Abstract
The relative importance of resource heterogeneity and quantity on plant diversity is an ongoing debate among ecologists, but we have limited knowledge on relationships between tree diversity and heterogeneity in soil nutrient availability in tropical forests. We expected tree species richness to be: (1) positively related to vertical soil nutrient heterogeneity; (2) negatively related to mean soil nutrient availability; and (3) more influenced by nutrient availability in the upper than lower soil horizons. Using a data set from 60, 20 × 40-m plots in a moist forest, and 126 plots in miombo woodlands in Tanzania, we regressed tree species richness against vertical soil nutrient heterogeneity, both depth-specific (0–15, 15–30, and 30–60 cm) and mean soil nutrient availability, and soil physical properties, with elevation and measures of anthropogenic disturbance as co-variables. Overall, vertical soil nutrient heterogeneity was the best predictor of tree species richness in miombo but, contrary to our prediction, the relationships between tree species richness and soil nutrient heterogeneity were negative. In the moist forest, mean soil nutrient availability explained considerable variations in tree species richness, and in line with our expectations, these relationships were mainly negative. Soil nutrient availability in the top soil layer explained more of the variation in tree species richness than that in the middle and lower layers in both vegetation types. Our study shows that vertical soil nutrient heterogeneity and mean availability can influence tree species richness at different magnitudes in intensively utilized tropical vegetation types.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Heterogeneous environments generally contain more niches compared with homogenous ones (MacArthur and MacArthur 1961; Ricklefs 1977; Chesson 2000). Habitat heterogeneity may be an important factor in explaining spatial heterogeneity in species richness (Huston 1979; Lundholm 2009; Stein et al. 2014). Indeed, numerous studies show that species richness of many groups of animals increases with habitat heterogeneity; for example, birds (Cody 1981), insects (Liu et al. 2015), and ungulates (Tews et al. 2004). Whether such a relationship also exists for plant species richness is less clear (Amarasekare 2003; Perroni-Ventura et al. 2006; Silva et al. 2013), partly because plants use, and interact within, a narrower spectrum of resources (Pérez-Ramos et al. 2012), and vary widely in morphology and size (Eilts et al. 2011). Nevertheless, some studies on forbs and grasses in grassland ecosystems have found positive relationships between small-scale heterogeneity in soil depth, soil nutrient availability, and species richness (Perroni-Ventura et al. 2006). However, few studies have examined if there is any relationship between habitat heterogeneity and tree species richness in forest ecosystems (Dufour et al. 2006; Chisholm et al. 2013), partly because it is difficult to conduct experimental manipulations over realistic spatial and temporal extents on these large, long-lived organisms.
Plant species richness and composition are strongly determined by availability and heterogeneity in resources, such as soil moisture, soil physical properties, and light availability (Murphy and Lugo 1986; Mooney et al. 1995). Soil nutrient availability in forest habitats depends on the underlying geology, plant species composition [e.g., the presence and abundance of legumes: (Gei and Powers 2013)], above-ground biomass production (Paoli et al. 2008), topography, climate, and anthropogenic disturbance (Lavelle and Spain 2001; Aponte et al. 2013). In addition, plant species richness varies across space, depending on the ability of species to acquire patchily distributed soil nutrients (Ricklefs 1977; Tilman and Pacala 1993; Hill and Hill 2001; Wright 2002; Questad and Foster 2008). The previous studies show that anthropogenic disturbance influences how plant species richness relates to soil nutrient availability, through altering plant nutrient demand and supply in forest (Fraterrigo et al. 2005; Bartels and Chen 2010) and grassland ecosystems (Abrams and Hulbert 1987; Silva et al. 2013).
The interactions between biotic and abiotic ecosystem components affect the inherent heterogeneity in nutrient availability among soil horizons (Lavelle and Spain 2001), which could affect plant growth and species distributions. For example, plant species are specialized in nutrient uptake and, therefore, have species-specific responses to soil nutrient availability (Huston 1979; Wijesinghe et al. 2005; Maestre et al. 2006; Lundholm 2009). Thus, plant species coexistence in forest and woodland may be influenced by the ability of species to avoid competition for resources, partly because of differences in their capacity to adapt and exploit available resources at different levels, such as soil nutrients at different soil depth layers (Sardans and Peñuelas 2013). In addition, differences in life forms and morphological characteristics enhance a plant species’ capacity to specialize in resource use across niche space (Schoener 1974; Scholes and Archer 1997).
Concentrations of most essential nutrients for plant growth, as well as microbial activity, generally decrease with soil depth (Jobbágy and Jackson 2001; Jumpponen et al. 2010). Moreover, vegetation change, such as tree growth, alters the quantity, availability, and vertical distribution of soil nutrients down a soil profile and thereby important determinants of the presence, abundance, and distribution of individual plant species across forest ecosystems (Jobbágy and Jackson 2004). To date, studies have focused mainly on how plant species richness varies with temporal and horizontal patterns of soil nutrient heterogeneity (Tilman and Pacala 1993; Stevens and Carson 2002; Musila et al. 2005; Perroni-Ventura et al. 2006; Reynolds et al. 2007). There is been ongoing debate among ecologists as to whether mean soil nutrient supply (Stevens and Carson 2002) or heterogeneity actually promotes plant species coexistence (Lundholm 2009; Schoolmaster Jr 2013). Studies in grasslands suggest that soil depth (Dornbush and Wilsey 2010) and vertical soil nutrient heterogeneity (Williams and Houseman 2014) positively affect plant species richness. However, we are not aware of any studies of the influence of vertical soil nutrient heterogeneity and depth-specific soil nutrient availability on tree species richness in tropical forests and woodlands. Given the high tree species diversity of these ecosystems, better knowledge about relationships between vertical soil nutrient heterogeneity and tree species richness could potentially shed light on conflicting findings in studies of resource heterogeneity and plant species coexistence (Eilts et al. 2011).
In this study, we explore the relationships between woody species richness in two vegetation types in Tanzania and both the vertical soil nutrient heterogeneity, and the mean soil and specific soil nutrient availabilities within the top three soil layers. We predict:
-
1.
A positive relationships between woody species richness and vertical soil nutrient heterogeneity, because theoretical models indicate that environmental heterogeneity promotes species coexistence and thereby species richness (Chesson 2000).
-
2.
