Abstract
Birds aim to optimize resources for feeding young and self-maintenance by timing reproduction to coincide with peak food availability. When reproduction is mistimed, birds could incur costs that affect their survival. We studied whether nesting phenology correlated with the apparent survival of American kestrels (Falco sparverius) from two distinct populations and examined trends in clutch-initiation dates. We estimated apparent survival using multi-state mark-recapture models with nesting timing, nesting success, sex, age, and weather covariates. Nesting timing predicted the apparent survival of successful adults; however, the effect differed between populations. Early nesting kestrels had higher apparent survival than later nesters in the western population, where kestrels have a relatively long nesting season. At the eastern site, where kestrels have a relatively short nesting season, the pattern was reversed—later nesters had higher apparent survival than earlier nesters. Nesting timing did not affect the apparent survival of adults with failed nests suggesting that the energetic cost of producing fledglings contributed to the timing effect. Finally, clutch-initiation dates advanced in the western population and remained static in the eastern population. Given that both populations have seasonal declines in productivity, population-specific survival patterns provide insight into seasonal trade-offs. Specifically, nesting timing effects on survival paralleled productivity declines in the western population and inverse patterns of survival and reproduction in the eastern population suggest a condition-dependent trade-off. Concomitant seasonal declines in reproduction and survival may facilitate population-level responses to earlier springs, whereas seasonal trade-offs may constrain phenology shifts and increase vulnerability to mismatch.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Optimal timing of avian reproduction coincides with peak food availability to exploit resources not only for feeding young but for self-maintenance (Lack 1968). Relationships between nesting phenology and productivity are well-researched. Earlier nesting birds typically have higher productivity than later-nesting birds (Reed et al. 2013; Saino et al. 2017). However, the consequences of nesting phenology on other fitness components, such as survival, and potential trade-offs between adult survival and productivity, are less-studied (Reed et al. 2013; Bastianelli et al. 2021). Understanding relationships between nesting phenology and fitness is increasingly important as climate change drives advances in spring green-up, and prey availability, in many regions (Dunn 2004). If birds do not advance nesting timing accordingly, phenological mismatch (i.e., when reproduction is out of synchrony with prey resources) could lead to fitness costs that culminate in population declines (Møller et al. 2008). Indeed, species that have shifted their timing of reproduction least in response to climate change have experienced more severe population declines than species that have shifted more (Møller et al. 2008). Therefore, it is important to understand the associations between nesting phenology and survival.
Breeding at suboptimal times (i.e., lower food availability compared to the peak of the season) can lead to decreased survival (Golet et al. 1998; Lof et al. 2012). Earlier nesting adults may increase survival by obtaining high-quality nesting sites with abundant resources, by synchronizing their reproduction with regional resource peaks, or both (Møller 1994). This may result in better physical condition at fledging for both early-breeding adults and their young, increasing their likelihood of survival. Adults who fledge young early in the breeding season have more time to complete molt and improve their physiological condition prior to migration, which may allow them to depart earlier for overwintering areas. Ultimately, this may result in early breeders obtaining higher quality winter territories than later breeding adults (Smallwood 1988). Additionally, earlier nesting adults may be more competitive, higher quality individuals than later-nesting adults, with inherent traits that result in higher survival and productivity (Verhulst and Nilsson 2008). Earlier hatched young have more time before autumn migration to gain foraging experience, establish breeding territories for subsequent years, and gain mass before migration, which could result in increased survival compared to later hatched young (Smallwood and Smallwood 1998; Catry et al. 2016). Because raising young is the most energetically expensive time of the nesting season for species with altricial young (Hillström and Moreno 1992), there may be trade-offs between adult survival and productivity if resources are limited. For example, adults raising young at suboptimal times (i.e., mismatched with peak food availability) may have to reallocate resources spent on self-maintenance to provisioning their young, which could result in poor body condition, increased metabolic stress, and decreased adult survival (Nilsson and Svensson 1996; Reed et al. 2013). Offspring reared at suboptimal times may be provisioned with less food, which can result in slow growth, poor body condition, high nestling mortality, and lower probabilities of fledging and recruitment (Visser et al. 2006).
The strength of timing effects on survival, and the capacity of individuals to shift their timing of reproduction, may depend on regional patterns of climate and resource availability, species life history traits, or variability in adaptive capability among individuals within a population. For example, thermoregulation costs, lasting snow cover, low food availability, and inclement spring weather may inhibit early nesting (Stevenson and Bryant 2000; Irons et al. 2017). Steeper seasonal declines in productivity (Garcia-Heras et al. 2016) and population declines (Both et al. 2010) are more pronounced for species that nest in regions with strong seasonality and shorter nesting windows than those nesting in regions with weaker seasonality and longer nesting windows. Migrant birds, particularly long-distance migrants that often nest in highly seasonal environments, have incomplete knowledge of breeding ground conditions, which may impede their ability to adjust the timing of migration departure, duration, and arrival to match resource peaks (Rubolini et al. 2010). Conversely, resident birds have real-time information about breeding area resources and do not need to time their migration to match resource peaks, which may facilitate a shift to earlier nesting in response to changing local conditions, like advancing green-up dates (Samplonius et al. 2018). Long-distance migrants show fewer and weaker phenological responses (Rubolini et al. 2010; Samplonius et al. 2018), and stronger negative population effects of climate change than residents (Møller et al. 2008; Both et al. 2010).
