photosynthesis in needles of yew growing in a range of light environments. Plants were categorised according to the light environment in which they grew: low light (LL), <5 % full daylight; medium light (ML), 5–20 % full daylight, and; high light (HL), >20 % full daylight. Within each of these light environments, plants were again selected from the four demographic size classes (seedlings 0–0.20 m; young saplings 0.21–0.50 m; saplings 0.51–2 m; and juveniles >2–6 m). Where sufficient replicates of plants in each treatment were not located in the study area, supplementary individuals were sampled from elsewhere within the GWNR. For example, low numbers of yew seedlings and young saplings were recorded growing in high light under natural conditions within the 1 ha study area. Therefore, supplementary plants were sampled from artificially open areas where canopy cover had been removed as part of forest management operations (>3 years prior to sampling). In total, 227 needles were sampled for analyses, with 15–20 replicates per treatment. In all cases, current year needles were collected from the uppermost shoot of each plant at the end of the growing season (late September/early October 2010). Mean maximum and minimum temperatures for the region in September 2010 were 18.3 and 11.3 °C, respectively, similar to 10 year averages for the period 2001–2010 (18.6–10.27 °C). For the 10 day period prior to sampling, there was a total of 37 sun hours (10 year average; 47.3 h) and 102.29 MJ m2 global solar radiation (10 year average; 106.09 MJ m2) (Met Eireann, Unpublished data). Immediately after removal from the plant, needles were stored in Petri dishes with wetted filter paper, covered with aluminium foil, and analysed within four hours. Chlorophyll-a fluorescence was measured using a modulated, imaging fluorometer equipped with ImagingWin software (IMAG-MAX/L with blue measuring light, PAM, Effeltrich, Germany). Needles were dark-adapted for 2–4 h (including transport time) prior to measurement and then placed on damp filter paper on the fluorometer stage. A weak measuring light (<1 µmol m−2 s−1) was used to determine the minimum fluorescence obtained in the dark-adapted state (Fo). Maximum fluorescence (Fm) was determined for the dark-adapted state by applying a saturating pulse of white light. Maximum quantum efficiency of photosystem II was calculated as (Fm−Fo)/Fm = (Fv/Fm), where Fv is variable fluorescence. The effective quantum yield of PSII (Φ PSII), photochemical quenching (qP) and electron transport rate (ETR) were also measured. Needles were illuminated using the in-built LED with up to eight levels of actinic light (1–800 µmol m−2 s−1), in order of increasing intensity. Needles were exposed for 20 s to each intensity, after which needles were exposed to a flash of saturating light. Analysis of acclimation kinetics showed that slow changes in chlorophyll fluorescence parameters occur over time-spans of many minutes. The 20 s acclimation period captures the fast initial change in these parameters. At each level of actinic light, quantum yield of PSII was calculated as Φ PSII = (Fm′−Fs)/Fm′) where Fs is steady state fluorescence (Genty et al. 1989). Photochemical quenching (qP) was calculated as qP = (Fm′−Fs)/(Fm′–Fo′) where Fo′ was calculated as Fo′ = Fo/(Fv/Fm + Fo/Fm′) following Oxborough and Baker (1997). The apparent rate of electron transport (ETR) was calculated as ETR = Φ PSII × 0.5 × 0.84 × PPFD (Maxwell and Johnson 2000).
Morphological differentiation of needles in relation to light availability
Current year needles used in the measurement of chlorophyll-a fluorescence parameters were used to investigate morphological characteristics. Needle thickness was measured using micro callipers. Needles were digitally scanned using a Hewlett Packard ScanJet 4300C and mean needle length, width and area was determined using ImageJ software. Dry weights of the needles were obtained after oven drying for 72 h at 50 °C and specific leaf area (SLA) was calculated as needle area: needle dry weight (cm2 g−1).
