Abstract
Background
Reference values for urinary calcium (Ca) and other solutes/creatinine (Cr) ratios in infants and young children are scarce. Its variation with type of lactation administered, breastfed (BF) or formula (F), is incompletely known.
Methods
A total of 511 spot urine samples from 136 children, aged 6 days to < 5 years, was collected. Urine was collected no fasting in infants < 18 months and first morning fasting in children aged 2.5–4 years. Urinary osmolality, Cr, urea, Ca, phosphate (P), magnesium (Mg), and uric acid (UA) were determined. Values are expressed as solute-to-Cr ratio.
Results
Urinary values were grouped according to the child’s age: 6–17 days (G1), 1–5 months (G2), 6–12 months (G3), 13–18 months (G4), and 2.5–4 years (G5). G1 was excluded; Ca/Cr and UA/Cr (95th percentile) decreased with age (G2 vs. G5) from 1.64 to 0.39 and 2.33 to 0.83 mg/mg, respectively. The P/Cr median rises significantly with age from 0.31 (G2) to 1.66 mg/mg (G5). Mg/Cr was similar in all groups (median 0.20, 95th percentile 0.37 mg/mg). Ca/Cr (95th percentile) of BF infants was 1.80 mg/mg (< 3 months) and 1.63 mg/mg (3–5 months), much higher than F infants (0.93 and 0.90 mg/mg, respectively). P/Cr and P/Ca were lower in BF infants.
Conclusions
Values for urinary Ca/Cr, P/Cr, Mg/Cr, and UA/Cr in infants and children < 5 years were updated. BF infants < 6 months showed higher Ca/Cr and lower P/Cr than F infants. New cutoff values to diagnose hypercalciuria in infants < 6 months, according to the type of lactation, are proposed.
Graphical abstract

A higher resolution version of the Graphical abstract is available as Supplementary information
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ontogenetic changes occur continuously during development. The standardization of metabolic and physiological values in relation to age is required, especially interesting being those obtained from studies in healthy population. Regarding kidney function, changes in urinary composition are particularly pronounced in the neonatal stage and up to 2–4 years of age, when relative values like those of older children and adults are reached.
The quantification of urinary elimination of the different solutes has traditionally been carried out in urine collected in 24 h, a sample that is difficult to obtain especially from the youngest children. For this reason, at present, we rely on the evaluation of the ratios between the urinary concentrations of different solutes and creatinine (Cr), after studies in children and adults have verified its correlation with the excretion in 24 h [1,2,3,4]. However, despite its usefulness, reference values in the early years are very scarce and vary with age, and most of them are not updated or have been obtained from populations with different living conditions, which modifies the range of normality (Table 1) [5,6,7,8,9,10,11,12,13,14,15].
Furthermore, the composition of breast milk or adapted formula influences the urinary elimination of some solutes (calcium, phosphorus) [15, 16] in infants, a fact that is not considered in the most frequently used references [5, 7, 10].
The purpose of this study was to determine values for urinary ratios of calcium (Ca), phosphorus (P), magnesium (Mg), and uric acid (UA), related to Cr in spot urine samples of Spanish healthy children < 5 years of age. We also analyzed the variation of urinary excretion of Ca, P, Mg, UA, Cr, osmolality, Na, and K depending on the type of lactation (breast or formula) in infants < 6 months, when milk is the only or main food intake. In addition, an evolutionary comparison of the excretion of Ca and P is made in the first 18 months of life in relation to the maintenance of breastfeeding.
Participants and methods
Participants
From January 2018 to December 2021, children of Caucasian origin from Valencia, Spain, with different ages (6–17 days old; 1, 2, 4, 6, 9, 12, 15 and 18 months old; and between 2.5 and 4 years old) were recruited from a primary health center at the time of their scheduled visits.
Inclusion criteria were healthy full-term newborns with an adequate birth weight. Children were excluded if they had kidney abnormalities observed on the antenatal ultrasound, Apgar score < 7 at 5 min of life, kidney disease, metabolic disorder, or intake of any drugs except for vitamin D prophylaxis, 400 IU per day, during the first year of life. All children were within normal weight and height limits according to their age and gender without pathological alterations. Children maintained a normal diet. In infants under 18 months, the type of lactation, breastfed (BF) or formula-fed (F), was recorded.
