Abstract
To fulfill their needs, animals are constantly making trade-offs among limiting factors. Although there is growing evidence about the impact of ambient temperature on habitat selection in mammals, the role of environmental conditions and thermoregulation on apex predators is poorly understood. Our objective was to investigate the influence of ambient temperature on habitat selection patterns of grizzly bears in the managed landscape of Alberta, Canada. Grizzly bear habitat selection followed a daily and seasonal pattern that was influenced by ambient temperature, with adult males showing stronger responses than females to warm temperatures. Cutblocks aged 0–20 years provided an abundance of forage but were on average 6 °C warmer than mature conifer stands and 21- to 40-year-old cutblocks. When ambient temperatures increased, the relative change (odds ratio) in the probability of selection for 0- to 20-year-old cutblocks decreased during the hottest part of the day and increased during cooler periods, especially for males. Concurrently, the probability of selection for 21- to 40-year-old cutblocks increased on warmer days. Following plant phenology, the odds of selecting 0- to 20-year-old cutblocks also increased from early to late summer while the odds of selecting 21- to 40-year-old cutblocks decreased. Our results demonstrate that ambient temperatures, and therefore thermal requirements, play a significant role in habitat selection patterns and behaviour of grizzly bears. In a changing climate, large mammals may increasingly need to adjust spatial and temporal selection patterns in response to thermal constraints.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
By the end of the twenty-first century, the global average temperature will have warmed by at least 1.5 °C since the 1900s, with greater warming in temperate and arctic regions (Intergovernmental Panel on Climate Change 2013). The body of research demonstrating the impacts of climate change on mammalian species is increasing (e.g. Walther et al. 2002; Molnár et al. 2011; McCain and King 2014) but there is a growing need to better understand how species interact and cope with their changing environment (Williams et al. 2008; Boyles et al. 2011). For species already threatened by anthropogenic factors such as over-exploitation, habitat encroachment, and fragmentation, additional stress incurred from climate change may exacerbate the negative effects of anthropogenic pressures and lead to further reduction in population sizes (Walther et al. 2002).
The ability of mammals to maintain thermoneutrality is affected by environmental conditions because heat generated through metabolism is continuously dissipated into the environment and heat from the environment is absorbed through radiation, conduction, and convection (Stelzner and Hausfater 1986). Individual differences in body size, sex and reproductive status can affect thermoregulatory needs (Poppitt et al. 1994), and for large mammals adapted to cold environments, thermal inertia, a thick hide, and abundant insulation limits their capacity to dissipate heat (Speakman and Król 2010; Riek and Geiser 2013). Although shade-seeking, postural adjustments, and reduced activity levels are probably the most common behavioural tactics of large mammals attempting to avoid heat stress (Stelzner and Hausfater 1986; Bourgoin et al. 2011; Duncan and Pillay 2013), little is known about how thermoregulatory needs and behavioural thermoregulation of large mammals affects daily activity patterns (Boyles et al. 2011; but see Bourgoin et al. 2011; Marchand et al. 2014).
Optimal foraging theory predicts that mammals should forage to maximise gains while taking into account all potential costs and benefits (Pyke 1984), and should therefore continue to forage as long as energetic gains exceed costs (Lima and Dill 1990). In accordance with the phenology of forage maturation, the digestible energy content of foods, energy maximization principles, and the need to balance macronutrients, individuals should also distribute themselves according to forage quality and abundance, and the availability of various food types (Wilmshurst et al. 1995; Erlenbach et al. 2014). The costs associated with foraging at a particular place and time can be related to predation risk, intra- and inter-specific competition, food availability and quality, digestion constraints, reproductive status, sex, and thermoregulation (Fortin et al. 2004; Steyaert et al. 2013). Abiotic factors (e.g. environmental conditions), biotic factors (e.g. competition and predation), and the heterogeneous distribution of resources over space and time influence trade-offs in habitat selection because individuals distribute themselves according to the availability of resources to satisfy their needs (Hebblewhite et al. 2008; Godvik et al. 2009). Habitat selection can be defined as the disproportionate use of a combination of abiotic and biotic resources in relation to availability and should be investigated at suitable spatial and temporal scales (Manly et al. 2002). Accordingly, fine spatial and temporal scales are typically best to investigate behaviours associated with thermoregulation (van Beest et al. 2012). To date, research on habitat selection for large mammals has focused on main trade-offs such as foraging and predator avoidance (Lima et al. 1985; Ordiz et al. 2011), but understanding the relationships among environmental conditions, thermoregulation, foraging efficiency, and habitat selection is becoming increasingly important to understanding species-specific vulnerabilities to climate change and improve management strategies that will allow for successful conservation efforts (Boyles et al. 2011; du Plessis et al. 2012).
Terrestrial bears (Ursus spp.) in North America are generally active during the day, forage in open areas and rest in closed-canopy forest stands with peaks of activity occurring in early morning and late evening (Munro et al. 2006; Heard et al. 2008). For bears, habitat selection is mainly driven by food availability, quality, and abundance (e.g. Nielsen et al. 2010), as well as sex and reproductive status including sexual segregation and searching for mates (Stenhouse et al. 2005), and avoidance of human activity and infrastructures (Gibeau et al. 2002; Nielsen et al. 2010). Still, time of day, season, and ambient temperatures can affect activity patterns and habitat selection (Moe et al. 2007; Martin et al. 2013). In human-dominated landscapes, daytime reductions in activity levels have been attributed to human avoidance (Gibeau et al. 2002; Ordiz et al. 2011) but in some cases, and as observed in other mammals, altered activity levels could also be attributed, in part, to thermoregulatory needs and avoidance of daytime heat (Garshelis and Pelton 1980; Heard et al. 2008).
The current and historical distribution of brown bears (Ursus arctos) covers a wide range of environmental conditions (Schwartz et al. 2003), and their persistence in extreme environments suggests that they are well adapted to extreme temperatures and temperature fluctuations. Phenotypic plasticity, genetic adaptation, and adaptive thermoregulation will likely provide some resilience to environmental changes associated with current climate change (Bradshaw and Holzapfel 2006; Boyles et al. 2011), but little is known about how resistant brown bears are to hyperthermia, and how plastic they are in response to a warming environment. Recent population estimates in Alberta, Canada, indicate that the province is now home to less than 500 mature grizzly bears (Ursus arctos horribilis) (Alberta Sustainable Resource Development and Alberta Conservation Association 2010) and understanding how current and changing weather conditions affect grizzly bear activity and trade-offs in habitat selection is therefore increasingly important. As observed in other species, high ambient temperature could reduce foraging efficiency of grizzly bears by pushing them into more thermally suitable habitats (thermal shelters), or by increasing their costs of active thermoregulation while foraging (du Plessis et al. 2012; van Beest et al. 2012). As was observed for herbivores, grizzly bears may also need to adjust their activity patterns to be active during the cool periods of the day (Dussault et al. 2004; Maloney et al. 2009). To understand the potential impacts of temperature on foraging efficiency, and as a first step to understanding the sensitivity of grizzly bears to climate change, we investigated the links among ambient temperature and habitat selection for grizzly bears in the boreal forest of Alberta.
