Abstract
Introduction
Pancreatic rest (PR) is an ectopic pancreatic lesion that is usually found incidentally on endoscopy or surgery. While most lesions do not have clinical significance, some patients are symptomatic and rarely, PR can predispose to malignancy. With the growing popularity of bariatric surgery, it has been unclear how to manage PR found on screening endoscopies, prior to bariatric surgery. Through review of the current literature, we propose an algorithm for clinicians to evaluate and manage PR found on screening endoscopies prior to bariatric surgery.
Methods
We performed a literature search in PubMed pertaining to PR, clinical characteristics, risk of malignant transformation, endoscopic characteristics, histological descriptions, and resection techniques. Using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA), we found 33 published articles from 2001 to 2019, including case reports, case series, retrospective cohorts, and a review paper.
Results
PR is commonly found incidentally in the gastric antrum. Larger lesions have a higher risk of being symptomatic or predisposing to malignant transformation. Endoscopic ultrasound (EUS) can assist in the diagnosis of PR and guide resection technique. Certain histological characteristics, such as Heinrich class, grading of neoplasia, and genetic alterations, can determine malignancy risk of PR. Resection technique, either endoscopically or surgically, should be based on lesion size, depth of wall invasion, and the endoscopists’ level of skill in endoscopic resection.
Conclusions
Proper evaluation and treatment of PR should be considered because of the risk for symptoms and malignant transformation. Symptomatic lesions and those at risk for malignant transformation should be considered for resection. EUS can guide the diagnosis and type of resection, either endoscopically through EMR or ESD or surgically through sleeve gastrectomy or Roux-en-Y gastric bypass (RYGB).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Pancreatic rest (PR), or heterotopic pancreas, is an uncommon finding of an ectopic pancreatic lesion that is usually found incidentally on endoscopy or surgery, or between 2 and 15% of all autopsies [1]. There is a slight male predominance (64% males), can occur in all age groups, and are predominantly found in the gastric antrum [2]. In the majority of cases, they pose no clinical significance as most patients are asymptomatic [2]. However, they can predispose to complications, such as pancreatitis, gastrointestinal obstruction, bleeding, anemia, abdominal pain, and nausea. In addition, there is a rare risk of malignant transformation of about 0.7 to 1.8% [3, 4].
With the increase in morbid obesity rates worldwide, bariatric surgery has been shown to be the most effective treatment in terms of weight loss and improving obesity-related comorbidities [5]. Sleeve gastrectomy (SG) has increased from 0 to 37% of all bariatric surgeries worldwide from 2003 to 2013 [5], and RYGB comprises of 45% of bariatric surgeries worldwide in 2013 [5]. In addition, PR can be found in 0.6% of bariatric surgeries [6]. After bariatric surgery, the potential complications and risk of malignant transformation in this population with PR is much more significant and difficult to manage given the need for repeat surgery and difficulty in surveillance or endoscopic resection in the setting of altered anatomy. Screening esophagogastroduodenoscopies (EGDs) are typically performed prior to bariatric surgery in order to evaluate for ulcers, erosions, hiatal hernias, or any other concerning lesions [7].
Currently, there is no guidance on how to evaluate patients with potential PR lesions prior to bariatric surgery. This manuscript performs a thorough analysis of the most current literature regarding incidence, diagnostic characteristics, potential complications, risk of malignant transformation, and resection techniques of PR. We aim to create an algorithm for clinicians to evaluate when PR should be resected and if so, what resection method should be utilized.
Methods
We performed a literature search in PubMed using keywords, “pancreas rest,” “heterotopic pancreas,” “pancreatic heterotopias,” “ectopic pancreas,” “pancreatic intraepithelial neoplasia,” “pancreatic ductal adenocarcinoma,” “Henreich system.” Given the limited studies published on heterotopic pancreas, we included case reports, case series, systematic reviews, retrospective reviews, and prospective studies in a list of eligible publications. We also identified articles from the references of the articles from our search results in order to expand our list of publications. IRB approval was not needed, so was not obtained because the study is a review of existing literature and does not involve participation of patients or patient chart review.
We filtered the publications which described the endoscopic, imaging, and histological features of HP, the clinical manifestations and risk of malignant transformation, as well as articles that describe various resection techniques for PR. For articles that include pancreatic ductal adenocarcinoma, we only considered them if the malignancy was attributed to heterotopic pancreas.
