Abstract
The “ecology of fear” framework was developed to describe the impacts predators have on potential prey and prey populations, outside of consumption/predation (i.e. non-consumptive effects, NCEs). This framework has recently been extended to symbiotic interactions such as host–parasite associations. Although the NCEs of predators and parasites on their individual victims can be measured experimentally, it is currently not known whether parasites can exert population-level effects on potential hosts through their NCEs. Modelling can be a useful tool for scaling individual-level NCEs to populations to determine impacts on host population growth. In this study, we used previously published data on the consumptive and non-consumptive effects of an ectoparasitic mite (Macrocheles subbadius) on a fruit fly (Drosophila nigrospiracula) to simulate populations experiencing fear (NCEs only), both fear and infection (consumption + NCEs) or neither. Population-level models indicate that NCEs alone were insufficient to reduce population growth. In fact, host populations experiencing NCEs but not infection had slightly larger final populations than unexposed populations (by ~ 550 flies). This result suggests there is compensation (i.e. increased daily reproduction that overcomes shorter lifespans) among exposed flies. By contrast, the consumptive effects of parasites suppressed the growth of simulated host populations, and this deleterious impact grew non-linearly with infection prevalence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Non-consumptive effects (NCEs) are the impacts predators have on potential prey outside of consumption, contrasted with predation itself (i.e. consumption) (Peacor and Werner 2008; Peckarsky et al. 2008). This “ecology of fear” can manifest as changes in the physiology, behaviour, and morphology of potential prey under predation risk (Peckarsky et al. 1993; Murray et al. 2020). NCEs can ultimately reduce the survival and reproductive success of potential prey (Peckarsky et al. 1993; Macleod et al. 2018). Furthermore, NCEs on individuals can scale up to population-level effects (Belgrad and Griffen 2016; DeWitt et al. 2019). In fact, one meta-analysis suggests NCEs may be responsible for over half of the impact of predators on prey populations (Preisser and Bolnick 2008). Laboratory studies, mesocosms, and statistical modelling have been used to study the NCEs of predators on prey populations (Peckarsky et al. 1993; Kindinger and Albins 2017; Macleod et al. 2018). However, field studies of NCEs are limited to traits that are readily measured, especially among long-lived species where longevity and lifetime fecundity may not be observable in the study period (Sheriff et al 2020). Given these challenges, few studies have empirically tested for suppression of wild populations through NCEs (Sheriff et al. 2020). Given these challenges, modelling provides a useful framework for estimating the scalability of individual-level NCEs to impacts on population growth (DeWitt et al. 2019). For example, models showed that changes in individual physiology among porcupines at risk of predation could lead to reduced birth rates and subsequently a reduction in population size (DeWitt et al. 2019).
Recent research has extended the concept of NCEs to describe interactions between hosts and parasites, as well as other natural enemies and their victims (e.g. parasitoids) (Fill et al. 2012; Abram et al. 2019; Daversa et al. 2021). Parasites have consumptive effects on hosts during infection when they feed on host tissues/resources (Buck 2019; Koprivnikar et al 2021). Laboratory studies have found there are physiological, behavioural, and fitness impacts of exposure to parasites on individual hosts even when infection does not occur (Koprivnikar and Penalva 2015; Horn and Luong 2018; Benoit et al. 2020). To date, it is unclear if these individual-level NCEs impact hosts on a population level. Parasite infection has smaller effects than predation, and this disparity may explain the smaller individual-level NCEs observed in tadpole–parasite interactions than tadpole–predator interactions in a recent meta-analysis (Daversa et al. 2021). However, almost all free-living organisms face the risk of parasitism, and small individual effects may scale up into substantial effects on host populations (Poulin and Morand 2000; Buck 2019). Thus, there is a need to study NCEs of parasites as they may be widespread yet underestimated (Buck 2019). In this study, we simulated host populations that experience either (1) fear (NCEs) only, (2) fear and infection (i.e. NCEs and consumptive effects), or (3) neither.
