Abstract
Background
Radial endoscopic ultrasound (EUS) is typically used to estimate the depth of rectal polyp invasion, however, there are no data on linear EUS in this setting and its relative accuracy compared to radial EUS.
Methods
In this prospective cohort study, 89 patients with non-pedunculated rectal polyp who underwent linear EUS or radial EUS were prospectively enrolled. The invasion depth was measured for each polyp and categorized as mucosal to shallow submucosal(SMs) or deep submucosal(SMd) invasion. Invasion measurements were compared with the final diagnosis on histopathology.
Results
A total of 58 patients underwent radial EUS and 31 patients underwent linear EUS examination. There were 38 lesions correctly diagnosed in the radial EUS group and 29 correctly diagnosed lesions in the linear EUS group. The diagnostic accuracy of SMd invasion for linear EUS was significantly higher than radial EUS (0.936 vs. 0.655, p = 0.003). A significant difference was also noted for specificity between the two groups (0.963 vs. 0.659, p = 0.003). Univariate analysis showed radial EUS type (OR 0.131, 95% CI 0.028–0.606, p = 0.009) to be an independent predictor for incorrect diagnosis. The area under the receiver operating curve (ROC) was 0.856 and 0.651 for linear EUS and radial EUS, respectively. It was noted that four patients underwent unnecessary surgery for radial EUS while there were no such patients in the linear EUS group.
Conclusions
Linear EUS was more accurate for determining SMd invasion and contributed to the selection of appropriate treatment modalities in patients with non-pedunculated rectal polyp.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Recent progress in endoscopic techniques and gastrointestinal cancer screening in China have contributed to an increase in identification of early-stage colorectal cancer (CRC) [1,2,3]. Non-pedunculated colorectal polyps (NPCP) are classified as sessile and flat colonic lesions, and can be further subclassified as laterally spreading type polyp (LST) [4]. NPCP is closely associated with increased risk of malignancy and more often presents as submucosal (SM) invasive cancer when compared to pedunculated colorectal polyp and requires endoscopic or surgical resection [4]. The most important aspect for choosing the appropriate therapeutic strategy is to determine invasion depth accurately before treatment [5]. Accurate determination of NPCP without deep SM (SMd) invasion is essential in guiding the decision to proceed with endoscopic resection. Endoscopic ultrasonography (EUS), Narrow-Band Imaging (NBI), and magnifying chromoendoscopy (MCE) are often utilized as diagnostic tools to predict the invasion depth of early CRC [6,7,8,9]. However, none of these procedures have sufficient accuracy to meet the clinical requirement. Currently, most results pertinent to EUS have been obtained using radial EUS or mini probe, and there are limited data on linear EUS for identifying invasion depth in the non-pedunculated rectal polyp. It has been widely recognized that linear array EUS performs better than radial EUS in the pancreas and biliary system in most clinical situations [10,11,12]. It is yet to be determined if the linear array EUS is superior for predicting tumor depth in non-pedunculated rectal polyp. Therefore, this prospective cohort study was conducted to compare linear EUS and radial EUS for preoperative diagnosis of SMd invasion in patients with non-pedunculated rectal polyp.
Materials and methods
Patients
Patients who met all of the following inclusion criteria were prospectively enrolled from November 2018 to May 2019 at Nanfang Hospital, Southern Medical University in this study: (1) Age 18–85 years; (2) Diagnosis of non-pedunculated rectal polyps larger than 10 mm or rectal LST determined by colonoscopy; (3) The patient who planned to undergo pre-treatment T staging using radial EUS or linear EUS; (4) The patient who planned to undergo endoscopic resection or surgical resection with detailed histopathological results to be available. Patients were excluded if any of the following conditions existed: (1) They had synchronous cancer in another organ; (2) Accompanied severe comorbidities, such as heart failure. (3) Pregnant patients; (4) The patient who had a contraindication to EUS examination; (5) The patient who could not provide informed consent.
Study design
The present study is a prospective observational study and the enrolled patients received linear EUS or radial EUS as determined by the endoscopist and technical expertise, equipment availability. Standard demographic and clinicopathologic data were collected among the EUS groups, and EUS-related procedural details were also recorded. The primary study endpoint was diagnostic accuracy for invasion depth. The secondary endpoints were sensitivity, specificity, and observation time. The study protocol was approved by the Institutional Review Board of Nanfang Hospital, Southern Medical University (IRB no. NFEC-2019-030) and was carried out according to the ethical guidelines of Declaration of Helsinki (6th revision, 2008). All patients provided their written informed consent prior to study enrollment. All the authors had access to the study data and had approved the final manuscript.
