Abstract
Background
Laparoscopy requires specific psychomotor skills and can be challenging to learn. Most proficiency-based laparoscopic training programs have used non-haptic virtual reality simulators; however, haptic simulators can provide the tactile sensations that the surgeon would experience in the operating room. The objective was to investigate the effect of adding haptic simulators to a proficiency-based laparoscopy training program.
Methods
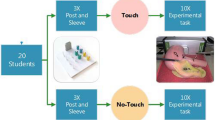
A randomized controlled trial was designed where residents (n = 36) were randomized to proficiency-based laparoscopic simulator training using haptic or non-haptic simulators. Subsequently, participants from the haptic group completed a follow-up test, where they had to reach proficiency again using the non-haptic simulator. Participants from the non-haptic group returned to train until reaching proficiency again using the non-haptic simulator.
Results
Mean completion times during the intervention were 120 min (SD 38.7 min) and 183 min (SD 66.3 min) for the haptic group and the non-haptic group, respectively (p = 0.001). The mean times to proficiency during the follow-up test were 107 min (SD 41.0 min) and 58 min (SD 23.7 min) for the haptic and the non-haptic group, respectively (p < 0.001). The haptic group was not faster to reach proficiency in the follow-up test than during the intervention (p = 0.22). In contrast, the non-haptic group reached the required proficiency level significantly faster in the follow-up test (p < 0.001).
Conclusion
Haptic virtual reality simulators reduce the time to reach proficiency compared to non-haptic simulators. However, the acquired skills are not transferable to the conventional non-haptic setting.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Laparoscopic surgery requires specific psychomotor skills of the surgeon who has to work in a three-dimensional space guided by two-dimensional images and has limited tactile feedback compared to open surgery. Performing laparoscopic procedures safely requires depth perception and excellent hand–eye coordination. These skills can be practiced safely outside the operating room, increasing the demand for efficient simulation-based training, e.g., using virtual reality simulators [1,2,3,4,5,6,7]. Previous studies have shown that virtual reality simulators can shorten the time for novice surgeons to reach proficiency, and the skills are transferable to the operating room [8, 9]. Therefore, virtual reality simulators are widely used for laparoscopy training and become a standard part of modern surgical education [10,11,12]. The virtual reality simulators can also provide an automated assessment of trainees’ performance, enabling them to use a mastery learning approach [13].
However, most laparoscopic training programs with demonstrated clinical transfer were created on virtual reality simulators without haptic feedback; this has been a concern from a clinical point of view as this differs from real surgeries. Although the tactile sensations in laparoscopic surgery are less evident than open surgery, haptic sensations can still be perceived through the instruments by the operating surgeon [14, 15]. These tactile sensations, especially pulling and grasping, are important as understanding them provides valuable information; excessive grasping can damage tissue, whereas inadequate tension can lead to tissue slippage. [16,17,18].
Therefore, virtual reality simulators with haptics for laparoscopy have been developed. These are more realistic but costly and more complex [19, 20], and there could be a risk for a higher frequency of technical malfunctions, disturbing training. [21, 22]. Previous studies have investigated the effect of haptic devices on virtual reality simulators and found that the haptic devices accelerate the initial learning curve of trainees [19, 23,24,25,26,27,28], but these studies only included very few repetitions or a limited amount of training time. The impact of haptic devices in a proficiency-based training program for laparoscopy has not been explored.
Instructor-based feedback has proven to be a valuable tool in reducing time to reach proficiency in laparoscopic simulator training programs without negatively impacting the retention of skills [29]. However, different training setups, in this case, using a haptic simulator versus the non-haptic simulator, might require different amounts of instructor assistance.
The objectives of this trial were to investigate if training with virtual reality with a haptic device compared with a non-haptic simulator resulted in reduced time to reach proficiency for surgical novices, how it affected their retention of skills and the need for instructor feedback and frequency of simulator malfunctions.