A negative relationship between tree species richness and mean soil nutrient availability, because the supply rate of the most limiting resource controls plant growth (Tilman 1985), thereby overriding resource limitations that cause species diversity to be more associated with quantity (i.e., mean) than the variation in resource availability in natural ecosystems (Stevens and Carson 2002). Moreover, in grasslands at least, adding nutrients experimentally tends to decrease richness, because a few species respond rapidly and outcompete others (Reynolds et al. 2007).
-
3.
Tree species richness is more strongly related to nutrient availability in the upper soil horizon than in lower horizons, because, in tropical forest ecosystems at least, most of the essential nutrients for plant growth and over 60 % of tree root mass occurs in the top 30-cm soil layer (Jackson et al. 1996; Gregory 2008).
Materials and methods
Study area
We measured tree species richness and soil nutrient availability along an elevational gradient in a moist forest in Hanang district, and in miombo woodlands in Kilombero, Kilolo, Mufindi, Iringa rural, Mbeya rural, Mbozi, and Chunya districts of Tanzania. These districts were selected to represent a wide range of topographic and climatic conditions in moist forest and miombo woodlands (Online Resource 1).
Miombo woodlands (hereafter ‘miombo’) cover about 90 % of the forested land in Tanzania (URT 1998). They occur over a wide range of tropical climates and are classified as either wet or dry, with a separation around 1000-mm annual rainfall (White 1983; Frost 1996). Our miombo study sites spanned an elevation range of 25–2000 m, a temperature range of 16–30 °C, and rainfall range of 400–650 mm (Frost 1996; Platts et al. 2014). They are dominated by the genera Brachystegia and Julbernardia, as elsewhere in southern East and Central Africa (Thomas and Packham 2007), and are strongly influenced by frequent fires and anthropogenic disturbances (Campbell et al. 1996; Furley et al. 2008; Tarimo et al. 2015). The soils underlying miombo are generally well drained, highly leached, and low in plant-available nutrients (Jeffers and Boaler 1966). Shallow soils occur around weathered rock outcrops and where laterite hard pans restrict plant rooting (MacClanahan 1996). These variations in soil depth and drainage, along with differences in local climate, fire regime, grazing, and land use, all produce considerable local heterogeneity in miombo species composition and vegetation structure (Frost 1996; Desanker et al. 1997). Anthropogenic disturbances include fuelwood extraction, settlement, and agricultural expansion (Dewees et al. 2010).
The moist forest spans an elevation range of 1860–3418 m on Mt Hanang, and is categorized as montane and upper montane forest on the wetter southern, eastern, and northern slopes, and dry montane forests on the drier western slopes. There are also extensive rocky areas covered by grassland, thickets, and upland moorlands (Lovett and Pocs 1993). The moist forest is dominated by species in the genera Albizia, Cassipourea, Hagenia, Prunus, Cussonia, Olea, and Vernonia. The underlying forest soils are derived mainly from volcanic parent material, but there is small-scale variation in both soils and associated vegetation, such as canopy forests with humus-rich loamy soils, bushlands with reddish–brown to orange–red soils, upland moorlands with dark brown sandy soils, and steep slopes with short stunted plant growth and bare soils on rocky areas with quartz pebbles (Lovett and Pocs 1993). The forest receives an annual rainfall of 750–2000 mm, with a mean annual temperature range of 16–25 °C (Lovett and Pocs 1993; Platts et al. 2014). This forest is relatively less disturbed by human activities.
Data collection
We surveyed 20 × 40-m vegetation plots in moist forest (n = 60) and miombo (n = 126). Plots were positioned at least 400 m apart along elevation gradients, and their geographic locations recorded using a hand-held GPS (Garmin Map76cx). We measured and recorded trees with dbh ≥5 cm, and identified them in the field. For unidentified trees species, voucher specimens were identified at the National Herbarium in Arusha, Tanzania. To account for the potential effects of disturbance on the relationships between species richness and aspects of soil nutrient availability, we counted cut tree stumps in each plot, and estimated distances (km) from the nearest road and human settlement. Stumps and distances from roads and settlements alone may not capture all elements of disturbances; because of a long history of anthropogenic disturbances (Campbell et al. 1996), wood also decays rapidly (Yang et al. 2010). However, the number of cut stumps and distance to roads provides an indication of recent human activities, such as logging, charcoal production, settlement, and agricultural expansions, and has previously been used to account for human disturbances in forest ecosystems (Ramı́rez-Marcial et al. 2001; Williams-Linera 2002).
We sampled soils between May 2011 and March 2012 during the vegetation survey in the two vegetation types. Soil samples were collected at 0–15-cm (top), 15–30-cm (mid), and 30–60-cm (lower) depths, from the four corners and centre of each plot, but we did not consider soil bulk density. Thereafter, the samples were aggregated into three composites of one sample per soil depth layer in each plot. A total of 378 soil samples were collected, but only 375 samples were analysed, because three samples from one plot were lost. Analyses were done at Seliani Agricultural Research Institute, Arusha, Tanzania. In the laboratory, all samples were air dried and sieved through a 2-mm wire mesh and subsequently analysed for soil pH (at 1:2.5 soil: H2O), percentage organic carbon (Walkley–Black method), available phosphorus (Bray II), total nitrogen (Kjeldahl method), potassium, calcium, sodium, and magnesium (ammonium acetate 1.0 M pH 7.0 extraction), as described in Page (1982). Soil particle size were classified into clay (<2 µm), silt (2–20 µm), fine sand (20–50 µm), and coarse sand (50–2000 µm) (Fullen and Catt 2004).
Statistical analysis
Tree species richness was determined by the total number of tree species occurring in each plot. We used the Chao2 estimator (Chao 1987; Colwell and Coddington 1994) to estimate overall species richness in the miombo woodland and forest sites using occurrence data across all samples from these sites.