We used published data on a Drosophila–Macrocheles association (reviewed in Perez-Leanos et al. 2017) to model the consumptive and non-consumptive effects of parasitism. The mite Macrocheles subbadius (Berlese) (Mesostigmata: Macrochelidae) is a naturally occurring ectoparasite of Drosophila nigrospiracula Patterson and Wheeler (Diptera: Drosophilidae) (Polak 1996). D. nigrospiracula feed and reproduce on rotting cacti, and migrate to new sites as the decaying cactus desiccates (Markow 1988). Mites use their chelicerae to attach to flies and feed on the haemolymph of adult flies (Polak 1996). Unlike most host–parasite systems, empirical data exist on both the consumptive and non-consumptive effects of M. subbadius on D. nigrospiracula fitness (measured in longevity and lifetime fecundity) under laboratory conditions (Polak 1996; Horn and Luong 2018). Infection with mites reduces the survival and fecundity of adult flies by up to ~ 60% (Polak 1996; Polak and Starmer 1998). Exposure to mites, without infection, induces costly defensive behaviours in flies (Luong et al. 2017; Horn and Luong 2019), reduces glycogen and lipid stores (Benoit et al. 2020), and ultimately shortens fly lifespans as well as lowering fecundity (Horn and Luong 2018). Since both NCEs and consumptive effects on fly fitness (defined in terms of survival and reproduction) have been measured, this fly–mite association provides a unique opportunity to model the ecology of fear in a host–parasite system.
We hypothesised that ectoparasites negatively impact host populations (growth and final size) through NCEs on individual fitness. Specifically, we predicted that simulated fly populations experiencing NCEs will have reduced growth rates/smaller final populations relative to populations not experiencing NCEs due to individual reductions in lifetime fecundity. Alternatively, many hosts increase reproductive effort when at risk of reduced survival due to parasites/infection, i.e. compensate (Forbes 1993; Adamo 1999; Parietti et al. 2020). For example, snails from populations with higher rates of castrating parasites have higher egg production than snails from low prevalence populations (Kirst 2001). Among female D. nigrospiracula, the decrease in lifespan during mite exposure (without infection) was larger than the reduction in lifetime fecundity, 22% shorter lifespans versus 13% lower fecundity, and the daily egg laying rate was higher in exposed flies even though lifetime fecundity was lower (Horn and Luong 2018). Combined, these results suggest compensation may prevent/reduce population-level impacts of parasite NCEs. To evaluate the potential for compensation to limit population-level NCEs, we made models where we varied the NCEs on fecundity relative to longevity (i.e. we varied the potential for compensation).
We created individual-based models to simulate populations of flies experiencing fear (NCE), fear and infection (NCE and consumptive effects), or no parasites over 100 days (~ 5 overlapping generations). A “consumption + no fear” condition was not modelled as it would require infecting flies while eliminating resistance as well as cues of mite presence/infection which is not possible in seminatural fly–mite interactions. For this reason, there is no experimental data that could be used to model a no fear with consumption condition. By modelling NCEs and consumptive effects of parasites on host populations, we also identify gaps in our current understanding of trait-mediated interactions between hosts and parasites.
Methods
We simulated fly populations under 3 scenarios: (1) in the presence of parasitic mites and their non-consumptive effect on host flies (“no consumption + fear”), (2) with both the consumptive (i.e. infection) and non-consumptive effects of parasitic mites on their fly hosts (“consumption + fear”), or (3) in the absence of parasite effects (“no consumption + no fear”). For each scenario, we constructed a stochastic matrix model of fly populations. The transition matrix considered flies living over 60 days, moving through pre-reproductive stages (eggs, larvae, pupae, pre-reproductive adults) for 20 days before becoming reproductively mature adults and producing eggs (model is illustrated in the supplemental file). Mites could infect pre-reproductive adults and mature adults. D. nigrospiracula have mean lifespans of ~ 2–4 weeks based on the environment and can live upwards of ~ 50 days post-adult-emergence in laboratory conditions (Polak 1996; Luong and Polak 2007). We ran each simulation 1000 times. Data on the survival and fecundity of individual adult female D. nigrospiracula flies infected by mites were derived from Polak (1996) (parameters used for modelling are summarised in Table 1). Fly survival drops precipitously and non-linearly with increased mite infections (Polak and Starmer 1998). Therefore, we did not vary the parasite load within infected individuals and assumed adult flies either had pathogenic levels of infection or were uninfected. Prevalence of infection in simulated populations was varied by changing the daily infection rate. The survival and fecundity of female adult D. nigrospiracula exposed to, but not infected with, mites (i.e. no consumption + fear) were derived from Horn and Luong (2018) (Table 1). NCEs of mites were measured in the previous study by housing flies in vials with mites separated by a mesh barrier preventing infection (Horn and Luong 2018, 2021). We used the demogR (Jones 2007), truncnorm (Mersmann et al. 2018) and Tidyverse packages (Wickhamn et al. 2019) in addition to R Core features (Ver. 3.5.1, R Core Team 2022). Code can be accessed online (https://doi.org/10.17605/OSF.IO/Z5A4S).
