Abstract
Honey bee is a social insect. Its colony is mainly coordinated by the chemical signals such as pheromones produced by queen or brood. Correspondingly, the worker bee developed numerous complicated olfactory sensilla in antennae for detection of these colony chemical signals and nectar/pollen signals in foraging. With the normal development of new emerged workers, young adults (nurse bee) worked in colony at the first 2–3 weeks and then followed by the foraging activity outside of the hive, which give rise to great change of the surrounding chemical signals. However, the olfactory adaption mechanism of worker bee in these processes of behavioral development is still unclear. In this study, we conducted a comprehensive and quantitative analysis of gene expression in Apis mellifera antenna of newly emerged workers, nurses and foragers using transcriptome analysis. Meanwhile, we constructed experimental colonies to collect age-matched samples, which were used to determine whether task is the principal determinant of differential expression. RNA sequencing and quantitative real-time polymerase chain reaction revealed that 6 and 14 genes were closely associated with nurse and forager behaviors, respectively. Furthermore, a broad dynamic range of chemosensory gene families and candidate odorant degrading enzymes were analyzed at different behavior statuses. We firstly reported genes associated with nursing/foraging behavior from antennae and the variations of expression of genes belonging to various olfactory gene families at different development stages. These results not only could contribute to elucidating the relationship between olfactory and behavior-related changes, but also provide a new perspective into the molecular mechanism underlying honey bee division of labor.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Division of labor is fundamental to the organization of social insects, contributing greatly to the growth, development and reproductive success of insect colonies. In honeybees (Apis mellifera), workers manifest a series of dramatic changes in behavioral status when adult workers emerge. Young adults (first 2–3 weeks of adult life) perform tasks in the hive such as nursing (bees engaging in brood care), whereas older bees (5- to 7-week life) switch to foraging outside the hive for nectar and pollen (Winston 1987). Such behavioral division can be accelerated, postponed, or reversed in respond to the changes of the environment (Robinson 1992). Previous studies have reported that division of labor in honey bee is controlled by many factors: the interaction between colony needs and worker physiological status, amount of food stores and brood, age structure of worker force, insulin signaling, the rate of juvenile hormone biosynthesis, and biogenic amines (Ament et al. 2008; Huang et al. 1991; Schulz and Robinson 2001). Recently, a study revealed that the expression of gustatory receptor 10 in A. mellifera was correlated with the division of labor (Paerhati et al. 2015). In honey bee, workers have evolved an intricate olfaction organs that produce and perceive complex pheromones and the olfaction system plays a fundamental role in mediating insect behavior such as food source identification, mate choice, kin recognition, social communication and regulating the specific tasks of each individual (Galizia and Szyszka 2008; Slessor et al. 2005). Antennae, the principal olfactory organs, are sensitive to a variety of volatile chemicals such as odors and pheromones, and play a fundamental role in regulating complex social interactions (Winston 1987). Antennae is crucial to nursing behaviors in hive and foraging behaviors, however, the molecular mechanism by which the olfactory system influences division of labor and behavioral development in honeybee is largely unexplored.