Statistical analysis
The relationship between the occurrence of yew in different size classes and light availability at the GWNR was assessed using one-way ANOVA with post hoc Bonferroni tests. Two-way ANOVA was used to test for significant differences between chlorophyll-a fluorescence parameters and needle morphological traits with respect to light environment (LL, ML, HL) and plant size (seedlings, young saplings, saplings, juveniles). Following Wyka et al. (2008), a single cardinal value for each light response curve was selected for statistical comparison of chlorophyll-a fluorescence parameters. For Φ PSII and qP values were measured using an actinic light of 281 µmol m−2 s−1. The maximum value for ETR was used (ETRmax). Significant results were tested for pair-wise comparisons using Bonferroni post hoc tests. Prior to performing ANOVAs, variables were tested for normality and homogeneity of variances using Kolmogorov–Smirnov and Levene’s tests, with transformations applied wherever necessary. All data were analysed using SPSS (PASW® Statistics 17) software package. Figures were generated using GraphPad Prizm software.
Results
The relationship between yew size and light availability
The relationship between the distribution of yew individuals in different size classes and light availability at the GWNR was assessed. 158 plants were sampled in total ranging in height from 0.07 m to 6 m. In general, seedlings and young saplings were associated with low to medium light availability, whereas saplings and juvenile trees were commonly associated with higher light availability. The mean percentage of full daylight available to seedlings and young saplings was 7.9 and 5.6 % respectively (Fig. 1). In comparison, mean percentage of full daylight available to saplings and juveniles was 27.9 and 50.6 % respectively (Fig. 1). Overall, mean light availability was significantly higher for yew saplings and juveniles in comparison to seedlings and young saplings (one-way ANOVA, F = 108.424, p < 0.001, Fig. 1).
The relationship between percentage availability of full daylight and the occurrence of yew in four demographic size classes; seedlings (0–0.20 m), young saplings (0.21–0.50 m), saplings (0.51–2 m) and juveniles (>2–6 m) in a 1 ha study area at the GWNR. Mean light availability at the top of plants was significantly higher for yew saplings and juveniles in comparison to seedlings and young saplings (one-way ANOVA, F = 108.424, p < 0.001). The box plots show the interquartile range (middle 50 % of data), with the median marked by a line, mean marked by plus, and the whiskers representing the full range of the data
Mean ± 1 SE maximum quantum yield of fluorescence (Fv/Fm) for first year yew needles from four demographic size classes; seedlings (0–0.20 m), young saplings (0.21–0.50 m), saplings (0.51–2 m) and juveniles (>2–6 m). Needles were sampled from plants growing in three light environments; LL- <5 % full daylight; ML–5–20 % full daylight; HL- >20 % full daylight. Two-way ANOVA was used to assess differences within categories (Table 2)
Chlorophyll-a fluorescence of yew needles in response to differing light availability
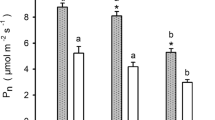
Light environment had a clear effect on measured chlorophyll-a fluorescence parameters of first year needles with values from HL environments being significantly different from other treatments (Table 1). Maximum quantum efficiency of photosystem II (Fv/Fm) was significantly reduced in yew needles growing in HL compared to those in LL and ML environments (Fig. 2; Table 1). Although the magnitude of Fv/Fm decrease in HL was less for juvenile individuals, size and interaction effects were not significant (Fig. 2; Table 1). High light environment had a significantly positive effect on the effective quantum yield of PSII (Φ PSII(281)) (Fig. 3; Table 1). Photochemical quenching (qP (281)) was significantly higher for both HL and ML needles in comparison to LL needles. On average, photochemical quenching (qP (281)) was found to be significantly greater for saplings in comparison to other size categories. A significant interaction between light environment and size was also recorded for qP (281) with differences between HL and LL values being greater for seedlings and young saplings compared to observed differences between HL and LL values for saplings and juveniles (Fig. 3; Table 1). For electron transport rate, ETRmax was significantly higher in HL needles compared to ML and LL needles.