We did not account for the influences of parasitism on male flies. Since female D. nigrospiracula can store sperm, the assumption that females were not sperm-limited is reasonable (Pitnick et al. 1999). Furthermore, mites preferentially infect female D. nigrospiracula over male conspecifics, infecting females 71% of the time in choice assays (Horn et al. 2020). Our model also assumes that flies are not food limited in the short term; however, these flies live on ephemeral habitats, rotting cacti (Markow 1988). Therefore, our model represents the ability of initial colonisers to exploit this food source. By not modelling the decline of the ephemeral food source, we avoid the confounding effect of food limitation on our analysis of the NCE of parasitism. In addition, there is currently no experimental data on a food–NCE interaction to use in models.
Daily survival was simulated out of a truncated normal distribution (ranging from 0–1) with a mean of 0.96, 0.94, and 0.93 for the no consumption + no fear, no consumption + fear, and consumption + fear scenarios, respectively (see Table 1). Models using these values matched the overall survival patterns (percentage of flies alive after 30 days) in the original data sources (Polak 1996; Horn and Luong 2018). In the absence of data, we set the standard deviation of survival to 10% of the mean survival (i.e. if survival was 0.96 the standard deviation was set to 0.096). Similarly, we calculated the per day egg production from the literature for each scenario as 4.38, 4.86, and 2.85 for the no consumption + no fear, no consumption + fear, and consumption + fear scenarios, respectively, and simulated daily values out of a Poisson distribution (Polak 1996; Horn and Luong 2018). Note that lifetime fecundity was still lower among flies experiencing only fear than flies experiencing no fear and no consumption (Horn and Luong 2018). The experimental evidence for infection impacting latency to ovipositing (age at first egg laying) was mixed and weak (Polak 1996). Thus, we assumed latency to ovipositing was equal between groups. There was no experimental data for daily survival rates of eggs, larva, and pupae with and without mites. Instead, we assigned a single value to all of them, tuned to reflect observations of fly population sizes on natural cactus rots (Breitmeyer and Markow 1998). Nor was there data on potential inter-generational NCEs of mites, e.g. changes in the quality of offspring from mite-exposed mothers.
We estimated the population growth rate (lambda) from the stochastic matrix by calculating the mean day-over-day growth in total fly numbers. Each simulated population was initiated with 50 competent adult flies (10 of each of 20–24 days old) who colonised a hypothetical cactus rot, dispersing from nearby populations. The simulation was run for 100 days; however, only the last 50 days of each simulation were used while calculating lambda to avoid early transient dynamics. We tracked and recorded the total number of uninfected and infected adult flies to determine population size. We did this to match the field data, which counted adult flies and not pre-adult stages. The stochastic matrices of the three scenarios were simulated 1000 times each and the adult fly populations plotted for each run. Likewise, the distribution of lambda for each scenario was plotted as a histogram across all simulations.
In a sensitivity analysis, we varied the consumptive effect of parasites by altering the daily probability of infection (ranging from 0–1). We set this value to 0 in mite-free scenarios. We tested the effect of a daily probability of infection on population growth by simulating 1000 populations for 100 days starting with 50 female dispersers, 20% of whom were infected with parasites (a prevalence reasonable to expect in nature, Polak and Markow 1995). We assumed that all subsequent adults were born uninfected but became infected at some daily probability, which we varied between 0 and 1. Uninfected females survived and reproduced using the parameters from the no consumption + fear scenario while infected females survived and reproduced using the parameters from the consumption + fear scenario. We recorded the final average population size and the proportion of the adult population that was parasitised.
Empirical data showed that the parasite-exposed females produced ~ 10% more eggs per day than unexposed females (4.86 vs. 4.38 eggs per day on average, respectively; Horn and Luong 2018), suggesting compensatory egg production may be occurring that offsets the survival detriment caused by NCEs. We investigated this possibility by running simulations of the effect of fear on egg production across a range of daily survival rates to determine compensatory egg production’s impact on population growth. In other words, we simulated populations with varying or no ability to increase egg production per day to compensate for shortened lifespan. As before, simulations were run over 100 days and 1000 populations were simulated at each combination of egg production and survival, then the average adult population size at the end of the simulation was recorded and compared to the “no fear + no consumption” scenario. We used these models to calculate the daily egg production required to compensate for reductions in longevity.