Vertical soil nutrient heterogeneity (VH) was estimated using coefficient of variation (CV) in soil nutrients from the three soil depth layers in each plot (Baer et al. 2005; Holl et al. 2013). We calculated mean soil nutrient availability (M) as the arithmetic mean of soil nutrient availability across each layer in a plot. The explanatory variables were the means and coefficients of variation of phosphorus (P), nitrogen (N), potassium (K), calcium (Ca), magnesium (Mg), sodium (Na), silt, fine sand, coarse sand, pH, and organic carbon (OC), along with the actual soil nutrient values in each soil depth layer. We included the number of cut tree stumps (stump-cuts), distance from the nearest settlement to each plot (settlement), distances from nearest road to each plot (road), and elevation as co-variables in the analyses (Table 1). Data exploration indicated high heteroscedasticity, and therefore, all continuous explanatory variables were checked for skewness, corrected where necessary to approximate zero skewness (Økland et al. 2001), and scaled to improve the interpretability of regression coefficients (Schielzeth 2010).
We used generalized least-square regression (gls) to explore the relationships between tree species richness and vertical soil nutrient heterogeneity (VH), mean soil nutrient availability (M), and depth-specific (DS) soil nutrient availability. Generalized least-square regression was preferred, because it accounts for the high heteroscedasticity in our data (Zuur et al. 2009, 2013).
We first explored the relationship between tree species richness and vertical soil nutrient heterogeneity (VH) or mean soil nutrients (M), and then with vertical soil nutrient heterogeneity and mean soil nutrient availability combined (VH + M). We then investigated relationships between tree species richness and soil nutrient availability at each soil depth separately. At each step, the models included stump-cuts, distance to settlement, distance to road, elevation, and their first-term interactions with all soil nutrient variables. Models were developed separately for moist forest and miombo. Data exploration indicated a non-linear relationship between tree richness and elevation in miombo; consequently, this relationship was fitted using a quadratic term. We checked for correlations within groups of explanatory variables, to ensure that all those included in the regression models had variance inflation factors ≤8.0 (Zuur et al. 2010) and Pearson correlation (r) ≤70 % (Dormann et al. 2013).
We used stepwise backward elimination and the Akaike Information Criterion (AIC) for final model selection (Zuur et al. 2009). Models with the lowest AIC and variables with significant contributions (p < 0.05) were selected as the most parsimonious (Zuur et al. 2009). We assessed each model’s goodness-of-fit, validated the final products by observing the spread of residuals, and compared the models using the likelihood ratio test (Zuur et al. 2009, 2010).
Results
Overall, we recorded 3003 tree stems of 240 tree species from 51 families in miombo and 3466 tree stems of 97 tree species from 46 families in moist forest. Miombo had higher estimated tree species richness (mean Chao2 = 279) than that of moist forest (mean Chao2 = 120 species) (Fig. 1). Moreover, there was a wider range and slightly higher values of soil nutrient availability in the middle and bottom soil depth layers compared with the top soil layers in both moist forest and miombo (Online Resource 2).
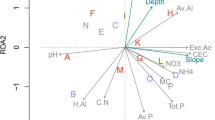
The most parsimonious model explaining tree species richness in miombo included only vertical soil nutrient heterogeneity (VH: AIC = 694, R 2 = 42 %), although it was not significantly different from models with mean soil nutrient availability only (M: AIC = 698, R 2 = 38 %), or the combination of mean soil nutrient availability and vertical soil nutrient heterogeneity (VH + M: R 2 = 42 %, AIC = 702, Table 2, Online Resources 3 and 4). In moist forest, the model with mean soil nutrient availability only (M: AIC = 316, R 2 = 59 %, LRT21.9, P = 0.001) explained significantly more of the variation in tree species richness compared with the model based on vertical soil nutrient heterogeneity alone (VH: AIC = 326, R 2 = 53 %), or the combination of mean soil nutrient availability and vertical soil nutrient heterogeneity (VH + M: AIC = 318, R 2 = 36 %; Table 2, Online Resources 3 and 4).
Tree species richness in miombo was strongly negatively associated with vertical soil nutrient heterogeneity (CV–pH and CV–OC; Table 2). CV–pH interacted positively with settlement and elevation (Table 2). In moist forest, tree species richness was significantly negatively associated with mean soil K and Na, and significantly positively associated with mean soil silt. Anthropogenic disturbance (distance from road) interacted negatively with the effect of silt (Table 2).
Relationships between species richness and soil nutrient availability differed substantially among the three soil depth layers (Table 3 and Online Resource 5). The variation in tree species richness was best explained by soil nutrient availability in the top soil layer in both vegetation types (montane forest: AIC = 308, R 2 = 66 %, miombo: AIC = 679, R 2 = 49 %), followed by soil nutrient availability in the middle (forest: AIC = 323, R 2 = 49 %, miombo: AIC = 690, R 2 = 35 %) and in the lower (forest: AIC = 325, R 2 = 46 %, miombo: AIC = 694, R 2 = 32 %) soil depth layers (Table 3, and Online Resource 5).
There were more negative relationships between tree species richness and soil nutrient availability in moist forest than in miombo across all the three soil depth layers (Table 3). However, the relationship between tree species richness and some of the soil nutrients showed opposite patterns among the three soil depths. For example, in moist forest, tree species richness had a strong positive relationship with Mg in the top soil layer but a negative relationship in the lower soil layer (Table 3, Online Resource 5). In miombo, tree species richness had a strong positive relationship with Ca in the topsoil layer, whereas it was negative in the middle soil layer (Table 3, Online Resource 5). In both vegetation types, soil nutrients and soil of different particle size interacted with many disturbance variables, illustrating a large complexity in the relationships (Table 3).
Discussion
Vertical soil nutrient heterogeneity (VH) explained more of the variation in tree species richness in miombo than did mean soil nutrient availability, which may reflect relatively high ranges in soil nutrient heterogeneity and anthropogenic disturbances (i.e., selective removal of trees, settlements, shifting cultivation, access roads, and frequent fires) in miombo woodlands. When compared with less herbaceous vegetation in moist forests, miombo has high mixture of woody-herbaceous communities that are highly diverse and exhibit large structural and functional differences within and between plant communities (House et al. 2003). Heterogeneity in soil nutrient (i.e., vertical soil nutrient availability) influences the relationships among plant species life forms and vice versa, which may subsequently influence species relative abundance in heterogeneous habitats like miombo woodlands. Our results also show that the ranges of nutrient content are generally larger in miombo than moist forest, suggesting high heterogeneity in miombo. Moreover, resource partitioning between the root systems of trees and herbaceous plants, and frequent disturbance (i.e., selective removal of trees, grazing, and frequent fires) in miombo, may also influence vertical heterogeneity in soil nutrients and thereby species coexistence (Hill and Hanan 2010). Furthermore, there is close association between species diversity and habitat heterogeneity (Huston 1994; Zhou et al. 2012).
