Abstract
FMRFamide-related peptides (FaRPs) are a class of neuropeptides that participate in a variety of physiological processes in invertebrates. They occur in nerves of stomatogastric ganglia and enteroendocrine cells of the insect digestive tract, where they may control muscle functions. However, their direct involvement in muscle function has never been shown in situ. We studied the relationship between FaRPs and midgut muscle during larval–pupal transition of the mosquito Aedes aegypti. In late L4, FaRP-positive neuronal extensions attach to the bundles of the external circular muscle layer, and muscle stem cells start to undergo mitosis in the internal circular layer. Thereafter, the external muscle layer degenerates, disappearing during early pupal development, and is completely absent in the adult mosquito. Our results indicate that FaRP-based neural signals are involved in the reorganization of the muscle fibers of the mosquito midgut during the larval–pupal transition. In addition to confirming FaRP involvement in muscle function, we show that the mosquito midgut muscles are largely innervated, and that circular and longitudinal muscle have specific neuron bodies associated with them.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
FMRFamide is a tetrapeptide that gives the name to the extended family of FMRFamide-related peptides (FaRPs). It was first isolated from the mollusk Macrocallista nimbosa and described as a heartbeat-accelerating neuropeptide (Price and Greenberg, 1977a, b). Later, peptides sharing similar sequences were found in other mollusks and members of other invertebrate groups, such as insects (Arthropoda) (Žitňan et al. 1993; Nassel et al. 1994; Price and Greenberg 2007; Elphick and Mirabeau 2014). All peptides sharing the RFamide C-terminal sequences are currently described as FaRPs. This neuropeptide family includes products of different genes (Strand et al. 2016; Hao et al. 2020), with the term FaRPs implying the RFamide signature and analogies to FMRFamides rather than genetic family. Neuropeptides F (NPF), short neuropeptides F (sNPFs), FMRFamides, myosupressins, and sulfakinins are members of the FaRPs group (Nassel and Winther 2010; Nagata 2016; Strand et al. 2016). They exert a wide range of basic physiological roles in invertebrates, such as regulating feeding, development, diapause, and reproduction (Orchard et al. 2001; López-Vera et al. 2008; Sedra and Lange 2016; Hao et al. 2020).
FaRPs are important neurotransmitter/neuromodulator peptides of invertebrates (Cottrell 1993; Predel et al. 2010; Taghert and Nitabach 2012; Tarr et al. 2019) and have been detected in the gastrointestinal tract of a variety of insects (Brown et al. 1986; Nichols et al. 1999; Onken et al. 2004; Haselton et al. 2008; Godoy et al. 2015; Souza et al. 2016; Oliveira et al. 2019), including the mosquitoes Aedes (Stegomyia) aegypti (Linnaeus 1762) (Brown et al. 1986; Stanek et al. 2002; Fernandes et al. 2014) and Toxorhynchites theobaldi (Godoy et al. 2015). In adults of these species, FaRPs are found in axons and in enteroendocrine cells of the anterior and posterior midgut, respectively (Brown et al. 1986; Stanek et al. 2002; Fernandes et al. 2014; Godoy et al. 2015). However, the distribution of these neuropeptides in the midgut of A. aegypti L4 larvae has not been reported.
FaRP-positive enteroendocrine cells have been reported to occur in the digestive tracts of insects, using immunofluorescence with anti-FMRFamide antibodies (Brown et al. 1986; Souza et al. 2016; Oliveira et al. 2019). However, the roles FaRPs play in these cells are unknown. It has been suggested that enteroendocrine cells may monitor the nutrient content of the midgut lumen or tension of the muscle wall, or both, thereby acting locally by regulating the production of digestive enzymes and contraction of muscle cells (Nichols et al. 1999). According to functional assays, FaRPs may also change the rates of rhythmic longitudinal and peristaltic waves of contractions in insect digestive tract (Kingan et al. 1996; Krajniak 2013).
Various types of neuropeptides, including FaRPs, regulate key biological processes involved in growth, reproduction, metabolism, and development of insects (Nassel and Winther 2010; Strand et al. 2016; Nassel and Zandawala 2019). Thus, these molecules have been targeted for use as a new generation of potential vector and insect pest control agents (Fónagy 2007; Scherkenbeck and Zdobinsky 2009; Bureau 2015). A. aegypti is a major vector of arboviruses, such as dengue, Zika fever, and chikungunya, that affect populations of countries worldwide (Weetman et al. 2018). In this context, the discovery of essential functions played by FaRP neuropeptides during mosquito metamorphosis could translate into the use of these molecules as insecticides, contributing to control strategies that focus on the mosquito immature stages.
As holometabolous insects, the mosquito life cycle includes the immature larval and pupal stages that lead to the adult (Peters et al. 2014). Mosquito larvae are aquatic and detritivorous, whereas adults are terrestrial and nectarivorous, while females feed on blood for egg production. At the end of the last larval stage, internal organs, such as the midgut, undergo deep structural changes and begin to be reshaped into adult organs, adapting the mosquito to its new ecological niche. These changes are regulated by several hormones, such as 20-hydroxyecdysone (20E) and juvenile hormone (JH), that coordinate cell-specific expression of nuclear receptors and transcription factors in larval tissues (Nishiura et al. 2003; Wu et al. 2006; Parthasarathy and Palli 2007). During pupation, the regenerative cells of the midgut epithelium (epithelial stem cells) start differentiation to replace the mature cells (enterocytes and enteroendocrine cells). The mature cells degenerate and detach towards the lumen, as the new digestive epithelium is formed (Nishiura et al. 2003; Wu et al. 2006; Fernandes et al. 2014). At the same time, the midgut muscles also undergo structural changes (Bernick et al. 2008), but the mechanisms involved in its remodeling are unknown.
The A. aegypti larval midgut is surrounded by circular and longitudinal muscles. Circular muscles form interconnected rings that intersect with the longitudinal muscles, wrapping the midgut in a supportive mesh (Bernick et al. 2007). During pupation, the larval midgut muscles show signs of degeneration and atrophy losing myofibril content but retaining intact cell membranes (Bernick et al. 2007). However, it is not clear whether muscle cells are generated de novo during pupal muscle remodeling, as is the case for midgut epithelial cells.
In situ labeling has not been used to investigate the role of FaRPs in insect muscle function. Previously, liquid chromatography and radioimmunoassay were used to investigate the action of FaRPs in muscles of migratory locust (Locusta migratoria), and in vitro assays were used to detect muscle contractions in the fruit fly (Drosophila melanogaster) (Duttlinger et al. 2002; Hill and Orchard 2004; Krajniak 2013) and in perfused midgut preparations of A. aegypti (Onken et al. 2004). In this study, we used in situ labeling to show that neuropeptides are directly involved in muscle function. We provide a new interpretation about the neuromuscular connections and enteric nervous system of mosquitoes.
Material and methods
Screening of the FaRP genes in A. aegypti
As previously described, FaRPs include five distinct subgroups of molecules, namely NPF, sNPF, FMRFamides, sulfakinins, and myosupressins. FMRFamide antibodies react with members of all five subgroups (detecting the RFamide epitope). For this reason, it was not possible to identify the subgroups using FaRP immunolabeling. Identification of A. aegypti FaRP genes was based on neuropeptide gene lists generated previously for A. aegypti and Drosophila spp. (Strand et al. 2016; Nassel and Zandawala 2019). The genes corresponding to NPF, sNPF, extended FMRFamides, myosupressin, and sulfakinin; and their predicted prepropeptide products were verified on NCBI (https://www.ncbi.nlm.nih.gov/), UniProtKB / Swiss—Prot (https://uniprot.org ), and VectorBase (https://vectorbase.org/vectorbase/app) platforms to generate an updated list of A. aegypti FaRP genes. The mature neuropeptide sequences for each FaRP gene are listed according to Stanek et al. (2002) and Predel et al. (2010).
Mosquitoes
Larvae of A. aegypti (PPCampos strain, Campos dos Goytacazes, RJ) were reared in the insectarium at the Departamento de Biologia Geral at the Universidade Federal de Viçosa and were collected at the following stages: L3 (randomly chosen), early (< 24 h) and late L4 (> 72 h), and early pupae (> 2 h). The insects were fed with turtle food (Nutral Ouro, Campinas, São Paulo, Brazil) ad libitum and kept in an incubator at 26 ± 3 °C and a 12-h photoperiod.
