Abstract
The perceived quality of habitat patches in complex landscapes is highly context dependent. Characteristics of neighboring patches in such complex landscapes can influence perceived habitat quality, altering colonization dynamics and community structure. Spatial contagion of predation risk across patches has been observed over smaller spatial scales in aquatic systems. Naturally colonizing aquatic beetles were used to examine the spatial dynamics of risk contagion by quantifying the size of predator shadows around fish patches across spatial scales potentially involving numerous patches in natural landscapes. These consisted of fish free, replicate experimental mesocosm arrays radiating from larger central mesocosms containing fish, and allowed examination of the effect of distance to fish on beetle abundance, rarified species richness, and variation in species responses. Overall, beetles avoided pools closer to fish, but species varied in colonization pattern, resulting in species-specific predator shadows and potential behavioral species sorting. The spatial and phylogenetic extent of contagion and other context-dependent effects has implications for the role of complex behavior in the dynamics of communities and metacommunities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Assembly of natural communities is driven jointly by processes of dispersal and colonization and by post-colonization species sorting. The composition of communities is thus determined by the classic dynamics of immigration and extinction. However, our understanding of these processes, particularly immigration, has undergone dramatic expansion since the Equilibrium Theory of Island Biogeography (MacArthur and Wilson 1967). While we have learned a great deal about how post-colonization processes impact the assembly of communities across space and time, we have also learned that habitat selection behavior can have effects on community assembly of equal magnitude to post-colonization sorting (Binckley and Resetarits 2005; Vonesh et al. 2009; Kraus and Vonesh 2010; Resetarits and Pintar 2016). Thus, a reevaluation of the paradigm that passive (or random) processes of dispersal and immigration, coupled with post-colonization species sorting, function as the primary drivers of variation in local species richness and community structure is underway (Morris 2003; Armsworth and Roughgarden 2005; Abrams et al. 2007; Golubski and Abrams 2011).
Habitat selection is driven by a colonizing organism’s perception of the relative quality (expected fitness) of available habitat patches (Fretwell and Lucas 1970). Recent work in freshwater systems has demonstrated that colonization dynamics of individual patches is highly context dependent. The perceived quality of given patch is dependent not only on inherent characteristics of the individual patch, but also on characteristics of the surrounding habitat matrix (Deans and Chalcraft 2016), and the quality, frequency, and spatial distribution of nearby habitable patches (Resetarits and Binckley 2009; Wesner et al. 2012; Hughey et al. 2012; Resetarits and Silberbush 2016).
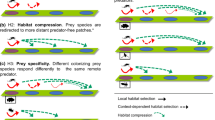
Spatial contagion, where effects of patch characteristics bleed over and affect perceived habitat quality in adjacent patches (Resetarits et al. 2005), has been demonstrated for both avoidance of risk and attraction to reward. Ovipositing treefrogs, mosquitoes, and colonizing aquatic beetles reduce oviposition/colonization in predator-free patches adjacent to predator patches (Resetarits 2005; Resetarits and Binckley 2009, Resetarits and Binckley 2013a; Wesner et al. 2012; Resetarits and Silberbush 2016), while predators of red-eyed treefrog eggs (frogflies) colonize undamaged egg clutches near their preferred habitat of damaged egg clutches at increased rates (Hughey et al. 2012). Attraction to low-quality patches spatially associated with high-quality patches can generate attractive sinks (Delibes et al. 2001) while avoiding high-quality habitats associated with high-risk habitats can generate repulsive sources (Resetarits and Binckley 2009). Contagion can also result in habitat compression at multiple spatial scales, wherein colonists repulsed by high risk or perceived high-risk patches are concentrated in remaining high-quality patches, thus increasing intraspecific densities and the changing the identity and intensity of interspecific interactions (Resetarits et al. 2005; Resetarits and Silberbush 2016).
An important, unanswered, question concerns the spatial scale and magnitude of such effects, e.g., distances across which patches of different types affect one another and degree to which colonization is affected by spatial context. Is spatial contagion a very localized phenomenon, or does it function over distances that amplify its effects at different spatial scales at the level of the metacommunity? The degree to which the perception of patch quality is altered by the quality of nearby patches has implications for how an organism “sees” the habitat landscape and the availability of suitable patches. It may prove maladaptive if an organism’s perception of patch quality and the actual expected fitness of a patch are out of sync. If multiple species share preferred habitats, but vary in their abilities to assess the quality of those habitats, or in their behavioral response to that perceived quality, the potential for species sorting at the colonization stage is expanded.