Tree species richness was higher in miombo than in moist forest, yet there were more tree stems in moist forest, contrary to some previous reports (Munishi and Shear 2004). These differences further suggest greater habitat heterogeneity in miombo than in moist forest. Most miombo sites that we surveyed appear to be recovering from earlier selective logging of trees and abandoned farms or fires (i.e., anthropogenic disturbance), which can increase tree species richness compared with undisturbed woodlands (Connell 1978; Frost 1996). In contrast, the moist forest sites were less disturbed and had taller and larger canopy trees of few dominant species. Lower disturbance and greater dominance by a few species are sometimes associated with lower overall diversity (Ruiz-Jaen and Potvin 2011). Nevertheless, this may also partly represent our sampling approach, given the larger number of plots from different sites in miombo compared with fewer plots from the more homogeneous and concentrated moist forest sites. However, extrapolation results from rarefaction curves (Fig. 1) show that miombo woodland in our study sites do have higher tree species richness than the moist forest.
Our results do not support our prediction that vertical soil nutrient heterogeneity promotes tree species richness. Thus, the strong negative relationship between species richness and vertical soil nutrient heterogeneity in miombo was unexpected, given the high species richness of these woodlands and the associated high incidence of anthropogenic disturbance (i.e., selective logging, fires, and grazing). In tropical forest ecosystems, tree species richness is positively correlated with the amount of soil organic carbon, because of high litterfall and rapid decay rates from different species (Islam et al. 2015). In dry subtropical forests (like miombo woodlands), however, soil organic carbon takes longer to recover after shifting cultivation, selective logging, and burning, at which over 50 % can be lost (Solomon et al. 2007), even though the plant community recovers relatively rapidly (Colón and Lugo 2006; Pinard et al. 2000). Nevertheless, longer soil organic carbon recovery and rapid plant recovery rate after disturbances may not explicitly explain the negative relationship between vertical soil nutrient heterogeneity (CV–OC) and tree species richness, and hence requires an in-depth study to understand the underlying mechanisms. The significant negative association that we found between tree species richness and CV–pH in miombo may also be associated with disturbance, such as frequent fires (Strømgaard 1992; Higgins et al. 2007; Tarimo et al. 2015), and the influence that soil pH has on other essential nutrients for plant growth (John et al. 2007). The rapid change in soil pH, phosphorus, and exchangeable bases (Mg and K) in the top soil, resulting from frequent burning of plant biomass, not only influences the dynamics of soil nutrients, but also plant species richness (Frost 1996; John et al. 2007). Controlled experiments are needed to determine at what level variability in soil pH results in changes to trees species richness. Phosphorus and nitrogen were not selected as important predictors in the most parsimonious models in our study, although some previous studies have shown that they are the most limiting soil nutrient elements to plant growth and do influence tree diversity in tropical ecosystems (Siddique et al. 2010; Xiankai et al. 2010), especially in high pH soils (i.e., phosphorus in regularly burned soil in miombo, see Campbell et al. 1996) and fertile volcanic soils (i.e., nitrogen in the moist forest, see Xiankai et al. 2010).
In moist forest, mean soil nutrient availability explained more of the variation in tree species richness than did vertical soil nutrient heterogeneity, a finding consistent with some studies (i.e., Holl et al. 2013) but not with others (i.e., Lundholm 2009). In line with our second prediction, we found that species richness decreased with soil nutrient availability. Strong resource limitations and interspecific competition characterize tropical moist forests (i.e., Hanang moist forest), where a dense canopy limits light availability (Bartels and Chen 2010) causing a few dominant species capturing available soil nutrients. Those tree species with a high biomass production and large canopies can dominate access to soil nutrients and light, probably making soil nutrient availability rather than heterogeneity more of a limiting factor for species coexistence (Stevens and Carson 2002). The Hanang moist forest is a primary forest, characterized by large stems and with canopies of some species tending to dominate, creating continuous, mono-dominant stands, a pattern found in some forests elsewhere in the tropics (Connell 1978; Ruiz-Jaen and Potvin 2011). In contrast, trees in miombo are typically shorter and their canopies are more open, because of frequent selective harvesting (Timberlake et al. 2010), and often found in areas with low precipitation (Murphy and Lugo 1986; Davenport and Nicholson 1993; Campbell et al. 1996), as also observed in our study sites.
There were more negative than positive relationships between tree species richness and both vertical soil nutrient heterogeneity and mean nutrient availability in the two vegetation types. This could reflect intensified competition as plants grow (Perroni-Ventura et al. 2006; Toledo et al. 2011). In the moist forest, tree species richness decreased linearly with an increase in nutrients essential for tree growth (e.g., the mean values for K and Na). Tree species richness in tropical forests decreases with increasing soil fertility due to an increase in the size of the dominant tree species (Huston 1980, 1994). The resulting increase in above-ground biomass production can limit resources, such as light available to smaller trees, thus leading to a decrease in tree species richness (Stevens and Carson 2002). Although we did not measure or observed changes in tree sizes and light during snapshot field survey, the previous studies show that the directions of the relationships between richness and soil nutrient availability are determined, therefore, not only by niche differentiation among plant species, but also by plant size (Tilman and Pacala 1993; Eilts et al. 2011).
In theory, heterogeneity in the availability of resources, such as soil nutrients, should explain much of the local variation in plant species richness, and hence together with topography, disturbance, and other factors, such as soil moisture, ought to promote high plant species richness in tropical woodlands and forests (Stein et al. 2014). We found that relationships between tree species richness, vertical soil nutrient heterogeneity, and depth-specific soil nutrient availability differed notably between miombo woodland and moist forest. In both vegetation types, soil nutrient availability in the top soil layer explained more of the variation in tree species richness than did availability in the middle and lower layers, likely reflecting the concentration of essential soil nutrients in the topsoil layer (Jobbágy and Jackson 2001). The patterns are complex, however, and require more detailed studies to uncover the underlying mechanisms. For example, in moist forest, tree species richness increased with increasing Mg in the topsoil layers but decreased with the concentration of Mg in the lower soil layers. Tree species richness in miombo showed the same pattern, but with Ca not Mg as the critical nutrient. We observed a wider range and slightly higher values of Mg and Ca in the middle and lower soil depths than at the topsoil layer in both moist forest and miombo. This vertical distribution of soil nutrients down the various soil profiles may reflect species-specific features of the trees related to nutrient cycling, such as root distribution, rooting depths, and nutrient uptake and return rates, as opposed to leaching and chemical weathering (Jobbágy and Jackson 2001). Thus, not only could soil depth be influencing plant species richness (Dornbush and Wilsey 2010), but also heterogeneity in nutrient availability at different depths. Overall, our results suggest possible species differences in root morphology and response to inter- and intra-specific competition for soil nutrients (e.g., Huston 1980, 1994).