Urine samples
Spot urine samples were obtained during the morning. Special plastic bags with adhesive were used to collect urine from infants < 18 months, no fasting, at the time of the scheduled visit in the primary health center. For children aged 2.5–4 years who were diurnal continent with no bed wetting, urine samples were obtained at home from the first morning fasting urine and were immediately carried to the pediatrics clinic. In those with urinary samples in all age groups from 1 to 18 months (repeated samples), the influence of the type of lactation was studied. A reagent strip test (Siemens Multistix® 10 SG) was performed, and samples showing protein, blood, or leukocytes were excluded.
All urine was stored at 4 ºC and transported to the hospital biochemistry laboratory within 24 h. Samples were centrifuged for 10 min at 2500 × g, and the following parameters were determined: osmolality (Osm, mOsm/kg H2O) and concentration of Cr (mg/dL), Ca (mg/dL), P (mg/dL), Mg (mg/dL), UA (mg/dL), urea (mg/dL), glucose (mg/dL), sodium (Na, mEq/L), and potassium (K, mEq/L). Osmolality was measured using the freezing point depression principle in an osmometer (Osmo Station OM-6060, Arkray, Inc.); Cr by Jaffe kinetic method (IDMS); P, Mg, and Ca by colorimetry using molybdate, xylidyl blue, and arsenazo-III as complexing agents, respectively; and Na and K by direct potentiometry with ion-selective electrodes and UA by uricase method. All analytes were normalized to Cr and expressed as analyte-to-creatinine ratios (mg/mg or mmol/mg). Ratios P/Ca (mg/mg) and Cr/Osm (µmol/mOsm) were calculated.
Statistics
Statistical analyses were performed using SPSS software (IBM SPSS Statistics v. 26.0, licensed to the University of Valencia). Descriptive results were expressed as mean ± standard deviation (SD) for continuous variables with normal distribution and as the median and interquartile range (IQR) for variables with non-normal distribution. The 5th and 95th percentiles were calculated, interpreting the 95th percentile as the upper reference limit. Comparisons between groups were made using ANOVA corrected with Bonferroni coefficient, or unpaired Student’s t-test, or non-parametric Kruskal–Wallis, or Mann–Whitney test, as appropriate. To account for differences by type of lactation, results of selected group with repeated samples, as indicated, were analyzed using a general linear model of repeated measures. A p value < 0.05 was used as the criterion for significance.
Ethics
Written informed consent was obtained from the parents, and approval was obtained from the Ethics Committee for Clinical Research of the Hospital Clínico Universitario, Valencia, Spain.
Results
A total of 511 urine samples were obtained from 136 children (369 from males and 142 from females). Urinary values were grouped according to the child’s age: 6–17 days (G1), 1–5 months (G2), 6–12 months (G3), 13–18 months (G4), and 2.5–4 years (G5) (Table 2). No sex-related significant differences were detected for any urine parameters. Glucosuria was lower than 15 mg/dL across all samples.
Urinary creatinine and creatinine-to-osmolality ratio
Table 2 shows urine Osm, urea, Cr, and Cr/Osm in the different groups. From the first month of age, there was a significant correlation (r = 0.89) between Cr and Osm. There was also a correlative increase of Osm (r = 0.77, p < 0.001), urea (r = 0.96, p < 0.001), and Cr (r = 0.88, p < 0.001) with age. The ratio Cr/Osm showed less difference between groups and less interindividual variability than Cr concentration (mg/dL), allowing the ratio solute/Cr to be used as a reliable indicator of solute excretion and allowing comparisons to be made between the analyzed age groups.
Urinary excretion of calcium, phosphate, magnesium, and uric acid
Ca/Cr was higher in infants ≤ 12 months of age (G1–G3) than in G4–G5, which showed similar values. G1 was excluded; Ca/Cr decreased significantly with age (groups G2 to G4; r = − 0.50, p < 0.001). By contrast, from the first month of age, phosphaturia increased with age (r = 0.27, p < 0.001). The P/Ca ratio followed a similar trend (Table 3). Mg/Cr was significantly lower in G1 when compared to other groups (p < 0.05), which showed no change with age. Newborn group (G1) was excluded; UA excretion decreased with age (r = − 0.58, p < 0.001) (Table 3).
