Abstract
Background
Fundoplication and medical management are current mainstays for management of Barrett’s esophagus (BE), however our understanding of differences in outcomes between these two treatments is limited. The aim of this study was to perform a systematic review and meta-analysis to evaluate the efficacy of these interventions on BE disease regression and progression.
Methods and procedures
A comprehensive search in MEDLINE, EMBASE, Scopus, Web of Science, and Cochrane Library databases was performed on February 22, 2021. Inclusion criteria were studies with both medical and surgical management comparators, BE diagnosis prior to treatment, patients aged ≥ 18 years, and studies with greater than five patients. Primary outcomes of interest included evaluating changes in histopathologic BE regression and disease progression between interventions. Meta-analysis was performed using a Mantel–Haenszel random-effects model (RevMan 5.4.1).
Results
A total of 7231 studies were retrieved after initial search with nine studies (1 randomized trial, 7 prospective cohorts, 1 retrospective cohort) meeting final inclusion criteria. Of included studies, 890 (65%) patients received medical management while 470 (35%) received surgical management. Medical management included proton pump inhibitors (n = 807, 91%; 6 studies), H2-receptor blockers (n = 40, 4% patients; 3 studies), and combination therapy (n = 43, 5%; 1 study). Nissen fundoplication was the most commonly performed type of fundoplication (n = 265, 93%). Median length of follow-up ranged from 1.5–7 years. Meta-analysis revealed that fundoplication was associated with improved histopathologic regression of metaplasia/low-grade dysplasia (OR 4.38; 95% CI 2.28–8.42; p < 0.00001) and disease progression to dysplasia/adenocarcinoma (OR 0.34; 95% CI 0.12–0.96; p = 0.04) compared to medical therapy.
Conclusion
Fundoplication is superior to medical therapy with regards to improved odds of histopathologic BE disease regression and disease progression. Additional randomized trials which directly compare medical management and surgical intervention are required to delineate the optimal delivery and timing of these interventions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Barrett’s esophagus (BE) is defined as intestinal metaplasia of the esophageal mucosa and occurs as a result of pathologic exposure of the esophageal mucosa to gastric contents [1]. BE is an important clinical entity because it is a precursor lesion to esophageal adenocarcinoma (EAC), occurring through the metaplasia-dysplasia-adenocarcinoma pathway [2]. The presence of dysplasia in patients with BE substantially increases the risk of developing EAC compared to individuals without dysplasia [3]. As the estimated five-year survival of EAC in Canada in 2019 was 15% [4], it is critical to identify the optimal interventions which incite regression of BE metaplasia and prevent the future progression of BE to dysplasia and EAC.
Current management of BE is aimed at reducing gastroesophageal reflux disease (GERD) which is the greatest modifiable risk factor in the metaplasia-EAC sequence [5]. There are presently two approaches for GERD management in patients with BE-medical therapy and surgical intervention [6, 7]. Medical therapy is the first-line treatment and consists of proton pump inhibitors (PPIs) or, less commonly, histamine-2 blockers (H2 blockers) which work by reducing gastric acid secretion [6]. Surgical management involves fundoplication, a procedure where the upper stomach is wrapped around the lower esophageal sphincter to reinforce the gastroesophageal junction and reduce reflux of gastric contents [8]. Surgical intervention is typically reserved for patients whose GERD is attributed to anatomic defects such as hiatal hernias, or who are symptomatic despite maximal medical therapy on twice a day anti-reflux medication [7]. However, debate exists regarding this current treatment paradigm as recent work suggests that surgery may be superior in preventing histopathologic progression of BE to EAC [9].
The aim of our study was to perform a systematic review and meta-analysis to evaluate the efficacy of fundoplication compared to medical therapy in inducing histopathologic regression of metaplasia or low-grade dysplasia (LGD) and preventing progression of BE to high-grade dysplasia (HGD) or EAC. Identifying the optimal treatment modality for patients with BE will ultimately reduce the morbidity and mortality associated with dysplasia and EAC.