In miombo, all these patterns were strongly modified by disturbance (fire, shifting cultivation, livestock grazing, and selective logging), caused by proximity to settlements and roads, and likely tree felling. Tree species richness strongly decreased with CV–pH, but interacted positively with disturbance (i.e., was higher closer to settlements), suggesting that disturbance may modify soil pH (i.e., alkaline deposition from charcoal ashes) and may influence tree recruitment patterns and growth (Hart et al. 2005). Miombo generally occurs on infertile, acidic soils, which may favor a few suitably adapted species (Frost 1996). However, increased anthropogenic disturbance (i.e., fires, charcoaling, and selective logging) close to settlement could increase soil nutrient availability through biomass burning and manuring by livestock, thereby promoting tree species richness. In addition, most of miombo species have the ability to regenerate from root suckers or coppice (Luoga et al. 2004), which increase their chance to survive after disturbances. Tree species richness in miombo also increased with CV-coarse sand, a variable that was negatively associated with disturbance, being higher further away from settlements (perhaps reflecting a disinclination for people to settle on sandy soils). Competition between plants for soil moisture is likely to be greater on coarse sands, favouring those tree species that can tolerate low water availability and compete successfully under such conditions. In contrast, close to settlement, on less sandy soils but with higher disturbance, competitive pressures among tree species could be lower (Woollen et al. 2012).
Tree species richness also increased with Ca, OC, and pH, although these relationships turned to negative upon interactions tree cutting, distance from roads and settlements, respectively. They do not support our prediction that vertical soil nutrient heterogeneity promotes tree species richness. Nevertheless our result suggests that the influence of anthropogenic disturbances on how tree species richness relates to soil fertility varies spatially on the woodland landscape (Pausas and Austin 2001). We do not have fire frequency data from our study area, but frequent recurring fires are common in miombo (Tarimo et al. 2015), and have an important influence on soil nutrient availability (Frost 1996; Williams et al. 2008), which, in turn, can influence plant growth and both species richness and composition (Shelukindo et al. 2014). The observed differences in the relationship between species richness and soil nutrient availability down the soil profile, especially when modified by disturbance, suggest that tree growth and species coexistence are likely influenced by species-specific adaptations to edaphic and physiographic conditions, disturbance, and associated vegetation.
We have shown that plant species richness is influenced by vertical heterogeneity in soil nutrient concentrations in miombo, mean soil nutrient in moist forest, and nutrient availability in the top soil layer in both vegetation types. Contrary to our expectation, most of the relationships between tree species richness and vertical soil nutrient heterogeneity and depth-specific soil nutrient availability were negative in the two vegetation types. Although often neglected, vertical soil nutrient heterogeneity and depth-specific soil nutrient availability have potential influence on woody species richness in forests and woodlands. The inherent vertical and depth-specific soil nutrient variability, especially in miombo (i.e., poor soil nutrient), indicate that soil depth and drainage have influence on water and nutrient dynamics in both vegetation types (Grundy 1996). However, we did not measure soil bulk density to account for soil water and nutrient dynamics in this study. The increase in anthropogenic disturbances (fire, selective logging, charcoaling, and livestock grazing) in miombo woodlands and other forests types in Tanzania (Luoga et al. 2002; Persha and Blomley 2009) will likely alter their vertical soil nutrient heterogeneity, depth-specific nutrient availability, structure, composition, and diversity. Thus, we call for increased soil conservation efforts and control of anthropogenic disturbances to maintain tree diversity and soil nutrient availability in miombo woodlands and moist forests across Eastern, Southern, and South-Central Africa.
References
Abrams MD, Hulbert LC (1987) Effect of topographic position and fire on species composition in tallgrass prairie in northeast Kansas. Am Midl Nat 117:442–445. doi:10.2307/2425988
Amarasekare P (2003) Competitive coexistence in spatially structured environments: a synthesis. Ecol Lett 6(12):1109–1122
Aponte C, García LV, Marañón T (2013) Tree species effects on nutrient cycling and soil biota: a feedback mechanism favouring species coexistence. For Ecol Manag 309:36–46
Baer SG, Collins SL, Blair JM, Knapp AK, Fiedler AK (2005) Soil heterogeneity effects on tallgrass prairie community heterogeneity: an application of ecological theory to restoration ecology. Restor Ecol 13:413–424
Bartels SF, Chen HY (2010) Is understory plant species diversity driven by resource quantity or resource heterogeneity? Ecology 91(7):1931–1938
Campbell B, Frost P, Byron N (1996) Miombo woodlands and their use: overview and key issues. In: Campbell B (ed) The miombo in transition: woodlands and welfare in Africa. Center for International Forestry Research (CIFOR), Bogor, pp 1–10
Chao A (1987) Estimating the population size for capture-recapture data with unequal catchability. Biometrics 43:783–791
Chesson P (2000) General theory of competitive coexistence in spatially varying environments. Theor Popul Biol 58:211–237
Chisholm RA, Muller-Landau HC, Abdul Rahman K, Bebber DP, Bin Y, Bohlman SA, Bourg NA, Brinks J, Bunyavejchewin S, Butt N, Cao H (2013) Scale-dependent relationships between tree species richness and ecosystem function in forests. J Ecol 101(5):1214–1224