American kestrels (Falco sparverius) are a widespread, generalist predator, breeding across much of North America (Smallwood and Bird 2020). Kestrel migration tendency and distance co-varies with latitude. At northern latitudes, kestrels are complete, long-distance migrants. Whereas at southern latitudes kestrels migrate shorter distances, and populations are partial migrants or complete residents (Smallwood and Bird 2020). There is extensive evidence of kestrel declines in eastern populations (Smallwood et al. 2009), but population trends in other regions are less clear and, in some cases, stable or increasing (McClure et al. 2017). The causes of declines and the reason for geographic differences in trends are still unclear, but regional differences in patterns of seasonality and climate change, and differing adaptive capacity of distinct genetic groups and migratory phenotypes (Smallwood et al. 2009; Ruegg et al. 2021) may contribute to these discrepancies. Western populations are advancing their nesting phenology in response to climate change (Heath et al. 2012; Smith et al. 2017), whereas this phenomenon has not been observed in eastern populations, creating a unique opportunity for comparative studies of the impacts of nesting phenology on survival. Nesting phenology of kestrels is positively correlated with the start of spring (estimated from the extended spring index, see supplement) and increasing Normalized Difference Vegetation Index (NDVI) values, which are a good proxy for small mammal and insect abundance (Lafage et al. 2014; Smith et al. 2017). Further, kestrels that nest before the start of spring (measured by extended spring index) have higher nesting success (i.e., ≥ 1 young fledged) and productivity (i.e., number of young fledged) than later nesters across their breeding range (Callery et al. 2022a). However, the relationship between nesting phenology and survival is not as well understood, even though the population dynamics of kestrels are particularly sensitive to changes in adult survival (McClure et al. 2021). Here we examined how nesting phenology relates to the apparent survival of adult and juvenile birds so that we can better understand how climate-driven changes in spring might impact populations.
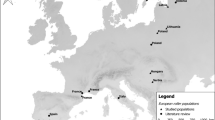
We used long-term mark-and-recapture data from sites in western (Idaho) and eastern (New Jersey) North America (Fig. 1a) to investigate how the timing of nesting relative to the start of spring affected the apparent survival of adult American kestrels and their offspring. We hypothesized that variation in nesting phenology and parental investment in raising young within sites, and regional differences in nesting windows between sites (~ 4 months in the West, ~ 2 months in the East) would explain the apparent survival of adults and that nesting phenology and regional differences would explain apparent survival of juveniles. We predicted that later-nesting adults and hatch-year (HY) birds from later nests would have lower apparent survival rates than early-nesting adults and hatch-year birds from earlier nests, respectively, and we expected this effect to be stronger at the eastern site, where the nesting window is shorter. We also expected a stronger negative effect of late nesting on the apparent survival of adults raising young, compared to adults with failed nests, because of higher energy demands. Finally, we included sex and winter temperatures in apparent survival analyses because of their effects on juvenile recruitment rates of kestrels (Steenhof and Heath 2013) and we examined temporal trends in clutch-initiation dates for both populations.
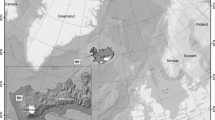
Locations (a) of the long-term mark-and-recapture studies used to estimate apparent survival of American kestrels with insets of annual Normalized Difference Vegetation Index (NDVI) from Jan 1–Dec 31 (2001–2020) from five locations within 1 km of nest boxes within the western site (Idaho, b) and eastern site (New Jersey, c). The blue lines represent a smoothed average. Though the study sites are at a similar latitude, growing seasons are more pronounced at the eastern site compared to the western site where vegetation green-up is more heterogeneous and less peaked
Methods
We collected mark-and-recapture data from birds nesting in nest box networks in Idaho and New Jersey (Fig. 1a) with long-term monitoring programs. The study site in southwestern Idaho (43° N, 116° W) encompasses approximately 1000 km2 within a mixture of sagebrush steppe, agriculture, and rangelands, alongside exurban and suburban areas in the municipalities of Kuna, Meridian, and Boise. Wooden nest boxes were erected on roadside utility poles or trees and spaced 1 km apart on average. The number of nest boxes at this study site ranged from 98 to 113, depending on the year. Occupancy averaged 42% (± 14.4 SD) and, on average, occupied boxes (i.e., American kestrels with ≥ 1 egg) were 1.5 km apart. The magnitude of seasonal changes in primary productivity is relatively low and variable across landscapes and years (Fig. 1b, see supplement for NDVI methods). The study site in northwestern New Jersey (41° N, 74° W) encompasses approximately 200 km2 and is comprised of agricultural lands with open fields embedded within forested areas in Sussex and Warren counties (Smallwood et al. 2009). Wooden nest boxes were erected on roadside utility poles, trees, and barns or other buildings, and spaced 1 km apart. The number of nest boxes at this study site ranged from 96 to 127, depending on the year. Occupancy averaged 28% (± 9.5 SD) and, on average, occupied boxes were 1.5 km apart. Seasonal changes in primary productivity at the New Jersey site are relatively higher in magnitude and consistent from year to year (Fig. 1c).
We captured and marked adult American kestrels nesting in nest boxes in Idaho from 2008 to 2017, and in New Jersey from 1997 to 2017. We monitored nest boxes from March to July and systematically checked for occupancy (every 1–3 weeks in Idaho; every 3–4 weeks in New Jersey). We hand-captured adults in nest boxes during the incubation stage. We then banded birds with US Geological Survey aluminum bands, measured and sexed (by plumage) adults, and returned them to the nest box. Recaptured adults, or birds that had been banded previously elsewhere, were recorded as already banded. On average, we trapped 80% (± 7.4 SD) and 39% (± 16.6 SD) of nesting adults in Idaho and New Jersey, respectively. Unless kestrels were first captured and marked as nestlings, we were unable to age after-hatch-year (adult, AHY) birds to more specific age categories. We returned to the nest box to band, measure, and sex nestlings when they were between 18 and 25 days old. We considered a nesting attempt to be successful if it produced at least one young that reached 25 days old (Anderson et al. 2016; Smallwood 2016).
We recorded the clutch-initiation date for each nesting attempt by different methods, depending on the status of the nest when it was discovered. When we discovered an incomplete clutch of eggs in a nest box, we back-calculated clutch-initiation dates assuming kestrels laid one egg every other day (Bird and Palmer 1988). For clutches that were discovered complete and the eggs hatched, we back-calculated 30 days from the hatching date and twice the clutch size to estimate clutch initiation. If complete clutches never hatched, we used the midpoint between the earliest and latest possible date of clutch initiation. The earliest possible date of clutch initiation was the date we last found the box empty or, if we removed another species nest (i.e., a European starling, Sturnus vulgaris, made the box unavailable to kestrels), the date we removed the nest material plus one. We estimated the latest possible date through back-dating, as described above. If the range of possible dates was > 15, we considered the clutch-initiation date as unknown. For clutches that were discovered complete and hatched, we used the ages of the nestlings, determined by plumage characteristics (Griggs and Steenhof 1993), to back-calculate the clutch-initiation date by subtracting the plumage age of the most mature nestling, 30 days for incubation, and twice the clutch size.