Responses of chlorophyll-a fluorescence parameters in first year needles from three light environments (HL–dashed line, ML–Grey line, LL–Black line) and four size classes (seedlings, young saplings, saplings, juveniles). Points represent mean values (±1 SE) from up to eight levels of actinic light (1–800 µmol m−2 s−1). Statistical comparisons for Φ PSII and qP were conducted at PPFD = 281 µmol m−2 s−1 and the maximum values for ETR (ETRmax) were compared to assess differences within size class and light environment (Table 2)
Morphological differentiation of needles in relation to light availability
Univariate analysis revealed significant differences and interactions in macroscopic needle morphological traits with respect to light environment and size. Significant differences between size classes were observed for all measured needle morphological traits. In general, needles were longer, wider, and consequently, of greater surface area in larger plant size classes (saplings and juveniles). Conversely, SLA was significantly greater in seedlings with respect to other size classes (Tables 1, 2). Needle morphology was also affected by light environment, with LL and ML needles being significantly wider and having a greater SLA than HL needles. For needle width, a weak interaction was found with respect to light environment and size class. Needles sampled from LL and ML had significantly greater surface area than HL needles. Light environment and plant size had a marked effect on needle thickness. Plants from HL and ML exhibited greater needle thickness than plants from LL environments (Tables 1, 2). A strong interaction between light environment and size occurred for needle thickness. Thickness was greater for HL and ML plants in sapling and juvenile size classes. Conversely, for seedlings, needle thickness was greatest in LL plants and lowest in HL plants.
Discussion
Light is a primary determinant of plant growth and survival (Walters and Reich 1996). However, due to the heterogeneous distribution of light in the environment many species will be exposed to a range of light intensities over the course of their life cycle (Valladares and Niinemets 2008). In forests, tree seedlings are more likely to be growing in a shaded position, whilst mature individuals of many species will be exposed to the full intensity and spectrum of solar radiation. Thus, to understand plant light responses, a life-cycle view is required (Valladares and Niinemets 2008; Kunstler et al. 2009). Here, we explored the relationship between light availability and the distribution and growth of yew Taxus baccata L.
The relationship between yew size and light availability
At the GWNR, a large proportion of yew seedlings (0–0.20 m) and young saplings (0.21–0.50 m) were found under relatively low light availability- on average less than 7 % of full sunlight. In contrast, saplings and juveniles were found in more illuminated conditions, where the light availability was often more than 50 % of full sunlight. This observation may reflect the dispersal pattern of yew seeds. Under natural conditions yew seeds are less likely to be deposited in open areas due to their dispersal mechanism (Martínez et al. 2008). Frugivorous birds (the primary dispersers of yew seeds) are attracted to perches on canopy trees and shrub vegetation and consequently seed deposition has been shown to be much greater beneath canopied areas than in open habitats (Smith 1980; Garcia et al. 2000, 2005; Farris and Filigheddu 2008). As yew height increases, it may grow above ground and understory shading vegetation and be gradually exposed to increased light availability. A predictable vertical gradient of diminishing light availability is well-established in forest habitats with light being intercepted by consecutive leaf layers within the forest profile (Poorter et al. 2005; Thomas and Packham 2007). Consequently, in undisturbed conditions, tree seedlings will often be subject to greater light availability as they increase in height (Poorter et al. 2005). In our study, the reduced number of sapling and juvenile yews in shaded conditions suggests that light availability is a limiting factor in survival and recruitment of yew to later demographic stages. Studies by Iszkulo and Boratynski (2004, 2006) have indicated that despite the presence of yew seedlings in deep shade (<2 % full daylight), survival to later stages is stimulated by more open canopy conditions. Similar transitions of shaded seedlings to higher light areas in subsequent demographic stages, as seen in yew (Fig. 1) were demonstrated for a range of other tree species, such as Nothofagus nitida (Coopman et al. 2008) and Aristotelia chilensis (Lusk et al. 2008).
Size-dependent photosynthetic shade adaptation of yew needles
Given that yew seedlings are typically exposed to low light intensities, we asked whether this developmental stage is inherently photosynthetically shade-adapted. The question of whether an ontogenetic component of shade acclimation in yew exists has previously been posed by Wyka et al. (2008). To address this, we analysed photosynthetic parameters of seedlings, saplings and juveniles that grew in low, medium or high light environments.