Our objective was to investigate the influence of daily ambient temperature on habitat selection patterns of grizzly bears in a managed landscape. We hypothesized that warm temperatures would force grizzly bears to select habitats reflecting thermoregulatory needs such as seeking canopy cover to avoid thermal stress. However, because foraging requirements drive habitat selection, we first considered the presence of daily bimodal selection patterns typical of the morning and evening activity peaks observed for grizzly bears (Moe et al. 2007), and considered that seasonal patterns of habitat selection would follow the seasonal abundance, availability, and quality of key bear foods (Nielsen et al. 2004a, b; Robbins et al. 2007). Therefore, following forage maturation and energy maximization principles, and the daily and seasonal patterns of habitat selection typically observed for grizzly bears, we expected that grizzly bears would address their thermoregulatory needs by selecting (1) cool habitats (closed canopy) more and warm habitats (open canopy) less during the warmest part of the day, and (2) warm habitats during the coolest part of the day. Grizzly bears are sexually dimorphic, males are generally at least 20 % larger than females (Schwartz et al. 2003), and sexual segregation is known to occur in this species (Steyaert et al. 2013). With their lower surface area to volume ratio, males may face higher thermoregulation costs than females in warm environments and we therefore expected that (3) the effect of thermoregulatory needs on habitat selection would be more pronounced for males than females.
Materials and methods
Study area
The study area includes 8828 km2 of boreal forest in the Weyerhaeuser Grande Prairie Forest Management Agreement Area south of Grande-Prairie, Alberta (54°32′N, 119°13′W; Fig. 1). The elevation ranges from 543 to 2440 m a.s.l. and the landscape is characteristic of the lower and upper foothills, and subalpine ecological subregions (Beckingham and Archibald 1996; Fig. 1). Forest stands are mainly dominated by lodgepole pine (Pinus cordata) and black spruce (Picea mariana), and to a lesser extent, white spruce (Picea glauca), balsam fir (Abies balsamifera), trembling aspen (Populus tremuloides), and balsam poplar (Populus balsamifera). Conifer stands are dominant but broadleaf, mixed forest, shrublands, and wetlands are also present. Forestry and oil and gas activities have extensively altered the landscape with roads, pipelines, well sites, and >1300 km2 of cutblocks. During the study period, the average daily temperatures recorded by the Alberta Provincial Forest Fire Center (PFFC) automatic weather station (Kakwa-G1; 54°10′29.9″N 119°03′33.1″W) was 9 °C and ranged from −11 to 32 °C from June to September. Average daily minimum and maximum temperatures for the same period were 0 and 17 °C, respectively (data available on request from PFFC: http://wildfire.alberta.ca/fire-weather/forecasts-observations/default.aspx; Fig. 1).
Grizzly bear data
Since 1999, the FRI Research Grizzly Bear Program has been trapping grizzly bears using standard aerial darting, leg snares, and culvert traps (Cattet et al. 2003). Capture and handling techniques were in accordance with guidelines of the American Society of Mammalogists (Sikes et al. 2011) and approved by the University of Saskatchewan Animal Care Committee (Cattet et al. 2003). A full description of capture and handling techniques is available in Cattet et al. (2003). Bears captured for this study were fitted with Followit (formerly Televilt; Lindesberg, Sweden) global positioning system (GPS) radio collars. Radio collars were programmed to obtain one GPS location per hour and for this study, we only used GPS locations associated with a maximum dilution of precision of less than eight and a three-dimensional fix (Lewis et al. 2007). This method removed 2.7 % of all potential locations and the removed locations were not biased towards land cover types (P > 0.4). We studied 11 male and 12 female grizzly bears between 2005 and 2011. We used data for more than 1 year on 12 individuals (eight females and four males) for a total of 45 bear-years. Within the 26 bear-years obtained from females, 17 were from lone females and nine from females with at least one dependent offspring. All individuals (bear-years) had a minimum of 100 collar locations within a particular season (seasons are described in full details in the “Temporal covariates” section below). In total, we used 43,224 locations acquired between 16 June and 15 September, and the elevation of locations from all individuals ranged between 729 m and 1804 m a.s.l.
We categorized GPS locations from (1) mature conifer stands, (2) 0- to 20-year-old cutblocks, (3) 21- to 40-year-old cutblocks, and (4) shrublands using geographical information system (GIS) land cover maps developed from layers generated by Landsat Thematic Mapper imagery, Moderate Resolution Imaging Spectroradiometer imagery, topographic derivatives of a digital elevation model, and cutblock GIS polygons obtained from Weyerhaeuser Grande-Prairie (Franklin et al. 2002a, b; McDermid et al. 2009; Table 1). We extracted GIS land cover data for all bear locations using ArcGIS Desktop 10.2 (ESRI 2011) and restricted analyses to conifer stands, 0–20 and 21- to 40-year-old regenerating cutblocks, and shrublands because the majority of bear locations were observed in these land covers (83.5 % of all locations). Larsen (2012) and Nielsen et al. (2004b) established that for this study area, crown closure in regenerating cutblocks occurs approximately 20 years post-harvest and causes a significant change in bear food composition and abundance. We therefore chose to separate cutblocks aged 0–20 and 21–40 years. Conifer stands aged >40 years were classified as a single land cover: mature conifer forest. Shrublands were defined as regenerating stands with >25 % shrub cover and ≤5 % tree cover based on McDermid et al. (2009).
Temperature differences among land covers
We obtained hourly temperature data recorded at the Kakwa-G1 PFFC weather station, and because solar radiation and wind differences among different forest stands influence ambient temperatures, we expected ambient temperature to vary among land covers. To assess temperature differences among the four main land covers selected by grizzly bears in the study area, we constructed operative temperature (T e ) sensors (Online Resource 1). These T e sensors recorded temperature every hour, were exposed to convection (wind) and radiation (sun) and therefore provided a proxy of the T e , i.e. the perceived temperature, within the respective land covers (Bakken 1992). To account for spatial variability inherent to variations in elevation, topography, and geographic locations within the study area, we installed 22 T e sensors across the study area with a minimum of three replicates and a maximum of ten replicates per land cover (Fig. 1; Online Resource 1, Fig. A2). The sites for the 22 T e sensors were selected to represent the natural subregions available, and the longitudinal and latitudinal gradients of the study area (Fig. 1). Elevation across sites varied between 940 and 1682 m.
We assessed differences in T e among land covers throughout the day using mixed-model ANOVAs (Littell et al. 2006; Fig. 2). We separated hourly T e measurements recorded between 16 June and 15 September as morning twilight, morning, midday, evening, evening twilight, or nighttime using civil twilight tables (period of the day; Table 1), and used the weekly average T e measurements for each period. We used these cut-off dates to match bear location data since bears are often captured in June, and also to allow for sufficiently warm temperatures to occur. We considered each sensor as the experimental unit, the weekly averages as repeated measures (Littell et al. 2006), and applied a step-down Bonferroni multiple comparison procedure to test for differences among land covers by period of the day (Table 1; Hochberg 1988).