Immunofluorescence
Midguts of L3, early and late L4, and early pupae (15, 15, 25, and 15 individuals, respectively) were dissected in phosphate-buffered saline or PBS (0.1 M NaCl, 20 mM KH2PO4, and 20 mM Na2HPO4; pH 7.6) and its food content (and the surrounding peritrophic matrix) were removed with the aid of tweezers. They were fixed with Zamboni fixative solution (4% paraformaldehyde and 0.4% picric acid in PBS), washed in PBST (0.1% Tween 20 in PBS) three times for 30 min each, and incubated for 24 h at 4 °C with primary antibody anti-FMRFamide (Rabbit polyclonal IgG, Genetex, USA) (1:400 v:v in PBS) in 1% PBST. After the incubation with the primary antibody, the samples were washed three times in PBS and incubated with FITC-conjugated secondary antibody (rabbit polyclonal IgG, Sigma-Aldrich, USA) (1:500 v:v in PBS) for 24 h at 4 °C, followed by three 10-min washes in PBS. After washing to remove excess secondary antibodies, the samples were stained with Phalloidin-Alexa 546 (Thermo Fisher Scientific, Eugene, USA) (1:100 v:v in PBS) for 60 min to identify muscle tissue; and TO-PRO-3 Iodide (Thermo Fisher Scientific, Eugene, USA) (1:1000 v:v in PBS) for 30 min to stain cell nuclei (DNA). Then, all samples were washed in PBS and mounted with mowiol solution (Sigma-Aldrich, USA) and analyzed under a laser scanning confocal microscope (LSCM) 510 META at the Núcleo de Microscopia e Microanálise at the Universidade Federal de Viçosa (NMM, UFV). The nuclei of some samples with triple labeling were artificially colored in blue to facilitate the observation of the structures. The Z-stack tool of the LSCM was used to obtain 3D images and videos to allow a better visualization of the samples. It is important to note that all the samples were incubated with the nuclear marker TOPRO-3; however, the labeling was not detected in some of them due technical limitations of the reagent.
Another set of midgut samples from 15 pre-pupal L4 and 10 early pupae were stained by means of a similar procedure of fixation, incubation, and mounting, but the primary antibody used was anti-phospho-histone H3 (S10) (rabbit polyclonal IgG, Cell Signaling, USA) (1:100 v:v in PBS).
Negative controls were prepared using the midgut of the abovementioned stages (n = 5 of each) submitted to the same process as the other samples; however, they were not incubated with the primary antibodies. Positive controls for FMRF were obtained using adult T. theobaldi mosquitoes, whose labeling was detected in a previous study (Godoy et al. 2015). Positive controls for phosphohistone H3 were obtained using the midguts of A. aegypti L3 larvae, which show mitotic divisions of the regenerative cells of the digestive epithelium.
Scanning electron microscopy
Complete midguts of L2, L3, early and late L4 larvae, and early pupae (early-1, 0–1 h post-pupation; and early-2, 3–4 h post-pupation) of A. aegypti (25 samples of each) were dissected in PBS, pH 7.6 (0.1 M NaCl, 20 mM KH2PO4, and 20 mM Na2HPO4), fixed in 2.5% glutaraldehyde solution (sucrose/cacodylate buffer 0.1 M, pH 7.2) for 24 h, post-fixed with osmium tetroxide (1% v:v in distillated water) for 30 min and washed in PBS. After washing, the samples were dehydrated in an ascending ethanol series, critically point dried using CO2, and sputter coated with gold. Samples were analyzed and photographed under the scanning electron microscope JEOL JSM 5600 at the Instituto René Rachou, Fiocruz, MG.
Results
A. aegypti FaRP genes
Table S1 lists the NPF, sNPF, extended FMRFamide, sulfakinin, and myossupressin genes detected in the A. aegypti genome, with each subgroup deriving from a single gene. The prepropeptide sequences and the predicted peptide products of the five genes are also listed. The mature NPF peptide sequence was detected by radioimmunoassay (Stanek et al. 2002), whereas for the other FaRP subgroups, the peptide products were sequenced by peptidomics (Predel et al. 2010). As the FaRP peptides labeled by the FMRFamide antibody have an RFamide C-terminal end, only peptide sequences with this tag are listed.
Muscle organization of the larval midgut
The organization of the circular and longitudinal muscles of midgut of A. aegypti larva is shown in the Fig. 1a, b. Two circular muscles can be identified: internal circular muscles (ICMs) and external circular muscles (ECMs). The first is more internal and thinner in relation to the ECM layer and forms a spiral that extends throughout the midgut, with each spiral ring sharing muscular fibers with the neighboring rings. This fiber sharing is seen at bifurcations that occur at specific points, which are close to the passage of two prominent and thicker longitudinal muscles (Fig. 1a, d, and Movie S1). The second type of circular layer is more external (ECMs) and has incomplete bundles, thick, and attached to the longitudinal muscles, forming cruciform regions (Fig. 1a, b and Movie S1). The cruciform regions are present only in two lateral portions of the midgut, ending exactly at the end points of the incomplete bands in the dorsoventral axis. The cruciform regions were not detected in the posterior midgut (Fig. 1e).
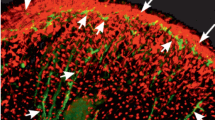
The larval midgut of Aedes aegypti. a The muscle organization of early L4 midgut. Longitudinal muscle bundles (LM) and two types of circular muscles, the external muscles (ECMs) and internal circular muscles (ICMs), are present in the anterior midgut. The ECMs (thin arrows) are incomplete and fuse with the LMs, originating cruciform cells (*) in two lateral portions (brackets) of the midgut. The ICM bifurcates (full arrow) at a common anteroposterior linear point of the midgut that is close to two prominent and thicker LMs (pLMs). Red: midgut muscles. b Cruciform (*) regions (bracket) formed by the fusion of the ECM (thin arrows) with LM bundles in a lateral portion of the midgut of early L4 larvae. c FaRP immunoreactivity (green) in numerous small points (thin arrow) throughout the cardia (Ca), gastric ceca (GC), and anterior midgut (AMG). Red: midgut muscles. d, e Detail of the bifurcations (full arrow) of the ICM close to the pLM passage, in the AMG of L3 larva. e, f FaRP-positive enteroendocrine cells (En, green) in the posterior midgut (PMG) of early L4 (e) and L3 larva (f). DC: digestive cell; red: midgut muscles; blue: cell nuclei; a, c, e, and f: confocal microscopy; b and d: scanning electron microscopy
In all larval stages, excluding the late L4, FaRPs are seen as small, rounded points located close to the muscles throughout the midgut (Figs. 1c and S1a), or inside enteroendocrine cells dispersed in the posterior midgut (Fig. 1e, f).
Mitotic nuclei were detected in the digestive epithelium of all larval instars, excluding the late stage of L4 (Fig. S1b). No muscle mitotic nuclei were observed.
Late L4
Using scanning electron microscopy, the general muscle morphology and organization were similar to those of earlier stages (Fig. 2a). However, FaRP-positive enteroendocrine cells were no longer found. Nevertheless, FaRP-positive structures were observed in the cruciform regions associated with the ECM layer. Of 25 late L4 midguts, such immunoreactive structures were observed in 21 midguts and were specifically located in the mid-position of the bundle fragment of the ECM, between their intersection points (cruciform points) with the longitudinal fibers (Figs. 2b, c and S1c and Movie S1). Most of these FaRP-positive structures corresponded to individually well-defined ring-shaped structures (Figs. 1d and 2b–d). In some of them, the site where the FaRP-positive rings surrounded the ECM fibers had continuous and intact actin filaments (Fig. 3a, b). At other points, gaps (dark non-stained regions) were seen in actin filaments close to the sites surrounded by FaRP-positive rings (Fig. 3c, e). Although the absence of actin was clear in some surrounding sites, scanning electron microscopy of the corresponding regions did not show physical breaks in the external structure of the ECM fibers, but sags were detected at some points (Fig. 3d, f).