Materials and methods
A single-center randomized superiority trial was planned according to the CONSORT statement [30]. The trial was exempt from ethical approval by the Regional Committee on Biomedical Research Ethics (H-20063093). The trial was registered at Clinicaltrials.gov (NCT05191589).
Setting
Data collection was done at the simulation center at Copenhagen Academy for Medical Education and Simulation (CAMES) in Copenhagen, Denmark [31].
Participants
Participants were surgeons without any previous laparoscopic experience. Participants were recruited from the surgical departments in the eastern part of Denmark and were invited by e-mail and received written and verbal information before filling out the informed consent forms. The inclusion and exclusion criteria used for the trial were as follows:
Inclusion criteria for both intervention and follow-up: (1) Residents working in the Eastern part of Denmark; (2) Provided informed consent before inclusion.
Exclusion criteria for intervention: (1) Having previously participated in studies involving laparoscopic training; (2) Having participated in laparoscopic training programs at any simulation center; (3) Having prior experience with laparoscopic surgery (having performed any laparoscopic procedures as primary surgeon, including supervised procedures); (4) Having performed any supervised laparoscopy procedures as primary surgeons during intervention; (5) Did not give consent; (6) Did not speak Danish on a conversational level.
All participants were given a unique trial identification number before randomization.
Intervention and control
Participants were randomized to proficiency-based training with either a haptic (haptic group) or non-haptic laparoscopy simulator (non-haptic group) during the intervention. All participants were invited back after a 3–6 weeks break for a follow-up test where both groups had to practice until they reached proficiency again using the non-haptic setup. No simulator training was allowed, nor any operations as primary surgeon in any laparoscopic procedures during this break.
The simulator training program consisted of four basic skills tasks (Instrument Navigation; Grasping; Lifting and grasping, and Fine dissection) and a procedural task (salpingectomy due to a bleeding ectopic pregnancy) on a virtual reality simulator [8, 13]. All participants had to reach proficiency for all modules, which entails passing twice within five consecutive attempts. The predefined proficiency level was established in previous trials and includes the parameters such as instrument time, instrument path length, instrument angular path length, tissue damage, bleeding, and energy damage [8, 13]. The metrics of these metrics are summarized in the Appendix. Participants booked training sessions by e-mail, and a maximum of one 2-h training session per day was allowed to minimize cognitive overload and fatigue.
Randomization
A 1:1 randomization was performed centrally using a web-based system from Sealed Envelope (London, United Kingdom). The allocation sequence was computer-generated and used varying block sizes of four and six, a sequence that was kept concealed from the principal investigator throughout the trial.
Materials and equipment
We used six LapSim® virtual reality simulators (Software version 2019.1) from Surgical Science (Gothenburg, Sweden) – three simulators with haptics and three simulators without. The only difference between the two setups was the tactile sensation provided by the haptic simulators. To prevent overworking the simulators leading to a potential risk of further technical malfunctions, participants were randomized to a different simulator for each training session within their allocation to minimize bias from the same person training on a simulator which could experience technical problems.
All simulators were connected to a 27″ screen and were height adjustable to ensure the best viewing condition and the most ergonomically correct working position. Each simulator was separated by dividing walls, and noise-canceling Bose Quiet Comfort III headsets were worn by all participants during the trial when training [31].
Outcome measures
The primary outcome was the total time (min) spent to reach the predefined proficiency level for all five tasks. The secondary outcome was the time (min) to reach proficiency on the conventional non-haptic setting after 3–6 weeks of no laparoscopic training.
Exploratory outcomes were instructor time (s) spent on feedback during training and number of malfunctions and time spent solving them during the intervention and follow-up test.
We also calculated the cumulative time (min) training to proficiency, meaning the sum of the time to proficiency during the intervention and the follow-up test.