Materials and methods
Search strategy
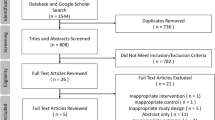
The medical librarian (JK) conducted comprehensive searches in MEDLINE (via Ovid), EMBASE (Ovid), Scopus, Web of Science Core Collection, and Cochrane Library (Wiley) on December 16, 2019 and was updated on February 22, 2021 to assess for additional studies. Search terms included “Barrett’s Esophagus OR esophageal cancer OR esophageal neoplasms” AND “fundoplication OR Nissen OR Dor OR Toupet OR Belsey Mark IV”. No language or date limits were applied. Refer to the appendices for full-text search strategies (Table S1). A total of 7231 results were retrieved and when all duplicates were removed, 3218 unique results remained for the initial title and abstract screening. In addition to subscription databases, the research team searched Google Scholar and evaluated the first 200 results for inclusion. This has been previously demonstrated as an appropriate number of results to screen since there is high overlap between Web of Science and Google Scholar [10]. The research team also reviewed bibliographies from included studies.
Study design and definition of outcomes
Inclusion criteria was defined as studies containing both surgery and medical therapy as comparators, studies with greater than five participants, studies evaluating individuals aged ≥ 18 years, diagnosis of BE made prior to treatment, and primary surgery was fundoplication. Animal studies, duplicate studies, studies published solely as abstracts, studies where patients had prior anti-reflux surgery, studies where full text could not be translated to English accurately, and studies that did not evaluate our primary outcomes were excluded. Participants were included in the surgical treatment group regardless of whether or not they previously received medical treatment.
The primary outcomes of interest included evaluating differences in histopathologic regression and disease progression in patients with BE undergoing either surgical or medical therapy.
Disease regression was defined as histopathologic improvement of metaplasia or LGD. Disease progression was defined as histopathologic progression of metaplasia to LGD, HGD, or EAC or by progression of LGD to HGD, and EAC. Secondary outcomes were to evaluate differences in morbidity or mortality between the two interventions.
Ethical approval
Due to the nature of this review, ethical approval was not required.
Data extraction
Titles and abstracts were screened by one author (HW), and two authors (HW and UJ) subsequently screened full-text articles. Discrepancies were resolved by consensus.
Pertinent data were collected from selected trials using an excel spreadsheet by one reviewer (HW) and verified by a second reviewer (WS). Included studies were then assessed for methodological quality and bias using the Cochrane Risk of Bias tool for randomized trials [11] and the MINORS tool for non-randomized trials [12].
Statistical analysis
Descriptive categorical data were expressed as percentages and continuous data were expressed as weighted means where appropriate. Meta-analysis was used to evaluate the odds of histopathologic BE regression and progression between interventions. Estimated effects were calculated using RevMan 5.4.1 software with a Mantel–Haenszel random-effects model. Heterogeneity was quantified by the I2 statistic: (1) low = 25%; (2) moderate = 50%; and (3) high = 75% [10]. Tests for statistical significance were two-tailed with significant p-values defined as < 0.05 a priori.
Results
Study selection
Preliminary database search of the literature yielded 3218 articles (Fig. 1). After initial screening of titles and abstracts, 157 studies underwent full-text assessment for eligibility. Nine manuscripts [13,14,15,16,17,18,19,20,21] met inclusion criteria and were included in the final systematic review. The included manuscripts comprised one randomized trial, seven prospective cohort studies, and one retrospective cohort study. Of those, only six studies were included in the meta-analysis [14, 17,18,19,20,21] as three [15,16,17] did not provided sufficient raw data to conduct analysis on our primary outcomes of interest.