Aquatic beetles (Coleoptera) in the families Dytiscidae and Hydrophilidae have proven excellent model organisms for studying the role of habitat selection in community assembly within a landscape context (Vonesh and Kraus 2009; Resetarits and Binckley 2009; Vonesh et al. 2009; Resetarits and Pintar 2016). Larvae and adults of both families are obligately aquatic, and females must choose habitats both for themselves and their offspring. Many aquatic beetles, especially the larvae, are highly fish intolerant, so much so that the presence of predatory fish often results in zero survival. As a consequence, reproduction is typically restricted to fishless waters, most of which are temporary habitats, and females must balance the risks posed by desiccation and predation. Fish signal long-standing water, but also potential reproductive failure. Thus, beetles may be attracted to the vicinity of permanent water containing fish as an indicator of pond duration while avoiding specific patches (ponds) containing fish. The initial decision of where to colonize and reproduce is critical, whether or not individuals secondarily disperse, because dispersal flights are energetically costly, and limited evidence suggests that once the initial settlement phase is complete, beetles typically undergo secondary dispersal only when conditions change dramatically (e.g., pond drying, temperature changes) (Zalom et al. 1979; Sheldon 1984; Layton and Voshell 1991; Lundkvist et al. 2001; Bilton 2014). Some species (especially the females) histolysize flight muscles to fuel reproduction and/or development of swimming muscles (oogenesis–flight syndrome) (Johnson 1969; Zera and Denno 1997).
Prior work with insects has documented spatial contagion of predation risk for adjacent patches over small, but ecologically relevant, distances of up to 1 m (Binckley and Resetarits 2005; Resetarits and Binckley 2009; Wesner et al. 2012; Resetarits and Silberbush 2016). The presence of high-risk patches adjacent to high-quality, low-risk patches has dramatic effects on the colonization rate of individual species and resulting community assembly. However, the spatial scale over which risk contagion may occur is unknown, and the extent of the zone of perceived risk (predator shadow) has important implications for the assembly of communities and metacommunities across natural landscapes. Use of the term “predator shadow” herein is a natural extension of the original concept dealing with the trophic effects of predators extending beyond their natal habitat as a consequence of complex life cycles and ontogenetic niche shifts (McCoy et al. 2009). Having established that the majority of colonizing beetle species avoid fish as well as fishless patches adjacent to those containing fish, the design specifically focused on the extent of risk contagion and the size of the resulting predator shadow over spatial scales that may involve numerous habitat patches (Binckley and Resetarits 2005; Resetarits and Binckley 2009; Kraus and Vonesh 2010). Fish free, replicate experimental mesocosm arrays were created around larger central mesocosms containing fish, thus allowing examination of the effect of distance to fish ponds on beetle abundance, species richness, and variation in responses among beetle species. The working hypothesis is that beetle abundance, in general, would show a positive correlation with distance from the central fish patch and that individual species would show variation in their spatial sensitivity to fish.
Materials and methods
Our experiment was conducted in a large, old-field at the University of Mississippi Field Station in Lafayette County, Mississippi, USA. UMFS is a 787 acre complex situated in the Eocene hills of the interior Gulf Coastal Plain that contains over 200 natural and experimental ponds along with multiple small streams, wetlands, fields, and mixed forests. The four arrays were situated in a field 20 m north and west of the nearest ponds, which contained fish. Each array (block) consisted of a “central” 2.7 m diameter (3100 L) cattle tank containing fish, with two arms consisting of four 1.2 m diameter (420 L) cattle tanks of the same material as the central tank, pointing at two adjacent primary intercardinal directions (SW, SE, NW, and NE). These pools were placed 1 m apart, edge to edge, so that distances from the edge of the central pool to the edge of each pool in the arm were 1 m (Distance 1), 3.2 m (2), 5.4 m (3), and 7.6 m (4). Using a partial spoke design equalized the possible effects of colonist sources, edge, and wind direction (Fig. 1). We do not have detailed wind data for UMFS, but US National Weather Service data indicate no prevailing wind direction for north Mississippi in any month of the year, so we simply equalized the potential contribution of each primary intercardinal direction (Fig. 1). Small pools were covered with screen lids (1.13 × 1.3 mm mesh), and all pools were filled with well-water on 7–9 July; on 10 July, the central pool received 3 kg of leaf litter, and each of the smaller pools received 0.5 kg. On 11 July, each central pool received two adult Lepomis macrochirus (bluegill sunfish). Lepomis