Abstract
Animal species often show substantial intraspecific trait variability (ITV), yet evidence for its flexibility across multiple ecological scales remains poorly explored. Gaining this knowledge is essential to better understand the different processes maintaining ITV in nature. Due to their broad geographic ranges, widespread invasive species are expected to display strong phenotypic variations across their distribution. Here, we quantified the scale-dependent patterns of morphological variability among invasive populations of two global freshwater invaders—red swamp crayfish Procambarus clarkii and pumpkinseed sunfish Lepomis gibbosus—both established in American and European lakes. We quantified patterns in body morphology across different ecological (Individual and Population) and spatial scales (Region). We then analyzed the scale-dependency of morphological variations among lake populations that span a diversity of abiotic and biotic conditions. Next, we used stable isotope analyses to test the existence of ecomorphological patterns linking morphology and trophic niche of individuals. We found that trait variations mainly accounted for at the regional and individual levels. We showed that populations of both species strongly differed between United States and Europe whereas habitat characteristics had a relatively minor influence on morphological variations. Stable isotope analyses also revealed that ecomorphological pattern for the trophic position of L. gibbosus was region-dependent, whereas no ecomorphological patterns were observed for P. clarkii. Overall, our study strongly supports the notion that the patterns of phenotypic variability among invasive populations are likely to modulate the ecological impacts of invasive species on recipient ecosystems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Across a species’ geographic range, individuals commonly exhibit considerable phenotypic variation (Pfennig et al. 2010; Richardson et al. 2014). Investigating the mechanisms associated with intraspecific trait variability (ITV) is fundamental to understanding the patterns and implications of ecological and evolutionary dynamics (Bolnick et al. 2011; Des Roches et al. 2018). Recent advances in plant ecology have demonstrated that the patterns of ITV frequently depend on both the spatial and ecological scales of investigation (e.g., Messier et al. 2010; Chalmandrier et al. 2017; Li et al. 2018). However, despite a growing number of studies quantifying ITV in animals (e.g., fish: Quevedo et al. 2009; insect: Araújo and Gonzaga 2007; mammal: Tinker et al. 2008; crustacean: Jackson et al. 2017; birds: Dubuc-Messier et al. 2017; reptiles: Bestion et al. 2015; amphibians: Costa-Pereira et al. 2018), we still know surprisingly little about phenotypic variations among nested hierarchical scales (i.e., “nested variation”; Foster et al. 1998). This insufficient knowledge likely reflects the challenge to acquire individual data at large spatial scales and across multiple populations (Moran et al. 2016), thus dampening our ability to reveal replicated divergence within taxon (Foster et al. 1998).
ITV may vary across different spatial scales in response to various ecological conditions faced by organisms. Trait variability at large spatial scales (e.g., regional scale) can manifest in response to biogeographical processes such as isolation and migration. At smaller scales (e.g., population scale), local processes such as species interactions and resource availability are key factors maintaining ITV, ultimately influencing the capacity of species to respond to heterogeneous local environmental conditions (Araújo et al. 2011). In addition, ITV is now considered as a key factor mediating the effects of individuals on community structure and ecosystem functioning (Des Roches et al. 2018). Therefore, elucidating the spatial structuring of ITV and its relationship with environmental conditions can provide new insight into global trends of ITV-induced ecological changes.
Determining the magnitude and geography of invasive species impacts remains a key challenge in ecology and conservation biology. Owing to their large distribution in their native and non-native ranges, invasive species often experience a wide range of local environmental conditions and display high phenotypic variability (Sol and Lefebvre 2000; Knop and Reusser 2012). At large scales, ecological success is also influenced by invasion history, i.e., propagule pressure and the origin of founders (Lockwood et al. 2009). For instance, higher propagule size (number of individuals) and increased number of introduction events are likely to promote phenotypic variability, especially if founders originate from different source populations (Ahlroth et al. 2003). Widespread non-native species include populations with diverse introduction histories experiencing heterogeneous environmental condition. Consequently, members of these populations are expected to display high variability in phenotype responses to environmental pressures across broad geographic scales. Knowledge regarding the scale-dependency of ITV may enhance our ability to predict the ecological impacts of invasive species across different invaded areas.
All major vertebrate groups demonstrate intraspecific morphological variability (Smith and Skùlason 1996). Body shape often depends strongly on local environmental conditions and the presence of competitors and predators can favor phenotypes associated with escape ability (Domenici et al. 2008) and/or promote investment into morphological defenses (Laforsch and Tollrian 2004). Habitat-induced morphological variability has also been documented in the past. For example, freshwater organisms inhabiting complex nearshore (littoral) lake habitats are often deeper-bodied to improve maneuverability (Svanbäck and Eklöv 2002). Such morphological variations may help to describe the functional role of freshwater organisms and notably their distinct foraging strategies (Wainwright 1996). Although ecomorphological patterns linking morphology and resource use of individuals are well studied along dichotomous habitats (e.g., littoral-pelagic axis; Quevedo et al. 2009; Faulks et al. 2015), our knowledge of this association along a gradient of environmental conditions is still limited.
In the present study, we first assessed morphological variability of two co-existing global invaders—red swamp crayfish Procambarus clarkii and pumpkinseed sunfish Lepomis gibbosus—across two ecological scales (i.e., individual, population) for lakes located in North America (northwestern United States) and Europe (southwestern France). We then quantified the spatial scale-dependency of phenotype-environment relationships, and finally explored associations between morphological traits and trophic niche of individuals to investigate the potential functional implication of morphological variations. We expected that morphological variations across individuals would contribute substantially to the overall ITV (Messier et al. 2010). Local environmental conditions would shape morphological variability at a local scale, but this would also differ between regions and reflect the importance of large-scale environmental filters for ITV (Moran et al. 2016).