In our study, it was found that the photosynthetic capacity of yew needles depends primarily on the light environment where the plant grew (Table 1). In general, seedlings growing in a particular light environment were photosynthetically indistinguishable from juveniles in the same light environment. There is little evidence that the developmental stage is an inherent determinant of photosynthetic performance, with the possible exception of a size-dependent effect on qP. Thus, based on a lack of significant interactions between light environmental and plant size (Table 1), yew plants acclimate to the light environment in which they occur, regardless of developmental stage. This finding is in contrast to other studies (Poorter et al. 2005; Coopman et al. 2008) that have indicated that the developmental stage itself is a determinant of photosynthetic performance. However, the degree to which photosynthetic capacity is mediated by development stage is likely to be species-specific with Poorter et al. (2005) recording all of the nine of their hypothesized height-light trajectories amongst 47 tropical tree species.
Interestingly, all yew plants displayed a decrease in the maximal efficiency of photosystem II (Fv/Fm) when growing in high light environments (Fig. 2). Such photoinhibition is caused when plants are subject to excess light beyond the photosynthetic capacity, leading to oxidative photodamage to photosystem II, and in some cases even a reduction in plant health (Mitamura et al. 2009). Conifers can be susceptible to photoinhibition, particularly when canopy openness is abruptly increased, or when relatively high light intensities in late winter are accompanied by low temperatures that impede biochemical reactions (Ball et al. 1991; Oquist and Huner 1991; Robakowski and Wyka 2009). In general, photoinhibitory stress can be observed when plants are exposed to moderate to high light intensities in combination with drought conditions, low nutrient levels, low temperatures, or various other stressors (Valladares et al. 2005; Takahashi and Murata 2008). For this study, PPFD (µmol m−2 s−1) above plants was not recorded on the day immediately before sampling. However, given that global solar radiation values and sunlight hours for the 10 day period prior to sampling (102.29 MJ m2 and 37 h, respectively) were both lower than 10 year averages for the same period (106.09 MJ m2 and 47.3 respectively), it is unlikely that climate conditions immediately prior to sampling were responsible for the observed potential photoinhibition in high light plants. For the current study, the question is raised whether the observed photoinhibitory effect (Fig. 2) directly reflects continued exposure to excess light, or rather a combined effect of high light with a secondary stressor. Devaney (2013) raised yew plantlets from seed ex situ under low (<5 % full daylight) and high light (100 % full daylight) conditions and measured a number of growth and photosynthetic parameters, including Fv/Fm. In these plants, 7 weeks after germination, Fv/Fm was also lower when plants were grown at the highest light intensity, despite the optimised growth conditions with adequate supply of water and nutrients. Thus, although yew seedlings, saplings and juveniles effectively adjust photosynthetic performance in response to the light environment where they grow (Table 1), there seems to be a maximum light intensity above which individuals from these demographic size classes are subjected to photoinhibition. Mitchell (1998) examined acclimation of mature yew trees (>10 m) to high light conditions and found no evidence of photoinhibition. In fact, yew T. baccata was found to be more tolerant of exposure to full light than T. brevifolia. Future work will need to examine whether the lack of photoinhibition in mature yew trees is an acclimatory response (as observed for seedlings, saplings and juveniles), or whether a development-dependent change in light-utilisation occurs between juvenile and mature stages.
It remains to be established to what extent photoinhibition affects overall performance of yew at higher light intensities. Evidence from the literature is contradictory. It has been reported that yew increases its growth rate in high light conditions (Svenning and Magård 1999; Thomas and Polwart 2003). In an experiment examining light limitation of yew seedlings in Poland, Iszkulo et al. (2007) found a positive correlation between seedling height increase and light availability. However, during the experiment, maximum light availability recorded in the study area was <35 % of full daylight, and hence, growth at high light intensities was not evaluated. In an ex situ study of light availability and yew growth, Perrin and Mitchell (2013) demonstrated that although yew plants grown below 27 % full daylight displayed a positive correlation between light availability and height increase, growth was suppressed in treatments exposed to 100 % of full daylight. Hence it is possible that yew offspring are capable of increasing growth under intermediate shade conditions but in higher light environments (>30–40 % daylight), photoinhibition of needles will lead to a reduction in growth.