We extracted data on demographics of patients with pancreatic rests, endoscopic, histological, and genetic/molecular characteristics, features at risk for malignant transformation, and outcomes of various resection endoscopic and surgical techniques. Supplementary Table 1 contains the compilation of all studies used in the review with descriptions of key points.
Results
Literature search
Figure 1 shows our Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) diagram. For our inclusion criteria, we searched for studies that were published in the year 2000 and afterwards. We searched for publications which discussed gastric PR, contained English text, were case reports, review papers, retrospective studies, and prospective studies. We excluded publications which were image reports, video reports, editorials, and contained pediatric cases. We also did not include studies which the full text was not available. Studies were reviewed by a single reviewer. Based on the literature search, we initially found 51 relevant published articles through literature search and 34 published articles from references among the literature search. After removing duplicate articles, we found 70 articles, which we removed 12 articles due to various reasons, such as not being able to find the full text or that the text was in a non-English language. Then we read through the full text and excluded 18 articles that did not pertain to gastric PR, were image or video reports, or small case reports which did not contribute much relevant information to our review. There were 7 articles which were referenced in our review but were not included in analysis for our algorithm.
Finally, we found 33 published articles relating to PR, published from 2001 to 2019. These included 7 case reports, 6 case series, 19 retrospective cohorts, and 1 review paper. 20 articles contained data on demographics and clinical characteristics of HP. 20 articles studied endoscopic ultrasonographic diagnosis. 15 articles discussed histological features. 25 articles discussed various resection techniques. There were no prospective studies given the rarity of PR. The grading of evidence is low given the lack of prospective randomized studies and the limited evidence on the topic of PR.
Demographics, endoscopic appearance, and clinical symptoms
Table 1 lists the demographic, endoscopic, and clinical characteristics of PR from the literature search. The pathogenesis of PR is unknown and there are no studied risk factors for its development. PR can be found from 0.25% of surgical explorations to 14% of autopsies [8]. One third of PR are found incidentally during procedures [9]. It can be seen in 0.6% of cases of bariatric surgeries [8] and 0.9% of gastrectomies [10]. Given the low incidence of PR found during surgery compared to those found during autopsies, a majority of PR may be missed, but exact numbers have not been discussed in existing literature. They are usually found in the stomach in 25–38% of cases, especially in the antrum, followed by the duodenum [10]. It can affect any age range, but is more common in ages 30 to 50 years and predominantly affects males [10]. Most patients are asymptomatic [8], but patients with symptoms usually had larger lesions [10]. However, when symptomatic, they can commonly present with nausea, vomiting, epigastric pain, and weight loss [11]. Rarely can it cause a gastric outlet obstruction [12]. In addition, malignant transformation can occur in 0.7% to 1.8% of cases [13,14,15], which demonstrates that it may be important to evaluate PR when noted.
Endoscopic ultrasonography characteristics
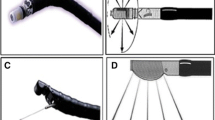
The endoscopic ultrasonographic characteristics of PR, as described in the literature, is listed in Table 2. Endoscopically, PR often appear as subepithelial lesions, primarily in the gastric antrum, followed by gastric body, then duodenum [10]. Size ranges from under 1 cm to over 4 cm [16,17,18]. Central dimpling, or umbilication, can sometimes be seen, which corresponds to the opening of the HP duct [19]. However, central dimpling is not found frequently and is usually seen in larger lesions [20]. Figure 2 demonstrates a typical PR lesion found on endoscopy.
However, endoscopic mucosal forceps biopsies are limited in diagnostic yield, often showing normal overlying mucosa [16, 21, 22], and EUS can help distinguish benign from malignant subepithelial lesions, which would guide endoscopic resection [23]. PR is a subepithelial lesion that mostly commonly arises from the submucosa (15 to 70%), followed by the muscularis propria (3 to 60%), then subserosa (0 to 10%) [18, 19, 24,25,26,27]. EUS can be helpful for diagnosis, given certain features. Typical ultrasound features include heterogeneous echogenicity with hypoechoic acinous tissue and anechoic ductal features [24], as well as indistinct borders [20]. However, lesions originating from the muscularis propria, as compared to those arising from the submucosa, may contain more of a homogenous pattern with hypoechoic features [19]. With added FNA, the sensitivity and specificity of cytological diagnosis can be 83.3% and 100%, respectively [12]. In addition, EUS can guide resection by determining risk of bleeding, extent of resection needed [28].
