Abstract
Predator–prey interactions primarily focus on prey life-stages that are consumed. However, animals in less vulnerable life-stages might also be influenced by the presence of a predator, making our understanding of predation-related impacts across all life-stages of prey essential. It has been previously demonstrated that Podisus maculiventris is a voracious predator of eggs and larvae of Leptinotarsa decemlineata, and that larvae will alter their behavior to avoid predation. However, the adult beetles are not readily consumed by P. maculiventris, raising the question of whether they will respond to predators to protect themselves or their offspring. Here, we examine the effect of predation risk by P. maculiventris, on three adult behaviors of L. decemlineata; colonization, oviposition, and feeding, and the resulting impact on host plant damage. In an open-field test, there was no difference in natural beetle colonization between plots with predation risk and control treatments. However, subsequent host plant damage by adult beetles was 63.9% less in predation risk treatments. Over the lifetime of adult beetles in field mesocosms, per capita feeding was 23% less in the predation risk treatment. Beetle oviposition was 37% less in the presence of predators in a short-term, greenhouse assay, and marginally reduced in longer term field mesocosms. Our results indicate that predation risk can drive relatively invulnerable adult herbivores to adjust behaviors that affect themselves (feeding) and their offspring (oviposition). Thus, the full impact of predator presence must be considered across the prey life cycle.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Predation has direct implications for prey biology through consumption which directly alters community and population dynamics. However, the risk of predation alone can also induce important changes in prey behavior and physiology (Lima and Dill 1990; Werner and Peacor 2003; Sheriff and Thaler 2014). The effects of predation risk on prey (known as non-consumptive effects, non-lethal effects, and trait-mediated interactions) have been shown to alter feeding rate and foraging behavior (Schmitz et al. 1997; Rypstra and Buddle 2013; Tan et al. 2013; Cherry et al. 2015), reproduction and life history (Xiong et al. 2015; Sheriff 2015; Rendon et al. 2016), and impact survival of prey—unrelated to direct consumption (Sitvarin et al. 2015; Wineland et al. 2015). To date, studies which examine the impact predation risk can have on prey in terrestrial arthropod systems have primarily focused on life-stages which are directly susceptible to predation [but see (Fill et al. 2012) for effects of predation risk on a non-prey insect]. Studies in other taxa demonstrate that invulnerable prey stages also evaluate predation risk when assessing suitability of habitats for the benefit of their offspring (Stav et al. 1999; Peckarsky et al. 2000; Munga et al. 2006). Throughout insect development, vulnerability to predators can shift due to changes in mobility, body structure, and effectiveness of antipredator defense mechanisms. In fact, since adult insects are generally more mobile than their juvenile counterparts, they have the ability to make larger scale habitat choices leaving them less vulnerable to predation.
Changes in adult behavior may serve to protect themselves and their offspring from predators. Alternative host selection for enemy-free space may benefit the adult and their offspring (Jaenike 1978; Thompson 1988; Peckarsky et al. 2000; Vonesh and Blaustein 2011). For example, the adult whitefly, Bemisia tabaci, is much less vulnerable to predation because of its ability to fly compared to their sessile juvenile counterparts. In this system, if predators are present on the host plant, adults will readily flee the plant and move to an alternative host (Lee et al. 2011). Offspring can be indirectly affected by parental predator exposure where reductions in parental feeding protect the adult but also affect the adult’s ability to produce young (Bond et al. 2005). In fact, feeding is one of the most vulnerable activities in an insect’s life, and herbivore prey often reduce feeding making them less conspicuous to predators (Bernays 1997; Rypstra and Buddle 2013). In addition, maternal stress may affect provisioning of developing offspring (Sheriff et al. 2009; Freinschlag and Schausberger 2016; Tigreros et al. 2017). Therefore, if invulnerable stages of prey might shift their behavior in response to predator presence, it is important to consider the impacts of predation risk across subsequent life-stages, rather than solely focusing on the most vulnerable life stage.
Adult insects have the potential to increase the safety of offspring by altering host-seeking, colonization, and oviposition behaviors (Tallamy 1984; Lefèvre et al. 2010). For adult Colorado potato beetles, the sequence of behavioral events following overwintering sets the stage for their success throughout the year. This includes: first, host location and colonization, subsequent feeding to acquire resources for future oviposition, mating, and, finally, oviposition. If adult beetles are solely concerned for themselves, and not the well-being of their offspring, we predict that feeding and, thus, host plant damage might be the only factors affected by increased risk. However, if adults change their behavior in response to potential risk to their offspring, then the initial colonization and oviposition could be impacted. Since the larval form of the Colorado potato beetle is negatively impacted by this predator through consumptive and non-consumptive effects (Kaplan et al. 2014; Hermann and Thaler 2014), we predicted that oviposition in habitats or patches of safety would be selected for.