macrochirus is one of the most abundant species at UMFS and is one of the most widespread and widely introduced fish species in North America. Fish were ranked by size (range 49.29–68.32 g) and paired to minimize variation in the final total mass, which then ranged from 117.66 to 120.22 g total mass/pool (~ 4 g/100 L). This was well within the range of natural densities of L. macrochirus at UMFS. Resources for colonizing beetles consisted of abundant zooplankton, phytoplankton, periphyton, and the larval stages of chironomids, mosquitoes, and other small organisms, whose propagules could pass through the screen mesh. Any larger organisms were removed during the weekly beetle sampling. The only larger organism colonizing in significant numbers was Notonecta irrorata (N = 111), but there was no relationship between the number of N. irrorata and the number of beetles, and subsequent work has shown that colonizing beetles do not avoid N. irrorata, perhaps because their presence is temporally unpredictable (unpubl. data). On 13 July, the screen lids were pushed down into the smaller pools to allow colonization and subsequent collection of beetles, while the large pools remained uncovered, simply providing a source of fish chemical cues. The uncovered central fish pools precluded comparable monitoring of colonization, but provided unimpeded diffusion of fish cues, and by allowing unimpeded colonization also allowed cues emanating from prey consumption. Doubtless some beetles colonized the central pools, but the previous work has established the strong avoidance of fish pools by both dytiscid and hydrophilid beetles at fish densities as low as 0.82 g/100 L (Binckley and Resetarits 2005, 2009), and densities of 2–8 g/L do not differ, indicating a threshold cue density (Resetarits and Pintar 2016). Beetles were removed weekly for 14 weeks (20 July–19 October 2015), preserved, and identified to species, with the exception of Paracymus, which was the only beetle taxa in which not all individuals were identified to species (95% of those identified from the UMFS are P. subcupreus, and the rest identified are P. confusus). One arm of four pools (Fig. 2) was compromised and was removed from the analysis, leaving seven replicates of each Distance from fish.
Mean ± 1 SE for the abundance of a 31 species of beetles (excluding the three most abundant species) at varying distances from central pool, b rarified beetle species richness, and c–e abundance for the three most numerous beetle species (species in y-axis title). Bars above graphs represent the three a priori contrasts with significant results indicated by black bars. Breaks separate treatment groups in each contrast
Hypothesis testing consisted of a priori contrasts parsing the variation in beetle colonization by distance from fish. Variables analysed were mean rarefied species richness, mean abundance of the three numerically dominant species, and mean combined beetle abundance of the 31 remaining species. Species richness was rarified using EstimateS (Version 9) (Colwell 2013). We tested for an interaction between Block and Distance for each variable with Tukey’s test for non-additivity (SAS code from Robert et al. 1997), and found no significant interaction for any variable. Data were analysed using a general linear mixed model ANOVA, with block as a random factor, on square root transformed (√x + 0.5) count data and square root transformed rarefied species richness, using PROC MIXED in SAS for Windows v.9.4 with α = 0.05. All transformed data met the assumptions of ANOVA. Block effects for Copelatus glyphicus and Enochrus ochraceus were estimated to be zero and were dropped from the model.
Results
Our experiment was colonized by 1977 individuals of 34 species of beetles: 716 individuals of 12 species of Dytiscidae, 1257 individuals of 19 species of Hydrophilidae, and one species each of Haliplidae (N = 2), Helophoridae (1), and Hydraenidae (1) (Table 1). This total represents a substantial proportion of the 115 species of aquatic beetles reported from the UM Field Station (unpublished data). Block effects were meaningful only for the combined abundance of the 31 less abundant species (Table 2a, Fig. 2a), and T. lateralis (Table 2c, Fig. 2c), simply as a result of more beetles colonizing blocks in the Southern half of the field.
Actual species richness among pools ranged from 8 to 15 and rarefied species richness from 6.86 to 15.16. Mean rarefied richness varied little with Distance, with none of the contrasts significant (Table 2b, Fig. 2c). Distance (Fig. 1) had a significant effect on the mean number of beetles (31 sp.), as revealed by the a priori contrasts (Table 2a, Fig. 2a). Distance 1, nearest to the central pool (40.71 ± 4.09, mean ± SE), was colonized by marginally significantly fewer beetles than Distances 2–4 (60.81 ± 10.33), Distances 1–2 received significantly fewer beetles (47.43 ± 7.13) and than Distances 3–4 (62.14 ± 10.42). Distance 4 (66.86 ± 8.98) was significantly different from Distances 1–3 (52.10 ± 8.70) (Figs. 1, 3b). Thus, a distinct breakpoint for contagion for the 31 combined beetle species occurred between Distances 1–2 and Distances 3–4.