Baseline demographics of study population
A total of 1360 patients were identified within the nine studies included (Table 1). Of those, 890 patients (65%) received medical therapy while 470 patients (35%) received surgical therapy. Evaluation of basic characteristics revealed a male predominance in both surgical (n = 275, 65%) and medical groups (n = 555, 65%). Mean weighted ages were 51.5 years for surgical patients and 58.3 years for medical patients. Only one study by Markar et al. provided a description of smoking and obesity prevalence while no studies provided information on other significant comorbidities like diabetes, hypertension, or alcohol use disorder [15].
Of the nine included studies, eight described the criteria and methodology used for the diagnosis of BE [13, 14, 16,17,18,19,20,21]. The majority of studies (n = 7) diagnosed BE based on histologic confirmation of intestinal metaplasia above the gastroesophageal junction. The remaining study by Attwood et al. [13] did not explicitly define the histologic diagnostic criteria. The Seattle protocol was followed in four of the included studies [14, 16, 18, 20, 22]. The remaining three studies described their protocol as a modification of the Seattle protocol, taking biopsies every 1–2 cm but did not specify whether they included four quadrants at every level [17, 19, 21].
Medical management included PPIs (n = 807, 91%; 6 studies), H2 blockers (n = 40, 4%; 3 studies), and combination therapy (n = 43, 5%; 1 study). Nissen fundoplication was the most commonly performed type of fundoplication (n = 265, 56%%; 7 studies) followed by Collis-Belsey (n = 14, 3%; 1 study), Toupet (n = 3, 0.6%; 2 studies), Hill (n = 2, 0.4%; 1 study), hemi-fundoplication (n = 1, 0.2%; 1 study), Collis-Nissen (n = 1, 0.2%; 1 study), and unspecified anti-reflux procedure (n = 185, 39%, 2 studies). The follow-up period ranged from 1.5 to 7 years in all included studies, and 1.5 to 6 years in the six studies included in the meta-analysis.
Histopathologic regression and progression associated with surgical and medical management of BE
The presence of dysplasia prior to surgery was reported by five studies [14, 16,17,18, 20] with patients receiving surgical intervention more likely to have low-grade dysplasia (n = 66, 28% surgical intervention vs n = 36, 18% medical intervention). Patients with medical intervention, on the other hand, were more likely to have non-dysplastic BE (medical intervention, n = 159, 82% vs. surgical intervention, n = 168, 72%).
Higher rates of regression and lower rates of progression in the surgical group were observed in the four studies [14, 17, 18, 20] that separated outcomes based on baseline dysplasia status. Regression of metaplasia in participants with nondysplastic BE occurred in 40.0% (n = 10) of medical participants and 39.1% (n = 27) of surgical participants. In patients with baseline LGD, 55.6% (n = 20) of medical participants and 83.3% (n = 55) of surgical participants had regression of LGD. Progression of metaplasia in nondysplastic BE participants occurred in 12.3% (n = 8) of those receiving medical treatment and 6.6% (n = 8) of those receiving surgery. Participants with baseline LGD showed progression in 13.9% (n = 5) of those receiving medical treatment and 4.5% (n = 3) of those receiving surgery.
Overall, histopathologic regression and progression outcomes favored patients receiving surgical intervention. Regression of metaplasia/LGD occurred in only 24.7% (n = 37) of those receiving medical therapy versus 43.5% (n = 104) of surgery patients. Similarly, progression to dysplasia/EAC was more common in the medical group (n = 12, 9.2%) than in those receiving surgery (n = 239, 4.6%).
Meta-analysis of regression and progression for surgical and medical management of BE
Meta-analysis revealed that fundoplication was associated with improved odds of histopathologic metaplasia regression (OR 4.38; 95% CI 2.28–8.42; p < 0.00001; 6 studies [14, 17,18,19,20,21]; I2 = 0%) in comparison to standard medical therapy alone (Fig. 2). Evaluation of disease progression further revealed that fundoplication was also associated with decreased odds of dysplasia progression (OR 0.34; 95% CI 0.12–0.96; p = 0.04; 6 studies [14, 17,18,19,20,21], I2 = 0%) (Fig. 3).