EUS equipment and examination procedure
We used a radial array echoendoscope (EG-580UR, Fujifilm, Tokyo, Japan or EU-ME2, GF-UE260-AL5, Olympus, Tokyo, Japan) for radial EUS examination and a linear array echoendoscope (EG-580UT, Fujifilm, Tokyo, Japan or EU-ME2, GF-UCT2600, Olympus, Tokyo, Japan) for linear EUS examination. Bowel preparation was composed of two liters of hypertonic polyethylene glycol solution taken in all patients 6–12 h prior to the procedure. Carbon dioxide was used for insufflation during the procedure. Normal saline was routinely used to cover the lesion and acquire better EUS image when necessary. All the EUS procedures were performed by experienced endoscopists who have performed over 1000 cases of EUS. The ultrasound frequency was set from 10 to 12 MHz.
EUS diagnosis of tumor invasion depth
The intestinal wall was assessed based on the standard five-layer sonographic structure and the T staging was determined according to AJCC 7th edition TNM staging system. In EUS, T1a was defined as a tumor confined to the first and second mucosal layer, T1b was a tumor that invaded into the third submucosal layer, T2 was a tumor that invaded the muscularis propria (fourth layer), T3 was a tumor that invaded the sub-serosa without interruption of serosa, T4a was a tumor that invaded the serosa (fifth layer), and T4b tumors invaded adjacent organs. In the present study, we mainly focused on the diagnostic accuracy of SMd invasion and EUS imaging was categorized into mucosal to shallow submucosal (M-SMs) invasion and ≥ SMd. A hypoechoic mass limited to within the first and second layers and slight irregularity on the upper side of the third layer was defined as M-SMs cancer. A hypoechoic mass that clearly invades and penetrates into the third layer was defined as SMd cancer (Fig. 1). All the EUS-based diagnoses were blinded and the EUS images were reviewed by a second senior EUS endoscopist. If disagreement occurred between the two diagnoses, a third endoscopist was consulted and the final impression was based on majorities. For M-SMs cancer, endoscopic resection such as endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD) was performed, while the tumor was SMd cancer or deeper, surgical resection was needed according to current recommendations for colorectal ESD.
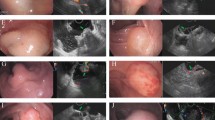
Non-pedunculated rectal polyps diagnosed as SMd cancers in radial EUS and linear EUS. Figure legend: A 68-year-old man with a sessile polyp (Is) about 15 mm in diameter located in rectum (A). Preoperative radial EUS examination showed that the lesion (red arrow) invaded and penetrated into with SM layer (B). Surgery was then performed and the pathological result revealed SMd carcinoma (C). A 60-year-old female patient with superficial protrude type (IIa) rectal lesion measuring about 20 mm in diameter (D). Preoperative linear EUS examination showed that the lesion penetrated into with SM with abruption of SM layer (red arrow) (E). The pathological result showed SMd carcinoma (F)
Histopathological diagnosis
Resected specimens were fixed in formalin and sectioned into 2-mm(mm)-thick slices and then embedded in paraffin. The resection margin status including vertical margin and horizontal margin were evaluated histopathologically. The depth of submucosal invasion was subclassified histologically into one of two grades: depth of invasion submucosal invasion depth < 1000 μm was defined as < pSMd and submucosal invasion depth ≥ 1000 μm was defined as ≥ pSMd. All the final histopathological diagnoses of tumor invasion were considered the true diagnoses and classified into < pSMd and ≥ pSMd. Complete resection was defined as negative horizontal and vertical margins in the pathological specimen and was determined on the basis of a review of postoperative pathology reports. All histological diagnoses were performed by an experienced pathologist who was blinded to the EUS diagnosis.
Statistical analysis
Data are presented as frequencies and percentages, means (standard deviation, SD), or median (range) as appropriate. Categorical data are presented as the number of cases. Data were analyzed by using the chi-square test, Fisher’s exact probability test, and Student t test as appropriate. Risk factors were evaluated using univariate and multivariate logistic regression. Results were presented as odds ratio (OR) and associated 95% CI. All tests for significance were two-tailed, and p values less than 0.05 were considered significant. All calculations were done in SPSS v19 (IBM, Armonk, NY, USA).