Chemical communication is crucial for regulating complex social interactions within honeybee colonies. For instance, the queen within a colony produces and releases queen substances that inhibit the fertility of workers, maintain the organization of the colony, and attract drones for mating (Brockmann et al. 2006; Gary 1962). Worker bees release alarm pheromones and various pheromones to defend the colony from enemies and recognize each other as colony members (Moritz and Bürgin 1987; Swanson et al. 2009). Brood produces and releases brood pheromone that stimulates the worker to take care of the larvae (Conte et al. 1990; He et al. 2016). There are different types of sensilla on the antennae. Among them, sensilla placodea, the main olfactory antennal structures in the honeybee, respond to a variety of plant and flower odorants as well as to components of the honeybee pheromones (Esslen 1976; Sandoz et al. 2007). Morphological observations have demonstrated that workers have considerably higher number of hair-like sensilla on their antennae than that of drones and queens (Fang et al. 2012). The higher number of hair-like sensilla in workers may assist food search and social communication. With the development of proteomics, a study of the antennal proteomes of drones, workers and queens has facilitated a better understanding of their social organizations and environmental interactions of honeybee (Fang et al. 2012; Feng et al. 2011; Woltedji et al. 2012). Some studies have reported that the detection of odor signals by insect antennae involves a complex perception system, including odorant binding proteins (OBPs), chemosensory proteins (CSPs), odorant receptors (Ors), gustatory receptors (Grs), and odorant degrading enzymes (ODEs). The recognition and discrimination of thousands of volatile compounds is mediated by olfactory sensory neurons. In insects, the chemosensory neurons are surrounded by an aqueous milieu acting as a barrier for hydrophobic compounds. OBPs can recognize many airborne molecules, such as hydrophobic odorants, and then facilitate their delivery to the olfactory receptors through the sensillum lymph (Forêt and Maleszka 2006). CSP, another major family of carrier proteins associated with insect sensory organs, shuttle odorants through the sensillar lymph to the olfactory receptors (Jin et al. 2005). Ors are located on the surface of olfactory sensory neuronal dendrites in the antennae and have crucial roles in chemosensory signal transduction processes that occur in olfactory receptor neurons (Liu et al. 2012). ODEs contribute to the signal deactivation within the olfactory sensilla of insect by quickly removing odorant molecules from olfactory receptors, thereby allowing the insect to rapidly respond to changes of the volatiles in its environment.
The A. mellifera genome provides us with a great deal of useful information that greatly promote the study of olfactory mechanism. The antennae as an olfactory organ have received considerable attention, including morphological observations and proteomics (Fang et al. 2012; Feng et al. 2011; Woltedji et al. 2012). Some OBPs and CSPs have been identified in antennae (Dani et al. 2010; Forêt et al. 2007). Four P450 genes were found to differ significantly in the antennae of worker bees according to age-related task performance (Mao et al. 2015). However, changes of gene expression in the olfactory system of workers at different behavior statuses are still unknown. In this study, we carried out an RNA-seq analysis of antennae from newly emerged workers (NEW), nurses and foragers. We focused on two main classes of genes: (1) genes associated with nurse and forager behaviors; (2) gene families that are connected with olfaction: OBPs, CSPs, ORs, Grs and ODEs. Meanwhile, we constructed a single cohort colony to collect old nurses and young foragers to confirm whether genes are associated with behavior development. The results of this study provide new insights into the gene expression of worker antenna at different behavioral statuses, and offer a valuable resource for studying the mutual relationship between olfactory system and behavior development in honey bees.
Methods
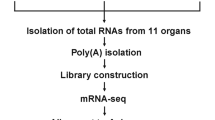
Honeybees and sample collection
The honeybee species A. mellifera ligustica was raised in the apiaries of College of Bee Science, Fujian Agriculture and Forestry University in June 2016. Three colonies of a queen-right were used in our experiment. Worker bees were gathered at three developmental stages (newly emerged worker, nurse and forager). Newly emerged bees were obtained from capped brood frames brushed clear of bees when adult bees emerged from their cells for 3–4 h. To generate nurses and pollen foragers of the same age, capped frames of worker brood were shook vigorously to get rid of bees, and then kept in a humid incubator at 34 °C; every morning adult bees emerged from their cells were marked on the thorax with paint and returned to the hive. 10-day-old nurses were collected from the hive when marked bees of tenth day entered the cells and were nursing the larvae. 21-day-old foragers were captured at the entrance of the source colony using soft forceps when marked bees of 21st day have pollen loads on their hind legs.
We constructed experimental colonies to enable us to collect age-matched samples of nurse and forager bees as previously described (McQuillan et al. 2012). Frames of emerging brood were sourced from the three colonies. We collected age-matched samples of nurses and forager bees when bees were 10 and 21 days old. All bees in this experiment were immediately snap-frozen in liquid nitrogen prior to removal of the antennae. Antennae were dissected from bee heads using fine dissection scissors.