Materials and methods
Study system
Native to southern United States and north-eastern Mexico, P. clarkii has been widely introduced to western United States as well as throughout all continents except Australia and Antarctica (Hobbs et al. 1989). The species was introduced to the west coast of United States initially as forage for frog farms in the 1930s but more recently has expanded its nonnative distribution via introductions from the aquarium and biological supply trades (Larson and Olden 2011). P. clarkii is widespread in Europe, being first introduced in the 1970s for aquaculture purpose and continuing to spread across France and at least 16 other countries via both human-induced and natural dispersal (Souty-Grosset et al. 2016). In the two studied areas, evidence suggests that P. clarkii may have similar dates of introduction: first documented around 1995 in south-western France (Changeux 2003) and 2000 in Washington State (Mueller 2001). P. clarkii is an opportunistic omnivore with a vegetable-based diet preference (Gherardi 2006; Jackson et al. 2017) and has been reported to induce important ecological impacts across entire aquatic ecosystems (Twardochleb et al. 2013; Alp et al. 2016).
Native to central and eastern North America, L. gibbosus was introduced into northwestern United States as early as 1890, and to Washington State between 1930 and 1945 when approximately 160 lakes were stocked to establish a panfish sport fishery (Washington Department of Fish and Wildlife 2005). Pathways for European introduction were primarily motivated by ornamental reason over a century ago and its establishment in Haute-Garonne occurred around 1945 (Copp and Fox 2007). The species is an opportunistic omnivore with an animal-based diet and preferentially forage on both littoral and pelagic invertebrates (García-Berthou and Moreno-Amich 2000). Consequently, the impacts of L. gibbosus mainly include reduction of native invertebrate species (van Kleef et al. 2008).
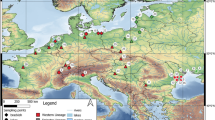
Our study examined lake ecosystems in the United States and in France (further description available in Larson and Olden 2013 and Zhao et al. 2016). A total of 15 lakes were included in two distant regions: 9 lakes in proximity to Seattle, Washington State (northwestern United States) and 6 lakes in proximity to Toulouse, Haute-Garonne (southwestern France) (Fig. 1). Littoral zones of the American lakes are characterized by a mixture of silt and detritus with patchy aquatic vegetation (e.g., macrophyte, water lily) and riparian forest dominated by native evergreen species and shrubs. By contrast, the French lakes are gravel pit lakes with steep banks and their substrates are dominated by a mix of sand, gravel and pebble. Moreover, aquatic vegetation (e.g., macrophytes) in gravel pit lakes is very limited and vegetable matter mainly occurs through allochthonous inputs of leaf litter from deciduous trees (mainly Populus sp.) (Alp et al. 2016). Lake size was estimated using GIS and lakes were selected to ensure comparable size (surface area ranging from 9 to 43 ha, mean France = 15 ha ± 2.5 SE; mean United States = 23 ha ± 3.7 SE) and similar ranges of environmental conditions (ESM Appendix A).
Samples collection
P. clarkii and L. gibbosus were sampled in summer 2014 from 04-Aug to 11-Aug in the United States and from 16-Sept to 24-Sept in France. In each lake, crayfish were sampled in the littoral zone using baited traps deployed overnight and active searching with dip nets when needed. L. gibbosus from the littoral zone were collected using combination of baited fyke nets, seine netting and electrofishing. Following capture, individuals were immediately euthanized with an overdose of anesthetic, stored on ice and then preserved at − 20 °C in laboratory for subsequent analyses. After defrosting, individuals were measured for carapace length (CL ± 0.1 mm) and fork length (FL ± 1 mm). To limit the potential effect of ontogeny in our analyses, a subsample of adult P. clarkii (CL from 30.0 to 71.0 mm) and L. gibbosus (FL from 65 to 170 mm) were taken in each lake to ensure consistent variability in morphological and trophic traits as well as body size overlap across the sampled lakes. These selected individuals (total number of P. clarkii per population = 209, average = 19.0 ± 0.7 SE; total number of L. gibbosus per population = 227, average = 18.9 ± 1.5) were weighed (W ± 0.1 g) and then used for subsequent analyses.
Habitat characteristics
For each lake, a set of habitat characteristics was selected to encompass biotic and abiotic conditions that were comparable between the two regions and based on their well-described influence on individual morphology (ESM Appendix A). Lake surface area (km2), lake perimeter (m), lake altitude (m) and lake depth (max and mean depth; m) were calculated using aerial images as well as, bathymetric and topographic data. Shoreline development index (SDI) was calculated following the formula SDI = perimeter/2√(π × surface area) (Hutchinson 1957). Secchi disk depth (m), chlorophyll-a concentration (µg L−1) and total phosphorus concentration (µg L−1) were calculated as average values measured on two occasions during early June and early September 2014. In one lake (noted U7), however, these three parameters were calculated using data collected in 2004 since no recent information was available. Land use practices have remained relatively unchanged over the past decade, making it possible to include this estimate. The integrative trophic status index (TSI) was also estimated using Secchi depth, chlorophyll-a and total phosphorus concentrations (Carlson 1977). Fish diversity was estimated as the number of fish species present in each lake according to Zhao et al. (2016) for France and the Washington Department of Fish and Wildlife (http://wdfw.wa.gov/) for the United States. The overall habitat characteristics were synthetized into a multidimensional space using a principal component analysis (PCA). The first two PCA axes (variance explained > 10% and eigenvalue > 1) explained 46.4% and 20.7% of the total variation across all habitat variables, respectively, and were used as composite variables in subsequent analyses. Increased PC1 values (environmental axis 1, hereafter) were primarily associated to shallow and productive lakes while increased PC2 values (environmental axis 2, hereafter) were mainly associated to large lakes with complex littoral zone and high fish diversity (see details in ESM Appendix A).