Influence of the type of lactation in the first 6 months after birth
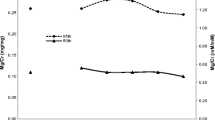
Data from infants under 6 months of age (exclusively milk-fed) were analyzed and divided into 2 groups according to the age (< 3 months and 3–5 months) (Table 4). Significant differences were detected in Ca/Cr, P/Cr, and P/Ca ratios between the two types of lactation in each age group. BF infants showed Ca/Cr ratios (median and 95th percentile) twice as high as formula-fed (p < 0.001), whereas P/Cr and P/Ca ratios were lower (p < 0.001) in BF infants. No differences in Cr/Osm, Urea/Cr, UA/Cr, and Na/Cr were found; in infants under 3 months, the Mg/Cr and K/Cr were higher in formula-fed (p < 0.05). No significant correlation (Pearson’s coefficient) was found between Ca/Cr vs. P/Cr and the type of lactation (BF or F) among infants < 3 months and among those 4–5 months of age. Figure 1 depicts the significant differences (p < 0.001) in the temporal variation of Ca/Cr, P/Cr, and P/Ca ratios according to type of lactation in 344 urine samples of 43 infants (29 breastfed, 14 formula-fed) by an analysis of variance with repeated measures.
Variation by age and type of lactation of urinary Ca/Cr (a), P/Cr (b), and P/Ca (c) ratios. General model of repeated measures: 344 urine samples of 43 healthy infants (29 BF, 14 F) aged 1–18 months. ANOVA p < 0.001 (within-subject means) in all three ratios of BF and F infants. Differences BF vs. F (*symbol: Mann–Whitney p < 0.05) were found in infants 1–9 months of age
Discussion
This study proposes urinary excretion values of Ca, P, Mg, and UA normalized to Cr in spot urine samples from healthy infants and children aged 6 days to less than 5 years. The samples were obtained from subjects with adequate feeding and growth, all of them living in Valencia, Spain. We have analyzed the influence of the type of lactation (BF vs. F) in infants. Finally, we provide updated normal limits of Ca/Cr ratios in infants and children < 5 years, considering the type of milk (BF or F) in the first 6 months of life. Although 24-h urinary solute excretion is considered the gold standard, there are difficulties in timed urine collections, especially in infants and young children. Measurements in random urine samples are an easy and practical method for pediatricians to assess solute excretion and very useful for epidemiologic studies. Recent studies have shown that solute/Cr ratios vary with age and geographic area; hence, reference ranges and 95th percentile values specific for each population are needed.
Calcium and phosphorus
There are few reports on urinary excretion of calcium [7, 10, 17] and phosphorus [7, 12] in healthy infants and young children. Studies that analyze the influence of the type of milk administered (BF or F) on urinary excretion of these solutes when the infant diet is exclusive (first 5–6 months) or predominantly milk (6–12 months) are also scarce [11, 13, 15, 18, 19].
Our results show a highly significant decrease in the urinary Ca/Cr ratio with age (Table 3) in agreement with reported observations by Sargent et al. [5] and Matos et al. [7], the first two studies that demonstrated that random urinary Ca/Cr is higher in infants and young children compared to older children and adolescents. This high Ca/Cr ratio has been linked to lower urinary creatinine at this age [6, 15, 20, 21]; however, the small variations in the urinary Cr/Osm ratio (Table 2) do not support this hypothesis [7]. Nevertheless, in our study, the median and 95th percentile for urine Ca/Cr ratio in children from 6 days to 18 months of age were higher than those reported in Europe [5, 7, 20,21,22] and in Caucasian children in the USA [10] and much higher than those observed in Iran [12], Turkey [11, 13, 23], and African-American infants in the USA [10]. In contrast, the 95th percentile of Ca/Cr in children aged 13–18 months and 2.5–4 years was comparable to those reported by Matos et al. in Swiss children used as a reference [7]. Since we have used similar analytical methods for the determination of urinary Ca, the variations must be related to ethnic origin or to the different content of Ca in foods by geographical or cultural variations [7, 10, 17, 22].
Contrary to calciuria, P/Cr ratio is lower in children under 6 months of age than in older ones (Table 3). However, Matos et al. [7] reported the highest values in infants from 1 to 12 months and a progressive decrease up to 4 years of age with values like those found in our children. A similar profile, with lower P/Cr values, was observed in Iranian children [12] (Table 1). The lack of knowledge about the type of lactation and the different age categorization < 12 months makes the comparison with our results difficult, despite using the same method of analysis for urinary P. However, a similar lower phosphaturia in the first 6 months of life was previously observed in healthy term infants in Greece [18] and in the USA [16, 19] being attributed to the low P content of human or formula milk. The increase in P/Cr ratio after 6 months coincides with the start of complementary feeding with a higher P content (Fig. 1), which remains elevated up to 4 years old with the diet of Western preschool children [7, 24, 25]. Such discrepancies highlight the need to provide reference values for phosphorus excretion in young children for the diagnosis of states with increased phosphaturia (Table 3).