Results
A total of 104 patients were screened with 89 patients fulfilling the inclusion criteria and were included in the analysis. Of these, 58 patients underwent radial EUS and 31 patients underwent linear EUS examination (Fig. 2). The mean age was 60.53 ± 11.76 years (range 18–85 years), and there were 49 male patients (49/89, 55.06%). Most of the non-pedunculated polyps were sessile and flat polyp (54/89, 60.67%), and the rest were LST (35/89, 39.33%) according to the Paris classification. The mean polyp size was 32.28 ± 16.97 mm (range 12–85 mm). Pathological results showed that majority of the lesions (63/89, 70.79%) were adenomatous polyp with the remaining 26 (29.21%) identified as differentiated adenocarcinoma. The baseline characteristics of patients in the two groups were well balanced (Table 1). The mean observation time of all EUS procedures was 15.44 ± 9.06 min (radial EUS 15.50 ± 7.79 vs. linear EUS 15.32 ± 11.21, p = 0.930). Based on the EUS evaluation, 61 patients (34 in radial EUS group and 27 in linear EUS group) were diagnosed as M-SMs and 28 patients (24 in radial EUS group and 4 in linear EUS group) were diagnosed as ≥ SMd. Eventually, 63 patients underwent endoscopic resection (including 54 ESD procedures and 9 EMR procedures) and 26 patients received surgery. For final histopathological diagnosis, 71 patients (44 in radial EUS group and 27 in linear EUS group) were diagnosed as M-SMs and 18 patients (14 in radial EUS group and 4 in linear EUS group) were diagnosed as ≥ SMd.
Comparison of diagnostic efficiency between linear EUS and radial EUS
There were 22 misdiagnosed lesions (22/89, 24.72%), with 20 lesions in the radial EUS group (20/58, 34.48%) and the remaining 2 lesions in linear EUS group (2/31, 6.45%) when we compared EUS-based diagnosis with the final histopathological diagnosis. Conversely, 67 lesions (67/89, 75.28%) were correctly diagnosed for all EUS procedures, with 38 lesions in radial EUS group (38/58, 65.52%) and 29 lesions in linear EUS group (29/31, 93.55%). As shown in Table 2, we found that the overall accuracy of linear EUS was significantly better than radial EUS (0.936 vs. 0.655, p = 0.003). The diagnostic sensitivity and the specificity were 0.750 (95% CI 0.301–0.954) and 0.963 (95% CI 0.817–0.993) for linear EUS, and 0.643 (95% CI 0.388–0.837) and 0.659(95% CI 0.511–0.781) for radial EUS, respectively. A significant difference was also noted for specificity between the two groups (0.963 vs. 0.659, p = 0.003). The false-positive rate was 0.037 (95% CI 0.007–0.183) for linear EUS and 0.341 (95% CI 0.219–0.489) for radial EUS. The false-negative rate was 0.250 (95% CI 0.046–0.699) for linear EUS and 0.357 (95% CI 0.163–0.612) for radial EUS. We also evaluated the diagnostic performance of linear EUS and radial EUS for SMd staging using receiver operating curve (ROC), and the area under the ROC was 0.856 and 0.651 for linear and radial EUS, respectively (Fig. 3).
Risk factors associated with incorrect diagnosis of SMd invasion in patients with non-pedunculated rectal polyp
We reviewed the incorrect diagnosis of all enrolled patients to identify risk factors related to the misdiagnosis. Possible factors for incorrect EUS diagnosis that were analyzed included tumor size, tumor location, endoscopic morphology, pathologic type, and EUS examination type. Univariate analysis showed that EUS type (OR 0.131, 95% CI 0.028–0.606, p = 0.009) was a risk factor of incorrect SMd invasion (Table 3). Furthermore, multivariate analysis confirmed that EUS type (OR 0.153, 95% CI 0.032–0.743, p = 0.020) was a risk factor of incorrect T1b staging in NPCP patients (Supplementary Table 1). Radial EUS examination was associated with more incorrect diagnosis compared with linear EUS. No complications such as perforation or bleeding were observed during the EUS examination procedures. The most common reason of the misdiagnosis was over-staging, which occurred in 16 lesions (16/22, 72.73%). It was noted that four misdiagnosed patients (4/20, 20%) underwent unnecessary surgery in the radial EUS group while there were no patients who underwent unnecessary surgery in the linear EUS group, suggesting that linear EUS contributed to accurate decision-making.