Sample size calculation
The sample size was calculated based on the primary outcome, time to reach proficiency, using data from a previous trial conducted by the same research group [32]. For the non-haptic group, it was assumed that a mean time of 320 min was needed to reach proficiency, while for the haptic group, a mean time of 240 min was expected. Standard deviations of 70 were assumed for both groups. Using a two-sided significance level of 0.05 and a power of 0.90, the minimum sample size required was 34 participants, 17 in each group.
Statistical analysis
The data was analyzed using SPSS® version 27.0 (IBM, Armonk, NY, USA) and RStudio 2021© (RStudio, Boston, MA, USA). Independent samples t-tests were used for intergroup comparisons for primary, secondary, and exploratory outcomes during the intervention and the follow-up test.
To analyze the training effect, over time, for both the haptic and the non-haptic group, the mixed model with repeated measurements and unstructured covariance matrix model was used for time to reach proficiency and instructor-based feedback. The basic model was Y = a + bI + ct + dt I, where I is the indication of the intervention, t is time (time1 corresponding to the intervention phase and time2 corresponding to the follow-up phase), and a through d are coefficients of the regression equation.
Finally, Fisher’s exact test was used to compare the number of technical malfunctions between the haptic and non-haptic simulator.
Results
The patient demographics for the trial are presented in Table 1 and the trial flowchart in Fig. 1. We found that the haptic group reached proficiency significantly faster than the non-haptic group during the intervention (p = 0.001). In contrast, the non-haptic group reached proficiency significantly faster than the haptic group (p < 0.001) during the follow-up test. Furthermore, we found that the haptic group had no significant improvement, regarding time spent, from the trial's intervention to the follow-up test (p = 0.22). However, the non-haptic group improved their time to reach proficiency significantly faster from the intervention to the follow-up test (p < 0.001). There was no significant difference between the two groups for the cumulated time spent reaching proficiency (p = 0.42) (Table 2) (Fig. 2).
During the intervention, we found that the haptic group required significantly less instructor assistance than the non-haptic group (p < 0.001). For the follow-up test, we found that the time needed for instructor assistance was significantly higher for the haptic group compared to the non-haptic group (p < 0.001).
The haptic group required significantly more feedback from the instructor for the follow-up test than the assistance needed during the intervention phase (p < 0.001). In contrast, the non-haptic group used significantly less help from the instructor during the follow-up phase (p < 0.001). We found that nine out of 17 participants in the non-haptic group needed no assistance from the instructor during the follow-up test. There was no significant difference in the cumulated time spent getting instructor-based feedback (p = 0.38) (Table 3) (Fig. 3).
Lastly, technical malfunctions were reported and registered during both the intervention and follow-up—five incidents were documented out of 4497 attempts resulting in a technical malfunction rate of 0.1% for the entire trial. Four of the five technical malfunctions were during the intervention; three occurred for the haptic group and one in the non-haptic group. Finally, only one technical malfunction occurred during the follow-up test using non-haptic simulators. There was no significant difference in the number of technical malfunctions between the haptic and non-haptic setup (p = 0.10). All technical malfunctions were solved on-site with a simple restart of the exercise.
Discussion
We found that training with a haptic compared with a non-haptic simulator reduced the time to reach proficiency in a simulation-based laparoscopy training program. Previous studies have examined the impact of haptic devices on virtual reality simulators but with contradictory results [21]. The effect of haptic devices on laparoscopic proficiency-based training programs has not been thoroughly examined, since most previous trials only tested the haptic device system over a few repetitions or a limited amount of training time before a post-test or transfer test, or did not look at the isolated influence of the haptic devices [19, 22, 24, 26, 27, 33]. We also minimized potential confounders using the same simulator software [19, 24,25,26,27, 33].
A trial by Hagelsteen et al. examined the combined effect of haptics and 3D-vision over non-haptics with 2D-vision on the performance curve of trainees and found this to accelerate skill acquisition [33]. However, 3D-vision alone has also been shown to reduce time to proficiency [32] and Hagelsteen’s study could not independently estimate haptics’ effect. To our knowledge, this is the first trial to examine the isolated effect of using haptic simulators on proficiency-based training. Using a proficiency-based design, compared with time- or repetition-based training, which only focuses on the initial part of the learning curve, is a strength of our trial.