Results
Baseline scenarios
In order to elucidate the relative impacts of NCEs on host populations, we simulated 1000 populations over 100 days in each of 3 scenarios: reflecting (1) the presence of parasitic mites and their non-consumptive effect on host flies (no consumption + fear), (2) both the consumptive and non-consumptive effects of parasitic mites on their fly hosts (consumption + fear), or (3) the absence of parasites (no consumption + no fear). We found that the estimated growth rate, lambda, was similar for the scenarios with no consumption (i.e. infection) with fear or without fear (\(\lambda\) = 1.051, and \(\lambda\) = 1.050, respectively). The mean final population size was larger in the simulations with NCEs than in groups absent parasitism: 4103 versus 3556, respectively (~ 15% increase) (Fig. 1). On the other hand, population growth rates (\(\lambda\) = 1.030) and final average population sizes were far lower in simulations including consumptive effects (Fig. 1).
Population trajectories (top panels) and growth rate (lambda, bottom panels) for each of the 3 simulated baseline scenarios: (1) absence of parasites (no consumption + no fear), (2) the presence of parasitic mites and their non-consumptive effect on host flies (no consumption + fear), or (3) the effects of consumptive (infection) and NCEs of parasites (consumption + fear). The trajectories represent 1000 simulations, with the shading indicative of where more of the simulations overlap and the red line is the average of all the simulation. Similarly, the histograms represent the estimated growth rate (lambda) from each of the 1000 simulations above them, the mean lambda from the last 50 days is recorded to avoid transient dynamics that may exist early in the simulation
Variation in fecundity within a sample simulation
There is substantial variation in lifetime fecundity among flies, especially in simulations with no consumption. This is illustrated in the results of a single simulated population (Fig. 2). In the simulation without any impacts of parasites, flies produced 71 (sd = 72.6) eggs over their lifespan and lived 16 (sd = 16.4) days. Simulated flies subject to only the non-consumptive effects of parasites produced 53 (sd = 67.3) eggs and lived 11 (sd = 13.8) days. Finally, in a simulated population where flies were exposed to both the consumptive and non-consumptive effects of parasitism, they produced 26 (sd = 36.8) eggs and lived 9 (sd = 12.7) days.
Example of the variation in fly lifetime reproductive success with or without parasite consumptive and non-consumptive effects in a single simulated population. Reproductive success was measured as the total number of eggs produced and the number of days lived, for a simulation run for a single population of 1000 flies in one of the three scenarios: (1) no consumption + fear, (2) consumption + no fear or (3) no consumption + no fear
Sensitivity to daily probability of infection
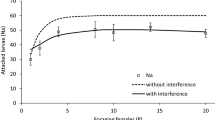
We also investigated how variation in the daily probability of infection affected population growth. These simulations showed that a daily probability of infection of approximately 0.05 resulted in a population size half that of the simulations with no mite effects (Fig. 3). Any daily probability of infection above ~ 0.3 results in > 75% of the population being parasitised and little difference in the overall average population size relative to a fully parasitised population (Fig. 3).
Sensitivity results for the effect of a daily probability of infection on population growth by simulating 1000 populations for 100 days starting with 50 female dispersers, 20% of whom were infected with parasites. Uninfected females survived and reproduced using the parameters from the “no consumption + fear scenario” while infected females survived and reproduced using the parameters from the “consumption + fear scenario”. The average of the 1000 populations is given by each line. We assumed that all subsequent adults born started out uninfected but became infected at a daily probability between 0 and 1 (left number); the right number shows the final proportion of flies infected with parasites (i.e. prevalence)
Compensation in egg laying could influence population growth
We varied the mite-mediated change in fecundity to consider the potential impacts of compensation (i.e. how/if flies compensate for early death with increased egg laying) for population growth across a range of daily survival rates. We empirically solved for the combination of egg production and survival rates that resulted in final adult population sizes that were equal to the baseline scenario of no fear + no consumption (contour line in Fig. 4). A daily egg production of ~ 4.6 eggs/day was required to compensate for reduced survival. Below this line (in cooler colours), daily female egg production was not able to compensate for the reduction in survival, while above this line (in hotter colours) egg production more than compensated for the reduction in survival associated with the fear of being parasitised. For comparison, the three original scenarios are plotted (symbols given in Fig. 4). We found that when survival was unchanged from the baseline scenarios (0.96 and 0.94 daily survival rate for uninfected and infected females, respectively), that population which compensated with 110% of the per day egg production of the baseline scenario (no fear and no infection) resulted in a population size of approximately 1000 additional adult flies, an increase of approximately 41% (Fig. 4).