RNA preparation
Total RNA was extracted using TRIzol reagent (Invitrogen, Burlington, ON, Canada) from the pool of 100 pairs of antennae for each sample according to the manufacturer’s protocol. RNA was monitored on 1% agarose gels to determine whether they were degraded or contaminated. RNA purity, RNA concentration and RNA integrity was checked using the NanoPhotometer® spectrophotometer (IMPLEN, CA, USA), Qubit® RNA Assay Kit in Qubit® 2.0 Flurometer (Life Technologies, CA, USA) and RNA Nano 6000 Assay Kit of the Bioanalyzer 2100 system (Agilent Technologies, CA, USA), respectively.
Library construction, clustering and transcriptomic sequencing
These were performed at the Novogene Bioinformatics Institute, Beijing, following procedures described previously with minor modification (Zhang et al. 2014). In brief, mRNA was enriched from total RNA (3 µg) and fragmented into short pieces. Double-strand cDNA was synthesized using the mRNA fragments as templates. First strand cDNA was synthesized using random hexamer primer and M-MuLV Reverse Transcriptase (RNase H). Second strand cDNA synthesis was subsequently performed using DNA Polymerase I and RNase H. The library fragments were purified with AMPure XP system (Beckman Coulter, Beverly, USA) to select cDNA fragments of preferentially 150 ~ 200 bp in length. After cluster generation, nine library preparations were sequenced on an Illumina Hiseq 2500 platform and 125 bp paired-end reads were generated.
Data analysis
Raw reads were firstly processed to remove reads containing adapter, reads containing poly-N and low-quality reads through in-house Perl scripts. Clean reads with high quality were aligned to the A. mellifera genome (Amel_4.5; ftp://ftp.ncbi.nih.gov/genomes/Apis_mellifera/) using TopHat v2.0.12 (Kim et al. 2013). HTSeq v0.6.1 was used to count the reads numbers mapped to each gene. And then FPKM of each gene was calculated based on the length of the gene and reads count mapped to this gene (Trapnell et al. 2009). Genes were considered to be expressed if the FPKM value was more than one in the corresponding sample (Wickramasinghe et al. 2015).
DESeq was carried out to identify differentially expressed genes (Anders and Huber 2010). Briefly, readcount was normalized using a model based on the negative binomial distribution. The resulting P values were adjusted using the Benjamini and Hochberg’s approach for controlling the false discovery rate (Benjamini and Hochberg 1995). Genes with an adjusted P value (Padj) < 0.05 found by DESeq were defined as differentially expressed genes (DEGs). The regulation of DEGs was determined by Log2Foldchange, in which the value of fold change was calculated by corrected readcount. Genes with Log2Foldchange > 0 were regarded as up-regulated; genes with Log2Fold change < 0 were regarded as down-regulated.
Gene Ontology (GO) annotations were obtained from Gene Ontology Consortium (http://www.geneontology.org/). GO enrichment analysis of DEGs was implemented by the GOseq R package based on Wallenius noncentral hypergeometric distribution (Young et al. 2010), which can be adjusted for gene length bias in DEGs. GO terms with corrected P value less than 0.05 were considered significantly enriched by DEGs. KEGG pathways of DEGs were obtained by KEGG (Kyoto Encyclopedia of Genes and Genomes) database (http://www.genome.jp/kegg/), and then the statistical enrichment of DEGs in KEGG pathways was determined using KOBAS software (Kanehisa et al. 2008; Mao et al. 2005).
Quantitative real-time reverse transcriptase-PCR (qRT-PCR)
qRT-PCR analysis of the samples was performed following methods described previously (Nie et al. 2014). Briefly, Total RNA was isolated from the worker antenna as described above. Using 1 μg of total RNA, cDNA was synthesized with the PrimeScript RT reagent Kit (RR037A, Takara). qRT-PCR was carried out with an ABI7500 real-time PCR system (Applied Biosystems) with a two-step reaction protocol of 40 cycles of 94 °C for 3 s and 60 °C for 30 s, followed by dissociation for quality control. The housekeeping gene actin (NM_001185146.1) was used as a control for normalization. Each sample was analyzed in triplicate. Data were calculated with the 2−△△Ct method (Livak and Schmittgen 2001). Primer information for qRT-PCR is described in Table S1. Using IMB SPSS statistics software, data were examined by one way ANOVA, followed by LSD’s multiple comparison tests.