Minimum predator shadow for a combined set of 31 species of beetles (excluding the three most abundant species) (N = 575), b Tropisternus lateralis (N = 559), c Copelatus glyphicus (N = 429), and d Enochrus ochraceus (N = 414) based on Fig. 2. Enochrus ochraceus has demonstrated strong fish avoidance and contagion effects in Virginia (Resetarits and Binckley 2009), but not in Mississippi, hence the range of effects shown. Black (red) = fish patches, gray (blue) = fishless patches, light gray (light red) shaded area = predator shadow. Actual size of fish patches is 5.73 m2 (color version available online)
The pattern of colonization varied considerably among the three numerically dominant beetle species, Tropisternus lateralis (N = 559), Copelatus glyphicus (429), and Enochrus ochraceus (414). Tropisternus lateralis showed a clear graded pattern of response, with colonization increasing linearly with Distance. Distance 1 was colonized by significantly fewer T. lateralis than Distances 2–4, Distances 1–2 received significantly fewer T. lateralis than Distances 3–4, and Distance 4 had significantly more beetles than the mean of Distances 1–3 (Table 1c, Fig. 2c), suggesting that contagion effects extend at least to Distance 3. For C. glyphicus, the pattern was quite different, with only Distance 1 showing significantly reduced abundance relative to Distances 2–4; neither of the other contrasts were close to significant (Table 2d, Fig. 2d). Thus, the critical breakpoint for C. glyphicus fell between Distances 1 and 2–4. Enochrus ochraceus showed a weak and non-significant pattern of increasing abundance in patches closer to fish; none of the three contrasts was close to significant (Table 2e, Fig. 2e).
Discussion
Colonizing organisms must rely on environmental cues as indicators of patch quality and expected fitness (Fretwell and Lucas 1970; Wiens 1976; Orians and Wittenberger 1991; Pulliam and Danielson 1991; Blaustein 1999; Schlaepfer et al. 2002). High-quality patches are a limited resource in most natural landscapes and organisms that fail to correctly assess risk and reward face the prospect of reduced fitness or even reproductive failure through adult/offspring mortality and/or reduced performance. A critical component of patch quality in aquatic habitats is predation risk (Wellborn et al. 1996), and colonization behavior is dependent upon how that risk is perceived across a landscape of patches. To examine how colonizing aquatic beetles respond to spatial variation in perceived predation risk, fish free, replicate experimental mesocosm arrays radiating from larger central mesocosms containing fish were created, allowing determination of the effect of distance to fish ponds on beetle abundance, species richness, and variation in responses among beetle species.
The presence of fish affected overall beetle abundance in patches within an area of at least 17 times the area actually containing fish, and the observed predator shadows for different species varied in extent from zero to up to at least 30 times the size of the fish patch itself. Thus, beetle species behaviorally sorted based not simply on the presence/absence of fish within a patch, but also on the spatial extent of the predator shadows cast. Individual beetle species differ in their specific response (or lack of response) to fish cues, sensitivity to fish cues, or the specific behavioral algorithm with regard to fish avoidance (Resetarits and Pintar 2016). For the 31 less abundant beetle species, the effect of spatial contagion of risk on abundance extends out to at least 4.4 m from a 2.7 m diameter patch with relatively low fish densities (Fig. 3a). The variation in specific responses is reflected in variation among our most abundant beetles; Tropisternus lateralis showed a graded response to distance from fish, affecting an area at least 30 times the size of the fish patch itself (Fig. 3b), while C. glyphicus had a threshold response which manifested itself between 2.2 and 3.2 m from the fish pool, affecting an area up to at least 6 times the area of the fish patch (Fig. 3c). Enochrus ochraceus did not respond to fish in the previous experiments in Mississippi (Resetarits and Pintar 2016; unpubl. data), where E. ochraceus appeared in low numbers, but showed strong avoidance and contagion in prior experiments in Virginia (Resetarits and Binckley 2009), where it was very abundant, hence the range shown in Fig. 3d. From a species sorting perspective, different responses to the presence of predators, presence or absence of spatial risk contagion, and different behavioral algorithms that define the extent of predator shadows, all serve as mechanisms producing species turnover across complex landscapes.
Independence of events in naturally colonized experimental landscapes (NCELs)
Prior work in our lab on context-dependent colonization has established that colonizing organisms can compare and choose among habitat patches at large spatial scales. However, our work also shows that contagion and other spatial processes can be precluded by eliminating choice at a given spatial scale. Thus, if we wish to compare among patches within a landscape, as in this experiment, each array (spatial block) must contain the same set of treatments. This eliminates any inherent basis of choice among arrays, leaving only largely spatial block effects, thus forcing organisms to choose among the available options within identical arrays. Specifically for this experiment [and many other such experiments (e.g., Resetarits and Silberbush 2016)], control arrays with no fish pools are infeasible, because they would very likely draw a heavily disproportionate number of colonists, while not contributing to the actual question being posed, because all patches are equally good, and the large central fishless patch may provide the best available habitat on the landscape. As a result, we cannot definitively exclude the alternative explanation that patches on the periphery of the arrays are somehow preferred, and the patterns have nothing to do with fish. However, based on prior evidence of contagion effects in beetles (Resetarits and Binckley 2009), as well as the lack of any observed, systematic positional bias in any previous experiment with beetles, that possibility is extremely unlikely, especially given that cues simply relating to water as an attractant would be strongest nearer the apex of the arrays.