The midgut of A. aegypti late L4 larva. a The late L4 midgut muscles have incomplete bundles of external circular muscles (ECM) in two lateral portions (brackets) of the anterior midgut (AMG) and internal circular bundles undergoing bifurcations (full arrow) close to the passage of two prominent and thicker longitudinal muscles (pLM), similarly to the previous instars of larva, and early L4. LM: longitudinal muscles. b Presence of FaRP-immunoreactivity (green) only in the lateral portion (bracket) that corresponds with the localization of the external circular muscles fused with longitudinal ones (cruciform regions, corresponding rectangles of a and b). c Detail of the FaRP-immunoreactivity (green) in specific ring-shaped structures (thin arrows) associated with the ECM. ICM internal circular muscles. d Detail of the ECM in the region that corresponds with the FaRP-positive ring-shaped structures showed in c. The ring-shaped structures (thin arrows) are easily visualized by scanning electron microscopy. Inset: 3D view of the FaRP-positive ring-shaped structures detected in association with the ECM at the place exactly between two cruciform points (*). Red: midgut muscles
FaRP-positive structures in confocal images (a, c, e, g, and h) and their corresponding regions using scanning electron microscopy (SEM) (b, d, f, i, and j) in the midgut of late L4 larvae of Aedes aegypti. a Some FaRP-positive ring-shaped structures (green) surround sites of the external circular muscle (ECM) fibers (thin arrow) that present intact actin filaments. They are seen surrounding the fibers in the mid-position between two cruciform points (*). b Structure corresponding to the ring-shaped connection seen in a. The ring-shaped structures are physically evident and have continuity with filamentous extensions (black arrow). c, d Other points surrounded by the ring-shaped structures have gaps in actin filaments (c), but the corresponding regions do not reveal physical breaks in the muscle fibers. However, sags were detected in the ECM sites surrounded by the ring-shaped structures (thin white arrow), which presented filamentous continuity (thin black arrow). e, f The gaps in actin filaments (thin white arrow) are clearly visualized in e, and the corresponding region also do not reveal physical breaks in the fiber structure in the surrounded sites, in f. g, h The end points of the incomplete bundles of ECM have thin and branched FaRP-positive structures. g The FaRP-positive thin branches (thin white arrow) are disposed in a linear conformation along the anteroposterior axis of the anterior midgut. Inset: Detail of the position of the thin branches. h Detail of the queued disposition of the FaRP-positive branches in the end points (thin white arrow) of the incomplete fibers of ECM. The internal circular muscles (ICMs) are seen below the grid of cruciform points (*). i, j The end points of the ECM seen using SEM in a wide field (i) and in detail (j). The numerous branches seen in h using confocal microscopy are not completely evident. However, some of these branches are detected (black arrow)
In addition to the ring-shaped structures, thin and dense FaRP-positive ramifications occur repeatedly but are restricted to a single line along the anteroposterior extension of the midgut (except in the posterior region). These thin and dense ramifications are located exactly at the end points of the cruciform (incomplete) circular fibers (Fig. 3g, h), but only a few of the branches were detected using scanning electron microscopy (Fig. 3i, j).
Mitotic nuclei were detected specifically in the ICM tissues (Fig. 4a, b). Some of these nuclei were seen in anaphase (Fig. 4b). In contrast, no mitosis was observed in longitudinal muscle tissues (Fig. 4a, b).
Mitotic nuclei are detected in the muscle tissue of the internal muscle layers (ICMs) in the late L4 larva (a, b) and early pupa (c, d) of Aedes aegypti. No mitotic nucleus is detected in the longitudinal muscle (LM) layers. a, b Muscle stem cells (MSCs) of the ICM of late L4 undergoing mitosis are showed in a wide field (a) and in detail (b). A mitotic nucleus (green) is detected in anaphase in b. Red: midgut muscles. c, d MSCs of the ICM of early pupa undergoing mitosis are showed in a wide field (c) and in detail (d). Red: midgut muscles; green: mitotic nuclei; blue: cell nuclei; pLM: prominent longitudinal muscles; ICM: internal circular muscles
Early pupa
In the early-1 pupa (0 to 1 h after pupation), the cruciform longitudinal and circular muscle bundles, similar to those of the larval stage, were also detected only in two lateral portions of the anterior midgut; they were larger than the corresponding structures of larval stage, with the cruciform intersection points close to each other (Fig. 5a–d). FaRP signal was no longer present in the cruciform circular muscles, but gaps (dark non-stained regions) were detected in actin filaments in the regions where FaRP-positive neurites had been attached in the late L4 (Fig. 5b, inset). The ICM layers had the same organization, running above the grid of cruciform cells formed by the fusion of ECM and the longitudinal fibers (Fig. 5a, e). Additionally, the ICM layers were visualized in the posterior midgut, maintaining the same configuration as that of larval stages (Fig. 5f).
The midgut of the early pupa of Aedes aegypti. The grids formed by cruciform cells (bracket) in the anterior midgut are seen in wide field (a, c) and in detail (b, d) using confocal (a, b) and scanning electron microscopy (c, d). The external circular muscles (ECMs) and the longitudinal muscles (LMs) of the grids are larger and with the cruciform regions (*) closer to each other than in the larval stages (b inset and d inset). e The internal circular muscle (ICM) layers run above the grid of cruciform cells (*) in the anterior midgut. f The ICM remain in the same position and configuration of the larval stages in the posterior midgut. Red: midgut muscles; blue: cell nuclei, green: residues of FaRP immunoreactivity; A → P: anteroposterior axis; GC: gastric cecum; pLM: prominent longitudinal muscle
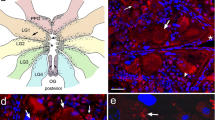
In some early-1 pupal midguts, FaRP-positive small rounded points were seen in rows (Fig. S1e), and FaRP-positive neuron bodies were detected in the anterior and anteroposterior transition region of the midgut (Fig. 6a–c). Among the neuron bodies, it was possible to discern three distinct types, depending on the orientation in the midgut and external/internal position in relation to the muscles: (a) one or two external neurons (ENs) (with neuron bodies not attached to the midgut muscles) with longitudinal orientation; and (b) internal circular neurons (ICNs), with the neuron bodies arranged as longitudinal rows and closely attached to the ICM bundles. Each muscle bundle of the spiral muscle ring was associated with at least two of these neuron bodies. Their position was close to the passage of the two prominent and thick longitudinal muscles previously described, with one of each pair located either in the left or right of muscle bifurcations; c) internal longitudinal neurons (ILNs), with neuron bodies attached to the longitudinal muscles. Their positions were not as well defined as the ICNs (Fig. 6a–c). All types of neurons presented extensions clearly visualized with FaRP labeling (Fig. 6a–c).
Distribution of three types of neurons in the midgut of immature stages of Aedes aegypti. a–c FaRP immunoreactivity (green) in neuron bodies and their ramifications in the midgut of an early pupa. A wide field view is showed in a. Three neuron types can be distinguished, depending on their orientation in the midgut and external/internal position in relation to the muscles: the external neurons (ENs), the internal circular neurons (ICNs), and the internal longitudinal neurons (ILNs). The thin white arrows show the extensions of the ENs in a, ICNs in b, and ILNs in c. Red: cell nuclei. d–f The midgut neurons in the larva of second instar using scanning electron microscopy. d The localization of the three neuron types in a wide field. e, f Details of the ICN (e) and ILN (f). g, h Redemonstration of the FaRP-positive ring-shaped (black full arrow) and densely branched (white full arrow) structures (g) to generate a scheme model of distribution of the internal neurons and FaRP-positive structures in the anterior midgut (AMG) (h). pLM: prominent longitudinal muscle; LM: longitudinal muscle; ECM: external circular muscle; ICM: internal circular muscle; DE: digestive epithelium; Fd: food in the midgut lumen; RC: regenerative cell; n: digestive cell nucleus
The location of the internal neuron bodies in the early pupa was difficult to visualize using scanning electron microscopy because they exhibited little difference in fiber dimensions. However, they were detected in some samples of early L4 (Fig. S1g, h) and were easily detected in L2 (second instar) larvae (Fig. 6d–f). In L2, the internal neuron bodies gave a swollen appearance to the ICM and to the longitudinal muscle fibers. The specific locations of the swollen structures corresponded with FaRP immunolabeling of the neuron bodies in the early-1 pupae (Fig. 6a, d, e).