Morphological analyses
For each species, individual morphological variations were quantified using a geometric morphometric technique (Zelditch et al. 2004). This landmark-based thin-plate spline (TPS) analysis is a powerful approach for quantifying body shape variation and covariation between body shape and environmental factors. P. clarkii were photographed dorsally whereas L. gibbosus were photographed on the left side. To describe body shape morphology, homologous landmarks (19 for P. clarkii and 18 for L. gibbosus; ESM Appendix B, Fig. B1) were digitized for each individual by the same operator using the software TpsDig2 v.2.17 (Rohlf 2015). Landmarks were selected to approximate the overall body shape of individuals with emphasis on head and were similar to other landmark-based crayfish and sunfish morphology studies (Parsons and Robinson 2007; Etchison et al. 2012). For each species, the landmark data were then imported into the software MorphoJ v.1.06d where a Generalized Procrustes Analysis (GPA) was performed to obtain superimposed landmark coordinates (Klingenberg 2011). GPA facilitated the removal of differences in size, position and orientation from the original landmark coordinates among individuals, although allometric relationships remained. The GPA coordinates were projected into a weight matrix to characterize shape using non-affine (partial warps) and affine (uniform) components of thin plate spline. For each species, morphological variations between populations were explored by computing a PCA of the weight matrix (relative warp analysis, RWA) in MorphoJ. The patterns of body shape variation along RW scores were visualized using outline diagrams generated in MorphoJ. Only the first two RW axes which explained the maximum of the variation in the data (RW1 = 36.2% and RW2 = 25.4% for red swamp crayfish; RW1 = 37.1% and RW2 = 13.1% for pumpkinseed sunfish) were used for further statistical analyses.
Stable isotope analyses
Carbon and nitrogen stable isotope (δ13C and δ15N) analyses of individuals and their putative resources were used to quantify the trophic niche of P. clarkii and L. gibbosus. Specifically, δ13C was used to determine the origin of the carbon in a consumer diet (e.g., terrestrial versus aquatic, littoral versus pelagic) and δ15N was used to indicate the trophic position of consumers within the food web (Layman et al. 2012). From each individual, a sample of white abdominal and dorsal muscle was collected for subsequent stable isotope analyses for P. clarkii and L. gibbosus, respectively. In addition, putative resources were collected in three different locations of each lake and during data collection sampling. Specifically, floating aquatic macrophytes and terrestrial leaf litter were collected by hand. In addition, periphyton samples were collected by gently brushing the surface of three randomly submerged cobbles and rinsing them with distilled water. Finally, arthropods from the littoral zone (Chironomidae, Ephemeroptera, Assellidae, Sialidae and Gastropoda; pooled samples with 1–23 specimens per sample) and zooplankton from the pelagic zone were collected with a pond net and a 100-μm mesh net, respectively. Stable isotope analyses for gastropods were performed on the soft muscle tissue. Once in the laboratory, periphyton samples were freeze-dried (− 50 °C for 5 days) while other samples were oven dried (60 °C for 48 h). All samples were then ground to a fine powder and analysed for stable isotope values (δ13C and δ15N) at the Cornell Isotope Laboratory (Ithaca, NY). Carbon and nitrogen stable isotope ratios are expressed relative to standards as δ13C and δ15N, respectively. Carbon isotope values of primary consumers were lipid-corrected for subsequent analyses because their average C:N were higher (invertebrates: mean = 4.57 ± 1.09 SD; zooplankton: 5.07 ± 1.09 SD) than the suggested limits (3.5 for aquatic organisms; Post et al. 2007) (ESM Appendix C).
δ13C values were converted to a corrected carbon isotopic ratio (δ13Ccor) adjusted for between-population variation in stable isotope baselines following Post (2002):
where δ13Cind is the stable isotope value of consumers (i.e., P. clarkii and L. gibbosus), δ13Cbase1 and δ13Cbase2 are the stable isotope values of baselines. Specifically, these baselines are aquatic primary producers (mixture of macrophyte and periphyton) and terrestrial leaf litter for P. clarkii; and littoral arthropods and pelagic zooplankton for L. gibbosus (ESM Appendix C). Trophic positions of each consumers were calculated following Vander Zanden et al. (1997):
where δ15Ni is the isotopic value of an individual i, δ15Nbase is the mean stable isotope values of resources (aquatic primary producers and terrestrial leaf litter for P. clarkii; littoral arthropods and pelagic zooplankton for L. gibbosus), ΔN is the TEF obtained from previous studies (3.8‰ and 3.3‰ for P. clarkii and L. gibbosus, respectively; see details in Jackson et al. 2017) and λbase is the trophic position of the resources used to estimate the baseline (λbase = 1 for primary producers and λbase = 2 for primary consumers). The δ15N values of putative resources in lake F3 were abnormal and samples from this lake were not used in subsequent trophic analyses (Fig. C2).
Statistical analyses
For each species, variance component analyses of morphological traits were assessed across different scales: Individual nested within Population (referring to the sampled lakes) and nested within Region (referring to North America or Europe). These three scales contain a mixture of ecological (Individual and Population) and spatial (Region) factors. The variance partitioning was accomplished using linear mixed effects models fitted with the method of restricted maximum likelihood (REML) and following Messier et al. (2010). Variance component analyses were then conducted on these models using the “varcomp” function from the “ape” package (v.5.1; Paradis et al. 2004).
For each species, linear mixed effects models (i.e., one for each morphological axis) with “Population” as random effect were also used to assess the effects of habitat characteristics and geographic location on the morphological scores RW1 and RW2 of all individuals (n = 209 and n = 227 for P.clarkii and L. gibbosus, respectively). In all models, the centroid size of each individual was used as a surrogate of overall body size to investigate allometry (i.e., non-independence of shape and size). Centroid size is the square root of the summed squared deviation of the coordinates from the centroid. The centroid was calculated as the center of gravity obtained by averaging the x and y coordinates of all landmarks. The first two PC axes (i.e., environmental axis 1 and environmental axis 2), geographic origin “Region” and their interactions, as well as interaction between “Region” and centroid size were included in the model to test for the scale-dependency of morphological variability along gradient of environmental conditions. In addition, linear models were used to assess the relationship between morphology and trophic niche within each species. Full models included the trophic position and δ13Ccor values of individuals as dependent variables, whereas morphology (i.e., RW1 and RW2 scores), “Regionˮ and their interactions were used as predictor variables.