To understand if exposure to predators by relatively invulnerable adult insects will have consequences within and across life-stages, we studied the changes in adult host selection, oviposition and feeding behavior, and host plant damage in response to predator presence. We used a predator–prey pair that co-occurs naturally in agroecosystems. The predatory-spined soldier bug (Podisus maculiventris) feeds on eggs and larvae of the Colorado potato beetle (Leptinotarsa decemlineata), but rarely feeds on adult beetles in the field (Hare 1990), leaving the adults at a low risk for predation themselves. Colorado potato beetle larvae have been shown to reduce feeding in the presence of predatory-spined soldier bugs (Kaplan et al. 2014; Hermann and Thaler 2014). Although Colorado potato beetle larvae are known to initiate general antipredator behaviors (Ramirez et al. 2010), changes in adult Colorado potato beetle behavior in response predation risk have not yet been demonstrated. Specifically, our study determined the behavioral and reproductive consequences of spined soldier bug predator exposure to relatively invulnerable adult Colorado potato beetles by asking the following questions: (1) Does the presence of predators alter natural beetle colonization and subsequent host plant damage in an open field? (2) Does predation risk alter short-term beetle oviposition? (3) Are there differences in host plant consumption or oviposition in response to predation risk over the adult life span?
Methods
Study system
Colorado potato beetles, L. decemlineata (Chrysomelidae: Coleoptera), were from a lab-reared colony obtained from a wild population in Tompkins County, NY, USA, and fed a diet of potato plants (Solanum tuberosum cv. Yukon Gold). Wild-collected beetles were introduced to the colony every season. Beetles were kept in 11-3/4″ cube BugDorm rearing cages (BioQuip Products Inc.) in the lab under an 16:8, L:D cycle.
Predatory-spined soldier bugs, P. maculiventris (Pentatomidae: Hemiptera), used in this study were lab-reared from a wild-collected population in Tompkins County, NY, USA, and reared on a diet of mealworms and potato plants under an 16:8, L:D cycle. Wild-collected spined soldier bugs were introduced to the colony each season. Spined soldier bug predation risk was manipulated in our experiments in one of the two ways: (1) by placing predators in breathable, see-through spun polyester bags on plants to physically separate them from prey but allow for volatile cues from the predators to persist or (2) by removing the final segment of the spined soldier bug’s rostrum with a sharp razor blade, so the predator could hunt but not kill Colorado potato beetles. The previous studies using this technique have demonstrated that physically manipulated (“risk”) predators are unable to consume prey but remain capable of surviving, mating, and plant-feeding normally (Griffin and Thaler 2006; Kaplan and Thaler 2010; Thaler et al. 2012; Hermann and Thaler 2014). However, direct interaction of the predator with its prey is not necessary, since the spined soldier bug pheromones are largely responsible for predator detection by these prey (Hermann and Thaler 2014). Therefore, the bagged predation risk treatment is also effective in eliciting non-consumptive predator effects.
Solanum tuberosum (cv. Yukon Gold potatoes) were planted in 4″ plastic pots with commercial potting soil. Plants were watered daily and fertilized weekly (21:5:20 N:P:K) in a greenhouse with an 16:8 light cycle. We used 2–3-week-old plants in the experiments.
Field colonization and host plant consumption in an open field
The effect of predation risk on beetle colonization and host plant damage was measured in an open-field, choice experiment. Yukon Gold potatoes were planted from tubers on 16 May 2013 in a field plot in Freeville, New York, 42°31′15.2″N: 76°19′18.9″W. Potatoes were spaced to follow commercial growing standards with 0.3 m between plants within a row and 1 m between plants across rows. Potatoes were left to grow undisturbed until breaking through the ground at which point they were covered (9 June 2013) with mesh fabric (Agrifabrics PRO17, Alpharetta, Georgia) to reduce colonization by insects. There were a total of 43 replicate plots, each plot consisting of five consecutive plants within a row: one treatment plant at one end and four subsequent surveyed plants to the left or right of the treatment plant. The treatment plant was always on the end of the plot, randomly assigned to the right end or left end, to account for possible spatial effects within the plot (differing colonization as you move further from the treatment plant). However, since we had low colonization in the field, there were not sufficient data to examine spatial effects. Instead, we pooled numbers of colonists across each replicate plot. When the plants were approximately 0.5-m tall (19-June-2013) the mesh fabric was removed and two treatments established: predation risk (five male and five female spined soldier bug adults bagged on the plant) and control (bagged plant). The predation risk treatment in this experiment utilized bagged predators rather than manipulated free-range predators to ensure confinement to our treatment plots and to allow for open plots where beetles could colonize naturally. Within the mesh bags that enclosed the entire treatment plant, predators were able to roam freely, mate, and plant feed. This design inhibited direct contact between the predators and prey, providing the colonizing beetles only a subset of cues to use as information. The plots were separated by at least 1 m within rows. The treatments were randomly assigned to plots throughout the field.