The issue of independence of patches within individual arrays in naturally colonized experimental landscapes is an interesting one. Inherent in many NCEL experiments is the idea that responses are context dependent and organisms are comparing at some level, and then making choices among the various options presented. Statistical independence of patches thus has two components: (1) the independence of each spatial choice from choices made by previously colonizing individuals and (2) independence of each spatial choice with respect to all available patches. One can think of our response variables, number of insects/patch, etc., as the total of repeated behavioral trials; the choice made by each individual insect is an event and we total the number of times an insect chooses a particular patch, without replacement. The first question is whether each individual event is affected by the previous event(s). If there is negative density-dependence [sensu the Ideal Free Distribution (Fretwell and Lucas 1970)] and initial colonists prefer patches further from fish, the contagion effect would be diluted by the responses of subsequent colonists to density. Conversely, if there is conspecific attraction with the initial colonists preferring more distant patches, effects ascribed to contagion would be enhanced. In either case, later events would not be independent of prior events. Thus, we remove colonists at intervals that should obviate the potential issue, in this experiment weekly. For each individual choice, the array is not a tabula rasa, but is as close as we can reasonably make it. Kraus and Vonesh showed that community assembly dampened fish avoidance over time in dytiscids, but not hydrophilids (Kraus and Vonesh 2010), while our own specific test with colonizing beetles (at UMFS) suggests that colonization history plays little role in behavioral choices among treatments (unpublished data). Thus, though the jury is still out on how sensitive colonization behavior is to colonization history, our weekly removals largely preclude, or at least dampen, any responses to community assembly.
With regard to the second question, spatial independence, colonizing organisms are given a choice among a set of pools that vary in one or more defining characteristics. The defining characteristics of a given patch include both inherent characteristics of that patch and the spatial context of that patch. The fact that part of the context is the other patches does not affect the statistical independence of choices among patches, because each “event” (choice) is not affected by any other event. In this specific experiment, the patches are identical except in their distance from a central, non-experimental patch, so the choices relate to the relationship of a specific experimental unit to the central patch; individuals may choose among a set of associated patches, but each of those choices is independent. A second point worth noting is that these are “open” communities; the total number of individuals in a given patch does not limit the total in any other patch, because the grand total is not fixed. Thus, while the decision of an individual colonist may integrate information on multiple patches, there are a potentially infinite number of colonists that are independently doing the same thing. We can also reference blocked designs, in which, by definition, units within a block should be more similar than those in different blocks. Thus, they are truly independent of units in other blocks, but not their own, which is true in NCEL behavioral choice experiments that utilize spatially blocked designs, as we have typically employed.
Mechanisms and consequences of spatial contagion
Previous work has established chemical cues as the primary driver of fish avoidance in ovipositing treefrogs, aquatic beetles, and ovipositing Culex mosquitoes (Binckley and Resetarits 2002; Resetarits and Binckley 2013b; Eveland et al. 2016); however, the identity of the cues used to identify fish remains elusive. One of the interesting questions regarding spatial contagion is whether chemical cues from non-preferred habitats simply drift over preferred habitats (or vice versa), causing avoidance or attraction by misperception of habitat quality. Organisms misperceive the inherent risk in a given pond, avoiding potential source habitats, or colonizing potential sink habitats. Ovipositing mosquitoes appear to use the spatial distribution of volatile cues from Notonecta to select habitat patches. Thus, Notonecta-free patches surrounded by patches containing Notonecta chemical cues receive fewer egg rafts, but the difference from controls is less than when patches themselves contain cues (Silberbush and Blaustein 2008). Frogflies ovipositing on red-eyed treefrog clutches actually have higher fitness on non-preferred (undamaged) clutches than on preferred (damaged) clutches (Hughey et al. 2012), suggesting that contagion actually aids in detection of suitable patches, perhaps via simple drift or diffusion of volatile cues. For frogflies, preferred patches may be those easiest to find via chemical cues, rather than those affording higher fitness. In our system, if cue drift is the mechanism, then larger bodies of water should generate larger predator shadows.