Histopathology and genetic/molecular marker characteristics
Table 3 details the histopathologic characteristics of PR from a review of the literature. Histolopathological examination of PR contains a mixture of pancreatic acini, ducts, and islets of Langerhans with normal overlying gastric mucosa and can involve the submucosa, muscularis propria, or subserosa [10]. The growth pattern can be either pushing or infiltrative, and there can be mucosal erosion, atrophy, as well as interstitial fibrosis or lymphoid cuffs [13].
Heinrich in 1909, then Gaspar-Fuentes in 1973 classified PR into three categories. Type 1 heterotopia consisted of normal pancreatic tissue with acini, ducts, and islet cells. Type II heterotopias showed pancreatic ducts only, and Type III only contained acinar tissue. Type IV is comprised of only islet cells [10]. The most common type is Type II heterotopia [22]. Most studied malignancies (42 to 58%) arose from Type I heterotopias, with the most common type being adenocarcinoma [1, 4].
Guillou, et al. proposed criteria for carcinoma to be arising from PR: (1) presence of neoplasm located in/or in the proximity of the PR site; (2) concurrent presence and continuity of normal ectopic pancreatic tissue and adenocarcinoma; (3) PR tissue with normal histological structure [29]. Most malignant transformation occurs with gastric PR and lesions were mostly greater than 4 cm [4]. Reported cases of adenocarcinomas arising from PR showed histological signs of desmoplastic stroma with most moderately differentiated, a few poorly differentiated [13].
Histopathological grading of neoplasia can be graded from Pancreatic Intraepithelial Neoplasia-1 (PanIN-1) to PanIN-3. Having PanIN or IPMN is associated with larger lesions, deeper wall involvement, and infiltrative growth. While low-grade PanINs can occur in normal pancreas tissue, high-grade PanINs contain higher number or genetic alterations, such as KRAS mutation, p53 accumulation, cyclin D1 overexpression, and loss of p16 expression, predisposing to malignancy [30]. In addition, KRAS mutation shows good evidence that the malignancy did arise from PR, so it may be helpful to perform KRAS mutational and SMAD4/DPC4 immunohistochemical studies in order to determine malignancy risk of PR [13]. Regarding IPMNs, which are usually larger or equal to 1 cm, progressive genetic alterations of KRAS, CDKN2A, TP53 may cause changes from low-grade to high-grade IPMN, then to pancreatic adenocarcinoma [13].
Endoscopic and surgical resection techniques
Table 4 reviews endoscopic and surgical resection modalities for PR. Usually asymptomatic lesions can be surveyed, but with the risk of malignancy and development of minimally invasive surgery and therapeutic endoscopy, it would be reasonable to resect lesions, which may develop into malignancy. It may be reasonable to resect concerning incidental lesions, such as high-risk PR, during bariatric procedures without increasing the post-operative complication rate [8]. Resection can serve as a diagnostic approach [4].
It may be reasonable to endoscopically resect gastric PR lesions given the low complication rate of EMR/ESD and difficulty of surveillance for post-bariatric surgery patients [18]. Park, et al. classified lesions that were of superficial type, which originate from the deep mucosa and/or submucosa, and those of the deep type, which originate from the submucosa and muscularis propria with or without extension into the subserosal or serosal layer, and proposed that superficial type lesions were appropriate for endoscopic resection [20]. In high-volume ESD centers, ESDs have been performed for small gastric lesions (≤ 20 mm) that originate from the muscularis propria with en bloc curative resection and no complications [19]. ESD with en bloc resection have also been performed for PR less than 3.5 cm in diameter [31]. The AGA Institute Clinical Practice states that indications for gastric ESD include moderately and well-differentiated superficial cancers ≤ 3 cm with ulceration or contain early submucosal invasion [32]. However, if lesions contained more vessels, this may result in longer procedure time and higher risk of muscle layer injury [28]. More superficial lesions, such as those originating from the submucosa, have been successful resected by standard EMR [12] or by endoscopic band ligation snare polypectomy without complications [16, 21]. It has been proposed that if the lesion is larger than 1 cm and there is no muscularis propria involvement on EUS, then EMR can be performed [18].