During the follow-up test where both groups trained using the non-haptic setup, we found that the haptic group performed worse than the non-haptic groups, meaning that the accelerated acquisition of skills using the haptic devices could not be transferred to the non-haptic setup, as they needed significantly longer time to reach proficiency than the non-haptic group.
This can be explained using the Guidance Hypothesis [34] which argues that providing trainees with guidance or help—in our case, the haptic device—when learning psychomotor skills such as those acquired during laparoscopy simulator training can be beneficial for faster skill acquisition [35, 36]. However, participants can become dependent upon the help, which can be detrimental to long-term motor learning [34].
We chose a follow-up test where both groups trained on the non-haptic simulators till proficiency because studies on clinical transfer from laparoscopy simulator training have used non-haptic simulators [9, 13, 37]. Our results show that the trainees who practice on the simulator with a haptic device may not necessarily achieve the same skill level and have reduced transfer of skills to the conventional non-haptic system, demonstrating that the trainees may not receive the full effect of the training program when training is accelerated using haptic simulators and proficiency levels from non-haptic simulators are used. Our findings illustrate that there might be a risk using proficiency levels defined for non-haptic simulators on haptic simulators, as skill acquisition might be reduced because proficiency levels are specific to the context in which they were determined [38].
Our findings are supported by the increased need for instructor assistance in the haptic group during in the follow-up test compared with the intervention phase, even though they are solving identical tasks as during the intervention, just on a non-haptic system. We observed that the participants from the haptic group became frustrated when transferred to the non-haptic simulator, as they perceived the tasks as more difficult, as seen by their increased need for instructor guidance. In contrast, the non-haptic group needed less instructor assistance during the follow-up test, and they trained more independently than during the intervention. During the follow-up test, we found that the participants from the haptic group spent almost the same amount of time training as during the intervention, demonstrating that to a large extent, they had to re-learn the skills necessary to reach proficiency again. This indicates the haptic device might have accelerated the participants’ skills acquisition too much by making it easier for them to reach proficiency during the intervention. This could result in lower consolidation of skills learned [39].
Because training without haptics is more difficult, the non-haptic group trained longer before reaching proficiency during the intervention, which could explain the better retention and lower need for instructor feedback during the follow-up test. Making skills acquisition faster does not necessarily make it better—simulation-based training should not focus on acquiring skills the fastest way possible if this compromises the quality of the targeted skills. A potentially unrecognized benefit of a non-haptic laparoscopy simulator might be the increased difficulty when training without haptics, leading to a higher level of skills acquisition. A study conducted by Ali et al. demonstrated that increasing the difficulty of laparoscopy tasks on a virtual reality simulator increased training quality, resulting in better skills acquisition [40].
Previous studies have examined how different interventions, 3D-vision, and instructor-based feedback, affected time to proficiency in a laparoscopy training program. Both 3D-vision and instructor-based feedback reduced time to proficiency, just as the haptic device did in this study. However, these studies demonstrated no negative impact on the retention of skills after using 3D-vision or instructor-based feedback when all the participants were invited back to do a retention test [32, 36, 41]. Using haptics compared with non-haptics might have accelerated training too much, resulting in a less effective learning process.
Dismissing the potential benefits of creating a more realistic training opportunity using haptic devices should be done with caution, as haptics has been described as a paradigm shift within laparoscopic virtual reality training [42]. However, direct implementation on already-established proficiency-based training programs should be done with caution also, as they create a new task. Instead, we would advise that implementation should be done by conducting further validation studies so that the proficiency setting would be redefined.
A limitation of our study is that we did not examine transfer to a clinical setting. Hence, we cannot conclude anything of the impact of haptic simulator training on actual clinical performance.
More research into the effect of haptic simulators, whether they result in better clinical transfer, and credible newly established pass/fail-standards are needed before they can be recommended as the standard for future simulation-based training for laparoscopy.