All four plants next to the bagged treatment or bagged control plant were monitored for adult Colorado potato beetle presence on six occasions starting on day 2 and every 3 days after that up to 18 days (the duration of the first beetle generation’s colonization window in 2013). On each occasion, beetles were removed along with any egg clutches, and at the end of the 18-day period, the total number of adults found in each replicate was tallied and pooled. The time between egg collections was not long enough for eggs to hatch, therefore, eliminating plant damage from hatching larvae.
At the end of the 18-day period, we assessed plant damage on the plant directly adjacent to the treatment or control plant to evaluate whether the predator treatment altered overall host plant damage by the beetles in the field. Only Colorado potato beetles and flea beetles (Epitrix cucurmeris) were found in significant numbers within the field; these two beetles create distinctly different feeding damage to potato foliage that was easily distinguishable using our methods. We quantified the amount of leaf area consumed by adult Colorado potato beetles feeding in each replicate by scanning all leaves (CanoScan LiDE 700F), and then, we analyzed images using the ImageJ, following methods in Hermann and Thaler (2014). There were a total of 41 replicates, 20 predator treatments, and 21 controls.
All experimental data were analyzed with JMP 10 (SAS Institute, Cary, NC, USA, 2012). Fisher’s exact test was used to determine whether the number of adult beetles colonizing treatment replicates differed between predation risk plots or control plots. To compare the amount of host plant damage in predation risk or control replicates, the data were log-transformed to correct for normality and analyzed using Student’s t test.
Short-term oviposition assay
To test the effects of predator presence on Colorado potato beetle oviposition, we set up a short-term, no-choice, experiment using potted potato plants. Individual plants were centered on top of a clear plastic deli lid (Solo®, product LGC88B). Each plant was then covered with a 30.5 × 12.5 cm clear cylinder made from plastic film (Grafix®, 0.005 Dura-Lar Film) that fit inside the lip of the plastic lid for a sealed arena. The clear plastic cylinder was closed on the top with a 700-μm-mesh fabric to prevent test insects from escaping and provide sufficient air exchange.
A single-mated adult female Colorado potato beetle was placed in each arena and randomly assigned one of two treatments: predation risk or control. For this experiment, the predation risk treatment consisted of two de-beaked spined soldier bug adults (one male and one female, manipulated as described above). The predators were able to roam the arena throughout the experiment. We chose to use non-lethal predators due to the possibility for predation in these small, no-choice arenas where no other food source is present for the predator. Control treatments were predator-free, receiving only one mated adult female Colorado potato beetle. Adult beetles were collected from the laboratory colony as they emerged, sexed, and placed in a small solo cup as male–female pairs for 24 h to mate. Mated females were used approximately 5–7 days post-emergence in laboratory experiments.
Experiments ran for 4 days in the greenhouse under a 16:8 L:D cycle between 24–27 °C. After 4 days, each plant arena was monitored for the number of egg clutches as well as the total number of eggs laid by each Colorado potato beetle. The beetles are polyandrous and will lay many egg clutches throughout their lifetime using resources acquired as larvae as well as during adult feeding (Hare 1990). Yet, females only need to mate with one male to reach reproductive potential (Boiteau 1988). If the beetle died or no eggs were found within an arena, the entire replicate was removed from the study. In total, 13 predation risk treatments and 15 control treatments were removed (3 due to death in the predation risk treatment and 5 from the control, as well as 10 from each treatment due to incorrect gender separation). Without the excluded replicates, a total of 68 replicates were completed, 34 from each treatment. A Student’s t test was conducted to compare the mean number of eggs as well as the mean number of clutches laid by beetles between treatments.