The median urinary P/Ca ratio in full-term newborns (2 mg/mg) drops to 0.65 mg/mg in infants aged 1–5 months (Table 3), with an important interindividual variability, attributable to the type of lactation. After 6 months, the increase in phosphaturia and the decrease in calciuria raise the P/Ca ratio in all the other groups (Table 3 and Fig. 1) with values comparable to 2-year-old or older children from Western countries [24, 25].
To summarize, the present study has found urinary Ca/Cr values higher and P/Cr lower than the reference values in healthy infants [5, 7, 9,10,11,12,13], especially in those under 6 months of age when food intake is exclusively milk, and similar values of calciuria and phosphaturia to those considered reference in children 1–4 years of age fed with a standard Western diet.
Magnesium
Ghazali and Barratt [2] were the first to report mean values for urinary Mg/Cr (0.21 ± 0.10 mg/mg) in the second morning urine sample in 29 British children aged 1 to 15 years. Most subsequent studies have been conducted in children older than 2 years, concluding that Mg/Cr displays a moderate inverse relationship with age [7, 26,27,28,29]. Upper limits for magnesium excretion have not yet been determined for all ages, and data are available from only 4 publications on infants from very different populations [6, 7, 12, 28].
Our study shows little variation in Mg/Cr in infants and children below 5 years of age excluding the group of newborns (Table 3). In neonates, Mg/Cr was lower than in the rest of the groups and slightly higher than those reported by Ariceta et al. in 33 hospitalized full-term newborns [29]. In infants (Table 3), our results show higher Mg/Cr ratios than those reported by Ariceta et al. [6] in Bizkaia, Spain, and by Safarinejad in Iranian infants (1–7 months of age) [12], but much lower than in the Swiss study [7] (Table 1). However, in children aged 1–4 years, our findings show similar and relatively stable values, comparable to other published data [7, 26, 27]. Analytical methods were similar in all studies except for those of Ariceta et al. [6, 29] and Paunier et al. [26]. This could explain the differences. However, factors such as the type of milk and the composition of the young child’s diet should also be considered [7]. Thus, Mg/Cr ratios in our infants are comparable to Yilmaz’s study [28] and lower than the Swiss study [7] which has an inferior percentage of infants on breast milk. Standard infant formulas have less Mg content than breast milk [30].
Uric acid
There are few studies providing normal values for UA/Cr in children younger than 5 years beyond the neonatal period, where it is a marker of the severity of asphyxia [31]. For older children, the UA/Cr ratio is used for the investigation of nephrolithiasis and recurrent hematuria [32, 33]. In Spain, there is only one study in children aged 2 to 6 years [34].
The median and 95th percentile of urine UA/Cr, excluding the neonates group, decrease with age (Table 3). An age-dependent decrease in urate excretion was also observed by Matos et al. [8] and Poyrazoglu et al. [14] in groups of children in the same age range and in other studies with older children [3, 8, 12, 14, 35,36,37].
Variation of urinary Ca, P, and Mg by type of lactation
The influence of the type of lactation (BF vs. F) on the urinary excretion of Ca, P, and Mg has not been considered in most classical references [5, 7, 10]. In the present study, Ca/Cr ratio in BF infants was twofold higher and P/Cr ratio was sevenfold lower compared with formula-fed infants (Table 4). In the long term, the analysis of repeated samples (Fig. 1) shows a progressive decrease in Ca/Cr and increase in P/Cr ratios in BF, reaching similar values in infants with both types of lactation from the age of 12–15 months.
Regarding calciuria, two studies in Turkey [11, 13] and a more recent multicenter European study [15] have provided information on very different populations. The influence of the type of lactation is contradictory among the two Turkish studies. One study [11] did not observe differences, and in the other [13], Ca/Cr was lower in BF infants. In contrast, the European multicenter study (Table 1), which includes a large number of spot urinary samples from infants at 3 and 6 months of age, reported higher Ca/Cr in BF, especially at 3 months [15], comparable to what has been observed in the present study (Table 4 and Fig. 1).
With respect to phosphaturia, the low P/Cr ratio found in BF infants (Table 4) is consistent with the findings of the first long-term longitudinal study on postnatal changes in phosphate excretion in healthy term newborns with different types of milk [19]. This and other studies suggest that the phosphate intake modulates urinary phosphate excretion and that the low P/Cr ratio found in BF infants can be explained by the low phosphate concentration in breast milk [16, 18, 19, 38].