The more complex alternative is that organisms possess a behavioral algorithm that spatially integrates observations of risk and reward. Organisms are not reacting directly to a gradient of cues, but to the identification, characterization, and frequency of “good” vs “bad” habitat patches on multiple spatial scales. Further detailed experiments are clearly needed on behavioral mechanisms underlying spatial contagion, but our work on treefrogs, mosquitoes, and beetles suggests that spatial contagion is driven by this type of complex behavioral algorithm, rather than simply the spatial distribution of airborne cues resulting in recognition errors. Selection likely has driven a conservative approach to assessing risk, given that predators have a greater probability of invading an empty patch close to occupied patches. For example, for fish in disconnected patches, potential linkages caused by flooding are greater at shorter inter-patch distances (Trexler et al. 2001; Petranka and Holbrook 2006). Thus, gray treefrogs that avoid patches of high potential fitness (fishless) adjacent to low fitness patches (fish) may or may not incur a real cost in avoiding these potential source patches, depending on the probability and cost incurred if fish subsequently invade (Resetarits 2005). The same may be true of ovipositing mosquitoes (Resetarits and Silberbush 2016) and colonizing aquatic beetles (Resetarits and Binckley 2009). Our beetle species clearly show variation in the level of “conservatism” in response to perceived predation risk, with T. lateralis being very conservative (or sensitive to cues), while C. glyphicus is either less conservative or better able to fine tune responses to small-scale variation (Fig. 3). If colonizers use a simple “distance to fish” rule, then the predator shadow should not scale with pond area, as with cue drift, but only pond perimeter.
How does this relate to possible effects on communities and metacommunities across complex natural landscapes? In the context of complex landscapes, many low-lying areas contain high densities of closely interspersed fish and fishless ponds (Trexler et al. 2001; Pearl et al. 2005; Petranka and Holbrook 2006), suggesting potentially dramatic effects of spatial contagion on colonization dynamics. This is especially true if contagion is driven by a behavioral algorithm based on patch boundaries and distance to fish. We used fish in this experiment because they play such an important role in freshwater systems (Wellborn et al. 1996), but other predators, as well as competitors, could be expected to generate similar responses. In addition, while spatial contagion could be expected to function over relatively limited spatial scales, it can generate habitat compression, wherein colonists repulsed by high risk or perceived high-risk patches are concentrated in nearby, high-quality patches. Thus, the initial effects cascade across the landscape, further linking multiple patches into metacommunities (Resetarits et al. 2005; Abrams et al. 2007; Abrams 2007; Orrock et al. 2010; Golubski and Abrams 2011; Resetarits and Silberbush 2016). For compression, the spatial scale affected is defined by nearest neighbor distances and the dispersal abilities of the colonists, which for beetles may be considerable. Such context-dependent colonization processes can generate behavioral species sorting, as illustrated by the variation among beetles in their responsiveness to fish and the size of the predator shadow generated. While these specific context-dependent mechanisms have thus far been identified in aquatic systems, such behavioral processes are applicable to any patchy system, and varied sources of risk. Spatial contagion, as well as other types of context dependence (Resetarits et al. 2005; Hughey et al. 2012; Deans and Chalcraft 2016; Resetarits and Silberbush 2016), have important implications for how communities are assembled across complex landscapes, and existence of behavioral algorithms based on spatial awareness of patch context speaks to the role of complex behavior in the dynamics of communities and metacommunities.