All FaRP-positive structures seen in late L4 and early pupae, including the ring-shaped and thin and dense branches (Fig. 6g), were combined to generate a model of their configuration in immature stages. The position of the neuron bodies attached to the ICM, close to their bifurcation point, and of the irregularly distributed neuron bodies of the LM, are shown (Fig. 6h).
In early-2 pupae (approximately 3–4 h after pupation), ECM bundles (cruciform), previously detected in the early (Fig. 7a, b and Movie S2) and late L4 (Fig. 7c, d) by labeling of actin filaments, were absent in the anterior positions of the midgut or seen as residual structures (Fig. 7e, f and Movie S3). In addition, comparing the midgut muscle structure among early larva, late larva and early pupa, it was seen that all stages present actin gaps in ECM mid-positions between the cruciform points (Fig. 7b, d, f).
Scanning electron microscopy of the entire midgut of early L4 (a), late L4 (c) and early-1 pupa (e) and confocal images of their respective stages showing the actin filaments (red) of the muscles in the anterior midgut (AMG) (b, d, f). The midgut of early L4 (a) decreases in thickness in the late L4 (c). From late L4 to early pupa (e), the gastric ceca undergo a drastic reduction. b, d, f The external circular muscle layer (ECM) of the AMG presents gaps in actin filaments (thin white arrow) since L4 stage (b), which is maintained in the late L4 (d), and in some portions of the early pupal midgut (f). In other portions of the early pupal midgut, the ECM is not detected or detected as a degenerative structure (full white arrow). Ca: cardia; PMG: posterior midgut; MT: Malpighian tubule; pLM: prominent longitudinal muscle; ICM: internal circular muscle; LM: longitudinal muscle
Finally, in both early-1 and early-2 pupae, muscle stem cells of the ICM bundles, which corresponded to the previous larval ICM, revealed continuous mitosis across the length of the midgut (Fig. 4c, d and Movie S4). The negative controls of the mitosis marker (PH3 immunolabeling) and FaRP detection (FaRP immunolabeling) were used to generate Fig. 7 b, d, and f, respectively.
Complementary findings and general results
To determine whether the adult midgut muscles have ECM bundles typical of the larval stages, adult mosquitoes were processed and whole-mounted to detect actin filaments. We found ICM-type muscles which bifurcate in a pattern similar to that of the larval instars and longitudinal muscle layers (Fig. S1i and Movie S5), which do not have ECM bundles.
The scanning electron microscopic images of Fig. 7 (Fig. 7a, c, e) and stereomicroscopic images of Fig. 8 show the morphology of the entire midgut of the early and late L4 and early pupae. Early L4 has a robust midgut and prominent gastric cecae, while the food could be clearly visualized in the lumen. In the late phase L4 larvae, the midgut tube and gastric ceca decreased in diameter and the food was absent in the lumen. In the early pupae, the gastric cecae were quite reduced, with only residual structures visible, and the midgut was wider in diameter compared with that of the late L4.
Summary scheme of the midgut muscle remodeling from early L4 larva (a–a”) to late L4 larva (b–b”), and early pupa (c–c”) in A. aegypti, showing the presence of FaRP-positive structures (a’, b’, c’) and muscle stem cells dynamics (a”, b”, c”). The mosquito midgut undergoes deep changes in morphology during the larval–pupal transition. (a) The early L4 shows food (Fd) ever present in the midgut lumen. (a’) The FaRP-positive structures in this stage are small, rounded points throughout the midgut (not shown) and enteroendocrine cells in the posterior midgut (PMG). (a”) The external circular muscles (ECMs) are present and have gaps in actin filaments in specific positions. It is not possible to state that quiescent muscle stem cells are present close to the internal circular layers, but their presence can be considered. (b) In the late L4, food is no longer found. (b’) FaRP-positive ring-shaped structures are seen associated to the ECM sites that have the gaps in actin filaments (maintained from the early L4), and FaRP-positive dense branches are seen in the end points of the ECM. (b”) Proliferative muscle stem cells are detected in the internal circular muscles (ICMs) throughout the midgut. (c–c”) In the early pupa, the FaRP-positive ring-shaped and branched structures are not detected (c’), the muscle stem cells keep proliferating, and the ECM bundles show signals of degeneration (c”). Bars 500 µm
The principal findings about muscle remodeling, with FaRP-positive structures and muscle stem cell dynamics, are presented in schematic form in Fig. 8.
Discussion
FaRP candidates expressed in the A. aegypti midgut
All the FaRP subgroups are found in the A. aegypti genome, as is the case for other insects such as Drosophila spp. and the mosquito Anopheles gambiae (Sedra and Lange 2016; Strand et al. 2016; Nassel and Zandawala 2019). Their expression is tissue-specific, with some subgroups not being detected in midguts. Only FaRPs of the sNPF subgroup were detected in the midgut peptidome of Drosophila spp. L3 larvae and adults (sequence SPSLRLRFamide) (Reiher et al. 2011), whereas FaRPs of the NPF and sNPF subgroups were identified in midgut transcriptomics (Nassel and Zandawala 2019; Hung et al. 2020). Considering mosquitoes, only NPF was detected in the midgut transcriptome of Anopheles gambiae adults (Warr et al. 2007) and only sNPF was detected in the midgut neuropeptidome of A. aegypti adults (sequences KAVRSPSLRLRFa, SPSLRLRFa, and APQLRLRFa) (Predel et al. 2010). Taking together results of transcriptomics, peptidomics, radioimmunoassays, and immunofluorescence experiments, the FaRP products of the NPF and sNPF subgroups were detected in mosquito midguts (Brown et al. 1994; Stanek et al. 2002; Stracker et al. 2002; Warr et al. 2007; Predel et al. 2010). As mentioned by Reiher et al. 2011, peptides not predicted from the genome possibly escape from peptidomics analysis, which may explain the lack of detection of NPF by this technique. Therefore, presently the presence of only NPF and sNPF has been confirmed in insect midguts.
Studies that detected the expression of only the FaRPs NPF and sNPF in insect midguts did not assess the larval–pupal transition. This period involves particular changes in gene expression of most organs and was not analyzed for neuropeptide expression. Thus, we cannot affirm that the other FaRPs subgroups (extended FMRFamides, myossupressins, and sulfakinins) are never synthesized in midguts.
NPF and sNPF, as well as other FaRP subgroups, have pleiotropic effects, including myotropic functions (Onken et al. 2004; Roller et al. 2016; Zels et al. 2015; Sedra and Lange 2016; Fadda et al. 2019). In the present study, we demonstrated the direct involvement of FaRPs in the midgut musculature development during the larval–pupal transition. The implications of these findings are discussed below.
Larval midgut muscle morphology and FaRP-positive structures
The midgut muscles have the same structural organization in all larval stages. The ICMs consist of rings of circular muscles that connect to each other through bifurcations. These bifurcations arise at specific points on the same longitudinal line and form a spiral structure throughout the midgut. The longitudinal muscle layer, in turn, is more external and comprises numerous linear bundles that extend along the anteroposterior axis. As first described by Bernick et al. (2007), two lateral regions of the anterior midgut of A. aegypti larvae have an additional layer of circular muscles, which morphologically differ from the ICM layer. It differs because of the discontinuous (incomplete bundles) are attached to the longitudinal muscle bundles at their intersections. The attachment point of these intersections is composed of “cruciform cells,” muscle cells that possess myofibrils running at 90º angles to one another (cells with longitudinal and circular myofibril orientation) (Bernick et al. 2007).Together, these muscle elements form a typical “squared grid” configuration, easily visualized with scanning electron microscopy. The muscles of the “squared grid” differ from other muscle regions in some aspects, such as the presence of carbonic anhydrase immunoreactivity (Seron et al. 2004) that is absent in the posterior midgut (Bernick et al. 2007).