Haptic devices are a costly add-on [43], and the use of more advanced simulators can potentially lead to a high frequency of technical malfunctions and thereby a need for technical support [44]; however, this was not the case, as technical malfunctions were very rare in our study.
Our initial assumptions regarding the time needed to reach proficiency for both groups during the intervention were not as expected. However, the effect is unaffected. There is a significant difference in the time to proficiency required in the two groups during both intervention and follow-up. For sample size calculation, assumptions were based on data from previous trials conducted by the same research group. There has been an update of the software since this trial [32] and this could potentially be the reason for the difference in training time (former version 2014 to the current version 2019), as some tasks may have become easier in newer versions. To our knowledge, no other studies have compared training on different versions of the same simulator software, and this merits further investigation in the future. Proficiency levels defined for one software version might not be applicable in a newer version of the software if it has undergone substantial changes, thereby changing the context for which it was defined [38].
Conclusion
The use of a haptic simulator reduces the time needed to reach proficiency in a proficiency-based laparoscopic training program. However, the acquired skills cannot be transferred to the conventional non-haptic simulator setup. Accelerating training too much using haptics may limit the effect of skills training if non-haptic proficiency levels are used on a haptic simulator.
References
Barnes RW, Lang NP, Whiteside MF (1989) Halstedian technique revisited. Innovations in teaching surgical skills. Ann Surg 210(1):118–121
Dankelman J, Grimbergen CA (2005) Systems approach to reduce errors in surgery. Surg Endosc 19(8):1017–1021
Reznick RK, MacRae H (2006) Teaching surgical skills–changes in the wind. N Engl J Med 355(25):2664–2669
Fried GM et al (2004) Proving the value of simulation in laparoscopic surgery. Ann Surg 240(3):518–525 (discussion 525–528)
Seymour NE et al (2002) Virtual reality training improves operating room performance: results of a randomized, double-blinded study. Ann Surg 236(4):458–463 (discussion 463–464)
Våpenstad C, Buzink SN (2013) Procedural virtual reality simulation in minimally invasive surgery. Surg Endosc 27(2):364–377
Zendejas B et al (2013) State of the evidence on simulation-based training for laparoscopic surgery: a systematic review. Ann Surg 257(4):586–593
Larsen CR et al (2009) Effect of virtual reality training on laparoscopic surgery: randomised controlled trial. BMJ 338:b1802
Ahlberg G et al (2007) Proficiency-based virtual reality training significantly reduces the error rate for residents during their first 10 laparoscopic cholecystectomies. Am J Surg 193(6):797–804
Bashankaev B, Baido S, Wexner SD (2011) Review of available methods of simulation training to facilitate surgical education. Surg Endosc 25(1):28–35
Gurusamy KS et al (2009) Virtual reality training for surgical trainees in laparoscopic surgery. Cochrane Database Syst Rev 1:Cd006575
Gallagher AG et al (2005) Virtual reality simulation for the operating room: proficiency-based training as a paradigm shift in surgical skills training. Ann Surg 241(2):364–372
Larsen CR et al (2006) Objective assessment of gynecologic laparoscopic skills using the LapSimGyn virtual reality simulator. Surg Endosc 20(9):1460–1466
Ottermo MV et al (2006) The role of tactile feedback in laparoscopic surgery. Surg Laparosc Endosc Percutan Tech 16(6):390–400
Lamata P et al (2006) Study of laparoscopic forces perception for defining simulation fidelity. Stud Health Technol Inform 119:288–292
Tholey G, Desai JP, Castellanos AE (2005) Force feedback plays a significant role in minimally invasive surgery: results and analysis. Ann Surg 241(1):102–109
Chmarra MK et al (2008) Force feedback and basic laparoscopic skills. Surg Endosc 22(10):2140–2148
Westebring-van der Putten EP et al (2009) Effect of laparoscopic grasper force transmission ratio on grasp control. Surg Endosc 23(4):818–824