We used extended spring index (SI-x) models to estimate the start of spring. SI-x provides a fine-scale (1 km), validated model of the start of spring across North America (Izquierdo-Verdiguier et al. 2018). These models were developed using daily maximum and minimum surface temperatures to predict the first-bloom dates of lilac (Syringa chinensis and S. vulgaris), and honeysuckle cultivars (Lonicera tatarica and L. korolkowii, Schwartz et al., 2006; Rosemartin et al., 2015). SI-x measures are highly correlated with land surface metrics (e.g., NDVI; Zurita-Milla et al. 2017), but in North America SI-x values have proven more predictive of bird phenology than NDVI (Kelly et al. 2016) and are positively associated with American kestrel nest initiation (supplemental information). We extracted SI-x dates derived from Daymet climate datasets (Thornton et al., 2018) at the latitude and longitude of each occupied nest box per year using Google Earth Engine code modified from Izquierdo-Verdiguier et al. (2018). We used NDVI to represent seasonal patterns in primary productivity because NDVI captures year-round green-up and senescence (methods in supplement information).
Ideally, nesting timing would be best represented as a continuous representation of the degree of overlap between seasonal resources and resource requirements of nesting birds (Miller-Rushing et al. 2010). However, multi-state survival models require individual time-varying covariates to be categorical. We calculated the difference (in days) between the clutch-initiation date and the year-specific SI-x date for each nest attempt and compared the difference to the median difference for each study site. Then, we categorized nesting attempts as “early” or “late” depending on whether the individual nesting attempt was before (early) or after (late) the median. If a bird attempted to nest more than once in a season (n = 16), the latest successful nesting attempt was considered when assigning the bird to a timing group for that year (n = 8), or if both nesting attempts were unsuccessful (n = 8), the latest nesting attempt was considered.
We calculated winter minimum temperature anomalies for each study site to determine winter severity. We used minimum temperatures because, especially in winter months, minimum temperatures limit species distributions (Root 1988; Zuckerberg et al. 2011) and lower minimum temperatures affect energetic requirements associated with thermoregulation (Meijer et al. 1999). The use of anomaly values allowed for standardized representation of climate change across locations with different minimum temperatures. We used Google Earth Engine (Gorelick et al. 2017) to extract minimum temperatures from the Daymet dataset, which provides daily gridded climate data at 1-km resolution (Thornton et al. 2018). For each year, we averaged daily minimum temperature values within a minimum bounding box of all nest box locations for each study area. For each study site, we calculated winter minimum temperature anomalies for each year as the difference between the mean winter minimum temperature and the mean winter minimum temperature from a 30-year (1981–2010) baseline period.
Statistical analysis
For the survival analysis, we created mark-recapture models using the multi-state model framework in Program MARK (White and Burnham 1999), using the RMark package and interface (Laake and Rexstad 2008). Multistate mark-recapture models estimate apparent survival (S), and capture probability (p) similar to Cormack–Jolly–Seber mark-recapture survival models (Lebreton and Pradel 2002); additionally, these models estimate transition probability (Ψ) between categorical states (Schwarz et al. 1993). We created six states, hereafter referred to as strata, to represent nesting timing, age, and nesting success of adults:
-
1)
HY from a brood initiated “early”
-
2)
HY from a brood initiated “late”
-
3)
a successful AHY that initiated egg-laying “early”
-
4)
a successful AHY that initiated egg-laying “late”
-
5)
an unsuccessful AHY that initiated egg-laying “early”
-
6)
an unsuccessful AHY that initiated egg-laying “late”
In addition to individual strata, we included sex as an individual-level, static covariate and winter minimum temperature anomaly as a population-level, time-varying covariate to explain apparent survival.
We created capture histories for each bird by coding their presence or absence in each year of the study and assigning the appropriate stratum according to their age, timing category, and nesting success, for each year they were present. Most (> 85%) American kestrels breed in their second year, and there was no difference between clutch-initiation dates between second year and older kestrels (Steenhof and Heath 2009). We had few known-age birds, so we did not consider age effects other than categorical hatch-year and after-hatch-year designations. We designed a model set for apparent survival that included all additive and interactive models of the multi-state variable stratum, sex, and annual minimum winter temperature anomaly (“winter temperature”). We examined models for p that included the covariates of winter temperature and sex because we typically capture a higher proportion of females compared to males (Steenhof and Heath 2013), and we created a candidate model set for Ψ that included an intercept-only model and an effect of current stratum membership. We did not have the sample size to build more complex models for Ψ. We fixed the Ψ estimates of transition from AHY to HY and between HY strata to 0 (Fig. S5). We used an iterative process to find the best model by selecting the top model for p, then the best model for Ψ, and used the top models for p and Ψ to build models for S. We ran separate mark-recapture analyses for western and eastern sites, using the same model set for each analysis. We ran goodness of fit tests on survival models with the variable ‘strata’ using functions from the package R2UCare (Gimenez et al. 2018). Specifically, we used the functions overall_JWV to test the fit of the model, test3Gsr to test for the presence of transients, and test3Gwbwa to test for the presence of memory. For all tests we failed to reject the null hypotheses (p > 0.05) indicating model fit, and lack of issues with transients and memory.
We examined temporal trends in clutch-initiation dates for each study site using generalized linear models with Gamma distributions and log link functions in the R package glmmTMB (Brooks et al. 2017). We used a Gamma distribution because clutch-initiation dates are positive data. We plotted residual dispersion to check model assumptions.