The FaRP-positive small rounded points seen on the surface of the larval midgut epithelium (except late L4) correspond to axon neurosecretory granules. These have also been shown in mosquito midgut axons by other studies (Brown et al. 1986; Brown and Lea 1988; Moffett and Moffett 2014; Godoy et al. 2015). The other FaRP-positive structures, the enteroendocrine cells, are typical of the posterior midgut of adult mosquitoes and other insects (Veenstra et al. 1995; Stanek et al. 2002; Micchelli et al. 2011; Beehler-Evans and Micchelli 2015; Godoy et al. 2015). The presence of these cells restricted to the posterior midgut in the A. aegypti larvae, as occurs in the adults, reveals a structural correspondence between the larval and adult midgut.
Mitosis was detected in regenerative cells of the digestive epithelium, but not in the midgut muscles of the L3 or L4 larva. The absence of mitosis in the midgut muscle bundles indicates no muscle growth or regeneration during the development of the mosquito larvae.
FaRP immunoreactivity during the larval–pupal transition
The midgut muscle structure of late L4 larvae did not show drastic differences from other larval stages except for the pattern of FaRP immunoreactivity. As late L4 is the stage close to pupation, these changes are probably related to the preparation for metamorphosis. Enteroendocrine cells were not detected in the midgut, and FaRP immunoreactivity was seen in two structures associated with the additional circular muscle layer of the squared grid regions: single ring-shaped structures, and thin and dense ramifications. Both FaRP-positive structures have the typical morphology of neurites, which comprise the neuronal extensions that carry and release neuropeptides (axons or dendrites) (Dowling 1991; Schinkmann and Li 1992; Wikgren and Fagerholm 1993; Nassel et al. 1994; Fónagy 2007; Elphick and Mirabeau 2014; Tuthill and Wilson 2016a, b; McLachlan et al. 2018; Xu et al. 2018).
The presence of ring-shaped neurites was confirmed and seen to be continuous with filamentous extensions by scanning electron microscopy (SEM), but the dense ramifications were not evident. Possibly, the neurites corresponding to the dense ramifications, found at the end of the incomplete bundles, branches toward the midgut epithelium, being hidden by its basal lamina.
Although FaRP-positive neurites were observed only in the late L4 midgut in our study, their structures are present in all larval stages, indicating that these neuronal connections are permanent. However, it is unknown if they belong to sensory or motor neurons. The reflex circuits in insect legs comprise basically three components: a sensory neuron, a motor neuron, and the target muscle. When the muscle is extended, the stretch is detected by the sensory neuron, which provides excitatory information to the motor neuron, eventually leading to muscle contraction (Tuthill and Wilson 2016a). This mechanism also exists in the muscles of the final portion of the D. melanogaster larval digestive tract, which has a stretch receptor (sensory) and motor neurons controlling the peristalsis responsible for defecation (Zhang et al. 2014). Therefore, it is possible that A. aegypti present a similar mechanism in which the midgut muscles are subjected to direct neural control by an enteric nervous system.
Spaced gaps in actin filaments were observed where the ring-shaped neurites are connected with the ECM and corresponding regions were visualized by SEM as sags in the ECM bundles. The H bands of the sarcomeres do not have actin filaments, but they are numerous and occur in series along all muscle fibers (Tune et al. 2020). As the spaced gaps were always seen in a unitary way and in the same position in the ECM bundles, they are unlikely to be caused by the presence of H bands. The occurrence of sags in the external structure at their positions reinforces the gaps that are not caused by sarcomere band differences. Therefore, a possible explanation for the actin gaps could be the presence of a boundary between two neighboring cruciform muscle fibers. In this context, we can hypothesize that the ring-shaped neurites connect with the ECM at the regions corresponding to the boundary between two ECM fibers.
The neuroenteric plexus and its participation in muscle remodeling during metamorphosis
FaRP-positive neuron bodies arranged in rows were detected in the anterior midgut soon after pupation (< 1 h), suggesting that midgut muscle remodeling is assisted by neurons during the larval–pupal transition. The presence of these neuron bodies was confirmed with SEM and, although they were not FaRP-positive in the larval stages, they are present as early as the L2 stage, with the same structural organization. Therefore, they are permanent muscle-associated neurons. It has already been described that the midgut enteric nervous system (ENS) of a generalized insect lacks ganglia and is composed of diffuse nerve plexuses across the muscles with distributed populations of enteric neurons (Nichols et al. 1999; Copenhaver 2007). Therefore, the muscle-associated neurons seen in A. aegypti correspond to enteric neurons of the midgut enteric plexus.
As demonstrated for A. aegypti, some insects, such as the hornworm Manduca sexta and the grasshopper Schistocerca americana, have a midgut innervated by nerve plexus, with several neuron bodies and long axons extending by them. Surprisingly, in D. melanogaster, which also belongs to the order Diptera, the midgut innervation is very scarce, comprising six nerves that extend a short distance onto the anterior midgut before branching, which leaves much of the midgut without direct innervation (Gonzalez-Gaitan and Jackle 1995). Thus, the structure and distribution of neural components of the midgut ENS strongly varies in insects, even in species with close phylogenetic relationships. Since the insect neurogenic program of the ENS presents malleability (Hartenstein 1997; Copenhaver 2007), this structural diversity seen in midgut neural components probably was directed by independent evolution.
To date, the mechanisms controlling midgut muscle contraction in mosquitoes are unknown. Previous studies have suggested that the peristaltic movements of the larval midgut muscles in the mosquito Anopheles gambiae could be controlled by self-excitable fibers, which would intercommunicate with neighboring fibers, passing the contractile stimulus forward (Jones and Zeve 2003). However, these fibers should communicate with each other through gap junctions, structures that were not detected by Bernick et al. (2007) between muscle cells of the A. aegypti midgut. Thus, it is probable that the muscle-associated enteric neurons, seen in the present work, act by initiating and coordinating the autonomous activity of midgut peristalsis in mosquitoes. Future studies focusing on the functional aspects of these neurons may verify this hypothesis.
In the early pupae-2 and in adult mosquitoes, the typical squared muscle grid of larval midgut, formed by the presence of ECM fibers, was no longer detected. This result indicates that this grid degenerates during the early stages of pupal development. As close to the ECM degeneration, the neurites associated with the ECM fibers are strongly FaRP-positive, the neural FaRPs are probably linked to the remodeling of circular midgut muscles. Signs of degeneration occur in the midgut muscles since the beginning of the metamorphosis of A. aegypti and include disorganization and myofibril loss (Bernick et al. 2008). Therefore, the FaRP-signal seems to be involved in the determination of muscle myofibril disorganization in the ECM bundles.
Reaffirming the occurrence of muscle degeneration in A. aegypti early pupae, Bernick et al. (2008) detected disorganization and destruction of myofilaments in proteasomes. A similar process occurs in Drosophila, where gut muscle fibers dedifferentiate and lose their myofibrils and extracellular matrix (Martin et al. 2001). Interestingly, in the Drosophila model, although the midgut muscle myofibrils are destroyed, the muscle cells do not die but are just remodeled, dissociating from syncytial form and/or fusing with each other. In A. aegypti, cell death was not detected in the midgut muscles, and the fibers containing myofibril disorganization kept their cell membranes intact (Bernick et al. 2008). Therefore, it is likely that, as in Drosophila, the muscle remodeling of A. aegypti midgut involves de-differentiation and structural reorganization of the pre-existing larval muscle fibers.
The absence of a squared muscle grid on the midgut of adult A. aegypti means that its function is necessary only in larval stages. The cruciform cells of the squared regions of A. aegypti larval midgut were suggested to have a profound impact on organ motility, probably allowing the wave of muscle contraction to act in concert (Bernick et al. 2007). As A. aegypti larvae feed on solid detritus that accumulates in the midgut, they probably need more intense peristaltic movements to digest the food than adult mosquitoes, who feed on liquid substances, such as nectar and blood. Therefore, the loss of some muscle components after metamorphosis could be explained by these physiological differences.