Results
Overview of transcriptional changes at three developmental stages
Nine digital gene expression (DGE) libraries were constructed and sequenced using the total RNA isolated from the antennae of A. mellifera at three developmental stages (NEW, nurse and forager) with three biological replicates. A total of 24.7–34.5 million raw reads for each library was generated (Table 1). These results demonstrated that the sequencing quality was robust (Figure S1 and Figure S2). After the low-quality reads were filtered, 79.63–83.30% clean reads were aligned to the A. mellifera genome using TopHat v2.0.12 software and 78.68–82.32% of the reads in the nine libraries were uniquely mapped to A. mellifera sequences (Table 1).
The expression level of annotated A. mellifera genes was determined by fragments per kilo base of exon per million fragments mapped (FPKM) method (Table S2). Differentially expressed genes (DEGs) were considered to be significant if Padj < 0.05. Using libraries of NEW as a comparison, the number of DEGs was 1732 and 3688 in nurse and forager, respectively, but with only 381 DEGs between foragers and nurses (Fig. 1). The higher number of DEGs between NEW and the other groups could be attributed to the immature status of NEW. More specifically, 1583 genes were significantly different in the NEW group compared with the nurse and forager groups; 227 genes were significantly different in the nurse group compared with the NEW and forager groups; and 356 genes were significantly different in the forager group compared to the NEW and nurse groups. Notably, 215 DEGs were identified among the NEW, nurse and forager groups (Fig. 1, and Table S3). Among them, 107 genes decreased significantly in nurse compared to NEW, and the same genes decreased significantly in forager compared to nurse (Fig. 2a); 104 genes increased significantly in nurse compared to NEW, and the same genes increased significantly in forager compared to nurse (Fig. 2b). Thus, these 107 and 104 genes appear to be continuously down-regulated and up-regulated, respectively, in development from NEW to forager. These data suggested that these genes may play a critical role in antenna development or chemical communication. To verify the RNA-seq results by quantitative real-time polymerase chain reaction (qRT-PCR), eight genes were randomly chosen from DEGs among NEW, nurses and foragers. The trends in expression level changes determined by qRT-PCR were similar to the trends we observed with RNA-seq (Figure S3).
Heat map of differentially expressed genes which were continuously down-regulated (a) or up-regulated (b) from the newly emerged workers to foragers. A hierarchical clustering of differentially expressed genes was obtained using RNA-seq data that was derived from the three antenna samples (newly emerged worker, nurses and foragers) based on log10(RPKM + 1) values. Red represents up-regulated expression and blue represents down-regulated
DEGs associated with nursing behaviors in the worker antennae
In the present study, eight DEGs (Table 2) that were up-regulated in the antennae of nurses compared with both NEW and foragers, but without differentially expression between NEW and foragers, were considered to be associated with nursing behaviors. We constructed experimental colonies to collect age-matched samples (10-day-old) of nurse and forager bees to confirm whether the eight DEGs have significant difference between nurse workers and forager bees. qRT-PCR analysis showed that six genes (odorant binding protein 17, UDP-glycosyltransferase, ammonium transporter Rh type B-like, urea transporter 2, GMC oxidoreductase 14, and mucin-6-like) were up-regulated in the antennae of 10-day-old nurses compared with those in both 10-day-old and 21-day-old foragers (Fig. 3), demonstrating that they are associated with nursing behaviors.