Size-dependent morphological responses to light availability in yew needles
Needles exhibited variations in morphology depending on both light environment where plants grew and their size (Table 1). Needle size (length and width) is predominantly dependent on the size of the yew individual (Table 1). Needle structure is an important determinant of light capture, and therefore photosynthetic performance. This creates an interesting dichotomy; the efficiency of the photosynthetic light reactions is largely linked to the light environment in which the plants grew, but needle area is mostly determined by the size of the plants. During the life cycle of yew, needle surface area will increase with plant size (Table 2), and this will be matched by an increase in light availability, as larger plants are exposed to more light (Fig. 1). The size-dependent increase in needle surface area may reflect the overall balance between above and below-ground biomass, i.e. larger individuals with a larger root system can develop and maintain greater needle surface area (Reich et al. 1998).
In this study, changes in needle thickness and related SLA were influenced by light environment and plant size. SLA decreases with both increasing plant size and increasing light availability. Mitchell also found that SLA was reduced in needles of yew trees growing in full sunlight (Mitchell 1998). In other coniferous species similar reductions in SLA in response to exposure to full sunlight were observed (Tucker and Emmingham 1977; Mitchell and Arnott 1995). Increases in SLA are associated with exposure to low light in many tree species and this response can be interpreted as an attempt to maximise light capture (Rozendaal et al. 2006). Conversely, decreases in SLA are often associated with growth under higher light intensities and drought acclimation in woody species (Monclus et al. 2006). In our study, the SLA data can be interpreted in the context of environmental pressure to increase light capture in small plants in low light environments, and to decrease transpiration in larger plants in high light environments. Thus, the lower thickness and higher SLA of needles of yew seedlings compared to juvenile yews, irrespective of light environment, suggests that seedlings are morphologically inherently shade-adapted.
For needle thickness, a strong interaction between plant size and light environment was evident with larger plants following expected patterns of greater thickness in higher light but seedlings being of lowest thickness in high light and greatest thickness in low light. The absence of shading vegetation for high light seedlings may have led to exposure to higher temperatures. Decreases in needle thickness in gymnosperms maybe related to increased atmospheric temperature (Luomala et al. 2005). However, for yew, potential size-dependent responses to environmental stresses other than light availability remain untested.
Yew regeneration and light availability
Our data show a strong association between seedlings and young saplings and shade, and hence, the question arises whether shade is a prerequisite for natural regeneration. Natural regeneration of yew seedlings is mostly absent in high light environments (Watt 1926; Smith 1980). Indeed, Watt (1926), recorded only one seedling in a grazer-excluded grassland area which was located near abundant mature yew trees at the South Downs in England. The relationship between recruitment success in yew and the presence of shade-giving shrubs has been highlighted in the literature (Smith 1980; Garcia et al. 2000; Farris and Filigheddu 2008). For example, the development of yew-dominated woodland in the British Isles is commonly associated with the presence of shrub species such as Crataegus monogyna and Ilex aquifolium (Rodwell 1991). In Mediterranean habitats, work by Garcia and others (Garcia et al. 2000; Farris and Filigheddu 2008) has also outlined the facilitation of yew regeneration by nurse plants. Nurse plants can facilitate yew recruitment by providing multiple microsite benefits such as increased seed input and protection from herbivore damage (Garcia and Obeso 2003; Garcia et al. 2005; Farris and Filigheddu 2008). The extent to which shaded conditions afforded by shrub species contribute to yew growth and development in early life history stages is not clear.
To enable effective conservation and silvicultural programs for tree species, further research is required to clarify the extent to which light availability regulates both photosynthetic capacity and needle morphology, and consequently species growth and distribution. The data in this paper highlight the importance of considering multiple plant traits and developmental stages when evaluating species environmental responses and distribution patterns.