Morbidity and mortality associated with surgical and medical interventions
Of the five studies [17,18,19,20,21] that commented on surgical complications, only Parrilla et al. [17] reported a major complication, with one patient requiring splenectomy. They also described mild complications including inability to belch or vomit (n = 13, 22%) and mild and transitory postoperative dysphagia (n = 17, 29%) that disappeared spontaneously. Rossi et al. reported mild postoperative complications, primarily dysphagia and gas-bloat syndrome. These complications, however, were rare and transient. No deaths were attributed to either intervention in any study. No adverse effects were reported with medical therapy.
Risk of bias for included studies
The single randomized controlled trial was assessed using the Cochrane Risk of Bias Tool (Appendix 1; Table S2). Parrilla et al. suffered from moderate risk of bias due to the ethical need to not blind participants and study personnel. This is somewhat mitigated by the evaluation of objective endpoints. Additionally, an unclear risk of bias was also noted for Parrilla et al. due to the exclusion of 12 participants who refused endoscopic follow-up.
The eight non-randomized trials were assessed for bias and methodology using the Methodological index for non-randomized studies (MINORS) criteria (Appendix 1; Table S3). No study met ideal criteria as all studies lacked a prospective calculation of study size. Further, the majority of studies lacked inclusion of consecutive patients and baseline equivalence of treatment groups.
Discussion
This study is the first to systematically evaluate differences in regression of metaplasia/LGD and progression of dysplasia/EAC in patients with BE receiving either surgical or medical management. Our findings demonstrate that both odds of histopathologic regression and disease progression were improved in patients receiving fundoplication compared to those receiving medical therapy (Table S1).
The development of BE and dysplasia has been suggested to be a two-step process [23] with the first step characterized by the transition of esophageal squamous epithelium to cardiac epithelium in response to pathologic GERD. The pathophysiology of GERD begins with gastric distension leading to relaxation of the lower esophageal sphincter and reflux of gastric contents into the distal esophagus, inducing cell transformation [6, 23, 24]. Objective markers of GERD, including an incompetent lower esophageal sphincter, hiatal hernias, increased esophageal acid exposure on 24-h pH monitoring, and erosive esophagitis are correlated with the transition to cardiac mucosa [25]. Prolonged exposure to both refluxed acid and the bile salts creates an inflammatory milieu, promoting the second step of development of BE-intestinal differentiation [23, 26]. Ongoing inflammation in this mucosa results in increased proliferation and decreased apoptosis, allowing for the accumulation of genetic abnormalities and ultimately the development of dysplasia in BE.
One proposed explanation for the improved outcomes observed with surgical intervention is that fundoplication reinforces the lower esophageal sphincter and creates a definitive mechanical barrier against all refluxed contents [27]. Both duodenal bile salt and gastric acid reflux are strong contributors to the development of metaplasia and the cellular atypia observed in BE [26, 28]. Yet, PPIs act solely through preventing gastric acid secretion by permanently inhibiting the hydrogen potassium ATPase proton pump on the luminal border of gastric parietal cells [6, 23, 29]. Failure to protect from bile reflux may be one explanation for why patients receiving PPI therapy are less likely to achieve complete response in their reflux symptoms. This suggests that the definitive mechanical barrier provided by fundoplication is perhaps more effective in facilitating BE disease regression and progression by addressing both bile salt and acid reflux contents which incite intestinal differentiation. Although fundoplication carries inherent perioperative risks that are not seen with medical treatment, morbidity, and mortality rates associated with the procedure are relatively low, resulting in an acceptable risk profile for many patients [30].