We compared models using Akaike’s information criterion corrected for small sample size (AICc) and considered the models with the lowest AICc to be most informative (Anderson and Burnham 2004). We estimated 85% confidence intervals for model parameters to be compatible with model selection criteria (Arnold 2010), and we considered effects statistically unclear if 85% confidence intervals overlapped zero (Dushoff et al. 2019). We report parameters as estimate ± standard error and with their 85% confidence intervals. We conducted all analyses in R (R Core Team 2021, version 4.0.5).
Results
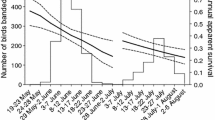
We captured and marked 1430 (first marked as AHY = 507, HY = 923) individual kestrels at the western site in Idaho and 1405 (first marked as AHY = 284, HY = 1121) individual kestrels at the eastern site in New Jersey. These individuals were associated with 369 nest attempts from 2008 to 2017, and 301 nest attempts from 1997 to 2017, at western and eastern sites, respectively. At the western site, clutch initiation ranged from early March through late June (Fig. 2a), and the median difference between clutch initiation and the start of spring was -17 days (std. deviation = 21 days, Fig. 2b). At the eastern site, clutch initiation ranged from late March through early June (Fig. 2a), and the median difference between clutch initiation and the start of spring was − 8 days (std. deviation = 12 days, Fig. 2b).
Density distributions of the clutch-initiation (CI) dates (a), and the difference between clutch-initiation date and extended spring index date (SI-x, b) for nests at western (Idaho, shaded orange, n = 369, 2008–2017) and eastern (New Jersey, shaded blue, n = 301, 1997–2017) sites. The orange dashed line represents the median overall CI date for western nests in “a” (April 12th) and the overall median difference between CI and SI-x in “b” (− 17 days). The blue dashed line represents the overall median CI date for eastern nesting attempts in “a” (April 27th) and the overall median difference between CI and SI-x in “b” (− 8 days)
The best-supported model for recapture probability contained a covariate for sex. Western males were more likely to be recaptured than females (β = 0.44; 85% CI 0.06–0.81, Table S2) and eastern males were less likely to be recaptured than females (β = − 1.21; 85% CI -1.7–0.71, Table S3). The best-supported model for the transition probability was the intercept-only model (Table S4, Table S5). Previous strata state did not explain the transition probability to alternate states.
The best-supported model for apparent survival at the western site included additive effects for the multi-state variable stratum (includes nesting timing, age, and nesting success), sex, and winter temperature (Table 1). The best-supported model for apparent survival at the eastern site contained the stratum and sex covariates (Table 2). Successful “early” nesting birds had higher apparent survival rates compared to successful “late” nesters in the West (Fig. 3, Table 3). Successful “late” nesting birds had higher apparent survival rates compared to successful “early” nesters in the East (Fig. 3, Table 3). At both sites, successful adults had higher apparent survival rates than unsuccessful adults, and there was no difference in apparent survival of unsuccessful “early” and “late” adults. Also, there was no difference in apparent survival of “early” versus “late” hatch-year birds. Overall, hatch-year birds had lower apparent survival estimates than after-hatch-year birds.
Apparent survival estimates for female western (Idaho) American kestrels from 2008 to 2017 (top) and eastern (New Jersey) American kestrels from 1997 to 2017 (bottom) categorized by age, whether or not an adult successfully raised young, and nesting timing category across winter minimum temperature anomalies. Circles and triangles are mean estimates and bars represent 85% confidence intervals. Apparent survival of successful adults depended on whether they were in the early (green) or late (coral) nesting timing category. Nesting timing did not affect apparent survival rates of hatch-year birds or adults that did not successfully rear offspring. Apparent survival rates of western kestrels increased as winter minimum temperature anomaly increased, but this result was statistically unclear for eastern kestrels
Winter minimum temperature anomalies were positively associated with higher apparent survival estimates (β = 0.43; 85% CI 0.21–0.64, Fig. 3) in the West, and males tended to have higher survival estimates than females, but the 85% confidence interval for sex crossed zero, so we considered this effect statistically unclear (β = 0.11; 85% CI − 0.23–0.45). The top model did not include winter temperature in the East. It did include sex, but the confidence interval for sex crossed zero (β = − 1.04; 85% CI − 1.30–0.01); therefore, we considered this effect statistically unclear.
Clutch initiation date advanced (β = − 0.009; 85% CI − 0.014–0.003) over the nine-year study period in the West (Fig. 4), whereas clutch-initiation dates did not change over the 20-year study period in the East (β = 0.003; 85% CI − 0.002–0.004, Fig. 4).
Trends in clutch-initiation dates of American kestrel nests at the western (Idaho) site from 2008 to 2017 (orange) and eastern (New Jersey) site from 1997 to 2017 (blue). The shaded areas represent the 85% confidence interval around the predicted line, and each point represents the clutch-initiation date at an occupied nest box. American kestrels are nesting earlier at the western site, but there is no change in nesting phenology at the eastern site
Discussion
Our comparative study on the effects of nesting phenology on the apparent survival of American kestrels showed differing trends at two distinct long-term monitoring sites in western and eastern North America. At both sites, nesting phenology affected the apparent survival of adult kestrels that raised young, but the direction of the effect differed between populations, with earlier nesters having higher apparent survival than later nesters in the West, and later nesters having higher apparent survival than earlier nesters in the East. Given that both populations have seasonal declines in productivity (Callery et al. 2022a), these results suggest that condition-dependent trade-offs between reproduction and survival exist for eastern kestrels, whereas this trade-off is not apparent for western kestrels. Furthermore, despite advancing springs across North America, clutch-initiation dates tended not to change at the eastern site, but they are advancing at the western site. Seasonal trade-offs may constrain shifts in nesting phenology in response to earlier springs in the eastern population, whereas the high apparent survival and productivity of early nesters in the West may make that population well-suited to respond to directional pressure to breed earlier (Fig. 5).