Discussion
Accurate prediction of invasion depth less than SMd is essential for proceeding with endoscopic resection. To date, this is the first prospective study comparing linear EUS and radial EUS for prediction of SMd invasion in non-pedunculated rectal polyp. Significant higher accuracy (0.936 vs. 0.655) and specificity (0.963 vs. 0.659) were observed in linear EUS.
Although estimation of the invasion depth based on the gross morphology, NBI or MCE has been reported to be useful in previous studies, its diagnostic accuracy was not established [7,8,9]. A recent study showed that the NBI classification system identified colorectal lesions with deep invasion with 58.4% sensitivity (95% CI 47.5–68.8) and 96.4% specificity (95% CI 95.5–97.2) [7]. Another study demonstrated that the sensitivity and specificity for optical diagnosis of endoscopically unresectable lesions (i.e., ≥ T1 CRC with deep invasion) in large NPCP were 63.3% (95% CI 43.9 to 80.1) and 99.0% (95% CI 97.1 to 100.0), respectively [13]. These studies suggest that NBI classification system is inadequate for clinical decision-making. A systematic review compared NBI and MCE in the differential diagnosis of SMd colorectal cancers from SMs colorectal cancers and found that these two methods had comparable specificities in diagnosing SMd, though the sensitivity of NBI was slightly lower than that of MCE [8]. There were also several studies comparing MCE with EUS for the prediction of invasion depth of early CRC [14,15,16,17,18]. Two such prospective studies demonstrated superiority of EUS (91.8–93%) to MC (59–63.3%) with regard to accuracy [16, 17]. Another study showed that MCE and EUS are equally accurate in estimating the invasion depth of early-stage CRC lesions, and the study also pointed out that neither procedure had sufficient diagnostic accuracy to be used as the standard [18]. All these data pertaining to EUS were obtained by using high-frequency 20 MHz miniprobe or radial EUS and the diagnostic accuracy of linear EUS for estimating the depth of invasion remains unclear. In our prospective study, linear EUS yielded a high accuracy of 0.936, a sensitivity of 0.750, and specificity of 0.963, demonstrating to be a promising modality with good diagnostic efficiency. The reason for better performance of the linear EUS may be attributed to better image resolution. The main problem of all the EUS diagnosis was over-staging, especially with radial EUS, with a false-positive rate of 34.1%. The over-staging of radial EUS directly lead to four cases of unnecessary surgery in this study, while there was no patient undergoing unnecessary surgery in linear EUS group. We also analyzed various factors affecting the diagnostic accuracy of EUS in non-pedunculated rectal polyp. As demonstrated by our data, EUS type was an independent risk factor of incorrect diagnosis and linear EUS was associated with less incorrect cases compared with radial EUS. It should be noted that linear array EUS can only be easily manipulated in the rectum, and it is not suitable for other parts of colon due to the high risk of perforation.
To our knowledge, this study is the first study to report the pre-resection accuracy of linear EUS for diagnosis of SMd invasion in non-pedunculated rectal polyp. There are a few limitations to this study. First, this prospective study was conducted without randomization and therefore selection bias should be considered. Second, this is not a tandem study with the two methods each performed in different patients, which may limit the generalizability of the study results.
In conclusion, linear EUS yielded higher accuracy and specificity than radial EUS for the diagnosis of SMd invasion in non-pedunculated rectal polyp, and it contributed to better clinical decision-making. The method could be widely applied in clinical practice for preoperative assessment in experienced centers. However, large-scale randomized controlled study is further needed to confirm the diagnostic efficiency of linear EUS in differentiating M-SMs from SMd invasion in non-pedunculated rectal polyp.