Lifetime oviposition and host plant consumption
To evaluate adult beetle oviposition and host plant consumption in a more natural setting, a no-choice experiment was done in small field mesocosms. Yukon Gold potato plants were sprouted in the lab and grown in 15.25-cm pots in enclosed field cages until they were between 20 and 40 cm in height. Recently emerged Colorado potato beetle adults were allowed to mate for 1 week in the lab, sexed, and females were collected for use in the experiments. A single female Colorado potato beetle was caged in a breathable sleeve on an individual potato plant (Agrifabrics PRO17, Alpharetta, Georgia), and allowed to settle for 24 h before the predation risk or control treatments began. Cages in the predation risk treatment received two risk manipulated spined soldier bugs (one male and one female). Control treatments did not receive a predator. Sixteen replicates per treatment were established.
Leaf consumption by the Colorado potato beetle was measured using an acetate grid, to estimate tissue removed to the nearest 2 mm2, three times over the beetle’s life (3, 16, and 24 days after the treatments started). The number of eggs laid by the adult beetle was counted on 4 days (days 3, 10, 16, and 24). This gave us a comparison of the how much the beetles ate in the control and predation risk treatments as well as a nearly complete estimate of lifetime egg production (adult beetles live approximately 30 days). After measuring eggs and damage, the beetles and spined soldier bugs, if present, were transferred to new, undamaged plants. If the beetle or spined soldier bugs were found dead or missing in the predation risk treatment, no further data were collected from that replicate. The total amount of leaf damage and number of eggs laid by treatment were analyzed using ANOVA. Beetle survival in the two treatments was analyzed using a G test and the days until death for the beetles that died was analyzed using ANOVA.
Results
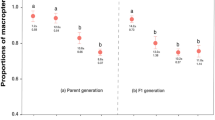
Host plant colonization in our open-field choice experiment showed that while there were 33% fewer beetles found in the risk treatment compared to control, overall colonization was not statistically different between these treatments (control: 21 individuals; predation risk: 14 individuals; p = 0.543; Fig. 1a). However, there was a 63.9% reduction in host plant damage in predation risk compared to control plots (t = − 2.23, p = 0.032; Fig. 1b).
Effect of spined soldier bug predators, Podisus maculiventris, on Colorado potato beetle, Leptinotarsa decemlineata, colonization, and host plant damage (leaf area removed) during an 18 days open-field, choice experiment. Treatments consisted of blocks of four plants in a row; one plant on the end (randomly selected to be on the left or right side) was covered with a mesh bag that was empty (control) or contained five male and five female spined soldier bug predators (predation risk). a Total number of adult Colorado potato beetles colonizing plants over 18 days (n.s. not significant) and b Mean ± SEM of potato leaf damage (*significant effects of p < 0.05)
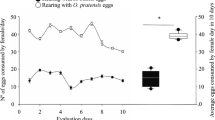
During the short-term no-choice oviposition assay, the total number of eggs laid by beetles was 36.6% lower in the presence of risk predators when compared to eggs laid in controls (t = − 2.94, p = 0.005; Fig. 2a). Similarly, there was a 26.4% reduction in the total number of clutches laid by beetles in predator present treatments compared to controls (t = − 2.40, p = 0.019; Fig. 2b).
Effect of spined soldier bug predators on Colorado potato beetle oviposition in a no-choice experiment over 4 days. Treatments were either no predators (control) or two manipulated risk predators (predation risk). Mean ± SEM of Colorado potato beetle a egg clutches or b eggs (*significant effects of p < 0.05)
Lifetime beetle oviposition followed the same pattern as the previous experiment, with a non-significant trend towards reduced egg-laying (27%) in the predation risk treatment compared to controls (F1,21 = 3.19; p = 0.089) (Fig. 3a). In addition, beetles in the predation risk treatment consumed 23% less leaf tissue compared to beetles in the control treatment (F1,23 = 18.35; p ≤ 0.001) (Fig. 3b). There was no difference in the mortality of beetles or the time until death for beetles between the treatments (mortality: G = 0.33, p = 0.57; days until death: F1,24 = 0.11, p = 0.74)
Effect of spined soldier bug predators on Colorado potato beetle oviposition and feeding in a no-choice field mesocosm experiment that lasted 24 days. Treatments were either no predators (control) or two manipulated risk predators (predation risk). Mean ± SEM of Colorado potato beetle a number of eggs laid or b leaf area removed (*significant effects of p < 0.05)
Discussion
The current research on host plant selection behavior evaluates factors such as food quality and host attractiveness in plants (Hufnagel et al. 2017), yet we are also beginning to understand how predation risk influences host choice in terrestrial insect systems (Sendoya et al. 2009). We predicted that host location in an open field would be a crucial decision for adult Colorado potato beetles, since eggs are deposited on the same plants where both adult and larval stages feed. In our experiment, natural colonization by adult beetles was not significantly affected by predation risk. However, overall plant damage was reduced in plots with the predator treatment, suggesting that influential behavioral changes occurred post-colonization. Perhaps, because the adult beetles are not very vulnerable to predation by this predator, they did not differentiate site choice based on predator presence. In other systems, colonization is affected by predation risk. For example, colonization of bird cherry-oat aphid, Rhopalosiphum padi, is reduced upon detection of cues from seven spot lady beetle (Coccinella septempunctata) predators (Ninkovic et al. 2013); however, this has only been investigated in laboratory settings. Since it is difficult to study the different stages of host selection in the lab, field studies are important in elucidating the effect of risk on these important choices. Field studies that span across all life stages of the prey should be a goal of future predator–prey and biological control studies (Hermann and Landis 2017).