Simulated final adult population size when host compensation (egg production) was varied relative to the baseline egg production from experimentation (4.38 eggs per day = 100%) along with daily survival rate relative to the baseline survival rate from experimentation (0.96 = 100%). The baseline scenarios are given for reference, where the circle is the no fear + no consumption scenario, the triangle is the fear + no consumption scenario, and the square is the fear + consumption scenario. The black line represents the contour of the final adult population size for the co-fear + no compensation scenario and indicates the daily egg production required to offset the reduction in survival that occurs as a result of NCE of parasitism
Discussion
We tested the hypothesis that NCEs at the individual-level scale up and impact population growth rates independent of infection by simulating 1000 fly populations experiencing no effects of parasites, just NCEs, or NCEs and consumptive effects (infection). The mean growth rates (\(\lambda\)) of populations experiencing both NCEs and consumptive effects were substantially lower (when mite prevalence = 100%, \(\lambda\) = 1.030). The higher the simulated prevalence of infection, the larger the impact on population growth rate (Fig. 3). In our study, ~ 25% final infection prevalence (daily infection chance = 0.05) corresponded to an average ~ 50% lower final simulated population size compared to mite-free populations (Fig. 3). The prevalence of M. subbadius infection among wild D. nigrospiracula generally ranges from ~ 10 to 40%, increasing as habitats deteriorate (Polak and Markow 1995). Our simulations suggest these rates of infection would have mild to moderate effects on population growth (Fig. 3). Contrary to our expectations, the growth rates of simulated populations experiencing only NCEs were slightly higher than in the populations without mite effects, \(\lambda\) = 1.051 and \(\lambda\) = 1.050, respectively (Fig. 1), and the average final population size was higher in the group experiencing only fear by ~ 550 flies (15% higher) (Fig. 1).
Although lab studies showed reductions in survival and lifetime fecundity among individual female flies exposed to mites without infection (Horn and Luong 2018), these effects when scaled up to the population level in our simulations did not reduce population growth rates (Fig. 1). Examination of an individual simulated population suggests the variation in fecundity among flies experiencing fear was large relative to the reduction in the mean number of eggs produced over the fly lifespan (Fig. 2). Substantial variation in fecundity among flies experiencing fear may, therefore, limit the effects of NCEs on populations. One potential explanation for the simulated results is that flies compensate for the presence of parasites and associated mortality, e.g. through earlier maturation/maximal reproduction or terminal reproductive output (Krist 2001; Duffield et al. 2017; Parietti et al. 2020). We modelled populations where flies were unable to compensate for reduced survival with increased daily fecundity (by setting fecundity in the fear group to match the fecundity of the uninfected female), on average these populations declined by approximately 650 flies or 19% relative to the mite-free scenario (Fig. 3). In our sensitivity analysis of the potential for compensation, we were able to derive the daily egg production required to offset the NCE of parasitism for survival in terms of the final adult population (~ 4.6 eggs/day, Fig. 4).
In our models, we assumed that flies exposed to and infected with mites would lay eggs at the same age as mite-free flies (i.e. have the same latency to ovipositing), based on observations of flies exposed to mites for 48 h (Polak 1996). It is possible that latency would have been affected by chronic exposure to mites or by exposure as larvae, as occurs at natural habitats. However, the long-term exposure experiment did not measure latency to ovipositing (Horn and Luong 2018). Alternatively, flies may vary in their daily fecundity over the course of their lifespan, although we did not account for that possibility in this model (Luong et al. 2007; Miller et al. 2014). Exposure and/or infection may alter the time of peak reproduction without changing the time of first ovipositing if there are constraints on reproductive maturation. Field studies are needed to test if early first/peak reproduction is a mechanism of compensation by examining the latency to ovipositing and peak reproductive age in fly populations with mites and without mites. Physiological mechanisms enabling compensatory egg production is a direction for future research.