Average weekly T e and 95 % confidence limits as a function of the period of the day for the main land covers selected by male and female grizzly bears in boreal forest of Alberta, during July and August 2008, 2009, 2010. Results and 95 % confidence limits are from mixed-model ANOVAs and different letters indicate significant differences among land covers following a step-down Bonferroni multiple comparison procedure, α = 0.05. 0–20 0- to 20-year-old cutblocks, 21–40 21- to 40-year-old cutblocks
Temporal covariates
To investigate diel patterns in habitat selection, we divided days into the same six periods mentioned above based on sunrise, sunset, and civil twilight tables (http://www.cmpsolv.com/los/sunset.html accessed 28 June 2012; Munro et al. 2006; Table 1). We divided summer into three sub-seasons (hereafter referred to as “seasons”) based on changes in bear activity and plant phenology (Nielsen et al. 2006). We defined the covariate referring to the different period of the day as Period and the covariate for the three summer seasons as Season (Table 1).
Defining availability
Spatial and temporal scales affect our ability to understand and measure behaviours, including habitat selection patterns (Johnson 1980; Ciarniello et al. 2007). At the 3rd order of selection defined by Johnson (1980), an individual may decide to remain at a site or leave it because of short-lived factors such as perceived temperature. In this case, a limited area surrounding each location represents the scale of alternative available locations for that individual at that time. Daily ambient temperature influences habitat selection within short temporal scales; we therefore defined the availability of land covers that were accessible to individuals based on their movement rates [3rd-order selection (Johnson 1980)]. For bears, movement rates vary with reproductive status and throughout the summer (Munro et al. 2006; Martin et al. 2013). To define availability at each observed bear location, we generated one random location per used location within a radius of available area defined as the average upper quartile distance travelled between consecutive hourly locations for season- and reproductive status-specific categories of individuals (Online Resource 3, Table A1; Compton et al. 2002).
Habitat selection
We used paired logistic regression as described in Compton et al. (2002) and chose a generalized estimating equations framework to make inferences at the population level using PROC PHREG with the STRATA and identity statements as grouping factors to account for the within-individual bear × year aggregate correlation structure and control for repeated observations of the same individuals. We specified a robust sandwich covariance matrix estimator adapted to Cox proportional hazard models [COVSANDWICH(aggregate) statement and TYPE = breslow options (Lin and Wei 1989; Koper and Manseau 2009; SAS Institute 2011)]. This sandwich estimator allows for model mis-specifications assuming the independence of observations and corrects variance estimates to account for the appropriate correlation structure (in our case, year-based correlations). This approach is therefore robust to type II errors (Koper and Manseau 2009). We evaluated statistical hypotheses using Wald tests, and all statistical analyses were performed using SAS statistical software (SAS Institute 2011).
We controlled for diel and seasonal variation in bear activity by including the Period and Season covariates in models (Table 1). Most covariates were categorical and we therefore separated models by land cover and sex to simplify model interpretation. We did not investigate differences in reproductive status because of low sample size, but performed model selection for lone females selecting 0- to 20-year-old cutblocks and 21- to 40-year-old cutblocks to confirm that including females of different reproductive status in analyses did not bias our results (Online Resource 3, Table A2). To avoid collinearity between temperature and period of the day, and to avoid the use of arbitrary temperature cut-offs to facilitate repeatability and comparison with other regions and species (e.g. Garshelis and Pelton 1980; McLellan and McLellan 2015), we used daily maximum temperature as a covariate (Maloney et al. 2005; du Plessis et al. 2012; Table 1). Daily maximum temperature is also a commonly used metric in the climate change literature (ex. Huey et al. 2012; Walther et al. 2002).
For each land cover and sex combination, we built a set of 16 candidate models following a complete—case design with all possible combinations of the covariates and their interactions (Burnham and Anderson 2002). We opted for a complete—case model selection design because we a priori expected the selection of land covers to change with periods of the day (Period) and throughout the summer (Nielsen et al. 2004a). All possible combinations of the Period, Season, and Temperature covariates, and their interactions, were therefore deemed biologically relevant (Burnham and Anderson 2002). We used the quasi-likelihood under the independence model criterion (QICU) developed by Pan (2001) as a selection criterion because Akaike’s information criterion (AIC) is not applicable to generalized estimating equations methods. Generalized estimating equations require the use of quasi-likelihood because no likelihood is defined (Pan 2001), and as with AIC, QICU weights are calculated from ΔQICU (Burnham and Anderson 2002). We performed model averaging when results revealed no obvious most supported model among the set of candidate models (Burnham and Anderson 2002).
We report selection as odds ratios, the ratio between the probability that an event will occur and the probability that the same event will not occur (Grimes and Schulz 2008). Here, an odd ratio >1 refers to a land cover that is selected more than expected from random sampling, while an odds ratio <1 indicates a selection below that expected from random sampling. We assessed the performance of the most supported model within each land cover and sex class using a modified k-fold cross-validation method based on Fortin et al. (2009). We generated models using 80 % of randomly selected data from individuals and estimated the relative probabilities with the remaining 20 %. We then ranked the relative probabilities of the observed locations against the relative probabilities of associated random locations, grouped the probabilities into ten categories, and performed Spearman rank correlation (r s ) between categories and their associated frequencies (Boyce et al. 2002). We used the same method for the random locations, repeated this process 100 times per model, and reported the mean and SD. High r s -values indicate good descriptive models (Boyce et al. 2002).
Results
Temperature differences among land covers
T e sensors improved our ability to distinguish temperature variations among structurally different land covers (Fig. 2; Online Resource 1). Maximum daily ambient temperature differed among land covers used by grizzly bears (morning, F 3,18 = 45.7, p < 0.001; midday, F 3,18 = 15.4, p < 0.0001; evening, F 3,18 = 39.6, p < 0.001; evening twilight, F 3,18 = 5.4, p = 0.008). Temperature among land covers diverged rapidly after sunrise with 0- to 20-year-old cutblocks, and to a lesser extent shrublands, warming up faster than other land covers. Maximum temperatures for all land covers were reached at midday (Fig. 2). After sunset, 0- to 20-year-old cutblocks showed a drastic drop in temperature and became the coolest land cover (Fig. 2).
Habitat selection
As expected from previous knowledge on bear activity and habitat selection, the Period and Season covariates were present in most of the models within ≤2 ΔQICu of the most supported model (Table 2). In all cases, the most supported models differed between sexes (Table 2). Conifer stands were mostly selected less than expected based on availability, and males and females selected shrublands more than their availability regardless of the period of the day or season (Fig. 3). In early summer, both male and female grizzly bears selected 0- to 20-year-old cutblocks less than or close to availability depending on the period of the day, while in mid and late summer both sexes selected for 0- to 20-year-old cutblocks at all times (Fig. 4; Online Resource 4). In early summer, males selected 21- to 40-year-old cutblocks close to availability, while females selected this land cover more than expected during the day. In midsummer, males and females selected 21- to 40-year-old cutblocks throughout the day, but in late summer selection for 21- to 40-year-old cutblocks was below availability at all times (Fig. 4; Online Resource 4).