References
Augspurger CK, Bartlett EA (2003) Differences in leaf phenology between juvenile and adult trees in a temperate deciduous forest. Tree Physiol 23(8):517
Ball M, Hodges V, Laughlin G (1991) Cold-induced photoinhibition limits regeneration of snow gum at tree-line. Funct Ecol 5:663–668
Canham CD, Denslow JS, Platt WJ, Runkle JR, Spies TA, White PS (1990) Light regimes beneath closed canopies and tree-fall gaps in temperate and tropical forests. Can J For Res 20(5):620–631
Coopman RE, Reyes-Díaz M, Briceño VF, Corcuera LJ, Cabrera HM, Bravo LA (2008) Changes during early development in photosynthetic light acclimation capacity explain the shade to sun transition in Nothofagus nitida. Tree Physiol 28(10):1561–1571
Devaney JL (2013) The natural regeneration ecology of yew Taxus baccata L., University College Cork
Devaney JL, Jansen MA, Whelan PM (2013) Spatial patterns of natural regeneration in stands of English yew (Taxus baccata L.); Negative neighbourhood effects. For Ecolog Manag 321:52–60
Dhar A, Ruprecht H, Klumpp R, Vacik H (2006) Stand structure and natural regeneration of English yew (Taxus baccata L.) at Stiwollgraben in Austria. Dendrobiology 56:19–26
Dhar A, Ruprecht H, Klumpp R, Vacik H (2007) Comparison of ecological condition and conservation status of English yew population in two Austrian gene conservation forests. J For Res 18(3):181–186
Dubreuil M, Sebastiani F, Mayol M, González-Martínez SC, Riba M, Vendramin GG (2008) Isolation and characterization of polymorphic nuclear microsatellite loci in Taxus baccata L. Conserv Genet 9(6):1665–1668
Farris E, Filigheddu R (2008) Effects of browsing in relation to vegetation cover on common yew (Taxus baccata L.) recruitment in mediterranean environments. Plant Ecol 199(2):309–318
Garcia D, Obeso RJ (2003) Facilitation by herbivore-mediated nurse plants in a threatened tree, Taxus baccata: local effects and landscape level consistency. Ecography 26(6):739–750
Garcia D, Zamora R, Hódar JA, Gómez JM, Castro J (2000) Yew (Taxus baccata L.) regeneration is facilitated by fleshy-fruited shrubs in mediterranean environments. Biol Conserv 95(1):31–38
Garcia D, Obeso JR, Martinez I (2005) Spatial concordance between seed rain and seedling establishment in bird dispersed trees: does scale matter? J Ecol 93(4):693–704
Genty B, Briantais J-M, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochimica et Biophysica Acta (BBA)-General Subjects 990(1):87–92
Givnish TJ (1988) Adaptation to sun and shade: a whole-plant perspective. Funct Plant Biol 15(2):63–92
Hilfiker K, Gugerli F, Schütz JP, Rotach P, Holderegger R (2004) Low RAPD variation and female-biased sex ratio indicate genetic drift in small populations of the dioecious conifer Taxus baccata in Switzerland. Conserv Genet 5(3):357–365
Iszkulo G (2010) Success and failure of endangered tree species: low temperatures and low light availability affect survival and growth of European yew (Taxus baccata L.) seedlings. Polish J Ecol 58(2):259–271
Iszkulo G, Boratynski A (2004) Interaction between canopy tree species and European yew Taxus baccata(Taxaceae). Polish J Ecol 52(4):523–531
Iszkulo G, Boratynski A (2006) Analysis of the relationship between photosynthetic photon flux density and natural Taxus baccata seedlings occurrence. Acta Oecol 29(1):78–84
Iszkulo G, Lewandowski A, Jasinska AK, Dering M (2007) Light limitation of growth in 10-year-old seedlings of Taxus baccata L. (European yew). Polish J Ecol 55(4):827–831
Iszkulo G, Jasinska AK, Giertych MJ, Boratynski A (2009) Do secondary sexual dimorphism and female intolerance to drought influence the sex ratio and extinction risk of Taxus baccata? Plant Ecol 200(2):229–240
Jackson G, Sheldon J (1949) The vegetation of magnesian limestone cliffs at Markland Grips near Sheffield. J Ecol 38–50
Jansen MAK, Mattoo AK, Edelman M (1999) D1-D2 protein degradation in the chloroplast. Eur J Biochem 260(2):527–532