References
Castaneda D et al (2018) New technologies improve adenoma detection rate, adenoma miss rate, and polyp detection rate: a systematic review and meta-analysis. Gastrointest Endosc 88(2):209–222.e211
Feng RM et al (2019) Current cancer situation in China: good or bad news from the 2018 Global Cancer Statistics? Cancer Commun (London, England) 39(1):22
Gu H et al (2017) Maneuverability and safety of a magnetic-controlled capsule endoscopy system to examine the human colon under real-time monitoring by colonoscopy: a pilot study (with video). Gastrointest Endosc 85(2):438–443
Rutter MD et al (2015) British Society of Gastroenterology/Association of Coloproctologists of Great Britain and Ireland guidelines for the management of large non-pedunculated colorectal polyps. Gut 64(12):1847–1873
Lee BI, Matsuda T (2019) Estimation of invasion depth: the first key to successful colorectal ESD. Clin Endosc 52(2):100–106
Mukae M et al (2015) Diagnostic performance of EUS for evaluating the invasion depth of early colorectal cancers. Gastrointest Endosc 81(3):682–690
Puig I et al (2019) Accuracy of the narrow-band imaging international colorectal endoscopic classification Ssstem in identification of deep invasion in colorectal polyps. Gastroenterology 156(1):75–87
Zhang QW et al (2017) Narrow-band imaging in the diagnosis of deep submucosal colorectal cancers: a systematic review and meta-analysis. Endoscopy 49(6):564–580
Backes Y et al (2017) Narrow band imaging, magnifying chromoendoscopy, and gross morphological features for the optical diagnosis of T1 colorectal cancer and deep submucosal invasion: a systematic review and meta-analysis. Am J Gastroenterol 112(1):54–64
Kaneko M et al (2014) Prospective, randomized, comparative study of delineation capability of radial scanning and curved linear array endoscopic ultrasound for the pancreaticobiliary region. Endosc Int Open 2(3):E160–170
Kanno Y et al (2018) Capability of radial- and convex-arrayed echoendoscopes for visualization of the pancreatobiliary junction. Clin Endosc 51(3):274–278
Shin EJ et al (2015) Linear-array EUS improves detection of pancreatic lesions in high-risk individuals: a randomized tandem study. Gastrointest Endosc 82(5):812–818
Backes Y et al (2019) Multicentre prospective evaluation of real-time optical diagnosis of T1 colorectal cancer in large non-pedunculated colorectal polyps using narrow band imaging (the OPTICAL study). Gut 68(2):271–279
Fu KI et al (2008) Staging of early colorectal cancers: magnifying colonoscopy versus endoscopic ultrasonography for estimation of depth of invasion. Dig Dis Sci 53(7):1886–1892
Haruki S et al (2012) Comparison of diagnostic accuracies of various endoscopic examination techniques for evaluating the invasion depth of colorectal tumors. Gastroenterol Res Pract 2012:621512
Hurlstone DP et al (2005) High magnification chromoscopic colonoscopy or high frequency 20 MHz mini probe endoscopic ultrasound staging for early colorectal neoplasia: a comparative prospective analysis. Gut 54(11):1585–1589
Matsumoto T et al (2002) Comparison of EUS and magnifying colonoscopy for assessment of small colorectal cancers. Gastrointest Endosc 56(3):354–360
Shimura T et al (2014) Magnifying chromoendoscopy and endoscopic ultrasonography measure invasion depth of early stage colorectal cancer with equal accuracy on the basis of a prospective trial. Clin Gastroenterol Hepatol 12(4):662–668
Acknowledgements
National Natural Science Foundation of China (No. 81600460); Clinical Research Startup Program of Southern Medical University by High-level University Construction of Guangdong Provincial Department of Education (LC2016YM002); Science and Technology Planning Project of Guangdong Province (2017A020215139); Natural Science Foundation of Guangdong Province (No.2016A030310382); Guangdong gastrointestinal disease research center (No. 2017B020209003); Special Scientific Research Fund of Public Welfare Profession of National Health and Family Planning Commission(Grant No.201502026).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosures
Author Zhixian Lan, Kangyue Sun, Yuchen Luo, Haiyan Hu, Wei Zhu, Wen Guo, Jing Wen, Wenting Mi, Junsheng Chen, Xiang Chen, Venkata Akshintala, Ying Huang, Side Liu, and Yue Li have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lan, Z., Sun, K., Luo, Y. et al. Linear-array EUS improves the accuracy of predicting deep submucosal invasion in non-pedunculated rectal polyps compared with radial EUS: a prospective observational study. Surg Endosc 35, 1734–1740 (2021). https://doi.org/10.1007/s00464-020-07566-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-020-07566-9