Surgical wedge resection has been reportedly used to curatively resect large, over 3 cm, gastric and small bowel PR, which can involve the muscularis propria and subserosa, without any complications [8, 10]. It has been recommended that smaller sized lesions, located in the stomach, superficial lesions, more pushing than infiltrative growth can be endoscopically resected [13]. Lesions that are larger in size, with deeper wall involvement, more infiltrative growth, with interstitial fibrosis should be resected surgically [13]. Pushing growth pattern refers to a well-demarcated pushing border with surrounding fibro adipose tissue, and an infiltrative growth pattern refers to an irregular infiltrating pattern into smooth muscle [13]. For patients with obesity, a laparoscopic wedge, followed by a RYGB, has been performed without complications for large (4 cm) submucosal PR [33]. In addition, for larger muscularis propria lesions, subtotal gastectomy with Billroth II anastomosis has also been performed without complications [15].
Discussion
Algorithm
Currently, there are no guidelines in managing PR during screening EGD prior to bariatric surgery. While most PR are asymptomatic and benign, there is a possibility of symptom development and malignant transformation. In addition, endoscopic surveillance after bariatric surgery may be difficult. Figure 3 presents a guideline on how to diagnose PR on a pre-bariatric surgery EGD and how to decide on resection, depending on symptoms, size, EUS characteristics, and histopathological findings. Once resection is determined, Fig. 4 guides clinicians on choice of resection, either endoscopic or surgical.
Screening EGD
A screening EGD prior to bariatric surgery is important in identifying concerning lesions that may require management prior to surgery. On pre-bariatric surgery screening EGD, PR is often seen in the gastric antrum as a subepithelial lesion without mucosal changes. Central umbilication or dimpling may be seen in larger lesions, which corresponds an opening of a PR duct [19, 20]. Mucosal forceps biopsies are usually non-diagnostic, so do not need to be done [1, 21, 26].
Further work-up with EUS
EUS provides further characterization of the subepithelial lesion and can distinguish between benign and malignant lesions. Most PR lesions are heterogeneous, with hypoechoi cacinous tissue and anechoic ductal features [24]. The borders may be distinct [19] or indistinct [20]. However, not all PR lesions may follow these characteristic features, as some may be more homogenous, depending on the wall layer they originate from. If the lesion is characteristic of other types of subepithelial lesions, then appropriate management should follow the appropriate guidelines. PR lesions may originate from the muscularis mucosa, submucosa, muscularispropria, serosa, or a combination, which will help determine the appropriate form of resection, if needed [20].
Symptomatology and size
Once there is suspicion for an PR lesion, the clinical characteristics of the patient must be taken into consideration. While most patients are asymptomatic, PR can cause or predispose to various symptoms, such as abdominal pain, obstruction, nausea, anemia, or pancreatitis [11, 12]. If the patient is symptomatic from PR, we recommend resection for clinical symptoms.
If the patient is not symptomatic, but the lesion is large, there is a possibility that the lesion may become symptomatic later on, as larger lesions are usually symptomatic [10]. In addition, PR has a risk of malignant transformation occurs in gastric PR when they are larger than 4 cm in diameter [4]. Therefore, we propose that asymptomatic lesions that are greater or equal to 4 cm in diameter should be resected.
Histopathological guidance
If the patient with suspected PR is asymptomatic and the lesion is smaller than 4 cm in diameter, then further work-up with histopathological analysis should help determine if resection is warranted. EUS-FNA can be used to obtain cytological diagnosis with a high sensitivity and specificity [12]. Most malignant transformation has occurred from Henreich Type I heterotopias [1, 4], classified as normal pancreatic tissue containing acini, ducts, and islet cells [10]. As a result, those lesions with Henreich Class I should proceed with resection, while patients with Type II to IV lesions may reasonably proceed to bariatric surgery.
Those PR lesions with high-grade PanIN have more infiltrative growth and higher risk of progression towards malignancy [30], so they should be considered for resection. If there are no signs of high-grade PanIN, then genetic/molecular analysis should be performed to evaluate for mutations that may predispose to malignant transformation. Genetic alterations, such as KRAS mutation, p53 accumulation, cyclin D1 overexpression, loss of p16 expression [30], and SMAD4/DPC4 mutation [13] may provide a stepwise progression to adenocarcinoma, so these PR lesions should be resected. Low-grade PanIN without those mutational alterations may not need to be resected because of the negligible risk of malignant transformation.