For each full model, interactions were removed when non-significant using a backward selection procedure. Linear mixed effects models were performed using “lme” from the “nlme” package (v.3.1.137; Pinheiro et al. 2018). Significance of all factors was evaluated with Type II tests using “Anova” from the “car” package (v.3.0.2; Fox and Weisberg 2011), calculating Wald Chi square statistics (χ2) for mixed effects models and F-ratio statistics for linear models (Langsrud 2003; Fox and Weisberg 2011). Given the multiplicity of comparisons involved, the false discovery rate (FDR; Benjamini and Hochberg 1995) procedure was applied to correct for alpha inflation using the “p.adjust” function (base-package v.3.1.2). The significant results after the FDR procedure are reported. Assumptions of linearity and homogeneity of variances on residuals from all models were checked visually. Trophic position and δ13Ccor values were log10 transformed to improve the fit of the models. For each linear mixed effect model, both the marginal R2 (\(R_{\text{M}}^{2}\), effect of the fixed variables) and conditional R2 (\(R_{\text{C}}^{2}\), effect of the fixed and random variables) were calculated (Nakagawa and Schielzeth 2013). All statistical analyses were performed using R v.3.5.1 (R Development Core Team 2018).
Results
Variation in body morphology
Substantial variation in P. clarkii and L. gibbosus body morphology was evident. P. clarkii with positive RW1 scores displayed a shorter rostrum, shorter head length, larger body length and reduced abdomen and telson width (Fig. 2a). Positive RW2 scores were associated with longer rostrum length, shorter head width, elongated cephalothorax and shorter abdomen and telson length (Fig. 2a). L. gibbosus with positive RW1 scores had a more rounded body with a wider anterior region compared to narrower anterior head region and a longer caudal peduncle for individuals with negative scores (Fig. 2b). Difference in eye position was also observed and positive RW1 scores were linked to smaller and upward-facing eyes, whereas negative RW1 scores depicted large and downward-facing eyes (Fig. 2b). Positive RW2 scores were associated with an elongated and narrow body, with large and terminal eyes (Fig. 2b).
Body shape variations between a eleven populations of Procambarus clarkii (n = 209) and b twelve populations of Lepomis gibbosus (n = 227) located in the United States (U1–U9, black symbols) and France (F1–F6, grey symbols). Landmark-based body shape is given for RW1 and RW2. Wireframe graphs represent the most extreme deviation from the consensus configuration and were generated using by regressing relative warp scores against landmark coordinates using MorphoJ
Morphological trait variations differed across the three scales (Table 1). For both species, most intraspecific variability in RW1 was between regions (65.0% and 77.4% of the total variability for P. clarkii and L. gibbosus, respectively) and individuals (34.3% and 17.3% for P. clarkii and L. gibbosus, respectively), while only a small part of morphological variation was accounted for at the population level (P. clarkii: 0.7%; L. gibbosus: 5.3%). The largest proportion of the variations of RW2 were explained at the individual level (P. clarkii: 74.5%; L. gibbosus: 78.1%). For P. clarkii, RW2 trait variation was slightly higher at the regional level (12.1%) than at the population level (13.5%). For L. gibbosus, the distribution of RW2 variations was better explained at the population level (21.9%) than at the regional level (< 0.01%).
Drivers of morphological variations
Individuals of both P. clarkii and L. gibbosus displayed considerable differences in their body morphology between regions (Fig. 2). P. clarkii from American lakes had a significantly lower RW1 scores that in France (χ2 = 24.76, P < 0.001; Table 2), indicating that American crayfish exhibited broader bodies compared to French crayfish (Fig. 2a). RW1 scores of L. gibbosus significantly differed between regions (χ2 = 61.93, P < 0.001; Table 2), with individuals in American lakes having a higher score than in France (Fig. 2b). Thus, American L. gibbosus displayed rounder bodies, while individuals in France had streamlined bodies. For both species, variations in RW2 scores did not significantly differ between countries (P. clarkii: χ2 = 0.41, P = 0.789; L. gibbosus: χ2 = 2.09, P = 0.317; Table 2).
There was no evidence for association between habitat characteristics and either RW1 or RW2 scores of individuals for the two species (Table 2; Fig. 3). The effect of centroid size on morphology was significant, indicating an allometric effect of size on morphological variations. RW1 and RW2 scores of P. clarkii and L. gibbosus significantly increased and decreased with increasing centroid size, respectively (χ2 = 17.14, P < 0.001 and χ2 = 142.97, P < 0.001 for P. clarkii and L. gibbosus, respectively; Table 2; Fig. 3a). There was also a significant region-dependent effect of centroid size on RW1 scores of L. gibbosus (χ2 = 8.17, P = 0.009; Table 2) and round-bodied L. gibbosus had a significantly longer body in American lakes, whereas it only slightly changed in French populations (Fig. 3d). RW2 scores of P. clarkii were not affected by centroid size (χ2 = 0.40, P = 0.789; Table 2).
Relationship between body shape (RW1 scores) and both a and d centroid body size (mm) and (b, c, e, f) habitat characteristics for Procambarus clarkii (circle; n = 209) and Lepomis gibbosus (triangle; n = 227). Significant relationships are displayed using continuous lines. Black and grey symbols represent individuals in the United States and France, respectively
Relationships between morphology and trophic niche
The trophic niche (i.e., trophic position and δ13Ccor values) of both species differed between regions and their observed ecomorphological patterns were also distinct. P. clarkii in American lakes had a significantly lower trophic position and δ13Ccor values than in France (F = 16.98, P < 0.001 and F = 9.96, P = 0.006; Table 3). However, there was no significant relationship between morphology and trophic niche (Table 3). L. gibbosus in American lakes displayed a significantly lower trophic position and δ13Ccor values than in France (F = 30.79, P < 0.001 and F = 18.27, P < 0.001; Table 3). The trophic position was significantly influenced by the interaction term between region and RW1 (F = 13.27, P = 0.001; Table 3). Specifically, the trophic position of round-bodied individuals increased and decreased in American and French lakes, respectively (Fig. 4b). The δ13Ccor values of L. gibbosus did not significantly differ with morphology (Table 3).