Cody ML (1981) Habitat selection in birds: the roles of vegetation structure, competitors, and productivity. Bioscience 31(2):107–113. doi:10.2307/1308252
Colón SM, Lugo AE (2006) Recovery of a subtropical dry forest after abandonment of different land uses. Biotropica 38(3):354–364. doi:10.1111/j.1744-7429.2006.00159.x
Colwell RK, Coddington JA (1994) Estimating terrestrial biodiversity through extrapolation. Philos Trans R Soc B 345:101–118
Connell JH (1978) Diversity in tropical rain forests and coral reefs. Science 199(4335):1302–1310
Davenport ML, Nicholson SE (1993) On the relation between rainfall and the normalized difference vegetation index for diverse vegetation types in East Africa. Int J Remote Sens 14(12):2369–2389
Desanker P, Frost P, Justice C, Scholes R (1997) The Miombo Network: framework for a terrestrial transect study of land-use and land-cover change in the miombo ecosystems of Central Africa. IGBP Report 41, International Geosphere-Biosphere Programme, Stockholm
Dewees PA, Campbell BM, Katerere Y, Sitoe A, Cunningham AB, Angelsen A, Wunder S (2010) Managing the miombo woodlands of southern Africa: policies, incentives and options for the rural poor. J Nat Resour Policy Res 2(1):57–73
Dormann CF, Elith J, Bacher S, Buchmann C, Carl G, Carré G, Marquéz JRG, Gruber B, Lafourcade B, Leitão PJ, Münkemüller T, McClean C, Osborne PE, Reineking B, Schröder B, Skidmore AK, Zurell D, Lautenbach S (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:27–46. doi:10.1111/j.1600-0587.2012.07348.x
Dornbush ME, Wilsey BJ (2010) Experimental manipulation of soil depth alters species richness and co-occurrence in restored tallgrass prairie. J Ecol 98:117–125
Dufour A, Gadallah F, Wagner HH, Guisan A, Buttler A (2006) Plant species richness and environmental heterogeneity in a mountain landscape: effects of variability and spatial configuration. Ecography 29(4):573–584
Eilts JA, Mittelbach GG, Reynolds HL, Gross KL (2011) Resource heterogeneity, soil fertility, and species diversity: effects of clonal species on plant communities. Am Nat 177:574–588
Fraterrigo JM, Turner MG, Pearson SM, Dixon P (2005) Effects of past land use on spatial heterogeneity of soil nutrients in southern appalachian forests. Ecol Monogr 75:215–230. doi:10.1890/03-0475
Frost PGH (1996) The ecology of miombo woodlands. In: Campbell B (ed) The miombo in transition: woodlands and welfare in Africa. CFIOR, Bogor, pp 11–57
Fullen MA, Catt JA (2004) Soil management: problems and solutions. Routledge, New York
Furley PA, Rees RM, Ryan CM, Saiz G (2008) Savanna burning and the assessment of long-term fire experiments with particular reference to Zimbabwe. Prog Phys Geogr 32:611–634
Gei MG, Powers JS (2013) Do legumes and non-legumes tree species affect soil properties in unmanaged forests and plantations in Costa Rican dry forests? Soil Biol Biochem 57:264–272
Gregory PJ (2008) Plant roots: growth, activity and interactions with the soil. Wiley, Oxford
Grundy IM (1996) Regeneration and management of Brachystegia spiciformis Benth. and Julbernardia globiflora (Benth). Troupin in miombo woodland, Zimbabwe. Ph.D thesis submitted for the degree of Doctor of Philosophy at the University of Oxford, UK
Hart SC, DeLuca TH, Newman GS, MacKenzie MD, Boyle SI (2005) Post-fire vegetative dynamics as drivers of microbial community structure and function in forest soils. For Ecol Manag 220(1):166–184
Higgins SI, Bond WJ, February EC, Bronn A, Euston-Brown DI, Enslin B, Govender N, Rademan L, O’Regan S, Potgieter AL, Scheiter S (2007) Effects of four decades of fire manipulation on woody vegetation structure in savanna. Ecology 88(5):1119–1125
Hill MJ, Hanan NP (2010) Ecosystem function in savannas: measurement and modeling at landscape to global scales. CRC Press, Boca Raton, New York
Hill J, Hill R (2001) Why are tropical rain forests so species rich? Classifying, reviewing and evaluating theories. Prog Phys Geogr 25:326–354
Holl KD, Stout VM, Reid JL, Zahawi RA (2013) Testing heterogeneity diversity relationships in tropical forest restoration. Oecologia 173:569–578
House JI, Archer S, Breshears DD, Scholes RJ (2003) Conundrums in mixed woody–herbaceous plant systems. J Biogeogr 30(11):1763–1777
Huston M (1979) A general hypothesis of species diversity. Am Nat 113:81–101. doi:10.2307/2459944
Huston M (1980) Soil nutrients and tree species richness in Costa Rican forests. J Biogeogr 7(2):147–157
Huston MA (1994) Biological diversity: the coexistence of species. Cambridge University Press, London
Islam M, Dey A, Rahman M (2015) Effect of tree diversity on soil organic carbon content in the homegarden agroforestry system of north–eastern Bangladesh. Small-scale For 14(1):91–101
Jackson R, Canadell J, Ehleringer J, Mooney H, Sala O, Schulze E (1996) A global analysis of root distributions for terrestrial biomes. Oecologia 108(3):389–411
Jeffers JNR, Boaler SB (1966) Ecology of a Miombo site, Lupa North Forest Reserve, Tanzania: i. weather and plant growth, 1962–64. J Ecol 54:447–463. doi:10.2307/2257961
Jobbágy EG, Jackson RB (2001) The distribution of soil nutrients with depth: global patterns and the imprint of plants. Biogeochemistry 53:51–77
Jobbágy EG, Jackson RB (2004) The uplift of soil nutrients by plants: biogeochemical consequences across scales. Ecology 85(9):2380–2389. doi:10.1890/03-0245
John R, Dalling JW, Harms KE, Yavitt JB, Stallard RF, Mirabello M, Hubbell SP, Valencia R, Navarrete H, Vallejo M, Foster RB (2007) Soil nutrients influence spatial distributions of tropical tree species. Proc Natl Acad Sci 104(3):864–869
Jumpponen A, Jones KL, Blair J (2010) Vertical distribution of fungal communities in tallgrass prairie soil. Mycologia 102:1027–1041. doi:10.3852/09-316
Lavelle P, Spain A (2001) Soil ecology. Springer Science and Business Media, Dordretch