Panait L et al (2009) The role of haptic feedback in laparoscopic simulation training. J Surg Res 156(2):312–316
Salkini MW et al (2010) The role of haptic feedback in laparoscopic training using the LapMentor II. J Endourol 24(1):99–102
Rangarajan K, Davis H, Pucher PH (2020) Systematic review of virtual haptics in surgical simulation: a valid educational tool? J Surg Educ 77(2):337–347
Våpenstad C et al (2017) Lack of transfer of skills after virtual reality simulator training with haptic feedback. Minim Invasive Ther Allied Technol 26(6):346–354
Hiemstra E et al (2011) Virtual reality in laparoscopic skills training: is haptic feedback replaceable? Minim Invasive Ther Allied Technol 20(3):179–184
Cao CG et al (2007) Can surgeons think and operate with haptics at the same time? J Gastrointest Surg 11(11):1564–1569
Zhou M et al (2012) Effect of haptic feedback in laparoscopic surgery skill acquisition. Surg Endosc 26(4):1128–1134
Kim HK, Rattner DW, Srinivasan MA (2004) Virtual-reality-based laparoscopic surgical training: the role of simulation fidelity in haptic feedback. Comput Aided Surg 9(5):227–234
Ström P et al (2006) Early exposure to haptic feedback enhances performance in surgical simulator training: a prospective randomized crossover study in surgical residents. Surg Endosc 20(9):1383–1388
Wottawa CR et al (2013) The role of tactile feedback in grip force during laparoscopic training tasks. Surg Endosc 27(4):1111–1118
Bjerrum F et al (2015) Effect of instructor feedback on skills retention after laparoscopic simulator training: follow-up of a randomized trial. J Surg Educ 72(1):53–60
Cuschieri S (2019) The CONSORT statement. Saudi J Anaesth 13(Suppl 1):S27–S30
Konge L et al (2015) The simulation centre at Rigshospitalet, Copenhagen, Denmark. J Surg Educ 72(2):362–365
Sørensen SMD, Konge L, Bjerrum F (2017) 3D vision accelerates laparoscopic proficiency and skills are transferable to 2D conditions: a randomized trial. Am J Surg 214(1):63–68
Hagelsteen K et al (2019) Performance and perception of haptic feedback in a laparoscopic 3D virtual reality simulator. Minim Invasive Ther Allied Technol 28(5):309–316
Winstein CJ, Pohl PS, Lewthwaite R (1994) Effects of physical guidance and knowledge of results on motor learning: support for the guidance hypothesis. Res Q Exerc Sport 65(4):316–323
Salmoni AW, Schmidt RA, Walter CB (1984) Knowledge of results and motor learning: a review and critical reappraisal. Psychol Bull 95(3):355–386
Strandbygaard J et al (2013) Instructor feedback versus no instructor feedback on performance in a laparoscopic virtual reality simulator: a randomized trial. Ann Surg 257(5):839–844
Larsen CR, Sørensen JL, Ottesen BS (2006) Simulation training of laparoscopic skills in gynaecology. Ugeskr Laeger 168(33):2664–2668
Andersen SAW et al (2020) Reliable assessment of surgical technical skills is dependent on context: an exploration of different variables using generalizability theory. Acad Med 95(12):1929–1936
Bjork EL, Bjork RA (2011) Making things hard on yourself, but in a good way: creating desirable difficulties to enhance learning. Psychol Real World: Essays Illustrat Fundam Contrib Soc 2:59–68
Ali MR et al (2002) Training the novice in laparoscopy. More challenge is better. Surg Endosc 16(12):1732–1736
Oestergaard J et al (2012) Instructor feedback versus no instructor feedback on performance in a laparoscopic virtual reality simulator: a randomized educational trial. BMC Med Educ 12:7
Favier V et al (2021) Haptic fidelity: the game changer in surgical simulators for the next decade? Front Oncol 11:713343
Våpenstad C et al (2013) Limitations of haptic feedback devices on construct validity of the LapSim® virtual reality simulator. Surg Endosc 27(4):1386–1396
Sakakushev BE et al (2017) Striving for better medical education: the simulation approach. Folia Med (Plovdiv) 59(2):123–131
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Anishan Vamadevan, Morten Stadeager, Lars Konge, Flemming Bjerrum have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Basic skill 1: Instrument navigation.