The density distributions of the difference between clutch-initiation (CI) dates and the start of spring (SI-x) in polygons, apparent survival estimates (mean and 85% confidence interval) of an early (green point and line) and late (coral triangle and line) successful, adult females, and seasonal trends in productivity (dark green line) based on Callery et al. (2022a) for the western (Idaho) site (a) and eastern (New Jersey) site (b). At the western site, seasonal declines in both productivity and apparent survival may be allowing for earlier nesting in response to climate change via directional selection, whereas at the eastern site an inverse pattern between apparent survival and productivity may create a constraint for earlier nesting
Results from the western site were consistent with our predictions that earlier nesting adults raising young during peak availability of resources would have higher apparent survival than later-nesting adults. Further, because early-nesting American kestrels also have high productivity (Callery et al. 2022a), results suggest that, in the West, resources do not create a trade-off between survival and reproduction, that early nesters are higher quality individuals, or both. Without experimental manipulation, the effects of nesting timing and individual quality are difficult to tease apart, and most likely a combination of nesting timing and individual effects contribute to the success of early nesters (Verhulst and Nilsson 2008). Winter temperatures have warmed over the past two decades at the western site (Heath et al. 2012, unpub data) and spring is advancing, especially on irrigated croplands within the study area (Smith et al. 2017). Warming winters and earlier springs may have removed previous constraints to early nesting, such as early-season cold temperatures that increase thermoregulation costs of survival and delay egg production, or low food availability that affects incubation and raising young (Stevenson and Bryant 2000; Woodworth et al. 2017). Indeed, our results show that western kestrels had higher apparent survival with increasing winter temperature anomalies, and Heath et al. (2012) showed earlier nesting after warmer winters. Late summer at the western site is subject to extreme heat and drought conditions, increasingly so with climate change (Sohrabi et al. 2013), which may have negative effects on both the survival and productivity of later breeders (Albright et al. 2010). Together, the removal of early constraints, additional costs of late nesting, and seasonal declines in survival and productivity may explain the advancement of nesting phenology in response to climate change in the West.
At the eastern site, results did not fit our predictions—later-nesting adults that raised young had higher apparent survival than earlier nesters. This survival pattern contrasts with the observed seasonal decline in productivity (Callery et al. 2022a). The narrow distribution of clutch-initiation dates and seasonal changes in primary productivity suggest that there is a very narrow window of high resources for raising young at the eastern site. Seasonally limited resources may cause condition-dependent trade-offs between survival and reproduction (Stearns 1989). Early in the season adults may allocate resources to raising young at a cost to survival, whereas later in the season this pattern is reversed. Alternatively, winter and early spring weather events, such as increased severity and frequency of blizzards (Cohen et al. 2018), and extreme precipitation events in spring (Huang et al. 2017) may decrease survival through delay in arrival time, and a decrease in prey activity, hunting efficiency, and provisioning rates (Olsen and Olsen 1992). Unpredictable early season conditions suggest that the potential survival costs of arriving at the breeding grounds and nesting early are high and nesting later may be a safer strategy (Stevenson and Bryant 2000). Inverse seasonal patterns in survival and reproduction may represent different investments by individuals of different ages or experiences. Though we did not find evidence of associations between age and timing. Hatch-year birds transitioned to both early and late states, and previous research at the western site found no difference in clutch-initiation dates between second-year birds and older birds (Steenhof and Heath 2009). Narrow resource windows and inverse seasonal patterns of survival and reproduction may constrain eastern American kestrels from nesting earlier in response to climate change.
Migratory phenotype is another individual-level trait that can affect nesting phenology, with long-distance migrants generally nesting later than residents and short-distance migrants because of arrival time constraints and incomplete knowledge of breeding ground conditions (Rubolini et al. 2010). In American kestrels, the arrival time of short-distance migrants is earlier in years with warmer spring temperatures, whereas the arrival time of long-distance migrants remains unchanged (Powers et al. 2021). The kestrel population at the western site is partially migratory (i.e., some individuals migrate, while others are year-round residents; Anderson et al. 2016), and migration distances are becoming shorter with warming winters (Heath et al. 2012). Hence, flexible migratory strategies may work in concert with reduced environmental constraints to allow for earlier nesting in Idaho. Less is known about migratory phenotypes and distances of kestrels from the eastern site, although Smallwood and Bird (2020) suggest that at these mid-latitudes, juveniles tend to be fully migratory, whereas adult migratory strategy may be dependent on weather patterns. Results showed that the apparent survival of eastern kestrels was not affected by winter temperatures, which could suggest that a large proportion of the population are migratory, so do not experience local winter weather conditions. Furthermore, because migration is the life history stage with the highest mortality risks (Klaassen et al. 2014; reviewed in Robinson et al. 2020), it is difficult to tease apart whether migratory strategy is directly affecting apparent survival or indirectly affecting apparent survival through nesting phenology. Finally, genetic differences between earlier and later breeding birds (Saino et al. 2017) and between migrants and residents (Ruegg et al. 2021) suggest that underlying genetic composition may be affecting nesting phenology directly or indirectly through migratory phenotype. American kestrels in western North America are genetically distinct from those in eastern North America and may have differing levels of adaptive capacity to shift phenology (Ruegg et al. 2021).
Overall, adults with failed nests had lower apparent survival rates than adults that raised young, which could reflect lower inherent individual fitness (Blums et al. 2005), or higher dispersal and permanent emigration rates of unsuccessful adults compared to successful adults (Steenhof and Heath 2009). We did not detect effects of nesting phenology in adults with failed nests, but only in those that raised young. The brood-rearing stage is when concurrent parental and offspring energy demand is at its highest (Hillström and Moreno 1992), so limited resources are most impactful during this stage. The majority of unsuccessful nests at both sites failed at the egg stage (Idaho: 87%, New Jersey: 78%), suggesting these individuals did not invest as much energy and resources as those raising young, so were unaffected by nesting timing. Additionally, nesting adults may mitigate their own mortality risk by abandoning nests when survival costs are too high compared to potential productivity benefits (Clutton-Brock 2019).