It is also important to acknowledge that anatomic defects like hiatal hernias (HH) are increasingly common and less likely to respond to medical therapy alone. Hiatal hernias left untreated also tend to enlarge over time, exacerbating the degree of reflux of gastroduodenal contents through the LES. Failure to identify such defects prior to initiation of PPI therapy may be one possible explanation for our findings. It has been reported that greater than 90% of patients with BE have concurrent HH [31, 32]. Indeed, HH are strongly associated with presence of BE despite their relationship to development of high-grade dysplasia and esophageal adenocarcinoma being less clear [32]. PPI trials are often started empirically prior to evaluation of HHs due to delays in obtaining confirmatory upper endoscopy or contrast swallowing studies. Delays in identifying these anatomic defects which are effectively addressed by surgical intervention may lead to increased BE disease progression due to potentially futile PPI trials.
Another factor which may contribute to the superior response of BE to surgical intervention is lack of medication adherence. GERD symptoms fluctuate and are often associated with frequency, type, and size of meals. It is not uncommon for patients to take medications on an as needed basis when they encounter inciting GERD factors [33]. Estimates of the medication possession adherence of PPIs for patients with GERD range from 55–84%, although patients with BE have been shown to be more compliant with their PPI regime than patients with uncomplicated GERD [33,34,35]. However, this leaves those with undiagnosed asymptomatic disease at higher risk of disease progression. In this regard, surgical correction may provide increased benefit by avoiding challenges with medication compliance associated with underlying asymptomatic disease.
Other studies have demonstrated similar superior outcomes when comparing surgical to medical intervention in patients with BE. A meta-analysis by Ouda et al. [9] found that surgical treatment decreased the incidence rate ratio of EAC in patients with BE compared to medical treatment, which became statistically significant only when including studies published after the year 2000. There was no difference in outcomes found in patients diagnosed with GERD. This highlights the importance of investigating outcomes specific to patients with BE, as the increased disease severity likely affects response to treatment. Corey et al. [36] published a meta-analysis in 2003 that found no significant difference in the rates of esophageal adenocarcinoma in patients with BE receiving either surgical or medical therapy. Overall, these studies are limited in that they only evaluated progression to EAC and did not evaluate the effect of intervention on BE regression.
While our findings indeed suggest that BE regression and lack of progression are superior in patients receiving fundoplication, it may well be possible that instances of disease recurrence may occur decades after the index surgical procedure. Unfortunately, the follow-up of our included studies ranged from 1.5 to 7 years which limits us from evaluating the long-term efficacy of these therapies and from providing recommendations on long-term surveillance. Nonetheless, given the chronic life-long cancer risk associated with BE, we believe that ongoing surveillance regardless of method of reflux control remains critical to prevent the morbidity and mortality of BE.
Our study is limited by the inherent limitations of systematic reviews. We found only six studies with adequate data for inclusion in our meta-analysis. Of these, the majority did not adequately describe their population, specifically regarding BMI, alcohol, tobacco use, or hiatal hernias which are known risk factors for BE and may confound our results and limit generalizability [4]. Older studies also included LGD patients who underwent fundoplication and did not undergo current standard of care therapies like endoscopic mucosal resection. Although all studies utilized acid suppression therapy, the class of drugs used was not consistent across studies, making it difficult to compare if fundoplication is superior to PPIs or H2 blockers specifically. Given the heterogeneity in endoscopic protocols used to evaluate BE, and the potential for sampling and diagnostic errors even with standardized methods, our findings should be interpreted with caution. Lastly, given the nature of our study design, we are not able to identify the specific mechanisms responsible for our findings. Together, these limitations preclude a recommendation of which specific patients would most benefit from surgical intervention, the optimal timing for fundoplication, or for timing of post-surgical endoscopic surveillance.
Despite these limitations, our study is novel because it is the first to identify the superiority of surgical intervention in optimizing both histopathologic regression and disease progression in patients with BE. These findings provide evidence supporting the increased adoption for surgical management of BE and suggest that patients with BE should have prompt workup and surgical referral at the time of diagnosis. Lastly, additional data are needed to evaluate the efficacy of fundoplication on long-term BE outcomes as well as to help guide the development of post-surgical screening guidelines.