Relationship between body shape (RW1 scores) and trophic niche (a–b trophic position and c–d δ13Ccor values) of Procambarus clarkii (circle; n = 189) and Lepomis gibbosus (triangle; n = 207). Significant region-dependent relationship is displayed using continuous lines. Black and grey symbols represent individuals in the United States and France, respectively
Discussion
Our study supports the existence of substantial morphological variations within two co-existing global invaders—red swamp crayfish Procambarus clarkii and pumpkinseed sunfish Lepomis gibbosus—across two ecological scales (i.e., individual, population) for lakes located in North America and Europe. Morphological trait variation was explained by regional and individual scales. Our results also highlighted that local habitat characteristics played a minimal role in generating trait variations and that ecomorphological associations were not consistently present between species.
Our study highlights the need to consider nested variation when investigating ITV in animals. We found that for both species, the partitioning of morphological trait variations varied greatly between the different scales (RW1: 0.72–64.97% within P. clarkii and 5.25–77.44% within L. gibbosus). However, for each morphological trait, patterns of variations across scales were consistent among the two species. We found that for both species, variation in RW1 was mainly explained by the region, whereas variation in RW2 was driven by individuals. This suggests that similar processes shape the structure of morphological variations between those two species and that large-scale processes may act more strongly than small-scale processes, hence inducing stronger morphological variations. Nonetheless, contrasting distributions of trait variation may arise when considering different types of traits (Li et al. 2018), suggesting that future studies should also integrate physiological and behavioral traits. Overall, understanding the spatial flexibility of ITV in response to variations in local and global ecological conditions requires further consideration (Araújo and Costa-Pereira 2013). Gaining this knowledge will help inform better predictions regarding both the patterns and relevance of ITV across ecosystems.
For both focal species, we observed distinct morphological differences between regions regardless of similarities in local habitat characteristics. Combined effects of the invasion process itself and large-scale climatic conditions can cause inter-regional phenotypic variability among conspecific invaders (Moran et al. 2016). For instance, distinct histories of colonization (e.g., introduction date, propagule pressure, ecological traits of founders) may trigger phenotypic variability among isolated invasive populations through agents of evolution (i.e., founder effect, genetic drift and mutation-order selection) (Langerhans and DeWitt 2004; Blount et al. 2008; Rosenblum and Harmon 2011). These findings highlight the need to investigate the genetic structure of invasive populations to determine the interplay between large-scale environmental conditions and genetic predisposition on phenotypic variations. This would provide new insight into how the evolutionary history of populations influence patterns of phenotypic variability (Weese et al. 2012).
It was striking that both biotic and abiotic attributes of lakes failed to explain morphological variations among populations. Previous studies have demonstrated that individuals inhabiting lakes with complex littoral habitat typically displayed robust and deeper bodies whereas streamlined bodies are expected to occur in lakes with more pelagic habitat (Svanbäck and Eklöv 2004; Riopel et al. 2008). It is worth noting that individuals studied here mainly occur in the littoral area of the lakes and were thus collected along the littoral shoreline, which may constrain the morphological divergence commonly observed along the littoral-pelagic axis (Faulks et al. 2015). However, we also highlighted that both deep-bodied and streamlined individuals can be found within the littoral habitat, suggesting that other agents of selection may operate to counterbalance the predictable habitat-induced morphological variations (Colborne et al. 2015). The overall increased body depth of American L. gibbosus might be the result of their preferred benthic mollusk consumption (Twardochleb and Olden 2016) that requires higher maneuverability and thus produce deep-bodied morphologies. This suggests that both differences in local environmental conditions between countries (e.g., different resource availability might favor different selection) and contrasting invasion histories may ultimately support contrasting pathways upon which morphological variability is expressed.
Although the presence of potential predators and competitors is predicted to generate morphological variations (e.g., elongated body, larger caudal region and smaller anterior head/body region; Robinson and Parsons 2002), we found little evidence for an association between the number of fish species and the morphology of individuals. Rather, we found that L. gibbosus in American lakes displayed a rounded body, which increased with increasing body size. This external body shape potentially favors survival against gape-size limited piscivorous fish species (Nilsson et al. 1995). Different predator species encountered in American and French lakes may induce different selection on morphology through the use of various attacking tactics and prey handling strategies. In addition, unlike the American lakes, L. gibbosus populations in the French lakes are subjected to high angling pressure that might interact with natural environmental conditions to alter phenotypic and evolutionary changes (Alós et al. 2014; Evangelista et al. 2015).
The patterns we observed may imply that ITV within invasive species modulate the impacts of invaders on recipient communities and ecosystems, since intraspecific trophic variability influences the effect of organisms on ecosystem functioning (Harmon et al. 2009; Bassar et al. 2010). We showed that, in the French populations, deep-bodied L. gibbosus had a lower trophic position than streamlined individuals, potentially highlighting the role of mobile predators to couple littoral and pelagic habitats (Vander Zanden and Vadeboncoeur 2002). Many fish species display phenotypic divergence between co-existing littoral and pelagic morphs, including L. gibbosus, where individuals foraging on benthic prey having deeper bodies, smaller eyes, shorter head and larger mouth (Parsons and Robinson 2007). In addition, fish specializing on littoral prey tend to have a lower trophic position than individuals specialized on pelagic prey because pelagic zooplankton is 15N-enriched relative to littoral arthropods (Vander Zanden and Rasmussen 1999; Matthews et al. 2010). P. clarkii is omnivorous and predominantly consume primary producers (Gherardi 2006). However, resource availability and intraspecific competition could modify individual diet (Evangelista et al. 2014; Jackson et al. 2017) and increase the trophic position through increased consumption of aquatic invertebrates. The higher density of macrophytes and the lower level of intraspecific competition observed in the American lakes might explain their lower trophic position than in the French lakes.
In conclusion, we demonstrated the importance of spatial structuring of ITV among invasive species for assessing their adaptive capacities and ecological impacts across their range. Although ecomorphological associations were not consistently present, the presence of such associations within invasive species strongly suggests that their impacts on recipient ecosystems differ between populations. Consequently, identifying the drivers of phenotypic divergence can inform more nuanced and effective management actions for invasive species and their recipient communities (Závorka et al. 2018).