While reviewing previous studies on the fitness effects of infection, we incidentally found additional evidence for individual-level NCEs of parasites. When measuring the effect of infection on fly longevity, Polak (1996) reported flies that resisted infection and those that were never exposed. Unexposed flies lived 29.3 days, whereas the resisted group lived 24.4 days on average, an 18% difference (Polak 1996). Although this difference was insignificant in the analyses, the magnitude of the reduction was comparable to the ~ 23% difference in longevity between flies chronically exposed to mites and unexposed flies reported in Horn and Luong (2018). A thorough review of the literature may find further examples of parasitic NCEs in studies not explicitly designed to test for them and may be a direction for future meta-analyses.
By building models of parasite-mediated NCEs, we identified gaps in our understanding of these trait-mediated interactions. For example, exposure to predators as larvae is known to affect the physiological and behavioural traits of adult flies, as well as other vertebrates and invertebrates (Davenport et al. 2014; Xiong et al. 2015; Krams et al. 2016). Female D. melanogaster exposed to spiders as larva have lower masses and reduced fat reserves relative to unexposed conspecifics as adults (Krams et al. 2016). Given the positive relationship between female body size and fecundity in Drosophila, deleterious NCEs on body size are likely to have deleterious effects on future reproduction (Lefranc and Bundgaard 2000; Krams et al. 2016). In addition, NCEs may directly impact larval survival. For example, larval dragonflies exposed to restrained fish during growth were then less likely to survive adult emergence (McCauley et al. 2011). NCEs can also impact future generations in the form of maternal effects. In a vertebrate system, the survival of offspring from mothers exposed to a sham-predator can be reduced even if the source of fear is removed post-birth (MacLeod et al. 2018). Further research is needed to determine if parasites have inter-generational or interstitial NCEs on Drosophila.
Our results here are consistent with a recent meta-analysis that found, relative to predators, the individual-level NCEs of parasites on amphibian hosts tend to be smaller and mixed (Daversa et al. 2021). However, data on only a small number of amphibians and their parasites were available (Daversa et al. 2021). Our results extend this trend to an insect host. Furthermore, the magnitude of parasitic NCEs may vary between parasite taxa. When there are few cues of parasite presence (e.g. with small, immobile infectious stages), hosts may be under less selection to have strong pre-infection defences. In turn, the potential for costly pre-infection defenses to drive NCEs is reduced. In host–parasite systems with limited pre-infection cues of parasites, consumptive effects may be present with few to no NCEs (Daversa et al. 2021; Koprivnikar et al. 2021). Comparative research across host and parasite taxa is an avenue for future research, in particular testing if transmission mode influences the magnitude of parasite NCEs.
Taken together, our results suggest that host compensation may reduce the impacts of individual-level NCEs on host population growth. NCEs may even have positive impacts on host population size, at least in the short term. Future studies should investigate biological mechanisms allowing host populations compensate for NCEs, and under which conditions compensation can have positive impacts on host population size. We also identify the need for future research on interstitial and inter-generational NCEs of parasites to improve future models and fully account for parasitic NCEs. Hosts live in an infectious world, but how this risk impacts host populations has implications for the ecology and coevolution of host–parasite symbioses.