Liu Y, Duan M, Zhang X, Zhang X, Yu Z, Axmacher JC (2015) Effects of plant diversity, habitat and agricultural landscape structure on the functional diversity of carabid assemblages in the North China Plain. Insect Conserv Divers 8(2):163–176. doi:10.1111/icad.12096
Lovett JC, Pocs I (1993) Assessment of the conditions of the catchment forest reserves, a botanical appraisal. Catchment Forest Project Report, vol 93.3. Forest Division ⁄ NORAD, Dar es Salaam
Lundholm JT (2009) Plant species diversity and environmental heterogeneity: spatial scale and competing hypotheses. J Veg Sci 20:377–391. doi:10.1111/j.1654-1103.2009.05577.x
Luoga EJ, Witkowski E, Balkwill K (2002) Harvested and standing wood stocks in protected and communal miombo woodlands of eastern Tanzania. For Ecol Manag 164(1):15–30
Luoga EJ, Witkowski ET, Balkwill K (2004) Regeneration by coppicing (resprouting) of miombo (African savanna) trees in relation to land use. For Ecol Manag 189(1):23–35
MacArthur RH, MacArthur JW (1961) On bird species diversity. Ecology 42:594–598. doi:10.2307/1932254
MacClanahan TR (1996) East African ecosystems and their conservation. Oxford University Press, New York
Maestre FT, Bradford MA, Reynolds JF (2006) Soil heterogeneity and community composition jointly influence grassland biomass. J Veg Sci 17:261–270
Mooney HA, Bullock SH, Medina E (1995) Introduction. In: Bullock SH, Mooney HA, Medina E (eds) Seasonally dry tropical forests. Cambridge University Press, Cambridge, pp 1–8
Munishi PK, Shear TH (2004) Carbon storage in afromontane rain forests of the Eastern Arc mountains of Tanzania: their net contribution to atmospheric carbon. J Trop For Sci 16(1):78–93
Murphy PG, Lugo AE (1986) Ecology of tropical dry forest. Ann Rev Ecol Syst 17:67–88
Musila W, Todt H, Uster D, Dalitz H (2005) Is geodiversity correlated to biodiversity? A case study of the relationship between spatial heterogeneity of soil resources and tree diversity in a Western Kenyan rainforest. In: African Biodiversity. Springer, Netherlands, pp 405–414
Økland RH, Økland T, Rydgren K (2001) Vegetation environment relationships of boreal spruce swamp forest in Østmarka Nature Reserve, SE Norway. Sommerfeltia 29:190
Page AL (ed) (1982) Methods of soil analysis. Part 2. Chemical and microbiological properties. American Society of Agronomy, Soil Science Society of America, Madison
Paoli GD, Curran LM, Slik J (2008) Soil nutrients affect spatial patterns of aboveground biomass and emergent tree density in southwestern Borneo. Oecologia 155(2):287–299
Pausas JG, Austin MP (2001) Patterns of plant species richness in relation to different environments: an appraisal. J Veg Sci 12:153–166. doi:10.2307/3236601
Pérez-Ramos IM, Roumet C, Cruz P, Blanchard A, Autran P, Garnier E (2012) Evidence for a plant community economics spectrum driven by nutrient and water limitations in a Mediterranean rangeland of southern France. J Ecol 100(6):1315–1327. doi:10.1111/1365-2745.12000
Perroni-Ventura Y, Montaña C, García-Oliva F (2006) Relationship between soil nutrient availability and plant species richness in a tropical semi-arid environment. J Veg Sci 17:719–728
Persha L, Blomley T (2009) Management decentralization and montane forest conditions in Tanzania. Conserv Biol 23(6):1485–1496
Pinard MA, Barker MG, Tay J (2000) Soil disturbance and post-logging forest recovery on bulldozer paths in Sabah, Malaysia. For Ecol Manag 130(1–3):213–225. doi:10.1016/S0378-1127(99)00192-9
Platts PJ, Omeny PA, Marchant R (2014) AFRICLIM: high resolution climate projections for ecological applications in Africa. Afr J Ecol 53:103–108
Questad EJ, Foster BL (2008) Coexistence through spatio-temporal heterogeneity and species sorting in grassland plant communities. Ecol Lett 11:717–726. doi:10.1111/j.1461-0248.2008.01186.x
Ramı́rez-Marcial N, González-Espinosa M, Williams-Linera G (2001) Anthropogenic disturbance and tree diversity in montane rain forests in Chiapas, Mexico. For Ecol Manag 154(1–2):311–326. doi:10.1016/S0378-1127(00)00639-3
Reynolds HL, Mittelbach GG, DARCY-HALL TL, Houseman GR, Gross KL (2007) No effect of varying soil resource heterogeneity on plant species richness in a low fertility grassland. J Ecol 95:723–733
Ricklefs RE (1977) Environmental heterogeneity and plant species diversity: a hypothesis. Am Nat 111:376–381. doi:10.2307/2460072
Ruiz-Jaen MC, Potvin C (2011) Can we predict carbon stocks in tropical ecosystems from tree diversity? Comparing species and functional diversity in a plantation and a natural forest. N Phytol 189(4):978–987
Sardans J, Peñuelas J (2013) Plant-soil interactions in Mediterranean forest and shrublands: impacts of climatic change. Plant Soil 365:1–33
Schielzeth H (2010) Simple means to improve the interpretability of regression coefficients. Methods Ecol Evol 1:103–113
Schoener TW (1974) Resource partitioning in ecological communities. Science 185(4145):27–39
Scholes R, Archer S (1997) Tree-grass interactions in savannas. Ann Rev Ecol Syst 28:517–544
Schoolmaster Jr DR (2013) Resource competition and coexistence in heterogeneous metacommunities: many-species coexistence is unlikely to be facilitated by spatial variation in resources. PeerJ 1:e136
Shelukindo HB, Semu E, Msanya B, Mwango S, Singh BR, Munishi P (2014) Characterization of some typical soils of miombo woodlands ecosystem of Kitonga forest reserve, Iringa, Tanzania: physico–chemical properties and classification. J Agric Sci Technol AB 4:224–234
Siddique I, Vieira ICG, Schmidt S, Lamb D, Carvalho CJR, Figueiredo RDO, Blomberg S, Davidson EA (2010) Nitrogen and phosphorus additions negatively affect tree species diversity in tropical forest regrowth trajectories. Ecology 91(7):2121–2131
Silva DM, Batalha MA, Cianciaruso MV (2013) Influence of fire history and soil properties on plant species richness and functional diversity in a neotropical savanna. Acta Botanica Brasilica 27:490–497