Consistent with previous studies (reviewed by Martin 1995), the apparent survival of hatch-year birds was lower than that of adults. However, contrary to previous studies of kestrels and other birds (Verhulst and Tinbergen 1991; Catry et al. 2016; Robinson et al. 2020), the apparent survival of hatch-year birds was not affected by the timing of nests they were hatched from. Nestlings hatched later in the season may have fewer siblings than those hatched earlier in the season because of reduced clutch size, hatching success, or nestling survival (Callery et al. 2022a). This could potentially mitigate the costs of later hatching on size, condition (Shutler et al. 2006), and subsequent survival of the remaining nestlings. Alternatively, the low overall apparent survival of hatch-year birds in our study may have limited our ability to detect a significant effect of nesting timing on apparent survival of this age class.
It is important to note that our model, like most survival models, estimates apparent survival as opposed to true survival (Lebreton and Pradel 2002; Schaub and Royle 2014) and hence cannot distinguish between death and permanent emigration (Schaub and Royle 2014). American kestrels with higher dispersal rates (e.g., unsuccessful breeders, Steenhof and Heath 2009) or longer dispersal distances (e.g., later hatched young, Miller and Smallwood 1997; migrants, McCaslin et al. 2020) have lower recapture likelihood and hence lower apparent survival estimates, so we cannot disentangle whether nesting phenology or dispersal patterns decreased apparent survival estimates.
In sum, although spring is advancing at both the western and eastern sites, American kestrel populations are responding differently. Western kestrels are nesting earlier, whereas nesting phenology did not change for eastern kestrels. In the West, flexible migratory strategies and warmer winters may reduce early-nesting constraints and allow kestrels to maximize fitness by nesting earlier when both productivity and survival rates are highest. Hence, this population may be well-suited to respond to directional pressure to breed earlier with a warming climate. Conversely, at the eastern site, potential constraints from early inclement weather and narrow seasonal availability of resources may result in trade-offs between seasonal declines in productivity and seasonal increases in survival. These trade-offs may constrain nesting phenology in eastern populations in response to climate-driven advances in spring, which is particularly concerning in light of steep declines in eastern populations.
Data availability
Data are available at Callery et al. (2022b).
References
Albright TP, Pidgeon AM, Rittenhouse CD, Clayton MK, Wardlow BD, Flather CH, Culbert PD, Radeloff VC (2010) Combined effects of heat waves and droughts on avian communities across the conterminous United States. Ecosphere 1:1–22
Anderson D, Burnham K (2004) Model selection and multi-model inference Second NY. Springer-Verlag 63:1007
Anderson AM, Novak SJ, Smith JF, Steenhof K, Heath JA (2016) Nesting phenology, mate choice, and genetic divergence within a partially migratory population of American kestrels. Auk: Ornithol Adv 133:99–109
Arnold TW (2010) Uninformative parameters and model selection using Akaike’s Information Criterion. J Wildl Manag 74:1175–1178
Bastianelli O, Charmantier A, Biard C, Bonamour S, Teplitsky C, Robert A (2021) Is earlier reproduction associated with higher or lower survival? Antagonistic results between individual and population scales in the blue tit. bioRxiv. https://doi.org/10.1101/2021.01.11.426202
Bird DM, Palmer RS (1988) American kestrel. In: Palmer RS (ed) Handbook of North American birds, vol 5: diurnal raptors, part 2. Yale University Press, New Haven, pp 253–290
Blums P, Nichols JD, Hines JE, Lindberg MS, Mednis A (2005) Individual quality, survival variation and patterns of phenotypic selection on body condition and timing of nesting in birds. Oecologia 143:365–376
Both C, van Turnhout CA, Bijlsma RG, Siepel H, van Strien AJ, Foppen RP (2010) Avian population consequences of climate change are most severe for long-distance migrants in seasonal habitats. Proc R Soc B: Biol Sci 277:1259–1266
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400
Callery KR, Schulwitz SE, Hunt AR, Winiarski JM, McClure CJ, Fischer RA, Heath JA (2022a) Phenology effects on productivity and hatching-asynchrony of American kestrels (Falco sparverius) across a continent. bioRxiv. https://doi.org/10.1101/2022.01.14.476385
Callery KR, Smallwood JA, Hunt AR, Snyder ER, Heath JA (2022b) Dataset for Seasonal trends in adult apparent survival and reproductive trade-offs reveal potential constraints to earlier nesting in a migratory bird [Data set]. https://doi.org/10.18122/bio_data.9.boisestate
Catry T, Moreira F, Alcazar R, Rocha PA, Catry I (2016) Mechanisms and fitness consequences of laying decisions in a migratory raptor. Behav Ecol 28:222–232
Clutton-Brock TH (2019) The evolution of parental care. Princeton University Press, Princeton
Cohen J, Pfeiffer K, Francis JA (2018) Warm Arctic episodes linked with increased frequency of extreme winter weather in the United States. Nat Commun 9:1–12
Dunn P (2004) Breeding dates and reproductive performance. Adv Ecol Res 35:69–87
Dushoff J, Kain MP, Bolker BM (2019) I can see clearly now: reinterpreting statistical significance. Methods Ecol Evol 10:756–759
Garcia-Heras MS, Arroyo B, Mougeot F, Amar A, Simmons RE (2016) Does timing of breeding matter less where the grass is greener? Seasonal declines in breeding performance differ between regions in an endangered endemic raptor. Nat Conserv 15:23–45
Gimenez O, Lebreton J-D, Choquet R, Pradel R (2018) R2ucare: an r package to perform goodness-of-fit tests for capture–recapture models. Methods Ecol Evol 9:1749–1754
Golet GH, Irons DB, Estes JA (1998) Survival costs of chick rearing in black-legged kittiwakes. J Anim Ecol 67:827–841