Relative change (odds ratio) in the selection of conifer stands (upper graphs) and shrublands (lower graphs) by male and female grizzly bears throughout the day and summer in the boreal forest of Alberta between 2005 and 2011. Odds ratios and 95 % confidence limits were estimated from the most supported candidate model using paired logistic regression. See Table 2 for the list of models ≤10 Δ quasi-likelihood under the independence model criterion (ΔQICu) of the most supported model. Season was not selected in the most supported model for males (Table 2). The most supported selected models for females included period of the day and season (Table 2) and odds ratios for each period of the day are shown for early summer, midsummer, and late summer. See Table 1 for a description of all covariates. Dashed lines emphasize changes in the odds ratios. MT Morning twilight, M morning, MD midday, E evening, ET evening twilight, N nighttime
Relative change (odds ratio) for the selection of 0- to 20-year-old (upper graphs) and 21- to 40-year-old (lower graphs) cutblocks by male and female grizzly bears throughout the day and summer in the boreal forest of Alberta, between 2005 and 2011. Odds ratios and 95 % confidence limits were estimated from the most supported candidate model using paired logistic regression. See Table 2 for the list of models ≤10 ΔQICu of the most supported model. The most supported models included period of the day and season (Table 2). Odds ratios for each period of the day are shown for early summer, midsummer, and late summer. See Table 1 for a description of all covariates. Dashed lines emphasize changes in the odds ratios. For abbreviations, see Fig. 3
Temperature-mediated effects
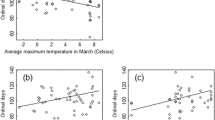
The influence of maximum daily ambient temperature on habitat selection patterns by male and female grizzly bears depended on land cover, period of the day, season, and sex (Table 2). We found no support for a relationship between maximum daily ambient temperature and the selection of conifer stands and shrublands by grizzly bears, but maximum daily ambient temperature influenced the selection of 0–20 and 21- to 40-year-old cutblocks (Table 2; Figs. 5, 6). Although our robust statistical approach to account for auto-correlation resulted in large confidence intervals (see “Habitat selection” section), we observed that with increasing maximum daily ambient temperatures, males increasingly selected 0- to 20-year-old cutblocks during the evening, evening twilight, and nighttime, while females increasingly selected these cutblocks during crepuscular periods (Fig. 5). The response to warming temperatures was more pronounced for males than females: females only slightly decreased their selection of 0- to 20-year-old cutblocks during midday, while males showed a strong decrease for selection of these during the same period (Fig. 5). The odds ratio for males selecting 0- to 20-year-old cutblocks during midday dropped to nearly half when maximum daily ambient temperatures increased from 20 to 30 °C, and while males did not select 0- to 20-year-old cutblocks when maximum daily ambient temperatures exceeded 25.3 °C, females still showed a slight selection for these cutblocks during midday (Fig. 5; Online Resource 4, Table A4).
Relative change (odds ratio) for the selection of 0- to 20-year-old cutblocks during morning twilight, morning, midday, evening, evening twilight, and nighttime as a function of daily maximum ambient temperature by male and female grizzly bears in the boreal forest of Alberta, between 2005 and 2011. Dotted lines are 95 % confidence intervals and the dashed line indicates even odds. Odds ratios and 95 % confidence limits were estimated from the most supported candidate model using paired logistic regression. See Table 2 for the list of models ≤10 ΔQICu of the most supported model and Online Resource 4, Table A4 for parameter estimates and SEs
Relative change (odds ratio) for the selection of 21- to 40-year-old cutblocks as a function of daily maximum ambient temperature by male and female grizzly bears in the boreal forest of Alberta between 2005 and 2011. There was no interaction between period of the day and temperature for the most supported models (Table 2). For males, odds ratios varied with season (Table 2) and confidence intervals are represented as different shades of grey: early summer (1), midsummer (2), and late summer (3). Dotted lines are 95 % confidence intervals and the dashed line indicates even odds. Odds ratios and 95 % confidence limits were estimated from the most supported candidate model using paired logistic regression. See Table 2 for the list of models ≤10 ΔQICu from the most supported model and Online Resource 4, Table A4 for parameter estimates and SEs
For both sexes, the selection of 21- to 40-year-old cutblocks was constant throughout the day and increased as daily maximum temperatures increased except for males in late summer (Fig. 6). In early and midsummer respectively, the odds ratio for males selecting 21- to 40-year-old cutblocks increased from 1.37 to 1.74 and from 1.29 to 1.76 with an increase in temperature from 20 to 30 °C, while in late summer, the selection for 21–40 year cutblocks by males showed a slight decrease (Fig. 6). For females, temperature also had a constant influence throughout the summer and the odds of selecting 21- to 40-year-old cutblocks increased from 0.95 to 1.09 with an increase in temperature from 20 to 30 °C (Fig. 6; Table 2). Even though we did not specifically investigate the influence of ambient temperature on the habitat selection patterns of females with and without dependent offspring because of low sample size, the most supported models for the selection of 0- to 20-year-old cutblocks and 21- to 40-year-old cutblocks for females without dependent offspring also included the effect of ambient temperature (Online Resource 3, Table A2). Overall, model performances within each land cover and sex categories were fair to good (Online Resource 5, Table A7).
Discussion
Male and female grizzly bears in the boreal forest of Alberta adjusted their patterns of habitat selection during summer according to ambient temperatures, suggesting that in addition to the main drivers of habitat selection (i.e. forage availability and quality, reproductive status, and predator avoidance), thermoregulatory constraints influence habitat selection. As predicted, grizzly bears decreased their selection for 0- to 20-year-old cutblocks during the warmest part of the day and increased their overall selection for 21- to 40-year-old cutblocks where higher crown closure provided shade and cooler temperatures. Patterns of habitat selection were similar between males and females but the influence of ambient temperature on the selection of cutblocks was more pronounced for males. Contrary to our expectations, however, ambient temperatures did not influence the selection of conifer stands and shrublands.
Our results demonstrate that grizzly bears respond to warm temperatures by altering their habitat selection patterns and seeking thermal shelters with likely foraging potential. In response to warm ambient temperatures, grizzly bears showed behavioural plasticity; they decreased their selection of young cutblocks with abundant high-quality forage during the warmest part of the day, and selected these cutblocks more during cooler periods. Similar responses to warming temperatures have been observed for ungulates such as moose (Alces alces) and wildebeest (Connochaetes gnou) (Dussault et al. 2004; Maloney et al. 2005) but to our knowledge, temporal adjustments in foraging behaviour as a response to thermal needs has never been observed for a large omnivore and apex predator. Also, bears are unique in that they are clear energy maximizers and forage constantly to gain weight for hibernation and reproduction (Farley and Robbins 1995; Erlenbach et al. 2014). Behavioural adjustments induced by thermal constraints could therefore affect foraging efficiency (du Plessis et al. 2012), especially during more energetically demanding periods associated with reproduction, or during periods of food scarcity associated with stochastic events. Owen-Smith (1998) observed that the influence of ambient temperature on activity levels of the greater kudu (Tragelaphus strepsiceros) was more pronounced during periods of low food abundance because of the compensatory increase in activity. In early and midsummer, grizzly bears selected old cutblocks that provided thermal cover and alternative high-quality forage (Nielsen et al. 2004a, b). In accordance with forage maturation and energy maximization principles (Robbins et al. 2007), forbs that are abundant within 21- to 40-year-old cutblocks during early and midsummer become less nutritious in late summer (Nielsen et al. 2004b), and selection for these cutblocks by males and females dropped accordingly. In late summer, male grizzly bears in our study area did not increase their selection for old cutblocks with increasing temperatures but females did, suggesting that females selecting these cutblocks were able to fulfill their foraging needs at least partly, but that males were not. These results suggest that thermal constraints may be exacerbated by seasonal fluctuation in food availability and associated energetic and macronutrient requirements. Although we did not seek to investigate changes in activity levels as a potential response to warm ambient temperature, it is likely that bears could also reduce the intensity of foraging activities to reduce heat production, thereby effectively reducing risks of hyperthermia while foraging in warm habitats (Maloney et al. 2005; Aublet et al. 2009). Because of the relatively low density of grizzly bears in our study area [18 bears/1000 km2 (Alberta Sustainable Resource Development and Alberta Conservation Association 2010; Mowat et al. 2013)], and because of the similarities observed in male and female grizzly bear habitat selection patterns in situations where temperature did not affect selection, we believe that sexual segregation and potential differences in human avoidance between sexes have a minimal impact on temperature-mediated habitat selection patterns in this area.