References
Ahlroth P, Alatalo RV, Holopainen A et al (2003) Founder population size and number of source populations enhance colonization success in waterstriders. Oecologia 137:617–620. https://doi.org/10.1007/s00442-003-1344-y
Alós J, Palmer M, Linde-Medina M, Arlinghaus R (2014) Consistent size-independent harvest selection on fish body shape in two recreationally exploited marine species. Ecol Evol 4:2154–2164. https://doi.org/10.1002/ece3.1075
Alp M, Cucherousset J, Buoro M, Lecerf A (2016) Phenological response of a key ecosystem function to biological invasion. Ecol Lett 19:519–527. https://doi.org/10.1111/ele.12585
Araújo MS, Costa-Pereira R (2013) Latitudinal gradients in intraspecific ecological diversity. Biol Lett 9:20130778. https://doi.org/10.1098/rsbl.2013.0778
Araújo MS, Gonzaga MO (2007) Individual specialization in the hunting wasp Trypoxylon (Trypargilum) albonigrum (Hymenoptera, Crabronidae). Behav Ecol Sociobiol 61:1855–1863. https://doi.org/10.1007/s00265-007-0425-z
Araújo MS, Bolnick DI, Layman CA (2011) The ecological causes of individual specialisation. Ecol Lett 14:948–958. https://doi.org/10.1111/j.1461-0248.2011.01662.x
Bassar RD, Marshall MC, Lopez-Sepulcre A et al (2010) Local adaptation in Trinidadian guppies alters ecosystem processes. Proc Natl Acad Sci USA 107:3616–3621. https://doi.org/10.1073/pnas.0908023107
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B 57:289–300. https://doi.org/10.2307/2346101
Bestion E, Clobert J, Cote J (2015) Dispersal response to climate change: scaling down to intraspecific variation. Ecol Lett 18:1226–1233. https://doi.org/10.1111/ele.12502
Blount ZD, Borland CZ, Lenski RE (2008) Historical contingency and the evolution of a key innovation in an experimental population of Escherichia coli. Proc Natl Acad Sci USA 105:7899–7906. https://doi.org/10.1073/pnas.0803151105
Bolnick DI, Amarasekare P, Araújo MS et al (2011) Why intraspecific trait variation matters in community ecology. Trends Ecol Evol 26:183–192. https://doi.org/10.1016/j.tree.2011.01.009
Carlson RE (1977) A trophic state index for lakes. University of Minnesota, Minneapolis, p 55455. https://doi.org/10.4319/lo.1977.22.2.0361
Chalmandrier L, Münkemüller T, Colace M-P et al (2017) Spatial scale and intraspecific trait variability mediate assembly rules in alpine grasslands. J Ecol 105:277–287. https://doi.org/10.1111/1365-2745.12658
Changeux T (2003) Evolution de la répartition des écrevisses en France métropolitaine selon les enquêtes nationales menées par le conseil supérieur de la pêche de 1977 à 2001. Bull Fr Pêche Piscic 370–371:15–41. https://doi.org/10.1051/kmae:2003002
Colborne SF, Garner SR, Longstaffe FJ, Neff BD (2015) Assortative mating but no evidence of genetic divergence in a species characterized by a trophic polymorphism. J Evol Biol 29:633–644. https://doi.org/10.1111/jeb.12812
Copp GH, Fox MG (2007) Growth and life history traits of introduced pumpkinseed (Lepomis gibbosus) in Europe, and the relevance to its potential invasiveness. In: Gherardi F (ed) Biological invaders in inland waters: profiles, distribution, and threats. Springer, Berlin, pp 289–306
Costa-Pereira R, Rudolf VHW, Souza FL, Araújo MS (2018) Drivers of individual niche variation in coexisting species. J Anim Ecol. https://doi.org/10.1111/1365-2656.12879
Des Roches S, Post DM, Turley NE et al (2018) The ecological importance of intraspecific variation. Nat Ecol Evol 2:57–64. https://doi.org/10.1038/s41559-017-0402-5
Domenici P, Turesson H, Brodersen J, Bronmark C (2008) Predator-induced morphology enhances escape locomotion in crucian carp. Proc R Soc B 275:195–201. https://doi.org/10.1098/rspb.2007.1088
Dubuc-Messier G, Réale D, Perret P, Charmantier A (2017) Environmental heterogeneity and population differences in blue tits personality traits. Behav Ecol. https://doi.org/10.1093/beheco/arw148
Etchison L, Jacquemin SJ, Allen M, Pyron M (2012) Morphological variation of rusty crayfish Orconectes rusticus (Cambaridae) with gender and local scale spatial gradients. Int J Biol 4:163–171. https://doi.org/10.5539/ijb.v4n1p163
Evangelista C, Boiche A, Lecerf A, Cucherousset J (2014) Ecological opportunities and intraspecific competition alter trophic niche specialization in an opportunistic stream predator. J Anim Ecol 83:1025–1034. https://doi.org/10.1111/1365-2656.12208
Evangelista C, Britton RJ, Cucherousset J (2015) Impacts of invasive fish removal through angling on population characteristics and juvenile growth rate. Ecol Evol 5:2193–2202. https://doi.org/10.1002/ece3.1471
Faulks L, Svanbäck R, Eklöv P, Östman Ö (2015) Genetic and morphological divergence along the littoral–pelagic axis in two common and sympatric fishes: perch, Perca fluviatilis (Percidae) and roach, Rutilus rutilus (Cyprinidae). Biol J Linn Soc. https://doi.org/10.1111/bij.12452
Foster SA, Scott RJ, Cresko WA (1998) Nested biological variation and speciation. Philos Trans R Soc Lond B Biol Sci 353:207–218. https://doi.org/10.1098/rstb.1998.0203
Fox J, Weisberg S (2011) An R companion to applied regression, 2nd edn. Thousand Oaks, SAGE Publications
García-Berthou E, Moreno-Amich R (2000) Food of introduced pumpkinseed sunfish: ontogenetic diet shift and seasonal variation. J Fish Biol 57:29–40. https://doi.org/10.1111/j.1095-8649.2000.tb00773.x