The expression of differentially expressed genes associated with nurse behaviors (a) and foraging behaviors (b) in the antennae of adult workers at normal colony and a single cohort colony. Expression levels are normalized using the reference gene Actin. Using IMB SPSS statistics software, data were analyzed by one way ANOVA with LSD’s multiple comparison tests. NS no significance
DEGs associated with foraging behaviors in the worker antennae
As shown in Table S4, in this study, there were 110 DEGs being up-regulated in forager group compared with both NEW and nurse but without significant difference between NEW and nurses. These DEGs were thought to be related to foraging behaviors. Gene Ontology (GO) analysis indicated that 80 of these DEGs had at least one matched GO in one of three categories (molecular function, cellular component and biological process) (Figure S4).
The Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis indicated that these DEGs were involved in 19 pathways (Table S5). The pathways (starch and sucrose metabolism, phenylalanine metabolism and tyrosine metabolism) were significantly enriched in antennae between forager and nurse (corrected P value < 0.05); seven were energy metabolism related pathways (citrate cycle, pyruvate metabolism, glycolysis/gluconeogenesis, phenylalanine metabolism, tyrosine metabolism, starch and sucrose metabolism, purine metabolism). This result is also consistent with the fact that foraging behavior tends to have higher energetic costs in a fluctuating environment (Stabentheiner and Kovac 2016).
To narrow the number of DEGs associated with foraging behavior, 38 DEGs with FPKM at forager > 2, log2Foldchange (forager vs. NEW) > 1 and log2Foldchange (forager vs. nurse) > 1, were chosen from the 110 DEGs (Table S6). We constructed experimental colonies to collect age-matched samples (21-day-old) of nurse and forager bees to examine whether these DEGs have significant difference between nurse workers and forager bees. The qRT-PCR analysis showed that 14 genes were up-regulated in the antennae of 21-day-old foragers compared with both 10-day-old nurses and 21-day-old nurses (Fig. 3), demonstrating that they were associated with foraging behaviors.
The expression profile of OBPs in the antennae at three development stages
The A. mellifera genome encodes 21 OBPs, three of which (OBP8, OBP19 and OBP20) are absent from the assembly (Forêt and Maleszka 2006), and the remaining 18 genes were detected in the antennae at three development stages (Fig. 4 and Table S7). Twelve genes were increasingly expressed from NEW to foragers, including OBP1, OBP2, OBP4, OBP5, OBP6, OBP7, OBP9, OBP12, OBP13, OBP14, OBP15 and OBP16; the expressions of four genes (OBP3, OBP11, OBP17 and OBP21) was firstly increased from NEW to nurses, and then decreased from nurses to foragers; the remaining two genes were continuously decreased from NEW to forages. Two genes (OBP1 and OBP2) were abundantly expressed in the antennae at three development stages, while the other four genes (OBP3, OBP9, OBP13 and OBP14) were weakly expressed or almost undetectable.
Heat map illustrating the various degrees of expression of odorant binding proteins (a), and chemosensory proteins (b) in the antennae at three developments (newly emerged worker, nurses and foragers) using RNA-seq data. The black units represent zero for FPKM value. “OBP and CSP represent odorant binding proteins (OBPs), chemosensory proteins (CSPs), respectively”
The expression profile of CSPs in the antennae at three development stages
Six CSPs have been identified in A. mellifera genome (Forêt et al. 2007), and their expression profiles were investigated in the antennae at three development stages in this study (Fig. 4 and Table S7). CSP1 in the A. mellifera antennae at NEW, nurses and foragers exhibited the highest expression levels, with FPKM values of 14702.09, 19963.51 and 20945.90, respectively. CSP2 and CSP4 had similar expression pattern that they were continuously down-regulated form NEW to foragers. CSP3 was heavily expressed in nurses (FPKM = 7887.91), and has much higher expression at NEW and foragers (FPKM = 13310.90 and FPKM = 14876.17, respectively). CSP5 and CSP6 were both less expressed in the antennae at three development stages. And CSP6 was down-regulated expressed from NEW to nurses and slightly increased from nurses to foragers, but there was no difference at foragers compared with NEW and nurses.