The response to warming temperatures was especially pronounced in males. Sexual dimorphism, with males being up to two times larger than females, likely explains this difference. Because of their low surface area to volume ratio, males face higher thermoregulation costs than females in warm environments, and could be more limited in their ability to cope with rising temperatures in open habitats (McNab 1983; McCain and King 2014). However, because of the high energy requirements of lactation, females with cubs likely generate more body heat than other females (Farley and Robbins 1995), and could also face high thermoregulation costs with increasing temperature. Because of low sample size, we were unable to investigate differences among reproductive status or body mass, and this shortcoming may have contributed to model selection uncertainty. Still, it is possible that because females have a higher surface area to volume ratio than males, they are better able to dissipate heat, but that the cost of active thermoregulation while foraging in productive 0- to 20-year-old cutblocks likely did not outweigh the benefits (Speakman and Król 2010). In addition, with warming temperatures in late summer, only females increased their selection for 21- to 40-year-old cutblocks. This difference may reflect the greater foraging requirements of males because succulent forbs that are abundant in this land cover become less nutritious as the summer progresses. As observed with captive grizzly bears by Rode et al. (2001), smaller individuals should be better able to make greater weight gain than larger bears while feeding on forbs.
We found no evidence that bears increased selection of cooler conifer stands on warm days; neither did we observe a reduction in the selection of warm shrublands. The lack of selection for conifer stands is not surprising because mature conifer forests are highly available and generally a poor source of food except for pine-dominant stands where Vaccinium species can be abundant (Ihalainen and Pukkala 2001; Larsen 2012). Conifer stands would provide thermal cover but the lack of forage in non-pine-dominant stands likely explains why bears did not increase their selection of conifer stands with increasing temperatures. Investigating selection patterns for pine-dominant vs. non-pine-dominant conifer stands would be a valuable continuation to this study but reliable within-stand species composition is still unavailable (Nijland et al. 2015). Shrublands were selected by males and increasingly selected by females throughout the summer, but temperature did not influence the selection of this land cover. Shrublands are similar to 0- to 20-year-old cutblocks (Table 1) but the presence of deciduous (Populus spp.) thickets typical of these stands could provide additional thermal shelter and allow for effective thermoregulation while bears forage. Microhabitat selection and intermixing of food and cover at a fine scale has been observed in other species (e.g. van Beest et al. 2012) but we were unable to investigate potential within-stand responses with the hourly GPS location data available for this study. We therefore recommend that future research assesses microhabitat selection of thermal shelters with the use of high-resolution GPS locations [i.e. 5-min intervals (Abrahms et al. 2015)]. Investigating microhabitat selection during specific activity bouts (i.e. foraging vs. resting) could also be accomplished with the use of activity sensors (Ware et al. 2015).
To our knowledge, there are no studies specifically linking ambient temperature to habitat selection patterns for terrestrial bear species. Still, Garshelis and Pelton (1980) observed that black bears (Ursus americanus) were virtually inactive when temperatures exceeded 23 °C, but in a similar study, Ayres et al. (1986) detected no such relationship. Ordiz et al. (2011) found that brown bears selected dense habitats for day beds more than for night beds, and also selected dense habitats more during the summer and autumn compared to the spring, especially near human settlements. They attributed these findings mainly to increased human activity but did not consider the potential for temperature-mediated selection of denser cover during daytime. In our study area, grizzly bears generally selected open areas more during the daytime than nighttime suggesting that human activity had a minimal influence on habitat selection patterns. Because ambient temperatures are high in the summer, and during the day, thermoregulatory needs could have played a role in the selection for dense cover observed by Ordiz et al. (2011). Even though we did not specifically seek to measure an upper critical temperature for grizzly bears, our results show that for males, the odds of selecting 0- to 20-year-old cutblocks at midday changed from positive to negative odds with maximum daily temperatures reaching 25.3 °C. This temperature threshold is similar to the threshold identified for black bear by Garshelis and Pelton (1980). Future research would benefit from looking into upper critical temperature for bear species allowing us to shed light on the mechanisms driving temperature-mediated changes in selection patterns and activity levels. Also, the use of heterothermy to cope with high ambient temperature has been observed for mammals living in arid environments (i.e. Weissenböck et al. 2011), and although this strategy is unlikely for large temperate mammals, the ability to cope with a high heat load by tolerating elevated body temperatures during warm days and dissipating excess heat during cool nights could also be investigated for terrestrial bear species.
Non-temperature-mediated selection patterns were similar between sexes. As observed by Munro et al. (2006) and Heard et al. (2008), grizzly bears in our area selected conifer stands following a diel bimodal pattern with the peak of selection occurring at nighttime and an increase in selection at midday. We observed the opposite diel selection pattern for 0- to 20-year-old cutblocks and shrublands where males and females reduced their selection at midday and nighttime but increased selection during other periods. Following forage maturation and the phenology of succulent forbs and berry availability, both sexes increased their selection of 0- to 20-year-old cutblocks throughout the summer and decreased their selection of 21- to 40-year-old cutblocks. Similar diel and seasonal patterns of selection have been observed in North America and Scandinavia (Munro et al. 2006; Moe et al. 2007). However, Scandinavian brown bears are mostly active during twilight and at night, likely because of human persecution (Moe et al. 2007; Zedrosser et al. 2011). In North America, Gibeau et al. (2002) and Nielsen et al. (2004a) also associated the selection of high-quality habitats during crepuscular and nocturnal period to high levels of human activity.
We did not strive to investigate the effects of human presence on the selection patterns of male and female grizzly bears. Even though the human footprint (roads, cutlines, and cutblocks) in the study area is substantial [more than 1300 km2 of cutblocks and >3500 km of road and truck trails within <9000 km2 (Laberee et al. 2014)], summer-time human activity in the area is mainly restricted to on-duty oil and gas and forestry workers. Except for a usual increase in human activity during the ungulate hunting season (September–November), human activity was not expected to vary throughout days and across seasons. Considering the temporal and spatial scale investigated, it is unlikely that human-activity introduced temporal, spatial, or sex-related biases in the habitat selection patterns that we observed for male and female grizzly bears.