Some components of a diet such as high protein and Na, low K intake, and vitamin D excess [22, 30, 33, 39] have been shown to favor urinary Ca excretion. Our data suggest that higher Ca/Cr in the BF group cannot be attributed to these factors, because there were no differences in urinary Na/Cr, K/Cr, and Urea/Cr ratio, a non-invasive index of protein intake [40] among the two types of lactation (Table 4).
It has been suggested that the lower P content of human milk compared to standard formula could increase calciuria in healthy term infants in the context of a relative nutritional P deficiency [15]. The nutritional P deficiency in newborns was described in small premature infants fed with breast milk without P and vitamin D supplementation [41]; and it is characterized by growth arrest, hypophosphatemia, hypercalciuria, undetectable P in urine, and rickets [41, 42]. To our knowledge, the quotation that the nutritional phosphate deficiency has not been described in any healthy term infants with exclusive BF in the first 6 months of life [30, 42] is still true. However, from the balance studies carried out by Fomon et al. [39], it is known that low P intake in healthy infants increases calcium absorption and elevates calciuria. Likewise, the supplementation of P to healthy term infants with BF in the first weeks of life (without vitamin D supplementation) decreases calciuria [19].
The present study does not include serum concentrations of Ca, P, 25-hydroxivitamin D, and other markers of bone metabolism; however, the normal growth and clinical status of infants and the verification of urinary P always detectable [19, 30] make it unlikely to attribute the high calciuria to a nutritional P deficiency. Thus, we propose that the higher calciuria of infants with exclusive BF can be attributed to the increased relative absorption of Ca of human milk compared with standard formula [39, 43,44,45], due to its low P concentration (and other compositional differences) and favored by adequate vitamin D supplementation, as already proposed by Hillman et al. [42].
Regarding the Mg urinary excretion, we found a slightly higher Mg/Cr ratio in formula-fed infants (Table 4) that can be explained by its higher concentration in standard infant formulas [30]. But we are not aware of other studies that compare these results, except for the study carried out in full-term newborns (age range 1–27 days), where no difference was observed between the two types of lactation [29].
Cutoff values for hypercalciuria in infants and young children
From the studies of Sargent et al. [5] and Matos et al. [7], the accepted upper limit values of 95th percentile of urinary Ca/Cr are < 0.8 mg/mg for infants aged 1–6 months, < 0.6 mg/mg for infants between 6 and 12 months, and 0.5–0.4 mg/mg for children aged 2–4 years. After this age, the limit of 0.21 mg/mg (0.60 mmol/mmol) is well accepted for schoolchildren, adolescents, and adults [2, 3, 7, 17, 20, 21, 24, 25, 33, 46].
In our study, the use of these limits would result in proportions of hypercalciuria of 39.3% (aged 1–5 months), 17.5% (aged 6–12 months), 1.5% (aged 13–18 months), and 4.2% (2.5–4 years), reaching 50.4% in BF infants below 6 months (8.1% in formula-fed). This high estimate indicates that the proposed cutoffs overestimate the hypercalciuria prevalence in children below 1 year of age, especially in infants with exclusive BF. The recent European multicenter study, limited to 3- and 6-month-old infants, has reported similar results to ours [15].
To summarize, and based on our data and using a statistical criterion for upper limit (> 95th percentile), in accordance with previous reference studies, we propose the following Ca/Cr (mg/mg) cutoff values for the diagnosis of hypercalciuria (Table 5): for infants < 3 months, 1.80 mg/mg if they are BF and 0.90 mg/mg in the case of standard F, and for infants from 3 to 6 months, 1.60 and 0.90 mg/mg for BF and F, respectively. From 7 to 12 months, 0.80 mg/mg is adequate, especially when the child continues with BF. After the first year, this limit should be 0.50 mg/mg from 1 to 2 years of age and 0.40 mg/mg up to 5 years of age if they have a normal Western diet for children.
Although we certainly believe that the new cutoff values can be used as prognosis at the clinical level, we cannot suggest any further use as, for instance, a marker of crystallization or lithogenesis, since additional urine composition data (oxalate, citrate) would be required.
The strengths of this work are as follows: (i) the study was carried out in healthy children with adequate growth in separate age groups and during their scheduled health exams, (ii) the difficulty in collecting urine samples at the different ages, (iii) the scarcity of reference excretion values at these ages, and (iv) the evaluation of the type of lactation on the urinary composition.