Parameter | Requirement for proficiency level |
|---|---|
Left instrument time (s) | < 25 |
Left instrument misses | < 2 |
Left instrument path length (m) | < 1.4 |
Left instrument angular path (degrees) | < 250 |
Right instrument time (s) | < 25 |
Right instrument misses | < 2 |
Right instrument path length (m) | < 1.4 |
Right instrument angular path (degrees) | < 250 |
Tissue Damage | < 5 |
Maximum Damage (mm) | < 10 |
Basic skill 2: Grasping.
Parameter | Requirements for proficiency level |
|---|---|
Left instrument time (s) | < 45 |
Left instrument path length (m) | < 2 |
Left instrument angular path (degrees) | < 300 |
Right instrument time (s) | < 45 |
Right instrument path length (m) | < 2 |
Right instrument angular path (degrees) | < 300 |
Tissue damage (frequency) | < 3 |
Basic skill 3: Lifting and grasping.
Parameter requirements for proficiency level | Requirements for proficiency level |
|---|---|
Total time (s) | < 120 |
Left instrument misses (%) | < 60 |
Left instrument path length (m) | < 3.2 |
Left instrument angular path (degrees) | < 600 |
Right instrument misses (%) | < 60 |
Right instrument path length (m) | < 3.2 |
Right instrument angular path (degrees) | < 600 |
Tissue damage (frequency) | < 5 |
Maximum damage (mm) | < 15 |
Grasper collided with left box (frequency) | < 10 |
Left box lifted (frequency) | < 15 |
Grasper collided with right box (frequency) | < 10 |
Basic skills 4: Fine dissection.
Parameters | Requirements for proficiency level |
|---|---|
Total time (s) | < 150 |
Ripped or burned blood vessels | < 0 |
Energy damaged on blood vessels (%) | < 20 |
Ripped small vessels (%) | < 25 |
Burned small vessels with or without stretch (%) | < 25 |
Grasper path length (m) | < 0.5 |
Grasper angular path (degrees) | < 120 |
Grasper outside view (frequency) | < 2 |
Grasper outside view (s) | < 4 |
Cutter path length (m) | < 0.9 |
Cutter path length (m) | < 0.8 |
Cutter angular path (degrees) | < 200 |
Cutter outside view (frequency) | < 2 |
Cutter outside view (s) | < 4 |
Procedural modul: Ectopic Pregnancy.
Parameters for the procedure: salpingectomy on the Lapsim® virtual reality simulator. To reach the proficiency level all the requirements for the proficiency level must be fulfilled using correct operation technique.
Parameters | Requirements for proficiency level |
|---|---|
Total time (s) | < 280 |
Left instrument path length (m) | < 2 |
Left instrument angular path (degrees) | < 350 |
Right instrument path length (m) | < 3 |
Right instrument angular path (degrees) | < 450 |
Blood loss (ml) | < 180 |
Pool of blood (ml) | < 10 |
Ovary Diathermy damage (s) | < 3 |
Tube Cut: Uterus distance (mm) | < 10 |
Removed dissected tissue (Yes/No) | Yes |
Bleeding vessel cut (Yes/No) | No |
Rights and permissions
About this article
Cite this article
Vamadevan, A., Konge, L., Stadeager, M. et al. Haptic simulators accelerate laparoscopic simulator training, but skills are not transferable to a non-haptic simulator: a randomized trial. Surg Endosc 37, 200–208 (2023). https://doi.org/10.1007/s00464-022-09422-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-022-09422-4