Our results show that effects of thermoregulation on habitat selection of grizzly bears vary according to sex, and that warm temperatures impose a trade-off in habitat selection. The use of mature or densely covered forest stands as thermal shelters by bear species has previously been suggested by Waller and Mace (1997) but to our knowledge, our results are the first to explicitly demonstrate active behavioural responses to warm temperatures for a terrestrial bear species. Because of their constant need to forage, thermally induced constraints on foraging could have potential detrimental effects on overall fitness of bears (du Plessis et al. 2012). Although measures of fitness are difficult to obtain for a species such as grizzly bear, we believe that our results offer a substantial contribution to our knowledge of grizzly bear ecology. Our research provides the first necessary steps towards an understanding of the influence of ambient temperature on patterns of habitat selection for grizzly bears, and the potential impacts of climate change on the ecology of the species. Grizzly bears seem to modify foraging behaviour in response to their thermoregulatory needs by increasingly foraging during cool periods, and by seeking alternative thermally adequate foraging opportunities. Although challenging, future research would benefit from investigating the impacts of thermally driven trade-offs on foraging efforts (fine-scale activity levels and duration), foraging efficiency, and ultimately, the fitness of individuals and population dynamics. In the context of climate change, thermally driven habitat selection and behavioural responses will undoubtedly have implications for the long-term conservation of vulnerable populations because the increased frequency of extreme climatic events, including heat waves and droughts, may exacerbate thermoregulatory constraints of large mammals (Speakman and Król 2010; Boyles et al. 2011). We therefore recommend that managers consider the availability of thermal cover while developing management strategies that could minimize the impacts of landscape modifications on animal populations in an increasingly warming world. Intermixing densely covered land covers and open stands that are abundant in high-quality forage would allow grizzly bears to forage in high-quality food patches regardless of temperature and therefore prevent risks of hyperthermia without reducing foraging opportunities.
References
Abrahms B, Jordan NR, Golabek KA, McNutt JW, Wilson JW, Brashares JS (2015) Lessons from integrating behaviour and resource selection: activity-specific responses of African wild dogs to roads. Anim Conserv. doi:10.1111/acv.12235
Alberta Sustainable Resource Development and Alberta Conservation Association (2010) Status of the grizzly bear (Ursus arctos) in Alberta: update 2010. Wildlife status report no. 37. Alberta sustainable resource development. Edmonton, Alberta
Aublet JF, Festa-Bianchet M, Bergero D, Bassano B (2009) Temperature constraints on foraging behaviour of male Alpine ibex (Capra ibex) in summer. Oecologia 159:237–247. doi:10.1007/s00442-008-1198-4
Ayres LA, Chow LS, Graber DM (1986) Black bear activity patterns and human induced modifications in Sequoia National Park. Ursus 6:151–154. doi:10.2307/3872819
Bakken GS (1992) Measurement and application of operative and standard operative temperatures in ecology. Am Zool 32:194–216. doi:10.1093/icb/32.2.194
Beckingham JD, Archibald JH (1996) Field guide to ecosites of Northern Alberta. Natural Resources Canada. Canadian Forest Service. Special report 5. Northern Forestry Centre, Edmonton, Alberta
Bourgoin G, Garel M, Blanchard P, Dubray D, Maillard D, Gaillard J-M (2011) Daily responses of mouflon (Ovis gmelini musimon × Ovis sp.) activity to summer climatic conditions. Can J Zool 89:765–773. doi:10.1139/Z11-046
Boyce MS, Vernier PR, Nielsen S, Schmiegelow FKA (2002) Evaluating resource selection functions. Ecol Model 157:281–300. doi:10.1016/S0304-3800(02)00200-4
Boyles JG, Seebacher F, Smit B, McKechnie AE (2011) Adaptive thermoregulation in endotherms may alter responses to climate change. Integr Comp Biol 51:676–690. doi:10.1093/icb/icr053
Bradshaw WE, Holzapfel CM (2006) Climate change—evolutionary response to rapid climate change. Science 312:1477–1478. doi:10.1126/science.1127000
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. 2nd ed. Springer, New York, doi:10.2307/3803155
Cattet MRL, Christison K, Caulkett NA, Stenhouse GB (2003) Physiologic responses of grizzly bears to different methods of capture. J Wildl Dis 39:649–654
Ciarniello LM, Boyce MS, Seip DR, Heard DC (2007) Grizzly bear habitat selection is scale dependent. Ecol Appl 17:1424–1440. doi:10.1890/06-1100.1
Compton BW, Rhymer JM, McCollough M (2002) Habitat selection by wood turtles (Clemmys insculpta): an application of paired logistic regression. Ecology 3:833–843. doi:10.1890/0012-9658(2002)083[0833:hsbwtc]2.0.co;2
du Plessis KL, Martin RO, Hockey PAR, Cunningham SJ, Ridley AR (2012) The costs of keeping cool in a warming world: implications of high temperatures for foraging, thermoregulation and body condition of an arid-zone bird. Glob Change Biol 18:3063–3070. doi:10.1111/j.1365-2486.2012.02778.x
Duncan LM, Pillay N (2013) Shade as a thermoregulatory resource for captive chimpanzees. J Therm Biol 38:169–177. doi:10.1016/j.jtherbio.2013.02.009
Dussault C, Ouellet JP, Courtois R, Huot J, Breton L, Larochelle J (2004) Behavioural responses of moose to thermal conditions in the boreal forest. Ecoscience 11:321–328
Erlenbach JA, Rode KD, Raubenheimer D, Robbins CT (2014) Macronutrient optimization and energy maximization determine diets of brown bears. J Mammal 95:160–168. doi:10.1644/13-MAMM-A-161
ESRI (2011) ArcGIS Desktop: release 10. Environmental Systems Research Institute, Redlands
Farley SD, Robbins CT (1995) Lactation, hibernation, and mass dynamics of American black bears and grizzly bears. Can J Zool 73:2216–2222. doi:10.1139/z95-262
Fortin D, Boyce MS, Merrill EH (2004) Multi-tasking by mammalian herbivores: overlapping processes during foraging. Ecology 85:2312–2322. doi:10.1890/03-0485
Fortin D, Fortin ME, Hawthorne LB, Duchesne T, Courant S, Dancose K (2009) Group-size dependent association between food profitability, predation risk and distribution of free-ranging bison. Anim Behav 78:887–892. doi:10.1016/j.anbehav.2009.06.026