A limitation of our study is the cohort of children, all of them of Caucasian origin and living in the east of Spain, with a different culture and climate than other population groups from other geographic regions. A study design that would allow results which are representative for a broader community and geographic area would be of great interest.
Conclusions
In conclusion, this study provides age-related 5th and 95th percentiles for urinary Ca/Cr, P/Cr, Mg/Cr, and UA/Cr in spot urine samples in healthy Spanish children between 6 days and 5 years of age.
The child’s age and the type of lactation, during the first months of life, should be taken into consideration when assessing calciuria and phosphaturia. In breastfed infants < 6 months of age, low phosphaturia coexists with high calciuria. This profile should be considered in pathologic situations of renal phosphate wasting.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Nordin BEC (1959) Assessment of calcium excretion from the urinary calcium/creatinine ratio. Lancet 74:368–371
Ghazali S, Barratt TM (1974) Urinary excretion of calcium and magnesium in children. Arch Dis Child 49:97–101. https://doi.org/10.1136/adc.49.2.97
Hernández Marco R, Nuñez Gomez F, Martinez Costa C, Fons Moreno J, Peris Vidal A, Brines Solanes J (1988) Urinary excretion of calcium, magnesium, uric acid and oxalic acid in normal children. An Esp Pediatr 29:99–104
García Nieto VM, Luis Yanes MI, Arango Sancho P (2014) Basic renal function tests reviewed. Is the 24-hour urine collection on the decline in paediatrics? An Pediatr (Barc) 80:275–277. https://doi.org/10.1016/j.anpedi.2014.03.009
Sargent JD, Stukel TA, Kresel J, Klein RZ (1993) Normal values for random urinary calcium to creatinine ratios in infancy. J Pediatr 123:393–397. https://doi.org/10.1016/S0022-3476(05)81738-X
Ariceta G, Rodriguez Soriano J, Vallo A (1996) Renal magnesium handling in infants and children. Acta Paediatr 85:1019–1023
Matos V, Van Melle G, Boulat O, Markert M, Bachmann C, Guignard JP (1997) Urinary phosphate/creatinine, calcium/creatinine and magnesium/creatinine ratios in a healthy pediatric population. J Pediatr 131:252–257. https://doi.org/10.1016/S0022-3476(97)70162-8
Matos V, Van Melle G, Werner D, Bardy D, Guignard JP (1999) Urinary oxalate and urate to creatinine ratios in a healthy pediatric population. Am J Kidney Dis 34:e1. https://doi.org/10.1053/AJKD034000e6
Vachvanichsanong P, Lebel L, Moore E (2000) Urinary calcium excretion in healthy Thai children. Pediatr Nephrol 14:847–850. https://doi.org/10.1007/s004679900229
So NP, Osorio AV, Simon SD, Alon US (2001) Normal urinary calcium/creatinine ratios in African-American and Caucasian children. Pediatr Nephrol 16:133–139. https://doi.org/10.1007/s004670000510
Ceran O, Akin M, Aktürk Z, Ozkozaci T (2003) Normal urinary calcium/creatinine ratios in Turkish children. Indian Pediatr 40:884–887
Safarinejad MR (1993) Urinary mineral excretion in healthy Iranian children. Pediatr Nephrol 18:140–144. https://doi.org/10.1007/s00467-002-1020-1
Erol I, Buyan N, Özkaya O, Sahin F, Beyazova SO, Hasanoglu E (2009) Reference values for urinary calcium, sodium and potassium in healthy newborns, infants and children. Turk J Pediatr 51:6–13
Poyrazoglu HM, Dussunsel R, Yazici C, Durmaz H, Dursum I, Sahin H, Gündüz Z, Gürgöze M (2009) Urinary uric acid:creatinine ratios in healthy Turkish children. Pediatr Int 51:526–529. https://doi.org/10.1111/j.1442-200X.2008.02785.x
Ferré N, Rubio-Torrents C, Luque V, Closa-Monasterolo R, Grote V, Koletzko B, Socha P, Gruszfeld D, Langhendries JP, Sengier A, Verduci E, Escribano J, European Childhood Obesity Project Group (2017) Influence of feeding types during the first months of life on calciuria levels in healthy infants: a secondary analysis from a randomized clinical trial. Ann Nutr Metab 70:139. https://doi.org/10.1159/000468156