Gherardi F (2006) Crayfish invading Europe: the case study of Procambarus clarkii. Mar Freshw Behav Physiol 39:175–191. https://doi.org/10.1080/10236240600869702
Harmon LJ, Matthews B, Des Roches S et al (2009) Evolutionary diversification in stickleback affects ecosystem functioning. Nature 458:1167–1170. https://doi.org/10.1038/nature07974
Hobbs HH, Jass JP, Huner JV (1989) A review of global crayfish introductions with particular emphasis on two North American species. Crustaceana 56:299–316. https://doi.org/10.1163/156854089X00275
Hutchinson GE (1957) A treatise on limnology, vol 1. Geography, Physics and Chemistry, Wiley, New York, p 1015
Jackson MC, Evangelista C, Zhao T et al (2017) Between-lake variation in the trophic ecology of an invasive crayfish. Freshw Biol 62:1501–1510. https://doi.org/10.1111/fwb.12957
Klingenberg CP (2011) MorphoJ: an integrated software package for geometric morphometrics. Mol Ecol Resour 11:353–357. https://doi.org/10.1111/j.1755-0998.2010.02924.x
Knop E, Reusser N (2012) Jack-of-all-trades: phenotypic plasticity facilitates the invasion of an alien slug species. Proc R Soc B 279:4668–4676. https://doi.org/10.1098/rspb.2012.1564
Laforsch C, Tollrian R (2004) Inducible defenses in multipredator environments: cyclomorphosis in Daphnia cucullata. Ecology 85:2302–2311. https://doi.org/10.1890/03-0286
Langerhans RB, DeWitt TJ (2004) Shared and unique features of evolutionary diversification. Am Nat 164:335–349. https://doi.org/10.1086/422857
Langsrud Ø (2003) ANOVA for unbalanced data: use Type II instead of type III sums of squares. Stat Comput 13:163–167. https://doi.org/10.1023/A:1023260610025
Larson ER, Olden JD (2011) The state of crayfish in the Pacific Northwest. Fisheries 36:60–73. https://doi.org/10.1577/03632415.2011.10389069
Larson ER, Olden JD (2013) Crayfish occupancy and abundance in lakes of the Pacific Northwest, USA. Freshw Sci 32:94–107. https://doi.org/10.1899/12-051.1
Layman CA, Araujo MS, Boucek R et al (2012) Applying stable isotopes to examine food-web structure: an overview of analytical tools. Biol Rev 87:545–562. https://doi.org/10.1111/j.1469-185X.2011.00208.x
Li T, Wu J, Chen H et al (2018) Intraspecific functional trait variability across different spatial scales: a case study of two dominant trees in Korean pine broadleaved forest. Plant Ecol 219:875–886. https://doi.org/10.1007/s11258-018-0840-4
Lockwood JL, Cassey P, Blackburn TM (2009) The more you introduce the more you get: the role of colonization pressure and propagule pressure in invasion ecology. Divers Distrib 15:904–910. https://doi.org/10.1111/j.1472-4642.2009.00594.x
Matthews B, Marchinko KB, Bolnick DI, Mazumder A (2010) Specialization of trophic position and habitat use by sticklebacks in an adaptive radiation. Ecology 91:1025–1034. https://doi.org/10.1890/09-0235.1
Messier J, McGill BJ, Lechowicz MJ (2010) How do traits vary across ecological scales? A case for trait-based ecology. Ecol Lett 13:838–848. https://doi.org/10.1111/j.1461-0248.2010.01476.x
Moran EV, Hartig F, Bell DM (2016) Intraspecific trait variation across scales: implications for understanding global change responses. Global Chang Biol 22:137–150. https://doi.org/10.1111/gcb.13000
Mueller KW (2001) First record of the red swamp crayfish, Procambarus clarkii (Girard, 1852) (Decapoda, Cambaridae), from Washington State, USA. Crustaceana 74:003–1007
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R 2 from generalized linear mixed-effects models. Methods Ecol Evol 4:133–142. https://doi.org/10.1111/j.2041-210x.2012.00261.x
Nilsson PA, Brönmark C, Petterson LB (1995) Benefits of a predator-induced morphology in crucian carp. Oecologia 104:291–296. https://doi.org/10.1007/BF00328363
Paradis E, Claude J, Strimmer K (2004) APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20:289–290. https://doi.org/10.1093/bioinformatics/btg412
Parsons KJ, Robinson BW (2007) Foraging performance of diet-induced morphotypes in pumpkinseed sunfish (Lepomis gibbosus) favours resource polymorphism. J Evol Biol 20:673–684. https://doi.org/10.1111/j.1420-9101.2006.01249.x
Pfennig DW, Wund MA, Snell-Rood EC et al (2010) Phenotypic plasticity’s impacts on diversification and speciation. Trends Ecol Evol 25:459–467. https://doi.org/10.1016/j.tree.2010.05.006
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2018) nlme: linear and nonlinear mixed effects models. R package version 3.1-137. http://CRAN.R-project.org/package=nlme. Accessed 07 April 2018
Post DM (2002) Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83:703–718. https://doi.org/10.1890/0012-9658(2002)083%5b0703:USITET%5d2.0.CO;2
Post DM, Layman CA, Arrington DA et al (2007) Getting to the fat of the matter: models, methods and assumptions for dealing with lipids in stable isotope analyses. Oecologia 152:179–189. https://doi.org/10.1007/s00442-006-0630-x
Quevedo M, Svanbäck R, Eklöv P (2009) Intrapopulation niche partitioning in a generalist predator limits food web connectivity. Ecology 90:2263–2274
R Development Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Richardson JL, Urban MC, Bolnick DI, Skelly DK (2014) Microgeographic adaptation and the spatial scale of evolution. Trends Ecol Evol 29:165–176. https://doi.org/10.1016/j.tree.2014.01.002
Riopel C, Robinson BW, Parsons KJ (2008) Analyzing nested variation in the body form of Lepomid sunfishes. Environ Biol Fishes 82:409–420. https://doi.org/10.1007/s10641-007-9303-9