Franklin SE, Hansen JM, Stenhouse GB (2002a) Quantifying landscape structure with vegetation inventory maps and remote sensing. For Chron 78:866–875. doi:10.5558/tfc78866-6
Franklin SE, Lavigne MB, Wulder MA, Stenhouse GB (2002b) Change detection and landscape structure mapping using remote sensing. For Chron 78:618–625. doi:10.5558/tfc78618-5
Garshelis DL, Pelton MR (1980) Activity of black bears in the Great Smoky Mountains National Park. J Mammal 61:8–19. doi:10.2307/1379952
Gibeau ML, Clevenger AP, Herrero S, Wierzchowski J (2002) Grizzly bear response to human development and activities in the Bow River Watershed, Alberta, Canada. Biol Cons 103:227–236. doi:10.1016/S0006-3207(01)00131-8
Godvik IMR, Loe LE, Vik JO, Veiberg V, Langvatn R, Mysterud A (2009) Temporal scales, trade-offs, and functional responses in red deer habitat selection. Ecology 90:699–710. doi:10.1890/08-0576.1
Grimes DA, Schulz KF (2008) Making sense of odds and odds ratios. Obstet Gynecol 111:423–426. doi:10.1097/01.AOG.0000297304.32187.5d
Heard DC, Ciarniello LM, Seip DR (2008) Grizzly bear behavior and global positioning system collar fix rates. J Wildl Manage 72:596–602. doi:10.2193/2007-175
Hebblewhite M, Merrill E, McDermid G (2008) A multi-scale test of the forage maturation hypothesis in a partially migratory ungulate population. Ecol Monogr 78:141–166. doi:10.1890/06-1708.1
Hochberg Y (1988) A sharper Bonferroni procedure for multiple tests of significance. Biometrika 75:800–802. doi:10.1093/biomet/75.4.800
Huey RB, Kearney MR, Krockenberger A, Holtum JAM, Jess M, Williams SE (2012) Predicting organismal vulnerability to climate warming: roles of behaviour, physiology and adaptation. Philos Trans R Soc B 367:1665–1679. doi:10.1098/rstb.2012.0005
Ihalainen M, Pukkala T (2001) Modelling cowberry (Vaccinium vitis-idaea) and bilberry (Vaccinium myrtillus) yields from mineral soils and peatlands on the basis of visual field estimates. Silva Fenn 35:329–340
Intergovernmental Panel on Climate Change Climate change (2013): the physical science basis. Contribution of Working Group I to the fifth assessment report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge
Johnson DH (1980) The comparison of usage and availability measurements for evaluating resource preference. Ecology 61:65–71. doi:10.2307/1937156
Koper N, Manseau M (2009) Generalized estimating equations and generalized linear mixed-effects model for modelling resource selection. J Appl Ecol 46:590–599. doi:10.1111/j.1365-2664.2009.01642.x
Laberee K, Nelson TA, Stewart BP, McKay T, Stenhouse GB (2014) Oil and gas infrastructure and the spatial pattern of grizzly bear habitat selection in Alberta, Canada. Can Geogr 58:79–94. doi:10.1111/cag.12066
Larsen TA (2012) The potential influence of mountain pine beetle (Dendroctonus ponderosae) control harvesting on grizzly bear (Ursus arctos) food supply and habitat conditions in Alberta. M.Sc. thesis, Department of Biological Science, University of Alberta, Edmonton, Alberta, Canada
Lewis JS, Rachlow JL, Garton OE, Vierling LA (2007) Effects of habitat on GPS collar performance: using data screening to reduce location error. J Appl Ecol 44:663–671. doi:10.1111/j.1365-2664.2007.01286.x
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation—a review and prospectus. Can J Zool 68:619–640. doi:10.1139/z90-092
Lima SL, Valone TJ, Caraco T (1985) Foraging-efficiency predation-risk trade-off in the grey squirrel. Anim Behav 33:155–165. doi:10.1016/s0003-3472(85)80129-9
Lin DY, Wei LJ (1989) The robust inference for the cox proportional hazards model. J Am Statist Assoc 84:1074–1078
Littell RC, Milliken GA, Stroup WW, Wolfinger RD, Schabenberger O (2006) SAS for mixed models, 2nd edn. SAS Institute, Cary
Maloney SK, Moss G, Cartmell T, Mitchell D (2005) Alteration in diel activity patterns as a thermoregulatory strategy in black wildebeest (Connochaetes gnou). J Comp Physiol A 191:1055–1064. doi:10.1007/s00359-005-0030-4
Maloney SK, Fuller A, Mitchell D (2009) Climate change: is the dark Soay sheep endangered? Biol Lett 5:826–829. doi:10.1098/rsbl.2009.0424
Manly BFJ, McDonald LL, Thomas DL, McDonald TL, Erickson WP (2002) Resource selection by animals—statistical design and analysis for field studies, 2nd edn. Springer, Dordrecht
Marchand P, Garel M, Bourgoin G, Dubray D, Maillard D, Loison A (2014) Sex-specific adjustments in habitat selection contribute to buffer mouflon against summer conditions. Behav Ecol 26:472–482. doi:10.1093/beheco/aru212
Martin J, van Moorter B, Revilla E, Blanchard P, Dray S, Quenette P-Y, Allaine D, Swenson JE (2013) Reciprocal modulation of internal and external factors determines individual movements. J Anim Ecol 82:290–300. doi:10.1111/j.1365-2656.2012.02038.x
McCain CM, King SRB (2014) Body size and activity times mediate mammalian responses to climate change. Glob Change Biol 20:1760–1769. doi:10.1111/gcb.12499
McDermid GJ, Hall RJ, Sanchez-Azofeifa GA, Franklin SE, Stenhouse GB, Kobliuk T, LeDrew EF (2009) Remote sensing and forest inventory for wildlife habitat assessment. For Ecol Manage 257:2262–2269. doi:10.1016/j.foreco.2009.03.005
Mclellan ML, Mclellan B (2015) Effect of season and high ambient temperature on activity levels and patterns of grizzly bears (Ursus arctos). Plos One 10:e0117734. doi:10.1016/j.biocon.2011.05.005
McNab BK (1983) Energetics, body size, and the limits to endothermy. J Zool 199:1–29. doi:10.1111/j.1469-7998.1983.tb06114.x
Moe TF, Kindberg J, Jansson I, Swenson JE (2007) Importance of diel behaviour when studying habitat selection: examples from female Scandinavian brown bears (Ursus arctos). Can J Zool 85:518–525. doi:10.1139/Z07-034
Molnár PK, Derocher AE, Klanjscek T, Lewis MA (2011) Predicting climate change impacts on polar bear litter size. Nature Commun 2:186. doi:10.1038/ncomms1183
Mowat G, Heard DC, Schwartz CJ (2013) Predicting grizzly bear density in western North America. PlosOne 8:e82757. doi:10.1371/journal.pone.0082757
Munro RHM, Nielsen SE, Price MH, Stenhouse GB, Boyce MS (2006) Seasonal and diel patterns of grizzly bear diet and activity in west-central Alberta. J Mammal 87:1112–1121. doi:10.1644/05-MAMM-A-410R3.1
Nielsen SE, Boyce MS, Stenhouse GB (2004a) Grizzly bears and forestry. I. Selection of clearcuts by grizzly bears in west-central Alberta, Canada. For Ecol Manage 199:51–65. doi:10.1016/j.foreco.2004.04.014