McCrory WW, Forman CW, McNamara H, Barnett HL (1952) Renal excretion of inorganic phosphate in newborn infants. J Clin Invest 31:357–366. https://doi.org/10.1172/JCI102616
Reusz GS, Dobos M, Byrd D, Sallay P, Miltényi M, Tulassay T (1995) Urinary calcium and oxalate excretion in children. Pediatr Nephrol 9:39–44. https://doi.org/10.1007/BF00858966
Bistarakis L, Voskaki I, Lambadaridis J, Sereti H, Sbyrakis S (1986) Renal handling of phosphate in the first months of life. Arch Dis Child 61:677–681. https://doi.org/10.1136/adc.61.7.677
Hillman LS, Chow W, Salmons S, Weaver E, Erickson M, Hansen J (1988) Vitamin D metabolism, mineral homeostasis, and bone mineralization in term infants fed human milk, cow milk-based formula, or soy-based formula. J Pediatr 112:864–874. https://doi.org/10.1016/s0022-3476(88)80206-3
Ubalde E, García de Jalón A, Abad A, Loris C (1988) Urinary excretion of calcium in healthy children. Multicenter collaborative study. Nefrologia 3:224–230
Butani L, Kalia A (2004) Idiopathic hypercalciuria in children - how valid are the existing diagnostic criteria? Pediatr Nephrol 19:577–582. https://doi.org/10.1007/s00467-004-1470-8
Hoppe B, Hesse A, Neuhaus T, Fanconi S, Blau N, Roth B, Leumann E (1997) Influence of nutrition on urinary oxalate and calcium in preterm and term infants. Pediatr Nephrol 11:687–690. https://doi.org/10.1007/s004670050366
Ozkaya O, Buyan N, Erol I, Atalay Y, Beyazova U, Sahin F, Söylemezoglu O (2005) The relationship between urinary calcium, sodium, and potassium excretion in full-term healthy newborns. Turk J Pediatr 47:39–45
Saez-Torres C, Rodrigo D, Grases F, García-Raja AM, Gómez C, Lumbreras J, Frontera G (2014) Urinary excretion of calcium, magnesium, phosphate, citrate, oxalate, and uric acid by healthy schoolchildren using a 12-h collection protocol. Pediatr Nephrol 29:1201–1208. https://doi.org/10.1007/s00467-014-2755-1
El Mallah C, Ghattas H, Shatila D, Francis S, Merhi K, Hlais S, Toufeili I, Obeid O (2016) Urinary magnesium, calcium, and phosphorus to creatinine ratios of healthy elementary school Lebanese children. Biol Trace Elem Res 170:264–270. https://doi.org/10.1007/s12011-015-0484-3
Paunier L, Borgeaud M, Wyss M (1970) Urinary excretion of magnesium and calcium in normal children. Helv Paediatr Acta 25:577–584
Simecková A, Zamrazil V, Cerovska J (1998) Calciuria, magnesuria and creatininuria-relation to age. Physiol Res 47:35–40
Yilmaz N, Yüksel S, Altintas F, Kocyigit A (2021) Nephrolithiasis during the first 6 months of life in exclusively breastfed infants. Pediatr Nephrol 36:1227–1231. https://doi.org/10.1007/s00467-020-04815-w
Ariceta G, Rodríguez-Soriano J, Vallo A (1995) Magnesium homeostasis in premature and full-term neonates. Pediatr Nephrol 9:423–427. https://doi.org/10.1007/BF00866716
Institute of Medicine (1997) Dietary reference intakes for calcium, phosphorus, magnesium, vitamin D, and fluoride. National Academies Press, Washington, DC
Bellos I, Fitrou G, Pergialiotis V, Perrea DN, Papantoniou N, Daskalakis G (2019) Random urine uric acid to creatinine and prediction of perinatal asphyxia: a meta-analysis. J Matern Fetal Neonatal Med 32:3864–3870. https://doi.org/10.1080/14767058.2018.1471677
van der Watt G, Omar F, Brink A, McCulloch M (2016) Laboratory investigation of the child with suspected renal disease. In: Avner ED, Harmon WE, Niaudet P, Yoshikawa N, Emma F, Goldstein SL (eds) Pediatric Nephrology, 7th edn. Springer, Heidelberg, pp 613–636
García Nieto VM, Moraleda T, Hernández González MJ (2021) Renal lithiasis in history. Diagnostic approach to renal lithiasis in childhood. Metabolic abnormalities causing stones. The concept of prelithiasis. Preventive treatment. In: Exeni R, García-Nieto V, Medeiros M, Santos F (eds) Paediatric nephrology. University of Oviedo and Autonomous University of Mexico, Oviedo, pp 991–997