Robinson BW, Parsons KJ (2002) Changing times, spaces, and faces: tests and implications of adaptive morphological plasticity in the fishes of northern postglacial lakes. Can J Fish Aquat Sci 59:1819–1833. https://doi.org/10.1139/f02-144
Rohlf FJ (2015) The tps series of software. Hystrix 26:9–12. https://doi.org/10.4404/hystrix-26.1-11264
Rosenblum EB, Harmon LJ (2011) “Same same but different”: replicated ecological speciation at white sand. Evolution 65:946–960. https://doi.org/10.1111/j.1558-5646.2010.01190.x
Smith TB, Skùlason S (1996) Evolutionary significance of resource polymorphisms in fishes, amphibian and birds. Annu Rev Ecol Evol Syst 27:111–133
Sol D, Lefebvre L (2000) Behavioural flexibility predicts invasion success in birds introduced to New Zealand. Oikos 90:599–605. https://doi.org/10.1034/j.1600-0706.2000.900317.x
Souty-Grosset C, Anastácio PM, Aquiloni L et al (2016) The red swamp crayfish Procambarus clarkii in Europe: impacts on aquatic ecosystems and human well-being. Limnologica 58:78–93. https://doi.org/10.1016/j.limno.2016.03.003
Svanbäck R, Eklöv P (2002) Effects of habitat and food resources on morphology and ontogenetic growth trajectories in perch. Oecologia 131:61–70. https://doi.org/10.1007/s00442-001-0861-9
Svanbäck R, Eklöv P (2004) Morphology in perch affects habitat specific feeding efficiency. Funct Ecol 18:503–510. https://doi.org/10.1111/j.0269-8463.2004.00858.x
Tinker MT, Bentall G, Estes JA (2008) Food limitation leads to behavioral diversification and dietary specialization in sea otters. Proc Natl Acad Sci USA 105:560–565. https://doi.org/10.1073/pnas.0709263105
Twardochleb LA, Olden JD (2016) Non-native Chinese mystery snail (Bellamya chinensis) supports consumers in urban lake food webs. Ecosphere 7:e01293. https://doi.org/10.1002/ecs2.1293
Twardochleb LA, Olden JD, Larson ER (2013) A global meta-analysis of the ecological impacts of nonnative crayfish. Freshw Sci 32:1367–1382. https://doi.org/10.1899/12-203.1
van Kleef H, van der Velde G, Leuven RSEW, Esselink H (2008) Pumpkinseed sunfish (Lepomis gibbosus) invasions facilitated by introductions and nature management strongly reduce macroinvertebrate abundance in isolated water bodies. Biol Invasions 10:1481–1490. https://doi.org/10.1007/s10530-008-9220-7
Vander Zanden MJ, Rasmussen JB (1999) Primary consumer δ13C and δ15N and the trophic position of aquatic consumers. Ecology 80:1395–1404. https://doi.org/10.1890/0012-9658(1999)080%5b1395:PCCANA%5d2.0.CO;2
Vander Zanden MJ, Vadeboncoeur Y (2002) Fishes as integrators of benthic and pelagic food webs in lakes. Ecology 83:2152–2161. https://doi.org/10.1890/0012-9658(2002)083%5b2152:FAIOBA%5d2.0.CO;2
Vander Zanden MJ, Cabana G, Rasmussen JB (1997) Comparing trophic position of freshwater fish calculated using stable nitrogen isotope ratios (δ15N) and literature dietary data. Can J Fish Aquat Sci 54:1142–1158. https://doi.org/10.1139/f97-016
Wainwright PC (1996) Ecological explanation through functional morphology: the feeding biology of sunfishes. Ecology 77:1336–1343. https://doi.org/10.2307/2265531
Washington Department of Fish and Wildlife (2005) Warmwater fishes of Washington. Report #FM93-9
Weese DJ, Ferguson MM, Robinson BW (2012) Contemporary and historical evolutionary processes interact to shape patterns of within-lake phenotypic divergences in polyphenic pumpkinseed sunfish, Lepomis gibbosus. Ecol Evol 2:574–592. https://doi.org/10.1002/ece3.72
Závorka L, Lang I, Raffard A et al (2018) Importance of harvest-driven trait changes for invasive species management. Front Ecol Environ 16:317–318. https://doi.org/10.1002/fee.1922
Zelditch ML, Swiderski DL, Sheets DH, Fink WL (2004) Geometric morphometrics for biologists. Elsevier Academic Press, New York
Zhao T, Grenouillet G, Pool T et al (2016) Environmental determinants of fish community structure in gravel pit lakes. Ecol Freshw Fish 25:412–421. https://doi.org/10.1111/eff.12222
Acknowledgements
We are very grateful to our numerous colleagues for their help during sampling and thank Florian Vincent for creating the map of the studied areas. We thank two anonymous reviewers and the editors for their valuable comments.
Funding
This work was supported by a CNRS-INEE PICS (Ind_Eco_Evo_Inva) and GDRI (EFF), the ONEMA (Projet ISOLAC and ERADINVA) and a PRES-Toulouse Grant (Inva_Eco_Evo_Lac). CE and JC are part of EDB, part of the French Laboratory of Excellence project “TULIP” (ANR-10-LABX.41; ANR-11-IDEX-0002-02). JDO was support by the University of Washington H. Mason Keeler Endowed Professorship.
Author information
Authors and Affiliations
Contributions
CE, JC and JDO designed and conceived the study and led the fieldwork. CE analysed the data and wrote the first draft of the manuscript. All authors contributed critically to the preparation of the manuscript and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Authorizations to perform this study were provided by the “Arrêtés Préfectoraux—18/09/2014 and 30/10/2014”.
Additional information
Communicated by Leon A. Barmuta.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Evangelista, C., Olden, J.D., Lecerf, A. et al. Scale-dependent patterns of intraspecific trait variations in two globally invasive species. Oecologia 189, 1083–1094 (2019). https://doi.org/10.1007/s00442-019-04374-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-019-04374-4