Nielsen SE, Munro RHM, Bainbridge EL, Stenhouse GB, Boyce MS (2004b) Grizzly bears and forestry. II. Distribution of grizzly bear foods in clearcuts of west-central Alberta, Canada. For Ecol Manage 199:67–82. doi:10.1016/j.foreco.2004.04.015
Nielsen SE, Stenhouse GB, Boyce MS (2006) A habitat-based framework for grizzly bear conservation in Alberta. Biol Cons 130:217–229. doi:10.1016/j.biocon.2005.12.016
Nielsen SE, McDermid G, Stenhouse GB, Boyce MS (2010) Dynamic wildlife models: seasonal foods and mortality risk predict occupancy-abundance and habitat selection in grizzly bears. Biol Conserv 143:1623–1634. doi:10.1016/j.biocon.2010.04.007
Nijland W, Coops NC, Nielsen SE, Stenhouse G (2015) Integrating optical satellite data and airborne laser scanning in habitat classification for wildlife management. Int J Appl Earth Obs Geoinform 38:242–250. doi:10.1016/j.jag.2014.12.004
Ordiz A, Støen OG, Delibes M, Swenson JE (2011) Predators or prey? Spatio-temporal discrimination of human-derived risk by brown bears. Oecologia 166:59–67. doi:10.1007/s00442-011-1920-5
Owen-Smith N (1998) How high ambient temperature affects the daily activity and foraging time of a subtropical ungulate, the greater kudu (Tragelaphus strepsiceros). J Zool 246:183–192. doi:10.1111/j.1469-7998.1998.tb00147.x
Pan W (2001) Akaike’s information criterion in generalized estimating equations. Biometrics 57:120–125. doi:10.1111/j.0006-341X.2001.00120.x
Poppitt SD, Speakman JR, Racey PA (1994) Energetics of reproduction in the lesser hedgehog tenrec, Echinops telfairi (Martin). Physiol Zool 67:976–994
Pyke GH (1984) Optimal foraging theory—a critical review. Annu Rev Ecol Syst 15:523–575
Riek A, Geiser F (2013) Allometry of thermal variables in mammals: consequences of body size and phylogeny. Biol Rev Camb Philos Soc 88:564–572. doi:10.1111/brv.12016
Robbins CT, Fortin JK, Rode KD, Farley SD, Shipley LA, Felicetti LA (2007) Optimizing protein intake as a foraging strategy to maximize mass gain in an omnivore. Oikos 116:1675–4682. doi:10.1111/j.0030-1299.2007.16140.x
Rode KD, Robbins CT, Shipley LA (2001) Constraints on herbivory by grizzly bears. Oecologia 128:62–71. doi:10.1007/s004420100637
SAS Institute (2011) SAS/STAT® 9.3 User’s guide. Release 9.3. SAS Institute, Cary, NC
Schwartz CC, Miller SD, Haroldson MA in Feldhamer GA, Thompson BC, Chapman JA (2003) Grizzly bear. Wild mammals of North America: biology, management, and conservation, second edition. Johns Hopkins University Press. Baltimore. MD
Sikes RS, Gannon WL, the Animal Care and Use Committee oof the American Society of Mammalogists (2011) Guidelines of the American Society of Mammalogists for the use of wild mammals in research. J Mammal 92:235–253. doi:10.1644/10-mamm-f-355.1
Speakman JR, Król E (2010) Heat dissipation and hyperthermia risk as limiting factors in endotherm ecology. Integr Comp Biol 51:E130–E130
Stelzner JK, Hausfater G (1986) Posture, microclimate, and thermoregulation in yellow baboons. Primates 27:449–463
Stenhouse G, Boulanger J, Lee J, Graham K, Duval J, Cranston J (2005) Grizzly bear associations along the eastern slopes of Alberta. Ursus 16:31–40. doi:10.2192/1537-6176(2005)016[0031:gbaate]2.0.co;2
Steyaert S, Kindberg J, Swenson JE, Zedrosser A (2013) Male reproductive strategy explains spatiotemporal segregation in brown bears. J Anim Ecol 82:836–845. doi:10.1111/1365-2656.12055
van Beest FM, Van Moorter B, Milner JM (2012) Temperature-mediated habitat use and selection by a heat-sensitive northern ungulate. Anim Behav 84:723–735. doi:10.1016/j.anbehav.2012.06.032
Waller JS, Mace RD (1997) Grizzly bear habitat selection in the Swan Mountains, Montana. J Wildl Manage 61:1032–1039. doi:10.2307/3802100
Walther GR, Post E, Convey P, Menzel A, Parmesan C, Beebee TJC, Fromentin JM, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395. doi:10.1038/416389a
Ware JV, Rode KD, Pagano AM, Bromaghin J, Robbins CT, Erlenbach J, Jensen S, Cutting A, Nicassio-Hiskey N, Hash A, Owen M, Jansen HT (2015) Validation of mercury tip-switch and accelerometer activity sensors for identifying resting and active behavior in bears. Ursus 26:86–96. doi:10.2192/URSUS-D-14-00031.1
Weissenböck NM, Arnold W, Ruf T (2011) Taking the heat: thermoregulation in Asian elephants under different climatic conditions. J Comp Physiol B 182:311–319. doi:10.1007/s00360-011-0609-8
Williams SE, Shoo LP, Isaac JL, Hoffmann AA, Langham G (2008) Towards an integrated framework for assessing the vulnerability of species to climate change. PLoS Biol 6:2621–2626. doi:10.1371/journal.pbio.0060325
Wilmshurst JF, Fryxell JM, Hudson RJ (1995) Forage quality and patch choice by wapiti (Cervus elaphus). Behav Ecol 6:209–217. doi:10.1093/beheco/6.2.209
Zedrosser A, Steyaert SMJC, Gossow H, Swenson JE (2011) Brown bear conservation and the ghost of persecution past. Biol Conserv 144:2163–2170. doi:10.1016/j.biocon.2011.05.005
Acknowledgments
We thank the Alberta Ecotrust, the Y2Y Sarah Baker Memorial Fund, the Alberta Conservation Association, the Natural Sciences and Engineering Research Council of Canada, and the FRI Research Grizzly Bear Program partners for providing research funds. L’Université Laval, the Centre d’Études Nordiques, and the Natural Sciences and Engineering Research Council of Canada provided scholarships and funding for conferences to K. E. P. We thank J. Duval and D. Weins for geographic information system support; A.-S. Julien for help with statistical analyses; R. Théorêt-Gosselin, R. Strong, A. Auger, E. Rogers, C. Curle, T. Larsen, P. Stenhouse, and A. Stenhouse for collecting field data; two anonymous reviewers for commenting on this manuscript; and J. Saunders and S. Wotton at Peregrine Helicopters.
Author contribution statement
K. E. P. and G. B. S. originally formulated the idea. K. E. P. developed the methodology under the supervision of S. D. C. and G. B. S. K. E. P. conducted fieldwork and collaborated with E. C. to perform the statistical analyses. K. E. P. and E. C. wrote the manuscript while G. B. S. and S. D. C. provided advice.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Andreas Zedrosser.
Electronic supplementary material
Below is the link to the Electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pigeon, K.E., Cardinal, E., Stenhouse, G.B. et al. Staying cool in a changing landscape: the influence of maximum daily ambient temperature on grizzly bear habitat selection. Oecologia 181, 1101–1116 (2016). https://doi.org/10.1007/s00442-016-3630-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-016-3630-5