References
Abram PK, Brodeur J, Urbaneja A, Tena A (2019) Nonreproductive effects of insect parasitoids on their hosts. Annu Rev Entomol 64:259–276
Adamo SA (1999) Evidence for adaptive changes in egg laying in crickets exposed to bacteria and parasites. Anim Behav 57:117–124. https://doi.org/10.1006/anbe.1998.0999
Belgrad BA, Griffen BD (2016) Predator-prey interactions mediated by prey personality and predator hunting mode. Proc R Soc B Biol Sci 283:20160408
Benoit JB, Bose J, Bailey ST, Polak M (2020) Interactions with ectoparasitic mites induce host metabolic and immune responses in flies at the expense of reproduction-associated factors. Parasitology 147:1196–1205
Breitmeyer CM, Markow TA (1998) Resource availability and population size in cactophilic Drosophila. Funct Ecol 12:14–21
Buck JC (2019) Indirect effects explain the role of parasites in ecosystems. Trends Parasitol 35:835–847
Davenport JM, Hossack BR, Lowe WH (2014) Partitioning the non-consumptive effects of predators on prey with complex life histories. Oecologia 176:149–155
Daversa DR, Hechinger RF, Madin E, Fenton A, Dell AI, Ritchie EG, Rohr J, Rudolf VHW, Lafferty KD (2021) Broadening the ecology of fear: non-lethal effects arise from diverse responses to predation and parasitism. Proc R Soc B Biol Sci 288:20202966
DeWitt PD, Visscher DR, Schuler MS, Thiel RP (2019) Predation risks suppress lifetime fitness in a wild mammal. Oikos 128:790–797
Duffield KR, Bowers EK, Sakaluk SK, Sadd BM (2017) A dynamic threshold model for terminal investment. Behav Ecol Sociobiol 71:185
Fill A, Long EY, Finke DL (2012) Non-consumptive effects of a natural enemy on a non-prey herbivore population. Ecol Entomol 37:43–50. https://doi.org/10.1111/j.1365-2311.2011.01333.x
Forbes MRL (1993) Parasitism and host reproductive effort. Oikos 67:444–450
Horn CJ, Luong LT (2018) Proximity to parasites reduces host fitness independent of infection in a Drosophila-Macrocheles system. Parasitology 145:1564–1569
Horn CJ, Luong LT (2019) Current parasite resistance trades off with future defenses and flight performance. Behav Ecol Sociobiol 73:1–10
Horn CJ, Luong LT (2021) Data from: Proximity to parasites reduces host fitness independent of infection in a Drosophila-Macrocheles system. Dryad. https://doi.org/10.5061/dryad.gmsbcc2pn
Horn CJ, Mierzejewski MK, Elahi ME, Luong LT (2020) Extending the ecology of fear: parasite-mediated sexual selection drives host response to parasites. Physiol Behav 224:1–7. https://doi.org/10.1016/j.physbeh.2020.113041
Jones JH (2007) demogR: a package for the construction and analysis of age-structured demographic models in R. J Stat Softw 22:1–28. https://doi.org/10.18637/jss.v022.i10
Kindinger TL, Albins MA (2017) Consumptive and non-consumptive effects of an invasive marine predator on native coral-reef herbivores. Biol Invasions 19:131–146
Koprivnikar J, Penalva L (2015) Lesser of two evils? Foraging choices in response to threats of predation and parasitism. PLoS One. https://doi.org/10.1371/journal.pone.0116569
Koprivnikar J, Rochette A, Forbes MR (2021) Risk-induced trait responses and non-consumptive effects in plants and animals in response to their invertebrate herbivore and parasite natural enemies. Front Ecol Evol. https://doi.org/10.3389/fevo.2021.667030
Krams I, Inwood SE, Trakimas G, Krams R, Burghardt GM, Bulter DM, Luoto S, Krama T (2016) Short-term exposure to predation affects body elemental composition, climbing speed and survival ability in Drosophila melanogaster. PeerJ 4:e2314
Krist AC (2001) Variation in fecundity among populations of snails is predicted by prevalence of castrating parasites. Evol Ecol Res 3:191–197
Lefranc A, Bundgaard J (2000) The influence of male and female body size on copulation duration and fecundity in Drosophila melanogaster. Hereditas 132:243–247
Luong LT, Polak M (2007) Costs of resistance in the Drosophila-Macrocheles system: a negative genetic correlation between ectoparasite resistance and reproduction. Evolution 61:1391–1402
Luong LT, Heath BD, Polak M (2007) Host inbreeding increases susceptibility to ectoparasitism. J Evol Biol 20:79–86
Luong LT, Horn CJ, Brophy T (2017) Mitey costly: energetic costs of parasite avoidance and infection. Physiol Biochem Zool 90:471–477
MacLeod KJ, Krebs CJ, Boonstra R, Sheriff MJ (2018) Fear and lethality in snowshoe hares: the deadly effects of non-consumptive predation risk. Oikos 127:375–380