References
Abrams PA (2007) Habitat choice in predator-prey systems: spatial instability due to interacting adaptive movements. Am Nat 169:581–594. https://doi.org/10.1086/512688
Abrams PA, Cressman R, Krivan V (2007) The role of behavioral dynamics in determining the patch distributions of interacting species. Am Nat 169:505–518. https://doi.org/10.1086/511963
Armsworth PR, Roughgarden JE (2005) The impact of directed versus random movement on population dynamics and biodiversity patterns. Am Nat 165:449–465. https://doi.org/10.1086/428595
Bilton DT (2014) Dispersal in Dytiscidae. In: Yee DA (ed) Ecology, systematics, and the natural history of predaceous diving beetles (Coleoptera: Dytiscidae). Springer, The Netherlands, pp 387–407
Binckley CA, Resetarits WJ Jr (2002) Reproductive decisions under threat of predation: squirrel treefrog (Hyla squirella) responses to banded sunfish (Enneacanthus obesus). Oecologia. https://doi.org/10.1007/s004420100781
Binckley CA, Resetarits WJ Jr (2005) Habitat selection determines abundance, richness and species composition of beetles in aquatic communities. Biol Lett 1:370–374. https://doi.org/10.1098/rsbl.2005.0310
Binckley CA, Resetarits WJ Jr (2009) Spatial and temporal dynamics of habitat selection across canopy gradients generates patterns of species richness and composition in aquatic beetles. Ecol Entomol 34:457–465. https://doi.org/10.1111/j.1365-2311.2008.01069.x
Blaustein L (1999) Oviposition site selection in response to risk of predation: evidence from aquatic habitats and consequences for population dynamics and community structure. In: Wasser S (ed) Evolutionary theory and processes: modern perspectives. Kluwer, Dordrecht, pp 441–456
Colwell RK (2013) EstimateS: Statistical estimation of species richness and shared species from samples. Version 9. User’s Guide and application published at: http://purl.oclc.org/estimates
Deans RA, Chalcraft DR (2016) Matrix context and patch quality jointly determine diversity in a landscape-scale experiment. Oikos 126:874–887
Delibes M, Gaona P, Ferreras P (2001) Effects of an attractive sink leading into maladaptive habitat selection. Am Nat 158:277–285. https://doi.org/10.1086/321319
Eveland LL, Bohenek J, Silberbush A, Resetarits WJ Jr (2016) Detection of fish and newt kairomones by ovipositing mosquitoes. In: Schulte BA, Goodwin T, Ferkin MH (eds) Chemical signals in vertebrates-volume 13. Springer, Berlin, pp 247–259
Fretwell SD, Lucas HLJ (1970) On territorial behavior and other factors influencing habitat distribution in birds. I. Theoretical development. Acta Biotheor 19:16–36
Golubski AJ, Abrams PA (2011) Modifying modifiers: what happens when interspecific interactions interact? J Anim Ecol 80:1097–1108. https://doi.org/10.1111/j.1365-2656.2011.01852.x
Hughey MC, McCoy MW, Vonesh JR, Warkentin KM (2012) Spatial contagion drives colonization and recruitment of frogflies on clutches of red-eyed treefrogs. Biol Lett 8:887–889. https://doi.org/10.1098/rsbl.2012.0468
Johnson CG (1969) Migration and dispersal of insects by flight. Methuen, London
Kraus J, Vonesh J (2010) Feedbacks between community assembly and habitat selection shape variation in local colonization. J Anim Ecol. https://doi.org/10.1111/j.1365-2656.2010.01684.x
Layton RJ, Voshell JRJ (1991) Colonization of new experimental ponds by benthic macroinvertebrates. Environ Entomol 20:110–117
Lundkvist E, Landin J, Milberg P (2001) Diving beetle (Dytiscidae) assemblages along environmental gradients in an agricultural landscape in southeastern Sweden. Wetlands 21:48–58. https://doi.org/10.1672/0277-5212(2001)021[0048:DBDAAE]2.0.CO;2
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton University Press, Princeton
McCoy MW, Barfield M, Holt RD (2009) Predator shadows: complex life histories as generators of spatially patterned indirect interactions across ecosystems. Oikos 118:87–100. https://doi.org/10.1111/j.1600-0706.2008.16878.x
Morris DW (2003) Toward an ecological synthesis: a case for habitat selection. Oecologia 136:1–13
Orians GH, Wittenberger JF (1991) Spatial and temporal scales in habitat selection. Am Nat 137:S29–S49. https://doi.org/10.2307/2678832
Orrock JL, Dill LM, Sih A et al (2010) Predator effects in predator-free space: the remote effects of predators on prey. Open Ecol 3:22–30. https://doi.org/10.2174/1874213001003030022
Pearl CA, Adams MJ, Leuthold N, Bury RB (2005) Amphibian occurrence and aquatic invaders in a changing landscape: implications for wetland mitigation in the Willamette Valley, Oregon, USA. Wetlands 25:76–88. https://doi.org/10.1672/0277-5212(2005)025[0076:AOAAII]2.0.CO;2
Petranka JW, Holbrook CT (2006) Wetland restoration for amphibians: should local sites be designed to support metapopulations or patchy populations? Restor Ecol 14:404–411. https://doi.org/10.1111/j.1526-100X.2006.00148.x
Pulliam HR, Danielson BJ (1991) Sources, sinks, and habitat selection: a landscape perspective on population dynamics. Am Nat 137:S50–S66