Despite the discovery of the close relationship between FaRPs and muscle control, it is difficult to determine the specific function exercised by the neuronal FaRPs in the ECM of A. aegypti larvae. Onken et al. (2004) demonstrated that the FaRPs NPF and sNPF have myoinhibitory effects on the anterior midgut of the A. aegypti larva. However, as discussed, FaRPs include additional three neuropeptide subgroups whose action on the mosquito midgut differentiation during metamorphosis cannot be disregarded. The extended FMRFamides, for example, have a direct myostimulatory function in the muscles of the body wall of D. melanogaster (Hartenstein, 1997), counteracting NPF and sNPF. Moreover, FaRPs may be involved in sensory mechanoreception, neurotransmission, and neuromodulation (Cottrell 1993; Krajniak 2013; Elphick and Mirabeau 2014; Milakovic et al. 2014; Lee et al. 2017). In this context, we cannot even state that the FaRPs detected in enteroendocrine cells of the posterior midgut in A. aegypti larvae are the same molecules detected in neurons. Based on these findings, it can be stated that FaRPs are related to visceral muscle function in insects and participate in the remodeling process of the midgut circular muscle of A. aegypti during metamorphosis. However, the exact function that they perform is unknown. The answer of these questions may require additional transcriptomic and neuropeptidomic studies assessing the expression of the FaRP genes in the A. aegypti midgut during the larval–pupal transition, and studies assessing the effects of FaRPs on midgut musculature development.
Muscle stem cell divisions during the muscle remodeling
In this study, stem cells associated with the ICM layers were seen undergoing mitosis in the late L4 and early pupae. As described in previous works (Bernick et al. 2007, 2008) and this study, the midgut muscular framework of A. aegypti undergoes a remodeling process during larval–pupal transition that explains the occurrence of myoblast mitosis. In Drosophila, the gut also undergoes muscle remodeling during metamorphosis, and the musculature of the adults are directly derived from the larval fibers; however, their myoblasts have little or no proliferation during gut metamorphosis (Aghajanian et al. 2016). This finding indicates that fruit flies and mosquitoes differ in the proliferative activity of their myoblasts, and midgut muscle remodeling probably occurs via distinct myogenic mechanisms.
Myoblast proliferation was detected in the circular muscle tissue of the A. aegypti midgut, but it is not possible to state if these myoblasts came from preexisting quiescent myoblasts. A previous study showed that in Drosophila, the muscle remodeling of the gut involves de-differentiation of pre-existing myocytes, which become “secondary myoblasts” and originate the new muscles of the adult gut (Aghajanian et al. 2016). In this context, it is possible that proliferative myoblasts detected in the ICM are derived from degeneration of mature myocytes, as occurs during gut remodeling in Drosophila (Agahajanian et al. 2016). The production of a stem cell marker for mosquitoes could help elucidate the myoblast dynamics in their midgut muscle remodeling.
No mitosis was detected in the longitudinal muscles, demonstrating that the remodeling of longitudinal muscles is different from that of circular muscles. As both insect muscles originate from different parts of mesoderm, it is possible that proliferative activity is a reflection of the embryonic origin (Klapper 2000; Rudolf et al. 2014). However, the lack of information about various aspects of the developmental biology of mosquitoes prevents further speculation. Further studies focusing on the determination and fate of midgut myoblast populations may clarify the present findings.
Hormonal regulation of midgut differentiation during the larval–pupal transition
The morphological changes that the mosquito midgut undergoes during the larval–pupal transition are regulated by several hormones (Nishiura et al. 2003, 2005; Taghert and Nitabach 2012). Here, we show that the midgut of the late A. aegypti L4 larvae show differences in patterns of FaRP immunolabeling and morphology relative to the early L4 midgut. The transition of late L4 to the early pupa is driven by the activation of genes that regulate metamorphosis by the steroid hormone 20E (Nishiura et al. 2003, 2005), such as the ecdysis triggering hormone (ETH) (Lenaerts et al. 2017), which, in turn, modulates the expression of a plethora of downstream genes, including FaRP genes (Taghert and Nitabach 2012). It is probable that the ETH acts on neurons and enteroendocrine cells (peptidergic elements), promoting the differential expression of FaRP neuropeptides. Later, neuropeptides may be released into the hemolymph to act on target cells or be delivered directly to midgut muscle cells. Identification of midgut cells that display ETH and FaRP receptors would help solve these issues.
In this study, the in situ detection of neuromuscular connections in the mosquito midgut was demonstrated for the first time. We showed that FaRPs are expressed in neurites located in specific regions of the ECMs of the late L4 midgut. After the detection of FaRPs in the neurites associated with these muscles, their fibers suffered degeneration, indicating that FaRP-based neural signals may be linked with the remodeling of the midgut muscles during metamorphosis. Simultaneously, mitosis was detected in the ICM, revealing that myoblasts associated with the circular muscles had a strong proliferative activity and participated in muscle renewal during immature to adult transition. In addition, we demonstrated that the mosquito midgut has a complex enteric nervous system, with a neuronal plexus closely associated with the muscle bundles, showing that the ENS may play a role in the peristaltic movements of the mosquito midgut. Finally, ours results can support future work for the understanding of the mosquito ENS organization and function. The knowledge about visceral muscle regulation and their contraction mechanisms in holometabolous insects, currently so scarce, is now closer to being revealed.
References
Aghajanian P, Takashima S, Paul M, Younossi-Hartenstein A, Hartenstein V (2016) Metamorphosis of the Drosophila visceral musculature and its role in intestinal morphogenesis and stem cell formation. J Development 420:43–59
Beehler-Evans R, Micchelli CA (2015) Generation of enteroendocrine cell diversity in midgut stem cell lineages. J Development 142:654–664
Bernick EP, Moffett SB, Moffett DF (2007) Organization, ultrastructure, and development of midgut visceral muscle in larval Aedes aegypti. J Tissue Cell 39:277–292
Bernick EP, Moffett SB, Moffett DF (2008) Ultrastructure and morphology of midgut visceral muscle in early pupal Aedes aegypti mosquitoes. J Tissue Cell 40:127–141
Brown MR, Crim JW, Lea AO (1986) FMRFamide-and pancreatic polypeptidelike immunoreactivity of endocrine cells in the midgut of a mosquito. J Tissue Cell 18:419–428
Brown MR, Lea A (1988) FMRFamide-and adipokinetic hormone-Like immunoreactivity in the nervous system of the mosquito, Aedes aegypti. J Comp Neurol 614:606–614
Brown MR, Klowden MJ, Crim JW, Young L, Shrouder LA, Lea AO (1994) Endogenous regulation of mosquito host-seeking behavior by a neuropeptide. J Insect Physiol 40:399–406
Bureau I (2015) Novel neuropeptides useful as insecticides Field. The present invention relates to peptide fragments and analogs derived from background of the invention neuropeptides play a key role in the regulation of a variety of physiological’, Israel Patente WO 2015/052701
Copenhaver PF (2007) How to innervate a simple gut: familiar themes and unique aspects in the formation of the insect enteric nervous system. J Dev Dyn 236:1841–1864
Cottrell GA (1993) The wide range of actions of the FMRFamide-related peptides and the biological importance of peptidergic messengers. Comparative Molecular Neurobiology, Birkhäuser Basel 279–285
Dowling JE (1991) Dopamine release from interplexiform cells in the retina: effects of bicuculline, and enkephalin on horizontal. J Neurosci 11:3034–3046