In our no-choice field mesocosms, per capita feeding by adult beetles was reduced in the presence of non-lethal predators, which limits crucial resources gained by feeding that would be available for long-term egg production. Beetles raised in mesocosms with predators also marginally reduced oviposition. If adults are nutritionally compromised, a cost in fecundity can occur directly or through lack of receptivity to males (Wheeler 2009). In the short-term oviposition assay, total egg deposition was reduced by 28% in the presence of predators. Here, adults might have reduced the number of eggs due to perturbation by the non-lethal predators in the arena or due to the host plant being unsafe for larval development. Reduced oviposition has been found in response to predator cues in other systems; the butterfly, Eunica bechina, can visually detect predator presence on host plants and will avoid depositing eggs on plants that house their ant predator (Sendoya et al. 2009). Thus, even the relatively invulnerable adult life stage can be affected by predation risk and these behavioral changes can affect the next generation.
In this study, we found the effects of the predator treatment on the number of offspring produced as well as on the amount of damage done to the host plant where offspring are deposited and would subsequently feed alongside adults. Predation risk-related maternal stress effects resulting from reduced feeding or moving to alternate host sites could be costly for the offspring. For example, the offspring of snowshoe hares (Lepus americanus) that experience predation threat during gestation are much less likely to survive and these impacts lead to significant reductions in population sizes compared to unthreatened hares (MacLeod et al. 2017). In fact, the more time mothers spend in risky situations can even lead to longer term effects in the next generation (Sheriff et al. 2015). In our system, maternal effects have the potential to play a role in how larvae respond to predation. It has been recently demonstrated that Colorado potato beetle adults will increase production of unviable eggs in the presence of predators which facilitates larval nutrition via increased cannibalism. This also results in a reduction in vulnerability to predation due to increased nutrition (Tigreros et al. 2017). Further studies that examine life-long influences of predation risk across all stages of the prey will help to elucidate cascading trans-generational effects.
It has been shown that adult Colorado potato beetles use plant or other cues/signals to identify the field and encounter a food source (Jermy et al. 1988). While it has been shown that the larval Colorado potato beetles are able to perceive and behaviorally respond to volatile cues from the stink bug predator (Hermann and Thaler 2014), how adults detect predators is not yet known. In our open-field experiment, we used mesh bags to house predators physically apart from adult beetles, which provide evidence that volatile cues may be driving this interaction. Further work is necessary to determine the mechanism for predator perception in the adult life stage as well as identification of the spatial and temporal range of cues that indicate potential risk versus safety in a habitat.
Many studies now acknowledge the importance of predation risk and resulting non-consumptive effects when attempting to understand predator–prey dynamics in their entirety (Werner and Peacor 2003; Schmitz et al. 2004; Preisser et al. 2005; Hermann and Landis 2017; Buchanan et al. 2017), though only a few recognize the potential to directly manipulate predator cues in applied insect systems (Blaustein et al. 2004; Thaler and Griffin 2008; Hermann and Thaler 2014; Hermann and Landis 2017). However, prey have been successfully manipulated using predator cues in various vertebrate wildlife systems (Sullivan et al. 1985; Swihart et al. 1991; Chamaillé-Jammes et al. 2014). Studies that focus on the effects of predation risk will lead to increased understanding of predator–prey dynamics in managed agroecosystems where augmentative predator release is already used to control specific insect pest life stages. In this system, the spined soldier bug biological control agent can be used to control the larval stages (Hough-Goldstein and McPherson 1996), and we know that for the larvae, non-consumptive and consumptive effects are strong (Hermann and Thaler 2014). Our study shows that a life stage which is typically unaffected by predator consumption and thus not considered for management through biological control (Hough-Goldstein et al. 1993) is in fact affected by predation risk in a direction that could possibly benefit plant yield. For the adult beetles, the non-consumptive effects of predators are much stronger than the consumptive potential. In addition to providing insight into possible population dynamics of prey in natural systems, this information could lead to more pinpointed and sustainable control methods within managed systems to control pests.