Markow TA (1988) Reproductive behavior of Drosophila melanogaster and Drosophila nigrospiracula in the field and in the laboratory. J Comp Psychol 102:169–173
McCauley SJ, Rowe L, Fortin MJ (2011) The deadly effects of “nonlethal” predators. Ecology 92:2043–2048
Mersmann O, Trautmann H, Steuer D, Bornkamp B (2018) truncnorm: truncated normal distribution. R package version 1.0-8. CRAN.R-project.org/package=truncnorm
Miller PB, Obrik-Uloho OT, Phan MH, Medrano CL, Renier JS, Thayer JL, Wiessner G, Bloch Qazi MC (2014) The song of the old mother: reproductive senescence in female Drosophila. Fly (austin) 8:127–139
Murray RL, Tah S, Koprivnikar J, Rowe L, McCauley SJ (2020) Exposure to potentially cannibalistic conspecifics induces an increased immune response. Ecol Entomol 45:355–363
Parietti M, Merlo MJ, Natal M, Mendez Casariego MA (2020) Reproductive compensation in female Palaemonetes argentinus (Decapoda: Natantia) due to Microphallus szidati (Trematoda) infection. J Helminthol 94:e204
Peacor SD, Werner EE (2008) Nonconsumptive effects of predators and trait-mediated indirect effects. Encyclopedia of life sciences (ELS). Wiley, Chichester, pp 1–8
Peckarsky B, Cowan C, Penton M, Anderson C (1993) Sublethal consequences of stream-dwelling predatory stoneflies on mayfly growth and fecundity. Ecology 74:1836–1846
Peckarsky BL, Abrams PA, Bolnick DI, Dill LM, Grabowski JH, Luttbeg B, Orrock JL, Peacor SD, Preisser EL, Schmitz OJ, Trussell GC (2008) Revisiting the classics: considering nonconsumptive effects in textbook examples of predator–prey interactions. Ecology 89:2416–2425
Perez-Leanos A, Loustalot-Laclette MR, Nazario-Yepiz N, Markow TA (2017) Ectoparasitic mites and their Drosophila hosts. Fly 11:10–18
Pitnick S, Markow T, Spicer GS (1999) Evolution of multiple kinds of female sperm-storage organs in Drosophila. Evolution 53:1804–1822
Polak M (1996) Ectoparasitic effects on host survival and reproduction: the Drosophila-Macrocheles association. Ecology 77:1379–1389
Polak M, Markow TA (1995) Effect of ectoparasitic mites on sexual selection in a Sonoran desert fruit-fly. Evolution 49:660–669. https://doi.org/10.2307/2410319
Polak M, Starmer WT (1998) Parasite-induced risk of mortality elevates reproductive effort in male Drosophila. Proc R Soc B Biol Sci 265:2197–2201
Poulin R, Morand S (2000) The diversity of parasites. Q R Biol 75:277–293
Preisser EL, Bolnick DI (2008) The many faces of fear: comparing the pathways and impacts of non-consumptive predator effects on prey populations. PLoS One 3:1–8
R Core Team (2022). R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. www.R-project.org/
Sheriff MJ, Peacor SD, Hawlena D, Thaker M (2020) Non-consumptive predator effects on prey population size: a dearth of evidence. J Anim Ecol 89:1302–1316
Wickhamn H, Averick M, Bryan J, Chang W, McGowan LD, François R, Grolemund G, Hayes A, Henry L, Hester J, Kuhn M, Pedersen TL, Miller E, Bache SM, Müller K, Ooms J, Robinson D, Seidel DP, Spinu V, Takahashi K, Vaughan D, Wilke C, Woo K, Yutani K (2019) Welcome to the tidyverse. J Open Source Softw 4:1686
Xiong XF, Michaud JP, Li Z, Wu PX, Chu YN, Zhang QW, Liu XX (2015) Chronic, predator-induced stress alters development and reproductive performance of the cotton bollworm, Helicoverpa armigera. Biocontrol 60:827–837
Acknowledgements
Kim Mathot provided feedback on an earlier version of this manuscript. We would like to thank 2 anonymous reviewers for constructive comments.
Funding
LTL is supported by the Natural Sciences and Engineering Research Council (RGPIN-2020–03910). CJH was supported by an Alexander Graham Bell Canada Graduate Scholarship (doctoral).
Author information
Authors and Affiliations
Contributions
CJH conceptualised the study, reviewed the literature, and wrote the manuscript. DRV conceptualised and wrote the code as well as co-wrote the manuscript. LTL acquired funding and helped write the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No authors have a conflict of interest to disclose.
Additional information
Communicated by George Heimpel.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Horn, C.J., Visscher, D.R. & Luong, L.T. Relative contributions of parasite consumptive and non-consumptive effects to host population suppression in simulated fly–mite populations. Oecologia 200, 339–347 (2022). https://doi.org/10.1007/s00442-022-05268-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-022-05268-8