Gorelick N, Hancher M, Dixon M, Ilyushchenko S, Thau D, Moore R (2017) Google earth engine: planetary-scale geospatial analysis for everyone. Remote Sens Environ 202:18–27
Griggs GR, Steenhof K (1993) Photographic guide for aging nestling American kestrels. USDI Bureau of Land Management Raptor Research Technical Assistance Center, Boise
Heath JA, Steenhof K, Foster MA (2012) Shorter migration distances associated with higher winter temperatures suggest a mechanism for advancing nesting phenology of American kestrels Falco sparverius. J Avian Biol 43:376–384
Hillström L, Moreno J (1992) Variation in time and energy budgets of breeding wheatears. Behaviour 120:11–39
Huang H, Winter JM, Osterberg EC, Horton RM, Beckage B (2017) Total and extreme precipitation changes over the northeastern United States. J Hydrometeorol 18:1783–1798
Irons RD, Harding Scurr A, Rose AP, Hagelin JC, Blake T, Doak DF (2017) Wind and rain are the primary climate factors driving changing phenology of an aerial insectivore. Proc R Soc B: Biol Sci 284:20170412
Izquierdo-Verdiguier E, Zurita-Milla R, Ault TR, Schwartz MD (2018) Development and analysis of spring plant phenology products: 36 years of 1-km grids over the conterminous US. Agric for Meteorol 262:34–41
Kelly JF, Horton KG, Stepanian PM, de Beurs KM, Fagin T, Bridge ES, Chilson PB (2016) Novel measures of continental-scale avian migration phenology related to proximate environmental cues. Ecosphere 7:e01434
Klaassen RH, Hake M, Strandberg R, Koks BJ, Trierweiler C, Exo KM, Bairlein F, Alerstam T (2014) When and where does mortality occur in migratory birds? Direct evidence from long-term satellite tracking of raptors. J Anim Ecol 83:176–184
Laake J, Rexstad E (2008) RMark—an alternative approach to building linear models in MARK. In: Cooch E, White GC (eds) Program MARK: a gentle introduction
Lack DL (1968) Ecological adaptations for breeding in birds. London, Methuen & Co
Lafage D, Secondi J, Georges A, Bouzillé JB, Pétillon J (2014) Satellite-derived vegetation indices as surrogate of species richness and abundance of ground beetles in temperate floodplains. Insect Conserv Divers 7:327–333
Lebreton JD, Pradel R (2002) Multistate recapture models: modelling incomplete individual histories. J Appl Stat 29:353–369
Lof ME, Reed TE, McNamara JM, Visser ME (2012) Timing in a fluctuating environment: environmental variability and asymmetric fitness curves can lead to adaptively mismatched avian reproduction. Proc R Soc B: Biol Sci 279:3161–3169
Martin K (1995) Patterns and mechanisms for age-dependent reproduction and survival in birds. Am Zool 35:340–348
McCaslin HM, Caughlin TT, Heath JA (2020) Long-distance natal dispersal is relatively frequent and correlated with environmental factors in a widespread raptor. J Anim Ecol 89:2077–2088
McClure CJ, Schulwitz SE, van Buskirk R, Pauli BP, Heath JA (2017) Commentary: research recommendations for understanding the decline of American kestrels (Falco sparverius) across much of North America. J Raptor Res 51:455–464
McClure CJ, Brown JL, Schulwitz SE, Smallwood J, Farley KE, Therrien JF, Heath JA (2021) Demography of a widespread raptor across disparate regions. Ibis 163:658–670
Meijer T, Nienaber U, Langer U, Trillmich R (1999) Temperature and timing of egg-laying of European Starlings. Condor 101:124–132
Miller KE, Smallwood JA (1997) Natal dispersal and philopatry of southeastern American Kestrels in Florida. Wilson Bull 1:226–232
Miller-Rushing AJ, Høye TT, Inouye DW, Post E (2010) The effects of phenological mismatches on demography. Philos Trans R Soc B: Biol Sci 365:3177–3186
Møller AP (1994) Phenotype-dependent arrival time and its consequences in a migratory bird. Behav Ecol Sociobiol 35:115–122
Møller AP, Rubolini D, Lehikoinen E (2008) Populations of migratory bird species that did not show a phenological response to climate change are declining. Proc Natl Acad Sci 105:16195–16200
Nilsson JǺ, Svensson E (1996) The cost of reproduction: a new link between current reproductive effort and future reproductive success. Proc R Soc Lond B 263:711–714
Olsen P, Olsen J (1992) Does rain hamper hunting by breeding raptors? Emu-Austral Ornithol 92:184–187
Powers BF, Winiarski JM, Requena-Mullor JM, Heath JA (2021) Intra-specific variation in migration phenology of American kestrels (Falco sparverius) in response to spring temperatures. Ibis 163:1448–1456
R Core Team (2021) R: A language and environment for statistical computing R Foundation for Statistical Computing. Vienna, Austria. https://wwwR-project.org/
Reed TE, Jenouvrier S, Visser ME (2013) Phenological mismatch strongly affects individual fitness but not population demography in a woodland passerine. J Anim Ecol 82:131–144
Robinson RA, Meier CM, Witvliet W, Kéry M, Schaub M (2020) Survival varies seasonally in a migratory bird: Linkages between breeding and non-breeding periods. J Anim Ecol 89:2111–2121
Root T (1988) Energy constraints on avian distributions and abundances. Ecology 69:330–339
Rosemartin AH, Denny EG, Weltzin JF, Lee Marsh R, Wilson BE, Mehdipoor H, Zurita-Milla R, Schwartz MD (2015) Lilac and honeysuckle phenology data 1956–2014. Sci Data 2:1–8
Rubolini D, Saino N, Møller AP (2010) Migratory behaviour constrains the phenological response of birds to climate change. Climate Res 42:45–55
Ruegg KC, Brinkmeyer M, Bossu CM, Bay RA, Anderson EC, Boal CW, Dawson RD, Eschenbauch A, McClure CJ, Miller KE, Morrow L, Morrow J, Oleyar MD, Ralph B, Schulwitz S, Swem T, Therrien J, van Buskirk RW, Smith TB, Heath JA (2021) The American kestrel (Falco sparverius) genoscape: Implications for monitoring, management, and subspecies boundaries. Ornithology 138:ukaa051
Saino N, Ambrosini R, Albetti B, Caprioli M, De Giorgio B, Gatti E, Leichti F, Parolini M, Romano A, Romano M, Scandolara C, Gianfranceschi L, Bollati V, Rubolini D (2017) Migration phenology and breeding success are predicted by methylation of a photoperiodic gene in the barn swallow. Sci Rep 7:1–10