The expression profile of Ors in the antennae at three development stages
A total of 180 Ors were discovered in the A. mellifera genome (Robertson and Wanner 2006), 104 of which were detected in the antennae at three development stages (Table S7). All the genes had low expression at the three stages, except Or2, which has the highest expression level with FPKM value of approximately 200, 83.65% (87 Ors) with the FPKM < 15 and 15.38% with FPKM ranging from 15 to 60.
The expression profile of Grs in the antennae at three development stages
In the present study, four Grs (Gr1, Gr2, Gr3 and Gr10) were detected in the antennae at NEW, nurses and foragers, with FPKM values less than 6. The expression profiles of Gr1, Gr2 and Gr3 were continuously decreased from NEW to foragers; the expression of Gr10 was firstly increased from NEW to nurse, and then lightly decreased from nurses to foragers (Table S7).
Putative candidate of odorant degrading enzymes (ODEs) in the antennae
Some insect pheromones can be rapidly degraded by candidate ODEs, including cytochromes P450s, some esterases and glutathione S-transferases (GSTs) (Vogt 2005). The expression profiles of these families were detected in the antennae of different behavior statuses in this study and some DEGs could affect olfaction as ODEs.
In our study, a total of 31 P450 genes were detected in the antennae, and most of them showed low expression levels (Table S7). Four genes (CYP4G11, CYP9Q1, CYP6BD1 and CYP49A1) were differently expressed between nurses and foragers. CYP4G11, CYP9Q1 and CYP6BD1 were expressed with significant differences along with age-related task performance, with expression levels increasing progressively from low detection in NEW, to significantly higher in nurses and highest in foragers. Several studies have shown that carboxylesterases in insect antennae act as ODEs (Chertemps et al. 2015; Ishida and Leal 2005, 2008; Younus et al. 2017). Here, ten carboxylesterases were found to be expressed weakly at antennae of bees with different behaviors, except for esterase FE4-like (409173) and juvenile hormone esterase (406066) (Table S7). The expression of esterase FE4-like (409173) in the antennae of nurses elevated by over threefold relative to its expression in the antennae of NEW, and then increased gradually from nurses to foragers without remarkable difference. Juvenile hormone esterase (406066) had the highest expression level at NEW, and was continuously down-regulated in nurses and foragers. In addition, esterase E4-like (409801) expression in the antennae of NEW, nurses and foragers increased significantly at each behavioral stage. The genes with higher expressions at special behavioral stages may have ODE function for bioactive ester odorants in hives or foraging to execute behavioral responses.
In insects, GSTs are also involved in signal termination (Rogers et al. 1999). Ten GSTs with low expression level, except for GSTD1 (409490) belonging to insect-specific delta classes and LOC102655694 (102655694), were detected in A. mellifera antennae (Table S6). GstU1 (725942) was differentially expressed between nurses and foragers, with expression profile down-regulated continuously from NEW to foragers. The highly expressed GstU1 at NEW suggested it may play a critical role in inactivating some odorant in hive.
Discussion
This study presented the first comprehensive transcriptome of worker antennae in A. mellifera at three development stages (newly emerged worker, nurses and foragers). These results provided a quantitative analysis of gene expression in the primary olfactory organ (antennae) of A. mellifera. In addition to transcriptome analysis, a single cohort colony was also constructed, 6 and 14 genes were confirmed to associate with nurse and foraging behaviors, respectively. Furthermore, this research investigated the antennal chemosensory gene families and candidate ODEs of A. mellifera via transcriptomic analysis. It provides a clue towards future studies regarding division of labor. The information achieved in this study will be beneficial for the elucidation of olfactory mechanism, and for the better understanding of olfactory system and behavior development.