References
Bernays EA (1997) Feeding by lepidopteran larvae is dangerous. Ecol Entomol 22:121–123. https://doi.org/10.1046/j.1365-2311.1997.00042.x
Blaustein L, Kiflawi M, Eitam A et al (2004) Oviposition habitat selection in response to risk of predation in temporary pools: mode of detection and consistency across experimental venue. Oecologia 138:300–305. https://doi.org/10.1007/s00442-003-1398-x
Boiteau G (1988) Sperm utilization and post-copulatory female-guarding in the Colorado potato beetle, Leptinotarsa decemlineata. Entomol Exp Appl 47:183–187. https://doi.org/10.1111/j.1570-7458.1988.tb01134.x
Bond J, Arredondo-Jimenez J, Rodriguez M et al (2005) Oviposition habitat selection for a predator refuge and food source in a mosquito. Ecol Entomol 30:255–263
Buchanan AL, Hermann SL, Lund M, Szendrei Z (2017) A meta-analysis of non-consumptive predator effects in arthropods: the influence of organismal and environmental characteristics. Oikos 126:1233–1240. https://doi.org/10.1111/oik.04384
Chamaillé-Jammes S, Malcuit H, Le Saout S, Martin J-L (2014) Innate threat-sensitive foraging: black-tailed deer remain more fearful of wolf than of the less dangerous black bear even after 100 years of wolf absence. Oecologia 174:1151–1158. https://doi.org/10.1007/s00442-013-2843-0
Cherry MJ, Conner LM, Warren RJ (2015) Effects of predation risk and group dynamics on white-tailed deer foraging behavior in a longleaf pine savanna. Behav Ecol 26:1091–1099. https://doi.org/10.1093/beheco/arv054
Fill A, Long EY, Finke DL (2012) Non-consumptive effects of a natural enemy on a non-prey herbivore population. Ecol Entomol 37:43–50. https://doi.org/10.1111/j.1365-2311.2011.01333.x
Freinschlag J, Schausberger P (2016) Predation risk-mediated maternal effects in the two-spotted spider mite, Tetranychus urticae. Exp Appl Acarol 69:35–47. https://doi.org/10.1007/s10493-016-0014-9
Griffin CAM, Thaler JS (2006) Insect predators affect plant resistance via density- and trait-mediated indirect interactions. Ecol Lett 9:335–343. https://doi.org/10.1111/j.1461-0248.2005.00880.x
Hare JD (1990) Ecology and management of the Colorado potato beetle. Annu Rev Entomol 35:81–100
Hermann SL, Landis DA (2017) Scaling up our understanding of non-consumptive effects in insect systems. Curr Opin Insect Sci 20:54–60
Hermann SL, Thaler JS (2014) Prey perception of predation risk: volatile chemical cues mediate non-consumptive effects of a predator on a herbivorous insect. Oecologia 176:669–676. https://doi.org/10.1007/s00442-014-3069-5
Hough-Goldstein J, McPherson D (1996) Comparison of Perillus bioculatus and Podisus maculiventris (Hemiptera: Pentatomidae) as potential control agents of the Colorado potato beetle (Coleoptera: Chrysomelidae). J Econ Entomol 89:1116–1123
Hough-Goldstein JA, Heimpel GE, Bechmann HE, Mason CE (1993) Arthropod natural enemies of the Colorado potato beetle. Crop Prot 12:324–334. https://doi.org/10.1016/0261-2194(93)90074-S
Hufnagel M, Schilmiller AL, Ali J, Szendrei Z (2017) Choosy mothers pick challenging plants: maternal preference and larval performance of a specialist herbivore are not linked. Ecol Entomol 42:33–41. https://doi.org/10.1111/een.12350
Jaenike J (1978) Optimal ovipostion behavior in phytophagous insects. Theor Popul Biol 14:350–356. https://doi.org/10.1016/0040-5809(78)90012-6
Jermy T, Szentesi Á, Horváth J (1988) Host plant finding in phytophagous insects: the case of the Colorado potato beetle. Entomol Exp Appl 49:83–98. https://doi.org/10.1007/BF00188242
Kaplan I, Thaler J (2010) Plant resistance attenuates the consumptive and non-consumptive impacts of predators on prey. Oikos 119:1105–1113. https://doi.org/10.1111/j.1600-0706.2009.18311.x