For IPMN lesions, then genetic/molecular testing should be performed to evaluate for alterations to KRAS, CDKN2A, TP53, which may predispose to progression to high-grade IPMN and then to pancreatic adenocarcinoma (Jun et al.). For these lesions, resection should be considered. Otherwise, it may be reasonable to proceed with bariatric surgery.
Histopathological findings of cancer should appropriately be managed.
Size and the choice of resection
Once the PR lesion has been decided for resection, then there are various methods which can be used. Endoscopic methods include EMR and ESD, and surgical resection is another option. Endoscopically, current technology allows for resection of early gastric cancers and small luminal lesions with limited wall layer infiltration through either endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD). When compared to ESD, EMR can only allow en bloc resection of smaller lesions, although still limited to superficial submucosal lesions [32].
Based on size, if the lesion is equal or less than 1 cm in diameter, then EMR is appropriate [18]. These lesions can be removed either by standard lift and resect EMR [12], but it also has been successfully removed by endoscopic band ligation snare polypectomy [16, 21]. Endoscopic resection, through EMR or ESD, has had a low complication rate [18], but for ESD, the studies have been at high-volume ESD centers [18, 19]. If the lesion is greater than 1 cm but equal or less than 3.5 cm in diameter and the center is a high-volume ESD center, then ESD with en bloc resection can be considered [31, 32]. If the center is not a high-volume ESD center or if the lesion is greater than 3 cm or 3.5 cm, then surgical resection should be considered [8, 10]. This can be in the form of a laparoscopic wedge resection, followed by a RYGB [33] or by a laparoscopic sleeve gastrectomy.
Depth of invasion or wall layer involvement is another important factor to consider in terms of resection options. Based on EUS characteristics, if there is only involvement of the muscularis mucosa or limited involvement of the submucosa, then EMR can be attempted through standard techniques or band-snare polypectomy [12, 16, 18, 21]. If there is significant involvement of the submucosa or if there is involvement of the muscularis propria, then ESD can be considered if it is performed at a high-volume ESD center [12, 18, 19]. Otherwise, surgical resection would be another option [8, 10, 15]. If subserosal involvement is suspected on EUS, then surgical resection should be attempted.
Surgical resection can be in the form of a wedge resection, followed by a RYGB [33] or laparoscopic sleeve gastrectomy. If the lesion is in the lesser curvature of the stomach, then it would be appropriate to perform a subtotal gastrectomy and RYGB [33]. If the lesion is in the antrum, which is the most common location, then a subtotal gastrectomy with Billroth II anastomosis, followed by a RYGB may be considered. For lesions along the greater curvature of the stomach, then a sleeve gastrectomy may be performed to resect the lesion. If a PR is noted during a screening EGD for bariatric surgery, we would recommend the surgeon follow the appropriate algorithm as noted in Figs. 3 and 4. If the bariatric patient is intended to undergo a RYGB, we recommend the PR is appropriately treated prior to the RYGB considering the PR will likely be excluded from proper follow-up using upper endoscopy.
Given the limited evidence on the diagnosis and management of PR, there are inherently limitations to our review study. Given the single reviewer methodology of selecting publications, there is a risk of bias. Given the rarity of PR and its malignant transformation, our review paper referenced existing literature on PR consisting of mostly case reports and retrospective cohorts, which unavoidably limits the level of evidence of our algorithm to a low level of evidence. However, most of the case reports and retrospective studies are consistent in the description of PRs, their malignant potential, and the endoscopic, ultrasonographic, and pathological findings. Nonetheless, our review paper is the first to create an algorithm on management of PR before bariatric surgery.
With the rising incidence of obesity and popularity of bariatric surgery, screening EGDs will demonstrate more findings of PR. Patients with these lesions may be symptomatic, become symptomatic, and there is a possibility of malignant transformation. What to do about these lesions prior to bariatric surgery has not been discussed previously. Based on extensive literature search, we created an algorithm to guide clinicians on the approach and management of patients with PR. Symptomatic lesions should be considered for resection and those at risk for malignant transformation, such as greater size, high degree of neoplasia, and genetic mutations. EUS can aid in extent of wall layer involvement in order to determine the type of resection, either endoscopically through EMR or ESD, or by surgery, either sleeve gastrectomy or RYGB. We also recommend proper treatment evaluation and treatment of the PR if a patient is destined for a RYGB.