Solomon D, Lehmann J, Kinyangi J, Amelung W, Lobe I, Pell A, Riha S, Ngoze S, Verchot L, Mbugua D (2007) Long-term impacts of anthropogenic perturbations on dynamics and speciation of organic carbon in tropical forest and subtropical grassland ecosystems. Glob Change Biol 13(2):511–530
Stein A, Gerstner K, Kreft H (2014) Environmental heterogeneity as a universal driver of species richness across taxa, biomes and spatial scales. Ecol Lett 17:866–880
Stevens MHH, Carson WP (2002) Resource quantity, not resource heterogeneity, maintains plant diversity. Ecol Lett 5:420–426
Strømgaard P (1992) Immediate and long-term effects of fire and ash-fertilization on a Zambian miombo woodland soil. Agric Ecosyst Environ 41(1):19–37
Tarimo B, Dick ØB, Gobakken T, Totland Ø (2015) Spatial distribution of temporal dynamics in anthropogenic fires in miombo savanna woodlands of Tanzania. Carbon Balance Manag 10(1):1–15
Tews J, Brose U, Grimm V, Tielbörger K, Wichmann M, Schwager M, Jeltsch F (2004) Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures. J Biogeogr 31(1):79–92
Thomas PA, Packham JR (2007) Ecology of woodlands and forests, description, dynamics and diversity. Cambridge University Press, Cambridge
Tilman D (1985) The resource-ratio hypothesis of plant succession. Am Nat 125(6):827–852
Tilman D, Pacala S (1993) The maintenance of species richness in plant communities. In: Ricklefs RE, Schluter D (eds) Species diversity in ecological communities. University of Chicago Press, Chicago, pp 13–25
Timberlake J, Chidumayo E, Sawadogo L (2010) Distribution and characteristics of African dry forests and woodlands. Managing for products and services, The dry forest and woodlands of Africa, pp 11–42
Toledo M, Poorter L, Peña-Claros M, Alarcón A, Balcázar J, Chuviña J, Leaño C, Licona JC, ter Steege H, Bongers F (2011) Patterns and determinants of floristic variation across lowland forests of Bolivia. Biotropica 43:405–413
URT (1998) United Republic of Tanzania National Forest Policy. Forestry and Beekeeping Division Ministry of Natural Resources and Tourism, Dar es Salaam
White F (1983) The vegetation of Africa, a descriptive memoir to accompany the UNESCO/AETFAT/UNSO vegetation map of Africa (3 Plates, Northwestern Africa, Northeastern Africa, and Southern Africa, 1: 5,000,000). United Nations Educational, Scientific and Cultural Organization, Paris
Wijesinghe DK, John EA, Hutchings MJ (2005) Does pattern of soil resource heterogeneity determine plant community structure? An experimental investigation. J Ecol 93:99–112. doi:10.1111/j.0022-0477.2004.00934.x
Williams BM, Houseman GR (2014) Experimental evidence that soil heterogeneity enhances plant diversity during community assembly. Plant Ecol 7:461–469. doi:10.1093/jpe/rtt056
Williams M, Ryan CM, Rees RM, Sambane E, Fernando J, Grace J (2008) Carbon sequestration and biodiversity of re-growing miombo woodlands in Mozambique. For Ecol Manag 254:145–155. doi:10.1016/j.foreco.2007.07.033
Williams-Linera G (2002) Tree species richness complementarity, disturbance and fragmentation in a Mexican tropical montane cloud forest. Biodivers Conserv 11(10):1825–1843
Woollen E, Ryan CM, Williams M (2012) Carbon stocks in an African woodland landscape: spatial distributions and scales of variation. Ecosystems 15(5):804–818
Wright JS (2002) Plant diversity in tropical forests: a review of mechanisms of species coexistence. Oecologia 130:1–14
Xiankai L, Mo J, Gilliam FS, Zhou G, Yunting Fang Y (2010) Effects of experimental nitrogen additions on plant diversity in an old-growth tropical forest. Glob Change Biol 16(10):2688–2700
Yang F-F, Li Y-L, Zhou G-Y, Wenigmann K, Zhang D-Q, Wenigmann M, Liu S-Z, Zhang Q-M (2010) Dynamics of coarse woody debris and decomposition rates in an old-growth forest in lower tropical China. For Ecol Manag 259(8):1666–1672
Zhou J, Dong B-C, Alpert P, Li H-L, Zhang M-X, Lei G-C, Yu F-H (2012) Effects of soil nutrient heterogeneity on intraspecific competition in the invasive, clonal plant Alternanthera philoxeroides. Ann Bot 109(4):813–818. doi:10.1093/aob/mcr314
Zuur A, Ieno EN, Walker N, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Statistics for biology and health, 1st edn. Springer, New York
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14. doi:10.1111/j.2041-210X.2009.00001.x
Zuur AF, Hilbe J, Ieno EN (2013) A beginner’s guide to GLM and GLMM with R: a frequentist and Bayesian perspective for ecologists. Highland Statistics, Newburgh
Acknowledgments
This research was supported by the Norwegian Government through the Climate Change Impacts, Adaptation and Mitigation programme (CCIAM) at Soikoine University of Agriculture, Tanzania, and the Norwegian State Education Loan Fund. We thankfully acknowledge the field assistance of H. Seki, R. Khasim, J. Herbet, G. Bulenga, O. Bakombezi, G. Swai, and G. Lema. We are indebted to Y. Abeid, M. Mwangoka, and C. Kayombo who helped in plant identification and P. K. T. Munishi for his comments and support at the designing stages. We thank two anonymous reviewers for their constructive comments, Peter Frost for his valuable comments, and copy-editing.
Author contribution statement
DS conceived the idea, DS, SM, and ØT designed the study, DS carried out the study and analysed the data, and DS, SM, and ØT wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors. For this type of study, formal consent is not required.
Additional information
Communicated by Russell K. Monson.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shirima, D.D., Totland, Ø. & Moe, S.R. The relative importance of vertical soil nutrient heterogeneity, and mean and depth-specific soil nutrient availabilities for tree species richness in tropical forests and woodlands. Oecologia 182, 877–888 (2016). https://doi.org/10.1007/s00442-016-3696-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-016-3696-0