Duttlinger A, Berry K, Nichols R (2002) The different effects of three Drosophila melanogaster dFMRFamide-containing peptides on crop contractions suggest these structurally related peptides do not play redundant functions in gut. J Peptides 23:1953–1957
Elphick MR, Mirabeau O (2014) The evolution and variety of RFamide-type neuropeptides: insights from deuterostomian invertebrates. J Front Endocrinol 5:93
Fadda M, Hasakiogullari I, Temmerman L, Beets I, Zels S, Schoofs L (2019) Regulation of feeding and metabolism by neuropeptide F and short neuropeptide F in invertebrates. Front Endocrinol 10:64
Fernandes KM, Neves CA, Serrão JE, Martins GF (2014) Aedes aegypti midgut remodeling during metamorphosis. Parasitol Int 63:506–512
Fónagy A (2007) Insect neuropeptides and their potential application for pest control. Acta phytopathologica et entomologica Hungarica 41:137–152
Godoy RSM, Fernandes KM, Martins GF (2015) Midgut of the non-hematophagous mosquito Toxorhynchites theobaldi (Diptera, Culicidae). Sci Rep 5:1–16
Gonzalez-Gaitan M, Jackle H (1995) Invagination centers within the Drosophila stomatogastric nervous system anlage are positioned by Notch-mediated signaling which is spatially controlled through wingless. J Development 121:2313–2325
Hartenstein V (1997) Development of the insect stomatogastric nervous system. J Trends Neurosci 20:421–427
Hao K, Ullah H, Jarwar AR, Nong X, Tu X, Zhang Z (2020) Functional identification of an FMRFamide-related peptide gene on diapause induction of the migratory locust, Locusta migratoria L. Genomics 112:1821–1828
Haselton AT, Yin CM, Stoffolano JG (2008) FMRFamide-like immunoreactivity in the central nervous system and alimentary tract of the non-hematophagous blow fly, Phormia regina, and the hematophagous horse fly, Tabanus nigrovittatus. J Insect Sci 8:1–17
Hill SR, Orchard I (2004) The influence of diet and feeding state on FMRFamide-related peptides in the gut of Locusta migratoria L. J Peptides 25:105–114
Hung RJ, Hu Y, Kirchner R, Liu Y, Xu C, Comjean A, Perrimon N (2020) A cell atlas of the adult Drosophila midgut. Proc Natl Acad Sci 117:1514–1523
Jones JC, Zeve VH (2003) The fine structure of the gastric caeca of Aedes aegypti larvae. J Insect Physiol 14:1567–1575
Kingan TG, Shabanowitz J, Hunt D, Witten J (1996) Characterization of two myotrophic neuropeptides in the FMRFamide family from the segmental ganglia of the moth Manduca sexta: candidate neurohormones and neuromodulators. J Exp Biol 199:1095–1104
Klapper R (2000) The longitudinal visceral musculature of Drosophila melanogaster persists through metamorphosis. J Mech Dev 95:47–54
Krajniak KG (2013) Invertebrate FMRFamide Related Peptides. J Prot Pept Lett 20:647–670
Lee JS, Shih PY, Schaedel ON, Quintero-Cadena P, Rogers AK, Sternberg PW (2017) FMRFamide-like peptides expand the behavioral repertoire of a densely connected nervous system. J Proc Natl Acad Sci 114:E10726–E10735
Lenaerts C, Cools D, Verdonck R, Verbakel L, Broeck JV, Marchal E (2017) The ecdysis triggering hormone system is essential for successful moulting of a major hemimetabolous pest insect, Schistocerca gregaria. Sci Rep 7:1–14
López-Vera E, Aguilar MB, Heimer de la Cotera EP (2008) FMRFamide and related peptides in the phylum mollusca. Peptides 29:310–317
Martin BS, Gomez MR, Landgraf M, Bate M (2001) A distinct set of founders and fusion-competent myoblasts make visceral muscles in the Drosophila embryo. J Development 128:3331–3338
McLachlan IG, Beets I, Bono M, Heiman MG (2018) A neuronal MAP kinase constrains growth of a Caenorhabditis elegans sensory dendrite throughout the life of the organism. J PLoS Gen 14:1–22
Micchelli CA, Sudmeier L, Perrimon N, Tang S, Beehler-Evans R (2011) Identification of adult midgut precursors in Drosophila. J Gene Exp Patt 11:12–21
Milakovic M, Ormerod KG, Klose MK, Mercier AJ (2014) Mode of action of a Drosophila FMRFamide in inducing muscle contraction. J Exp Biol 217:1725–1736
Moffett SB, Moffett DF (2014) Comparison of immunoreactivity to serotonin, FMRFamide and SCPb in the gut and visceral nervous system of larvae, pupae and adults of the yellow fever mosquito Aedes aegypti. J Insect Sci 5:20
Nagata S (2016) FMRFamides. In: Handbook of hormones. Academic Press 432–433
Nässel DR, Winther ÅM (2010) Drosophila neuropeptides in regulation of physiology and behavior. Prog Neurobiol 92:42–104
Nässel DR, Zandawala M (2019) Recent advances in neuropeptide signaling in Drosophila, from genes to physiology and behavior. Prog Neurobiol 179:101607
Nassel RD, Bayraktaroglu E, Dircksen H (1994) Neuropeptides in neurosecretory and efferent neural systems of insect thoracic and abdominal ganglia. J Zool Sci 11:15–31
Nichols R, McCormick J, Lim I (1999) Regulation of Drosophila FMRFamide neuropeptide gene expression. J Neurobiology 39:347–358
Nishiura JT, Ho P, Ray K (2003) Methoprene interferes with mosquito midgut remodeling during metamorphosis. J Med Entomol 40:498–507
Nishiura JT, Ray K, Murray J (2005) Expression of nuclear receptor-transcription factor genes during Aedes aegypti midgut metamorphosis and the effect of methoprene on expression. Insect Biochem Mol Biol 35:561–573
Oliveira AH, Gonçalves WG, Fernandes KM, Barcellos MS, Sampaio WMS, Lopes MP, Martins GF, Serrão JE (2019) Morphology and morphometry of the midgut in the stingless bee Friesella schrottkyi (Hymenoptera: Apidae). J Insects 10:73
Onken H, Moffett SB, Moffett DF (2004) The anterior stomach of larval mosquitoes (Aedes aegypti): effects of neuropeptides on transepithelial ion transport and muscular motility. J Exp Biol 207:3731–3739
Orchard I, Lange AB, Bendena WG (2001) FMRFamide-related peptides: a multifunctional family of structurally related neuropeptides in insects. Adv Insect Physiol 28:267–329
Parthasarathy R, Palli SR (2007) Stage-and cell-specific expression of ecdysone receptors and ecdysone-induced transcription factors during midgut remodeling in the yellow fever mosquito, Aedes aegypti. J Insect Physiol 53:216–229
Peters RS, Meusemann K, Petersen M, Mayer C, Wilbrandt J, Ziesmann T, Donath A, Kjer KM, Aspöck U, Aspöck H (2014) The evolutionary history of holometabolous insects inferred from transcriptome-based phylogeny and comprehensive morphological data. J BMC Evol Biol 14:52
Predel R, Neupert S, Garczynski SF, Crim JW, Brown MR, Russell WK, Nachman RJ (2010) Neuropeptidomics of the mosquito Aedes aegypti. J Proteome Res 9:2006–2015
Price DA, Greenberg MJ (1977a) Purification characterization of a cardioexcitatory neuropeptide from the central ganglia of a bivalve mollusc. J Prep Bioch 7:261–281
Price DA, Greenberg MJ (1977b) Structure of a molluscan cardioexcitatory neuropeptide. J Science 197:670–671
Price DA, Greenberg MJ (2007) The Hunting of the FaRPs: The distribution of FMRFamide-related peptides. J Biol Bull 177:198–205
Reiher W, Shirras C, Kahnt J, Baumeister S, Isaac RE, Wegener C (2011) Peptidomics and peptide hormone processing in the Drosophila midgut. J Proteome Res 10:1881–1892
Roller L, Čižmár D, Gáliková Z, Bednár B, Daubnerová I, Žitňan D (2016) Molecular cloning, expression and identification of the promoter regulatory region for the neuropeptide trissin in the nervous system of the silkmoth Bombyx mori. Cell Tissue Res 364:499–512
Rudolf A, Buttgereit D, Jacobs M, Wolfstetter G, Kesper D, Pütz M, Berger S, Renkawitz-Pohl R, Holz A, Önel SF (2014) Distinct genetic programs guide Drosophila circular and longitudinal visceral myoblast fusion. J BMC Cell Biol 15:27
Scherkenbeck J, Zdobinsky T (2009) Insect neuropeptides: structures, chemical modifications and potential for insect control. J Bioorg Med Chem 17:4071–4084
Schinkmann K, Li C (1992) Localization of FMRFamide-like peptides in Caenorhabditis elegans. J Compar Neurol 316:251–260
Sedra L, Lange AB (2016) Cloning and expression of long neuropeptide F and the role of FMRFamide-like peptides in regulating egg production in the Chagas vector, Rhodnius prolixus. Peptides 82:1–11
Seron TJ, Hill J, Linser PJ (2004) A GPI-linked carbonic anhydrase expressed in the larval mosquito midgut. J Exp Biol 207:4559–4572