Kobe RK, Pacala SW, Silander JA Jr, Canham CD (1995) Juvenile tree survivorship as a component of shade tolerance. Ecol Appl 5(2):517–532
Krol S (1978) An outline of ecology. In The yew—Taxus baccata L. Foreign Scientific Publication, Department of the National Centre for Scientific and Technical, and Economics Information (for the Department of Agriculture and the National Science Foundation, Washington, DC), Warsaw, Poland, pp 65–86
Kunstler G, Coomes DA, Canham CD (2009) Size dependence of growth and mortality influence the shade tolerance of trees in a lowland temperate rain forest. J Ecol 97(4):685–695
Little DJ (1994) Occurences and characteristics of podzols under oak woodland in Ireland PhD Thesis, Trinity College Dublin
Luomala E, Laitinen K, Sutinen S, Kellomäki S, Vapaavuori E (2005) Stomatal density, anatomy and nutrient concentrations of Scots pine needles are affected by elevated CO2 and temperature. Plant, Cell Environ 28(6):733–749
Lusk C (2004) Leaf area and growth of juvenile temperate evergreens in low light: species of contrasting shade tolerance change rank during ontogeny. Funct Ecol 18(6):820–828
Lusk CH, Jorgensen M (2013) The whole-plant compensation point as a measure of juvenile tree light requirements. Funct Ecol 27(6):1286–1294
Lusk C, Falster D, Jara Vergara C, Jimenez Castillo M, Saldaña Mendoza A (2008) Ontogenetic variation in light requirements of juvenile rainforest evergreens. Funct Ecol 22(3):454–459
Martínez I, García D, Obeso JR (2008) Differential seed dispersal patterns generated by a common assemblage of vertebrate frugivores in three fleshy-fruited trees. Ecoscience 15(2):189–199
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51(345):659–668
McKenna B (2003) An inventory of Arbutus unedo (Arbutus) and Taxus baccata (Yew) at Glengarriff wood nature reserve-A GIS approach Higher Diploma Dissertation, University College Cork
Messier C, Doucet R, Ruel JC, Claveau Y, Kelly C, Lechowicz MJ (1999) Functional ecology of advance regeneration in relation to light in boreal forests. Can J For Res 29(6):812–823
Mitamura M, Yamamura Y, Nakano T (2009) Large-scale canopy opening causes decreased photosynthesis in the saplings of shade-tolerant conifer Abies veitchii. Tree Physiol 29(1):137
Mitchell A (1998) Acclimation of Pacific yew (Taxus brevifolia) foliage to sun and shade. Tree Physiol 18(11):749
Mitchell A, Arnott J (1995) Effects of shade on the morphology and physiology of amabilis fir and western hemlock seedlings. New For 10(1):79–98
Monclus R, Dreyer E, Villar M, Delmotte FM, Delay D, Petit JM et al (2006) Impact of drought on productivity and water use efficiency in 29 genotypes of Populus deltoides × Populus nigra. New Phytol 169(4):765–777
Myking T, Vakkari P, Skrøppa T (2009) Genetic variation in northern marginal Taxus baccata L. populations. Implications for conservation. Forestry 82(5):529
Niinemets Ü, Valladares F (2006) Tolerance to shade, drought, and waterlogging of temperate Northern Hemisphere trees and shrubs. Ecol Monogr 76(4):521–547
O’Neill FH (2003) Regeneration of Native Oak Woodland following Rhododendron ponticum L. clearance from Glengarriff Woods Nature Reserve, Co. Cork, Ireland. PhD. Thesis, University College Cork
Oquist G, Huner N (1991) Effects of cold acclimation on the susceptibility of photosynthesis to photoinhibition in Scots pine and in winter and spring cereals: a fluorescence analysis. Funct Ecol 5:91–100
Oxborough K, Baker NR (1997) Resolving chlorophyll a fluorescence images of photosynthetic efficiency into photochemical and non-photochemical components—calculation of qP and Fv-/Fm-; without measuring Fo. Photosynth Res 54(2):135–142
Perrin PM, Mitchell FJ (2013) Effects of shade on growth, biomass allocation and leaf morphology in European yew (Taxus baccata L.). Eur J For Res 132:211–218
Perrin PM, Kelly DL, Mitchell FJG (2006) Long-term deer exclusion in yew-wood and oakwood habitats in southwest Ireland: natural regeneration and stand dynamics. For Ecol Manag 236(2–3):356–367