Resetarits WJ Jr (2005) Habitat selection behaviour links local and regional scales in aquatic systems. Ecol Lett 8:480–486. https://doi.org/10.1111/j.1461-0248.2005.00747.x
Resetarits WJ Jr, Binckley CA (2009) Spatial contagion of predation risk affects colonization dynamics in experimental aquatic landscapes. Ecology 90:869–876. https://doi.org/10.1890/08-0613.1
Resetarits WJ Jr, Binckley CA (2013a) Patch quality and context, but not patch number, drive multi-scale colonization dynamics in experimental aquatic landscapes. Oecologia 173:933–946. https://doi.org/10.1007/s00442-013-2644-5
Resetarits WJ, Binckley CA (2013b) Is the pirate really a ghost? evidence for generalized chemical camouflage in an aquatic predator, pirate perch. Am Nat 181(5):690–699
Resetarits WJ Jr, Pintar MR (2016) Functional diversity of non-lethal effects, chemical camouflage, and variation in fish avoidance in colonizing beetles. Ecology 97:3517–3529. https://doi.org/10.1002/ecy.1593
Resetarits WJ Jr, Silberbush A (2016) Local contagion and regional compression: habitat selection drives spatially explicit, multiscale dynamics of colonisation in experimental metacommunities. Ecol Lett 19:191–200. https://doi.org/10.1111/ele.12553
Resetarits WJ Jr, Binckley CA, Chalcraft DR (2005) Habitat selection, species interactions, and processes of community assembly in complex landscapes: a metacommunity perspective. In: Holyoak M, Leibold MA, Holt RD (eds) Metacommunities: spatial dynamics and ecological communities. University of Chicago Press, Chicago, pp 374–398
Robert S, Torrie JH, Dickey D (1997) Principles and procedures of statistics: a biometrical approach. McGraw-Hill, New York
Schlaepfer MA, Runge MC, Sherman PW (2002) Ecological and evolutionary traps. Trends Ecol Evol 17:474–480. https://doi.org/10.1016/S0169-5347(02)02580-6
Sheldon AL (1984) Colonization dynamics of aquatic insects. In: Resh HV, Rosenberg DM (eds) The ecology of aquatic insects. Praeger, New York, pp 401–429
Silberbush A, Blaustein L (2008) Oviposition habitat selection by a mosquito in response to a predator: are predator-released kairomones air-borne cues? J Vector Ecol 33:208–211
Trexler JC, Loftus WF, Jordan CF et al (2001) Ecological scale and its implications for freshwater fishes in the Florida Everglades. In: Porter JW, Porter KG (eds) The Everglades, Florida Bay, and coral reefs of the Florida keys: an ecosystem sourcebook. CRC Press, Boca Raton, pp 153–181
Vonesh JR, Kraus JM (2009) Pesticide alters habitat selection and aquatic community composition. Oecologia 160:379–385. https://doi.org/10.1007/s00442-009-1301-5
Vonesh JR, Kraus JM, Rosenberg JS, Chase JM (2009) Predator effects on aquatic community assembly: disentangling the roles of habitat selection and post-colonization processes. Oikos 118:1219–1229. https://doi.org/10.1111/j.1600-0706.2009.17369.x
Wellborn GA, Skelly DK, Werner EE (1996) Mechanisms creating community structure across a freshwater habitat gradient. Annu Rev Ecol Syst 27:337–363. https://doi.org/10.1146/annurev.ecolsys.27.1.337
Wesner JS, Billman EJ, Belk MC (2012) Multiple predators indirectly alter community assembly across ecological boundaries. Ecology 93:1674–1682. https://doi.org/10.1890/11-2061.1
Wiens JA (1976) Population responses to patchy environment. Annu Rev Ecol Syst 7:81–120. https://doi.org/10.1146/annurev.es.07.110176.000501
Zalom FG, Grigarick AA, Way MO (1979) Seasonal and diel flight periodicities of rice field Hydrophilidae. Environ Entomol 8:938–943
Zera AJ, Denno RF (1997) Physiology and ecology of dispersal polymorphism in insects. Annu Rev Entomol 42:207–230. https://doi.org/10.1146/annurev.ento.42.1.207
Acknowledgements
M. Pintar, J. Bohenek, and L. Eveland helped with numerous aspects of the experiment. M. Pintar identified the beetles. The manuscript benefitted from the comments of J. Bohenek, M. Pintar, and T. Breech. Special thanks to D. Chalcraft for stimulating discussions on analysis and the nature of independence. The work was supported by The University of Mississippi and the Henry L. and Grace Doherty Foundation. Logistical support provided by the UM Field Station.
Author information
Authors and Affiliations
Contributions
WJR conceived and designed the experiment, analysed the data, and wrote the manuscript. No other person is entitled to authorship.
Corresponding author
Ethics declarations
Ethical approval
All applicable institutional and national guidelines for the care and use of animals were followed.
Additional information
Communicated by Jill Lancaster.
Rights and permissions
About this article
Cite this article
Resetarits, W.J. Giving predators a wide berth: quantifying behavioral predator shadows in colonizing aquatic beetles. Oecologia 186, 415–424 (2018). https://doi.org/10.1007/s00442-017-4024-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-017-4024-z