Samplonius JM, Bartošová L, Burgess MD, Bushuev AV, Eeva T, Ivankina EV, Kerimov AB, Krams I, Laaksonen T, Magi M, Mand R, Potti J, Torok J, Trnka M, Visser ME, Zang H, Both C (2018) Phenological sensitivity to climate change is higher in resident than in migrant bird populations among European cavity breeders. Glob Change Biol 24:3780–3790
Schaub M, Royle JA (2014) Estimating true instead of apparent survival using spatial Cormack-Jolly-Seber models. Methods Ecol Evol 5:1316–1326
Schwartz MD, Ahas R, Aasa A (2006) Onset of spring starting earlier across the Northern Hemisphere. Glob Change Biol 12:343–351
Schwarz CJ, Schweigert JF, Arnason AN (1993) Estimating migration rates using tag-recovery data. Biometrics 49:177–193
Shutler D, Clark RG, Fehr C, Diamond AW (2006) Time and recruitment costs as currencies in manipulation studies on the costs of reproduction. Ecology 87:2938–2946
Smallwood JA (1988) A mechanism of sexual segregation by habitat in American Kestrels (Falco sparverius) wintering in southcentral Florida. Auk 105:36–46
Smallwood JA (2016) Effects of researcher-induced disturbance on American Kestrels breeding in nest boxes in northwestern New Jersey. J Raptor Res 50:54–59
Smallwood JA, Bird DM (2020) American Kestrel (Falco sparverius), version 10. In: Poole AF, Gill FB (eds) Birds of the World. Cornell Lab of Ornithology, Ithaca. https://doi.org/10.2173/bow.amekes.01
Smallwood PD, Smallwood JA (1998) Seasonal shifts in sex ratios of fledgling American kestrels (Falco sparverius paulus): the early bird hypothesis. Evol Ecol 12:839–853
Smallwood JA, Causey MF, Mossop DH, Klucsarits JR, Robertson B, Robertson S, Mason J, Maurer MJ, Melvin RJ, Dawson RD, Bortolotti G, Parrish JW, Breen TF, Boyd K (2009) Why are American kestrel (Falco sparverius) populations declining in North America? Evidence from nest-box programs. J Raptor Res 43:274–282
Smith SH, Steenhof K, McClure CJ, Heath JA (2017) Earlier nesting by generalist predatory bird is associated with human responses to climate change. J Anim Ecol 86:98–107
Sohrabi MM, Ryu JH, Abatzoglou J, Tracy J (2013) Climate extreme and its linkage to regional drought over Idaho, USA. Nat Hazards 65:653–681
Stearns SC (1989) Trade-offs in life-history evolution. Funct Ecol 3:259–268
Steenhof K, Heath JA (2009) American Kestrel reproduction: evidence for the selection hypothesis and the role of dispersal. Ibis 151:493–501
Steenhof K, Heath JA (2013) Local recruitment and natal dispersal distances of American kestrels. Condor 115:584–592
Stevenson IR, Bryant DM (2000) Climate change and constraints on breeding. Nature 406:366–367
Thornton PE, Thornton M, Mayer BW, Wei Y, Devarakonda R, Vose RS, Cook RB (2018) Daymet: Daily Surface Weather Data on a 1-km Grid for North America, Version 3. ORNL DAAC, Oak Ridge
Verhulst S, Nilsson JA (2008) The timing of birds’ breeding seasons: A review of experiments that manipulated timing of breeding. Philos Trans R Soc B 363:399–410
Verhulst S, Tinbergen JM (1991) Experimental evidence for a causal relationship between timing and success of reproduction in the great tit Parus major. J Anim Ecol 60:269–282
Visser ME, Holleman LJ, Gienapp P (2006) Shifts in caterpillar biomass phenology due to climate change and its impact on the breeding biology of an insectivorous bird. Oecologia 147:164–172
White GC, Burnham KP (1999) Program MARK: survival estimation from populations of marked animals. Bird Study 46:S120–S139
Woodworth BK, Wheelwright NT, Newman AE, Schaub M, Norris DR (2017) Winter temperatures limit population growth rate of a migratory songbird Nature. Communications 8:1–9
Zuckerberg B, Bonter DN, Hochachka WM, Koenig WD, DeGaetano AT, Dickinson JL (2011) Climatic constraints on wintering bird distributions are modified by urbanization and weather. J Anim Ecol 80:403–413
Zurita-Milla R, Goncalves R, Izquierdo-Verdiguier E, Ostermann FO (2017) Exploring vegetation phenology at continental scales: linking temperature-based indices and land surface phenological metrics. In: Proceedings of the 2017 Conference on Big Data from Space, Toulouse, pp 28–30
Acknowledgements
We thank Jason Winiarski for the extended spring index analysis. Jason Winiarski and Chris McClure provided statistical advice and thoughtful comments on early drafts of the manuscript. Comments from our handling editor, Rob Robinson, and two anonymous reviewers greatly improved the manuscript. We thank landowners for allowing access to their property.
Funding
Funding for this project was provided by a grant from the Strategic Environmental Research and Development Program (SERDP) of the US Department of Defense (US DoD; Award Number: RC-2702) and the Boise State and American Kestrel Partnership Adopt-A-Box Partners. Release time for JAS was provided by the Faculty Scholarship Program, Montclair State University.
Author information
Authors and Affiliations
Contributions
KRC, JAH, JAS, ARH originally formulated the idea, all authors conducted fieldwork and curated data for analysis, KRC and JAH analyzed data, KRC wrote the first draft of the manuscript and all authors contributed to revision.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethics approval
American kestrels were handled and marked under the authority of federal bird banding permits (JAH: 23307, JAS: 21378), New Jersey scientific collecting permit SC 2017023, and its predecessors, Idaho scientific collecting permits, and institutional IACUCs.
Additional information
Communicated by Thomas Koert Lameris.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Callery, K.R., Smallwood, J.A., Hunt, A.R. et al. Seasonal trends in adult apparent survival and reproductive trade-offs reveal potential constraints to earlier nesting in a migratory bird. Oecologia 199, 91–102 (2022). https://doi.org/10.1007/s00442-022-05169-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-022-05169-w