Conclusion
Surgical management of BE was associated with improved odds of histopathologic disease regression and less progression to dysplasia compared to standard medical therapy alone. Further large randomized controlled trials which directly compare medical management and surgical intervention are required to further delineate the optimal delivery and timing of these interventions.
References
Hvid-Jensen F, Pedersen L, Drewes AM, Sørensen HT, Funch-Jensen P (2011) Incidence of adenocarcinoma among patients with Barrett’s esophagus. N Engl J Med 365:1375–1383. https://doi.org/10.1056/NEJMoa1103042
Marques de Sá I, Pereira AD, Sharma P, Dinis-Ribeiro M (2020) Systematic review of the published guidelines on Barrett’s esophagus: should we stress the consensus or the differences? Dis esophagus Off J Int Soc Dis Esophagus. https://doi.org/10.1093/dote/doaa115
Kestens C, Offerhaus GJA, van Baal JWPM, Siersema PD (2016) Patients with Barrett’s esophagus and persistent low-grade dysplasia have an increased risk for high-grade dysplasia and cancer. Clin Gastroenterol Hepatol Off Clin Pract J Am Gastroenterol Assoc 14:956-962.e1. https://doi.org/10.1016/j.cgh.2015.12.027
Canadian Cancer Society (2019) Survival Statistics for Esophageal Cancer. Available at https://www.cancer.ca/en/cancer-information/cancer-type/esophageal/prognosis-and-survival/survival-statistics/?region=qc. Accessed 16 Jan 2020
Runge TM, Abrams JA, Shaheen NJ (2015) Epidemiology of Barrett’s esophagus and esophageal adenocarcinoma. Gastroenterol Clin North Am 44:203–231. https://doi.org/10.1016/j.gtc.2015.02.001
Maret-Ouda J, Markar SR, Lagergren J (2020) Gastroesophageal reflux disease: a review. JAMA 324:2536–2547. https://doi.org/10.1001/jama.2020.21360
Society of American Gastrointestinal and Endoscopic Surgeons (2020) Survival Statistics for Esophageal Cancer. https://www.sages.org/publications/guidelines/guidelines-for-surgical-treatment-of-gastroesophageal-reflux-disease-gerd/. Accessed 3 Dec 2020
Attwood SEA, Lundell L, Ell C, Galmiche J-P, Hatlebakk J, Fiocca R, Lind T, Eklund S, Junghard O (2008) Standardization of surgical technique in antireflux surgery: the LOTUS trial experience. World J Surg 32:995–998. https://doi.org/10.1007/s00268-007-9409-4
Maret-Ouda J, Konings P, Lagergren J, Brusselaers N (2016) Antireflux surgery and risk of esophageal adenocarcinoma: a systematic review and meta-analysis. Ann Surg 263:251–257. https://doi.org/10.1097/SLA.0000000000001438
Haddaway NR, Collins AM, Coughlin DKS (2015) The role of google scholar in evidence reviews and its applicability to grey literature searching. PLoS ONE. https://doi.org/10.1371/journal.pone.0138237
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (2020) Cochrane handbook for systematic reviews of interventions version 6.1. Available at www.training.cochrane.org/handbook. Accessed 15 Dec 2020
Higgins JPT, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560. https://doi.org/10.1136/bmj.327.7414.557
Attwood SE, Barlow AP, Norris TL, Watson A (1992) Barrett’s oesophagus: effect of antireflux surgery on symptom control and development of complications. Br J Surg 79:1050–1053. https://doi.org/10.1002/bjs.1800791021