References
Emerson L, Layfield LJ, Rohr LR et al (2004) Adenocarcinoma arising in association with gastric heterotopic pancreas: a case report and review of the literature. J Surg Oncol 87:53–57
Dolan RV, Remine WH, Dockerty MB (1974) The fate of heterotopic pancreatic tissue. A study of 212 cases. Arch Surg 109:762–765
Yan ML, Wang YD, Tian YF et al (2012) Adenocarcinoma arising from intrahepatic heterotopic pancreas: a case report and literature review. World J Gastroenterol 18(22):2881–2884
Cazacu IM, Chavez AAL, Gonzalez GMN et al (2019) Malignant transformation of ectopic pancreas. Dig Dis Sci 64:655–668
Angrisani L, Santonicola A, Iovino P et al (2015) Bariatric surgery worldwide 2013. Obes Surg 25:1822–1832
Finnell CW, Madan AK, Temovits CA (2007) Unexpected pathology during laparoscopic bariatric surgery. Surg Endosc 21:867–869
Loewen M, Giovanni J, Barba C (2008) Screening endoscopy before bariatric surgery: a series of 448 patients. Surg Obes Relat Dis 4(6):709–712
Montalvo D, Hernandez P, Larrazaba A (2016) Unexpected ecotopic pancreatic tissue during laparoscopic bariatric surgery. Case report and literature review. Surg Obes Relat Dis 12:E87–e88
Zhang Y, Sun X, Gold JS et al (2016) Heterotopic pancreas: a clinicopathological study of 184 cases from a single high-volume medical center in China. Hum Pathol 55:135–142
Christodoulidis G, Zacharoulis D, Barbanis S et al (2007) Heterotopic pancreas in the stomach: a case report and literature review. World J Gastroenterol 13(45):6098–6100
Eisenberger CF, Gocht A, Knoefel WT et al (2004) Heterotopic pancreas—clinical presentation and pathology with review of literature. Hepatogastroenterology 51(57):854–858
Liu X, Wang G, Ge N et al (2013) Endoscopic removal of symptomatic gastric heterotopic pancreas: a report of nine cases. Surg Innov 20(6):NP40–NP46
Jun SY, Son D, Kim MJ et al (2017) Heterotopic pancreas of the gastrointestinal tract and associated precursor and cancerous lesions: systematic pathologic studies of 165 cases. Am J Surg Pathol 41:833–848
Vitiello GA, Cavnar MJ, Haidu C et al (2017) Minimally invasive management of ectopic pancreas. J Laparoendosc Adv Surg Technol 27(3):277–282
Huang YC, Chen HM, Jan YY et al (2002) Ectopic pancreas with gastric outlet obstruction: report of two cases and literature review. Chang Gung Med J 25(7):485–490
Bain AJ, Owens DJ, Tang RS et al (2011) Pancreatic rest resection using band ligation snare polypectomy. Dig Dis Sci 56:1884–1888
Endo S, Saito R, Ochi D et al (2013) Effectiveness of an endoscopic biopsy procedure using EUS-FNA and EMR-C for diagnosing adenocarcinoma arising from ectopic pancreas: two case reports and a literature review. Intern Med 53:1055–1062
Zhong YS, Shi Q, Yao LQ et al (2013) Endoscopic mucosal resection/endoscopic submucosal dissection for gastric heterotopic pancreas. Turk J Gastroenterol 24(4):322–329
Chou JW, Cheng KS, Ting CF et al (2014) Endosonographic features of histologically proven gastric ectopic pancreas. Gastroenterol Res Pract 2014:160601
Park SH, Kim GH, Park DY et al (2011) Endosonographic findings of gastric ectopic pancreas: a single center experience. J Gastroenterol Hepatol 26(9):1441–1446
Khashab MA, Cummings OW, DeWitt JM (2009) Ligation-assisted endoscopic mucosal resection of gastric heterotopic pancreas. World J Gastroenterol 15(22):2805–2808
Chen HL, Chang WH, Shih SC et al (2008) Changing pattern of ecotopic pancreas: 22 years of experience in a medical center. J Formos Med Assoc 107(12):932–936
Cantor MJ, Davila RE, Faigel D (2016) Yield of tissue sampling for subepithelial lesions evaluated by EUS: a comparison between forceps biopsies and endoscopic submucosal resection. Gastrointest Endosc 65:29–34