Piovesan G, Saba EP, Biondi F, Alessandrini A, Di Filippo A, Schirone B (2009) Population ecology of yew (Taxus baccata L.) in the Central Apennines: spatial patterns and their relevance for conservation strategies. Plant Ecol 205(1):23–46
Poorter L, Bongers F, Sterck FJ, Woell H (2005) Beyond the regeneration phase: differentiation of height–light trajectories among tropical tree species. J Ecol 93(2):256–267
Reich P, Tjoelker M, Walters M, Vanderklein D, Buschena C (1998) Close association of RGR, leaf and root morphology, seed mass and shade tolerance in seedlings of nine boreal tree species grown in high and low light. Funct Ecol 12(3):327–338
Robakowski P, Wyka T (2009) Winter photoinhibition in needles of Taxus baccata seedlings acclimated to different light levels. Photosynthetica 47(4):527–535
Rodwell JS (1991) British plant communities, woodlands and scrub. Cambridge University Press, Cambridge
Rozendaal D, Hurtado V, Poorter L (2006) Plasticity in leaf traits of 38 tropical tree species in response to light; relationships with light demand and adult stature. Funct Ecol 20(2):207–216
Smith CJ (1980) Ecology of the English chalk. Academic Press, London
Svenning J, Magård E (1999) Population ecology and conservation status of the last natural population of English yew Taxus baccata in Denmark. Biol Conserv 88(2):173–182
Takahashi S, Murata N (2008) How do environmental stresses accelerate photoinhibition? Trends Plant Sci 13(4):178–182
Thomas P, Packham JR (2007) Ecology of woodlands and forests: description, dynamics and diversity. Cambridge Univ Press, Cambridge
Thomas P, Polwart A (2003) Taxus baccata L. J Ecol 91(3):489–524
Tucker G, Emmingham W (1977) Morphological changes in leaves of residual western hemlock after clear and shelterwood cutting. For Sci 23(2):195–203
Valladares F, Niinemets Ü (2008) Shade tolerance, a key plant feature of complex nature and consequences. Annu Rev Ecol Evol Syst 39:237–257
Valladares F, Dobarro I, Sánchez-Gómez D, Pearcy RW (2005) Photoinhibition and drought in mediterranean woody saplings: scaling effects and interactions in sun and shade phenotypes. J Exp Bot 56(411):483–494
Walters MB, Reich PB (1996) Are shade tolerance, survival, and growth linked? Low light and nitrogen effects on hardwood seedlings. Ecology 77(3):841–853
Watt A (1926) Yew communities of the South Downs. J Ecol 14(2):282–316
Wayne P, Bazzaz F (1993) Birch seedling responses to daily time courses of light in experimental forest gaps and shadehouses. Ecology 1500–1515
Wyka T, Robakowski P, Zytkowiak R (2008) Leaf age as a factor in anatomical and physiological acclimative responses of Taxus baccata L. needles to contrasting irradiance environments. Photosynth Res 95(1):87–99
Author contribution statement
All authors conceived and designed the work presented in this paper, and contributed substantially to analysis and interpretation of data. J.L.D. carried out field sampling, with assistance from P.M.W. Chlorophyll fluorescence and needle morphology work was carried out and analysed by J.L.D., with assistance from M.A.K.J. The manuscript was prepared and edited by J.L.D., P.M.W., and M.A.K.J.
Acknowledgments
The authors thank Grace Cott and Alan O’Connor for their assistance in fieldwork. Acknowledgement to National Parks and Wildlife Service, Department of the Environment, Heritage and Local Government for access to some lands. Particular thanks to Declan O’Donnell and Clare Heardman for facilitating research at the Glengarriff Wood Nature Reserve. This study was funded by a University College Cork departmental postgraduate fellowship.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by W. Bilger.
Rights and permissions
About this article
Cite this article
Devaney, J.L., Whelan, P.M. & Jansen, M.A.K. Light responses of yew (Taxus baccata L.); does size matter?. Trees 29, 109–118 (2015). https://doi.org/10.1007/s00468-014-1095-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-014-1095-x