Gurski RR, Peters JH, Hagen JA, DeMeester SR, Bremner CG, Chandrasoma PT, DeMeester TR (2003) Barrett’s esophagus can and does regress after antireflux surgery: a study of prevalence and predictive features. J Am Coll Surg 196:703–706. https://doi.org/10.1016/S1072-7515(03)00147-9
Markar SR, Arhi C, Leusink A, Vidal-Diez A, Karthikesalingam A, Darzi A, Lagergren J, Hanna GB (2018) The influence of antireflux surgery on esophageal cancer risk in England: national population-based cohort study. Ann Surg 268:861–867. https://doi.org/10.1097/SLA.0000000000002890
Oberg S, Wenner J, Johansson J, Walther B, Willén R (2005) Barrett esophagus: risk factors for progression to dysplasia and adenocarcinoma. Ann Surg 242:49–54. https://doi.org/10.1097/01.sla.0000167864.46462.9f
Parrilla P, Martínez de Haro LF, Ortiz A, Munitiz V, Molina J, Bermejo J, Canteras M (2003) Long-term results of a randomized prospective study comparing medical and surgical treatment of Barrett’s esophagus. Ann Surg 237:291–298. https://doi.org/10.1097/01.SLA.0000055269.77838.8E
Rossi M, Barreca M, de Bortoli N, Renzi C, Santi S, Gennai A, Bellini M, Costa F, Conio M, Marchi S (2006) Efficacy of Nissen fundoplication versus medical therapy in the regression of low-grade dysplasia in patients with Barrett esophagus: a prospective study. Ann Surg 243:58–63. https://doi.org/10.1097/01.sla.0000194085.56699.db
Trentino MDP, Trifero MDM, Marzullo MDA, Arcese MDR, Correnti MDFS, Pappalardo MDG, Castrini MDG (1990) Biomarkers’ evaluation for early neoplastic degeneration in Barrett’s esophagus. A preliminary report on 12 cases. Dis Esophagus 3:29–36. https://doi.org/10.1093/dote/3.1.29
Tolone S, Limongelli P, Romano M, Federico A, Docimo G, Ruggiero R, Brusciano L, Del Genio G, Docimo L (2015) The patterns of reflux can affect regression of non-dysplastic and low-grade dysplastic Barrett’s esophagus after medical and surgical treatment: a prospective case-control study. Surg Endosc 29:648–657. https://doi.org/10.1007/s00464-014-3713-5
Zaninotto G, Parente P, Salvador R, Farinati F, Tieppo C, Passuello N, Zanatta L, Fassan M, Cavallin F, Costantini M, Mescoli C, Battaglia G, Ruol A, Ancona E, Rugge M (2012) Long-term follow-up of Barrett’s epithelium: medical versus antireflux surgical therapy. J Gastrointest Surg Off J Soc Surg Aliment Tract 16:5–7. https://doi.org/10.1007/s11605-011-1739-8
Spechler SJ, Sharma P, Souza RF, Inadomi JM, Shaheen NJ, Association AG (2011) American gastroenterological association technical review on the management of Barrett’s esophagus. Gastroenterol 140:e18–e13. https://doi.org/10.1053/j.gastro.2011.01.031
Oh DS, Demeester SR (2010) Pathophysiology and treatment of Barrett’s esophagus. World J Gastroenterol 16:3762–3772. https://doi.org/10.3748/wjg.v16.i30.3762
Tobey NA, Hosseini SS, Argote CM, Dobrucali AM, Awayda MS, Orlando RC (2004) Dilated intercellular spaces and shunt permeability in nonerosive acid-damaged esophageal epithelium. Am J Gastroenterol 99:13–22. https://doi.org/10.1046/j.1572-0241.2003.04018.x
Oberg S, Peters JH, DeMeester TR, Chandrasoma P, Hagen JA, Ireland AP, Ritter MP, Mason RJ, Crookes P, Bremner CG (1997) Inflammation and specialized intestinal metaplasia of cardiac mucosa is a manifestation of gastroesophageal reflux disease. Ann Surg 226:522–532. https://doi.org/10.1097/00000658-199710000-00013