Sanchez Bayle M, Martínez Jiménez AL, Ruiz-Jarabo C, Asensio J, Amaiz P, Vila S (1992) Urinary excretion of uric acid in childhood and adolescence. Nefrologia 12:239–243
Chen YH, Lee AJ, Chen CH, Chesney RW, Stapleton FB, Roy S 3rd (1994) Urinary mineral excretion among normal Taiwanese children. Pediatr Nephrol 8:36–39. https://doi.org/10.1007/BF00868256
Stapleton FB (1978) Uric acid excretion in normal children. J Pediatr 92:911–914. https://doi.org/10.1016/S0022-3476(78)80359-X
Vázquez Martul M, Sánchez Bayle M, Écija JL, Montalvo N, Sánchez Medina D, Otero J (1988) Normal values of urinary excretion of uric acid in childhood. Nefrologia 8:250–254
Karlén J, Aperia A, Zetterström R (1985) Renal excretion of calcium and phosphate in preterm and term infants. J Pediatr 106:814–819. https://doi.org/10.1016/s0022-3476(85)80364-4
Fomon SJ, Nelson SE (1993) Calcium, phosphorus, magnesium, and sulfur. In: Fomon SJ (ed) Nutrition in normal infants. Mosby-Yearbook Inc, St. Louis, pp 192–216
Mathes M, Maas C, Bleeker C, Vek J, Bernhard W, Peter A, Poets CF, Franz AR (2018) Effect of increased enteral protein intake on plasma and urinary urea concentrations in preterm infants born at <32 weeks gestation and <1500 g birth weight enrolled in a randomized controlled trial - a secondary analysis. BMC Pediatr 18:154. https://doi.org/10.1186/s12887-018-1136-5
Rowe J, Rowe D, Horak E, Spackman T, Saltzman R, Robinson S, Philipps RJ (1984) Hypophosphatemia and hypercalciuria in small premature infants fed human milk: evidence for inadequate dietary phosphorus. J Pediatr 104:112–117. https://doi.org/10.1016/s0022-3476(84)80606-x
Hillman LS (1990) Mineral and vitamin D adequacy in infants fed human milk or formula between 6 and 12 months of age. J Pediatr 117:S134-142. https://doi.org/10.1016/s0022-3476(05)80011-3
Dror DK, Allen LH (2018) Overview of nutrients in human milk. Adv Nutr 9(Suppl 1):278S-294S. https://doi.org/10.1093/advances/nmy022
Academy American, of Pediatrics. Committee on Nutrition, (1998) Pediatric Nutrition Handbook, 4th edn. American Academy of Pediatrics, Elk Grove Village, pp 655–658
ESPGHAN Committee on Nutrition, Agostoni C, Braegger C, Decsi T, Kolacek S, Koletzko B, Michaelsen KF, Mihatsch W, Moreno LA, Puntis J, Shamir R, Szjewska H, Turck D, van Goudoever J (2009) Breast-feeding: a commentary by the ESPGHAN Committee on Nutrition. J Pediatr Gastroenterol Nutr 49:125. https://doi.org/10.1097/MPG.0b013e31819f1e05
Metz MP (2006) Determining urinary calcium/creatinine cut-offs for the paediatric population using published data. Ann Clin Biochem 43:398–401. https://doi.org/10.1258/000456306778520106
Author information
Authors and Affiliations
Contributions
A. Peris Vidal and R. Hernández contributed to the study conception and design. Material preparation and data collection were performed by A. Peris Vidal and statistical analysis by R. Hernández. The first draft of the manuscript was written by A. Peris Vidal and R. Hernández, and all authors commented on the previous versions of the manuscript. All authors interpreted the data, revised the manuscript, provided intellectual content, and approved the submitted version.
Corresponding author
Ethics declarations
Ethics approval
The procedures used in this study adhere to the tenets of the Declaration of Helsinki. Approval was obtained from the Ethics Committee for Clinical Research of the Hospital Clínico Universitario, Valencia, Spain.
Consent to participate
Written informed consent was obtained from the parents of the patients.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Peris Vidal, A., Ferrando Monleón, S., Marín Serra, J. et al. Urinary excretion of calcium, phosphate, magnesium, and uric acid in healthy infants and young children. Influence of feeding practices in early infancy. Pediatr Nephrol 39, 761–770 (2024). https://doi.org/10.1007/s00467-023-06145-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-023-06145-z