Matsushita M, Okazaki HK, Takakuwa H (1999) Gastric aberrant pancreas: EUS analysis in comparison with the histology. Gastrointest Endosc 49(4):493–497
Attwell A, Sams S, Fukami N (2015) Diagnosis of ectopic pancreas by endoscopic ultrasound with fine-needle aspiration. World J Gastroenterol 21(8):2367–2373
Chen SH, Huang WH, Feng CL et al (2008) Clinical analysis of ectopic pancreas with endoscopic ultrasonography: an experience in a medical center. J Gastrointest Surg 12(5):877–881
Kim JH, Lim JS, Lee YC et al (2008) Endosonographic features of gastric ectopic pancreases distinguishable from mesenchymal tumors. J Gastroenterol Hepatol 23(8):e301–e307
Xin L, Jun LQ, Hua XL et al (2016) Endoscopic color Doppler ultrasonography in predicting the safety of endoscopic submucosal dissection for antral heterotopic pancreas. Saudi J Gastroenterol 22:380–384
Guillou L, Nordback P, Gerber C et al (1994) Ductal adenocarcinoma arising in a heterotopic pancreas situated in a hiatal hernia. Arch Pathol Lab Med 118(5):568–571
Zhang L, Sanderson SO, Lloyd RV et al (2014) Pancreatic intraepithelial neoplasia in heterotopic pancreas: evidence for the progression model of pancreatic ductal adenocarcinoma. Am J Surg Pathol. 31:1191–1195
Zhang Y, Huang Q, Zhu LH et al (2014) Endoscopic excavation for gastric heterotopic pancreas: an analysis of 42 cases from a tertiary center. Wien KlinWochenschr 126:509–514
Draganov PV, Wang AY, Othman MO et al (2019) AGA Institute clinical practice update: endoscopic submucosal dissection in the United States. Clin Gastroenterol Hepatol 17(1):16–25
Haidar AH, Saliba C, Nicolas G et al (2019) Unexpected gastric ectopic pancreas during sleeve gastrectomy: a case report. Am J Case Rep 30(20):1966–1968
Ormarsson OT, Gudmundsdottir I, Marvik R (2008) Diagnosis and treatment of gastric heterotopic pancreas. World J Surg 30:1682–1689
Faigel DO, Gopal D, Weeks DA et al (2001) Cap-assisted endoscopic submucosal resection of a pancreatic rest. Gastrointest Endosc 54(6):782–784
Ryu DY, Kim GH, Park DY et al (2010) Endoscopic removal of gastric ectopic pancreas: an initial experience with endoscopic submucosal dissection. World J Gastroenterol 16(36):4589–4593
Jeong HY, Yang HW, Seo SW et al (2002) Adenocarcinoma arising from an ectopic pancreas in the stomach. Endoscopy 12:1014–1017
Galvez-Valdovinos R, Mendoza-Rodriguez A, Coronado-Perez JH et al (2006) Laparoscopic treatment of heterotopic pancreas in the prepyloric region. J Minim Access Surg 2(4):224–226
Dias de Castro F, Magalhaes J, Monteiro S et al (2016) The role of endoscopic ultrasound in the diagnostic assessment of subepithelial lesions of the upper gastrointestinal tract. GE Port J Gastroenterol 23(6):287–292
Iwamuro M, Tsuzuki T, Ohya S et al (2015) Ectopic pancreas in the stomach successfully resected by endoscopic submucosal dissection. Case Rep Med 2015:147927
Otani Y, Furukawa T, Saikawa Y et al (2006) Operative indications for relatively small (2–5cm) gastrointestinal stromal tumor of the stomach based on analysis of 60 operated cases. Surgery 139(4):484–492
Kakeji Y, Nakanoko T, Yoshida R et al (2012) Laparoscopic resection for gastrointestinal stromal tumors in the stomach. Surg Today 42:554–558
Funding
No sources of funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Salvatore Docimo—Consultant, Boston Scientific. Galen Leung, John Mills, Juan Carlos Bucobo declares that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Leung, G., Mills, J., Bucobo, J.C. et al. Evaluation and management of a pancreatic rest noted during pre-bariatric surgery screening endoscopy. Surg Endosc 35, 536–561 (2021). https://doi.org/10.1007/s00464-020-08040-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-020-08040-2