Souza DLL, Zanuncio JC, Serrão JE (2016) FMRFamide-cells in the midgut of Scaptotrigona xanthotricha (Apidae: Meliponini) of different ages and fed different diets. J Apicul Research 55:428–432
Stanek DM, Pohl J, Crim JW, Brown MR (2002) Neuropeptide F and its expression in the yellow fever mosquito, Aedes aegypti. Peptides 23:1367–1378
Strand MR, Brown MR, Vogel KJ (2016) Mosquito peptide hormones: diversity, production, and function. Adv Insect Physiol 51:145–188
Stracker TH, Thompson S, Grossman GL, Riehle MA, Brown MR (2002) Characterization of the AeaHP gene and its expression in the mosquito Aedes aegypti (Diptera: Culicidae). J Med Entomol 39:331–342
Taghert PH, Nitabach MN (2012) Peptide neuromodulation in invertebrate model systems. Neuron 76:82–97
Tarr EA, Fidler BM, Gee KE, Anderson CM, Jager AK, Gallagher NM, Fabian-Fine R (2019) Distribution of FMRFamide-related peptides and co-localization with glutamate in Cupiennius salei, an invertebrate model system. Cell Tissue Res 376:83–96
Tune TC, Irving T, Sponberg S (2020) Nanometer-scale structure differences in the myofilament lattice spacing of two two cockroach leg muscles correspond to their different functions. J Exp Biol 9:223
Tuthill JC, Wilson RI (2016a) Mechanosensation and adaptive motor control in insects. J Curr Biol 26:R1022–R1038
Tuthill JC, Wilson RI (2016b) Parallel transformation of tactile signals in central circuits of Drosophila. J Cell 164:1046–1059
Veenstra JA, Lau GW, Agricola HJ, Petzel DH (1995) Immunohistological localization of regulatory peptides in the midgut of the female mosquitoAedes aegypti. Histochemistry and Cell Biology 104:337–347
Warr E, Aguilar R, Dong Y, Mahairaki V, Dimopoulos G (2007) Spatial and sex-specific dissection of the Anopheles gambiae midgut transcriptome. BMC Genomics 8:1–11
Weetman D, Kamgang B, Badolo A, Moyes CL, Shearer FM, Coulibaly M, Pinto J, Lambrechts L, McCall PJ (2018) Aedes mosquitoes and Aedes-borne arboviruses in Africa: current and future threats. Int J Environ Res Public Health 15:220
Wikgren M, Fagerholm HP (1993) Neuropeptides in sensory structures of nematodes. J Acta Biol Hung 44:133–136
Wu Y, Parthasarathy R, Bai H, Palli SR (2006) Mechanisms of midgut remodeling: juvenile hormone analog methoprene blocks midgut metamorphosis by modulating ecdysone action. Mech Dev 123:530–547
Xu Y, Cleary L, Byrne J (2018) Identification and characterization of pleural neurons that inhibit tail sensory neurons and motor neurons in Aplysia: correlation with FMRFamide immunoreactivity. J Neuroscience 14:3565–3577
Zels S, Dillen S, Crabbé K, Spit J, Nachman RJ, Broeck JV (2015) Sulfakinin is an important regulator of digestive processes in the migratory locust, Locusta migratoria. Insect Biochem Mol Biol 61:8–16
Zhang W, Yan Z, Li B, Jan LY, Jan YN (2014) Identification of motor neurons and a mechanosensitive sensory neuron in the defecation circuitry of Drosophila larvae. J eLife 3:e03293
Žitňan D, Šauman I, Sehnal F (1993) Peptidergic innervation and endocrine cells of insect midgut. J Arch Insect Bioch Physiol 22:113–132
Acknowledgements
We thank the Núcleo de Microscopia e Microanálise (NMM, UFV) for letting us use the confocal microscope. We are grateful to Nágila Francinete Costa Secundino and Paulo Fillemon Paolucci Pimenta for their support with the reagents and use of scanning electron microscopes at the Instituto René Rachou, Fiocruz, MG.
Funding
This work was supported by the Universidade Federal de Viçosa, Comissão de Aperfeiçoamento de Pessoal de Nível Superior (CAPES)—support to RSMG; the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (Fapemig; APQ-00560–17)—support to GFM, and by the National Institutes of Health (USA) R01AI031478 and the Bloomberg Philanthropies to MJL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was performed in accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health and the Animal Use Manual (FIOCRUZ, Ministry of Health of Brazil, national decree, no. 3179). The protocol was approved by the Ethics Committee of Universidade Federal de Viçosa (UFV-Protocol 561/2016).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 Movie S1 Specificity of the FaRP immunoreactivity (green) in the midgut of late L4 of Aedes aegypti. The grid formed by the external circular muscles (ECMs) fused with the longitudinal muscles (LMs) are shown in a more external position in relation to the midgut epithelium; therefore, they are seen first in the z-stack video. The internal muscle (ICM) layer is more internal and shown above the ECM/LM grid. The FaRP immunoreactivity is inside ring-shaped structures attached to the mid-position of the ECM between two cruciform points (fusion regions of ECM and LM). (MOV 1129 KB)
Supplementary file2 Movie S2 Muscle framework (red) of the anterior midgut of early L4 of Aedes aegypti. The external circular muscle (ECM) layer is shown. (MP4 253 KB)
Supplementary file3 Movie S3. Muscle framework (red) of the anterior midgut of the early pupal Aedes aegypti. The external circular muscle (ECM) layer in the process of degeneration is shown. (MP4 424 KB)
Supplementary file4 Movie S4 Mitotic nuclei (green) in the internal circular muscle layer (ICM) of the early pupal midgut of Aedes aegypti. All labeled nuclei were found in the ICM bundles. No mitosis labeling was observed in the midgut epithelium. Red: midgut muscles; blue: cell nuclei. (MOV 626 KB)
Supplementary file5 Movie S5 Muscle framework (red) of the anterior midgut of adult female Aedes aegypti. The external circular muscle (ECM) layer is not present. (MP4 344 KB)
441_2021_3462_MOESM7_ESM.tiff
Supplementary file7 Fig. S1 FaRP and PH3 immunolabeling, neurite and neuron body identifications in immature stages of Aedes aegypti; and the absence of ECM layer in the adult mosquito. (a) FaRP-positive (green) small, rounded points (white arrow) in the posterior midgut of Aedes aegypti early L4. ICM: internal circular muscle layer. Red: midgut muscles; Blue: cell nuclei. (b) Mitotic nuclei (green) of regenerative cells (RCs) from the midgut epithelium in the early L4 of A. aegypti. DC: digestive cell nuclei. (c) 3D view of the FaRP immunoreactivity (green) in the grid formed by the presence of an external circular muscle (ECM) layer fused with the longitudinal muscle (LM) layers in the L4 of A. aegypti. Ring-shaped structures (arrow) are shown in the mid-position of the ECM just between two cruciform points (*) (where ECM fuses with LM). The internal circular muscle (ICM) fibers are shown between two ECM bundles. Red: midgut muscles. (d) Scanning electron microscopy (SEM) of the ring-shaped structures (white arrows) attached to the mid-position of the ECM between two cruciform points (*). Their extensions are also shown (black arrow). (e) FaRP-positive (green) small, rounded points (arrow) lined up and in close association with LM bundles in the anterior midgut of A. aegypti early pupa. (f–h) SEM of the early pupal (f) and early L4 (g, h) midgut, showing the region where the ICM bifurcates (full arrow), close to the two prominent longitudinal muscles (pLMs). (f) In the early pupa, it is difficult to identify the internal neurons using SEM, however, the predicted regions of their presence are shown with the thin arrows. (g, h) In the late L4, both bifurcations of the ICM (full arrow) and the internal neurons (thin arrows) are identified in g. Details of the ICM neurons are shown in h. (i) Midgut muscle organization (red) in the anterior midgut of the adult female of A. aegypti. The pLM; LM; and ICM bundles and their bifurcations (full arrow) are detected in a similar configuration of the larval midgut; however, the ECM is not present (no grids of cruciform regions are detected) (TIFF 5705 KB)
Rights and permissions
About this article
Cite this article
Godoy, R.S.M., Barbosa, R.C., Procópio, T.F. et al. FMRF-related peptides in Aedes aegypti midgut: neuromuscular connections and enteric nervous system. Cell Tissue Res 385, 585–602 (2021). https://doi.org/10.1007/s00441-021-03462-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-021-03462-3