Kaplan I, McArt SH, Thaler JS (2014) Plant defenses and predation risk differentially shape patterns of consumption, growth, and digestive efficiency in a guild of leaf-chewing insects. PLoS One. https://doi.org/10.1371/journal.pone.0093714
Lee DH, Nyrop JP, Sanderson JP (2011) Avoidance of natural enemies by adult whiteflies, Bemisia argentifolii, and effects on host plant choice. Biol Control 58:302–309. https://doi.org/10.1016/j.biocontrol.2011.06.005
Lefèvre T, Oliver L, Hunter MD, De Roode JC (2010) Evidence for trans-generational medication in nature. Ecol Lett 13:1485–1493. https://doi.org/10.1111/j.1461-0248.2010.01537.x
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640. https://doi.org/10.1139/z90-092
MacLeod KJ, Krebs CJ, Boonstra R, Sheriff MJ (2017) Fear and lethality in snowshoe hares: the deadly effects of non-consumptive predation risk. Oikos 127:375–380. https://doi.org/10.1111/oik.04890
Munga S, Minakawa N, Zhou G et al (2006) Effects of larval competitors and predators on oviposition site selection of Anopheles gambiae sensu stricto. J Med Entomol 43:221–224
Ninkovic V, Feng Y, Olsson U, Pettersson J (2013) Ladybird footprints induce aphid avoidance behavior. Biol Control 65:63–71. https://doi.org/10.1016/j.biocontrol.2012.07.003
Peckarsky B, Taylor B, Caudill C (2000) Hydrologic and behavioral constraints on oviposition of stream insects: implications for adult dispersal. Oecologia 125:186–200. https://doi.org/10.1007/s004420000446
Preisser E, Bolnick D, Benard M (2005) Scared to death? The effects of intimidation and consumption in predator–prey interactions. Ecology 86:501–509
Ramirez RA, Crowder DW, Snyder GB et al (2010) Antipredator behavior of Colorado potato beetle larvae differs by instar and attacking predator. Biol Control 53:230–237. https://doi.org/10.1016/j.biocontrol.2010.01.004
Rendon D, Whitehouse MEA, Taylor PW (2016) Consumptive and non-consumptive effects of wolf spiders on cotton bollworms. Entomol Exp Appl 158:170–183. https://doi.org/10.1111/eea.12390
Rypstra AL, Buddle CM (2013) Spider silk reduces insect herbivory. Biol Lett 9:20120948. https://doi.org/10.1098/rsbl.2012.0948
Schmitz OJ, Beckerman AP, O’Brien KM (1997) Behaviorally mediated trophic cascades: effects of predation risk on food web interactions. Ecology 78:1388–1399
Schmitz OJ, Krivan V, Ovadia O (2004) Trophic cascades: the primacy of trait-mediated indirect interactions. Ecol Lett 7:153–163. https://doi.org/10.1111/j.1461-0248.2003.00560.x
Sendoya SF, Freitas AVL, Oliveira PS (2009) Egg-laying butterflies distinguish predaceous ants by sight. Am Nat 174:134–140. https://doi.org/10.1086/599302
Sheriff MJ (2015) The adaptive potential of maternal stress exposure in regulating population dynamics. J Anim Ecol 84:323–325. https://doi.org/10.1111/1365-2656.12334
Sheriff MJ, Thaler JS (2014) Ecophysiological effects of predation risk; an integration across disciplines. Oecologia 176:607–611. https://doi.org/10.1007/s00442-014-3105-5
Sheriff MJ, Krebs CJ, Boonstra R (2009) The sensitive hare: sublethal effects of predator stress on reproduction in snowshoe hares. J Anim Ecol 78:1249–1258. https://doi.org/10.1111/j.1365-2656.2009.01552.x
Sheriff MJ, Mcmahon EK, Krebs CJ, Boonstra R (2015) Predator-induced maternal stress and population demography in snowshoe hares: the more severe the risk, the longer the generational effect. J Zool 296:305–310. https://doi.org/10.1111/jzo.12249
Sitvarin MI, Breen K, Rypstra AL (2015) Predator cues have contrasting effects on lifespan of Pardosa milvina (Araneae: Lycosidae). J Arachnol 43:107–110. https://doi.org/10.1636/J14-48.1
Stav G, Blaustein L, Margalith J (1999) Experimental evidence for predation risk sensitive oviposition by a mosquito, Culiseta longiareolata. Ecol Entomol 24:202–207. https://doi.org/10.1046/j.1365-2311.1999.00183.x