Nasr AO, Dillon MF, Conlon S, Downey P, Chen G, Ireland A, Leen E, Bouchier-Hayes D, Walsh TN (2012) Acid suppression increases rates of Barrett’s esophagus and esophageal injury in the presence of duodenal reflux. Surgery 151:382–390. https://doi.org/10.1016/j.surg.2011.08.021
Jürgens S, Meyer F, Spechler SJ, Souza R (2012) The role of bile acids in the neoplastic progression of Barrett’s esophagus - a short representative overview. Z Gastroenterol 50:1028–1034. https://doi.org/10.1055/s-0032-1312922
Kauer WK, Peters JH, DeMeester TR, Ireland AP, Bremner CG, Hagen JA (1995) Mixed reflux of gastric and duodenal juices is more harmful to the esophagus than gastric juice alone. The need for surgical therapy re-emphasized. Ann Surg 222:525–533. https://doi.org/10.1097/00000658-199522240-00010
Stein HJ, Kauer WKH, Feussner H, Siewert JR (1998) Bile reflux in benign and malignant Barrett’s esophagus: effect of medical acid suppression and Nissen fundoplication. J Gastrointest Surg 2:333–341. https://doi.org/10.1016/S1091-255X(98)80072-3
Niebisch S, Fleming FJ, Galey KM, Wilshire CL, Jones CE, Litle VR, Watson TJ, Peters JH (2012) Perioperative risk of laparoscopic fundoplication: safer than previously reported—analysis of the American college of surgeons national surgical quality improvement program 2005 to 2009. JACS 215:61–68. https://doi.org/10.1016/j.jamcollsurg.2012.03.022
Cameron AJ (1999) Barrett’s esophagus: prevalence and size of hiatal hernia. Am J Gastroenterol 94:2054–2059. https://doi.org/10.1111/j.1572-0241.1999.01277.x
Roman S, Kahrilas PJ (2015) Mechanisms of Barrett’s oesophagus (clinical): LOS dysfunction, hiatal hernia, peristaltic defects. Best Pract Res Clin Gastroenterol 29:17–28. https://doi.org/10.1016/j.bpg.2014.11.002
Van Soest EM, Siersema PD, Dieleman JP, Sturkenboom MCJM, Kuipers EJ (2006) Persistence and adherence to proton pump inhibitors in daily clinical practice. Aliment Pharmacol Ther 24:377–385. https://doi.org/10.1111/j.1365-2036.2006.02982.x
Hungin APS, Hill C, Molloy-Bland M, Raghunath A (2012) Systematic review: patterns of proton pump inhibitor use and adherence in gastroesophageal reflux disease. Clin Gastroenterol Hepatol Off Clin Pract J Am Gastroenterol Assoc 10:109–116. https://doi.org/10.1016/j.cgh.2011.07.008
El-Serag HB, Fitzgerald S, Richardson P (2009) The extent and determinants of prescribing and adherence with acid-reducing medications: a national claims database study. Am J Gastroenterol 104:2161–2167. https://doi.org/10.1038/ajg.2009.312
Corey KE, Schmitz SM, Shaheen NJ (2003) Does a surgical antireflux procedure decrease the incidence of esophageal adenocarcinoma in Barrett’s esophagus? A meta-analysis. Am J Gastroenterol 98:2390–2394. https://doi.org/10.1111/j.1572-0241.2003.08702.x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Ms. Wilson and Kung, and Drs. Mocanu, Sun, Deng, Jogiat, Switzer, Wong and Karmali have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.



Rights and permissions
About this article
Cite this article
Wilson, H., Mocanu, V., Sun, W. et al. Fundoplication is superior to medical therapy for Barrett’s esophagus disease regression and progression: a systematic review and meta-analysis. Surg Endosc 36, 2554–2563 (2022). https://doi.org/10.1007/s00464-021-08543-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-021-08543-6