Sullivan T, Nordstrom L, Sullivan D (1985) Use of predator odors as repellents to reduce feeding damage by herbivores: IV. Northern pocket gophers (Thomomys talpoides). J Chem Ecol 14:379–389. https://doi.org/10.1007/BF01012078
Swihart R, Pignatello J, Mattina M (1991) Aversive responses of white-tailed deer, Odocoileus virginianus, to predator urines. J Chem Ecol 17:767–777
Tallamy DW (1984) Insect parental care. Bioscience 34:20–24. https://doi.org/10.2307/1309421
Tan K, Hu Z, Chen W et al (2013) Fearful foragers: honey bees tune colony and individual foraging to multi-predator presence and food quality. PLoS One. https://doi.org/10.1371/journal.pone.0075841
Thaler JS, Griffin CAM (2008) Relative importance of consumptive and non-consumptive effects of predators on prey and plant damage: the influence of herbivore ontogeny. Entomol Exp Appl 128:34–40. https://doi.org/10.1111/j.1570-7458.2008.00737.x
Thaler JS, McArt SH, Kaplan I (2012) Compensatory mechanisms for ameliorating the fundamental trade-off between predator avoidance and foraging. Proc Natl Acad Sci 109:12075–12080. https://doi.org/10.1073/pnas.1208070109
Thompson JN (1988) Evolutionary ecology of the relationship between oviposition preference and performance of offspring in phytophagous insects. Entomol Exp Appl 47:3–14. https://doi.org/10.1111/j.1570-7458.1988.tb02275.x
Tigreros N, Norris RH, Wang EH, Thaler JS (2017) Maternally induced intraclutch cannibalism: an adaptive response to predation risk? Ecol Lett 20:487–494. https://doi.org/10.1111/ele.12752
Vonesh JR, Blaustein L (2011) Predator-induced shifts in mosquito oviposition site selection: a meta-analysis and implications for vector control. Isr J Ecol Evol 56:263–279. https://doi.org/10.1560/IJEE.56.3-4.263
Werner EE, Peacor SD (2003) A review of trait-mediated indirect interactions in ecological communities. Ecology 84:1083–1100. https://doi.org/10.1890/0012-9658(2003)084[1083:Arotii]2.0.Co;2
Wheeler D (2009) Suitability of different artificial diets for development and survival of stages of the predaceous ladybird. Annu Rev Entomol 41:10–12. https://doi.org/10.1146/annurev.en.41.010196.002203
Wineland SM, Kistner EJ, Joern A (2015) Non-consumptive interactions between grasshoppers (Orthoptera: Acrididae) and wolf spiders (Lycosidae) produce trophic cascades in an old-field ecosystem. J Orthoptera Res 24:41–46. https://doi.org/10.1665/034.024.0101
Xiong X, Michaud JP, Li Z et al (2015) Chronic, predator-induced stress alters development and reproductive performance of the cotton bollworm, Helicoverpa armigera. BioControl 60:827–837. https://doi.org/10.1007/s10526-015-9689-9
Acknowledgements
We thank Marie Russel, Elizabeth Davidson-Lowe, Alyssa Cowles, Scott Nelson, Stephen Pecylak, Evan Hoki, and Dylan Beal for assistance in running experiments. Special thanks to Charlie Linn, Katja Poveda, Jared G. Ali, Douglas A. Landis, and two anonymous reviewers for thoughtful comments on the working manuscript. This project was supported by NIFA 2013-02649 and NYC-139851.
Author information
Authors and Affiliations
Contributions
SH and JT formulated the study, performed the experiments, designed the experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Sven Bacher.
This work explores how predation risk alters herbivore life history. These lab and field studies provide evidence that risk influences oviposition and feeding of invulnerable life-stages of insects.
Rights and permissions
About this article
Cite this article
Hermann, S.L., Thaler, J.S. The effect of predator presence on the behavioral sequence from host selection to reproduction in an invulnerable stage of insect prey. Oecologia 188, 945–952 (2018). https://doi.org/10.1007/s00442-018-4202-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-018-4202-7