Combined with transcriptome analyses and qRT-PCR, six genes in the worker antennae were found to be associated with nursing behaviors. It was reported that OBPs are thought to facilitate the transport of odorants through the aqueous surrounding to the olfactory receptors (Taylor et al. 2008). In this study, odorant binding protein 17 was up-regulated in the antennae of nurses compared with both NEW and foragers. We speculated that some odorants in the surrounding environment enter the sensillum pore of the nurse’s antennae, bind to odorant binding protein 17, and transport their cargo to the surface of olfactory neurons, thereby ultimately causing nurse bees to exhibit nursing behaviors. As young larvae cannot excrete after feeding, excessive nitrogenous wastes, such as ammonia, uric acid and urea, which are toxic to the tissues, tend to accumulate in their bodies. These toxins may be eliminated by releasing ammonia, a critical olfactory cue for many insects (Braks et al. 2001; Geier et al. 1999), from the pores of young larvae integument or transport urea to the surface of the integument. Previous studies have reported that ammonium transporter plays a critical role in olfactory signaling for ammonia response (Menuz et al. 2014; Pitts et al. 2014). In our study, up-regulation of ammonium transporter Rh type B-like in the antennae of nurse bees suggests a role in the detection of ammonia surrounding young larvae. Increased expression of ammonium transporter Rh type B-like may increase the proportion of nurse visits to cell with young larvae. Besides, behavioral tasks result in increased glucose metabolism (Libyarlay et al. 2014), and glucose-methanol-choline oxidoreductase family (GMC oxidoreductase family) genes are involved in insects (Sun et al. 2012). In honeybees, a glucose oxidase, belonging to GMC oxidoreductase, was detected in the hypopharyngeal gland of the foragers but not in the nurses, indicating its importance in the forager bee’s task of converting nectar into honey (Ohashi et al. 1999). Given that nurses frequently visit cell with or without brood, we assumed that the up-regulated GMC oxidoreductase 14 in antennae of nurses may involve in glucose metabolism to provide abundant energy for nursing behaviors. A previous study showed that UDP-glycosyltransferase can be used as odorant degrading enzymes to terminate diverse odorant signals (Lazard et al. 1991). Therefore, UDP-glycosyltransferase up-regulated in antennae of nurses may be used to prevent the accumulation of stimulants, thereby allowing nurse bees to rapidly respond to changes of volatiles in its environment.
In our study, 14 genes in worker antennae were associated with foraging behaviors. Among them, venus kinase receptor (VKR) belongs to receptor tyrosine kinases family. According to phylogenetic studies, VKR is close to the insulin receptor and exhibits highly conserved IR-like tyrosine kinase domains, indicating that VKR and insulin receptor could transduce similar pathways (Dissous 2015; Vanderstraete et al. 2013). Moreover, it was reported that insulin signaling is involved in the regulation of worker division of labor in honey bee colonies (Ament et al. 2008). Therefore, the up-regulated expression of venus kinase receptor in foragers in our study implied that they might affect insulin-signaling status, causing the change of their social behavior.
A previous study reported that nine OBPs (OBP1, OBP2, OBP4, OBP5, OBP6, OBP8, OBP11, OBP12 and OBP15) are expressed exclusively in the antennae of adult bees (Forêt and Maleszka 2006). In our study, five genes (OBP1, OBP2, OBP5, OBP6 and OBP12) were up-regulated from NEW to nurses; however, no difference was detected from nurses to foragers. OBP5 is highly expressed in the antennae of 10-day-old and 15-day-old workers, who perform task in the hive, such as nursing, storing and processing food (Trhlin and Rajchard 2011). In our study, OBP5 had higher up-regulated expression in nurses compared with NEW, which could be assumed that OBP5 may be involved in nursing behavior in the hive in response to olfactory signals for tasks.
Ozaki et al. (2005) have reported that, in Camponotus japonicus, CSP1 was highly expressed in the worker antennae and was found to possess binding spectrum to cuticular hydrocarbon components that cause the colony-specific odor of workers (Ozaki et al. 2005). CSP1 of A. mellifera showed high similarity to the homologous genes CSP1 of Camponotus japonicus (Hojo et al. 2015). Therefore, CSP1 may play an important role in recognizing discriminations via binding to the colony-specific odor in A. mellifera workers. CSP2 and CSP4 were found to exhibit a strong affinity for binding to the large aromatic molecules (p-tert-butylbenzophenone and 4-hydroxy-4′-isopropyl azobenzene) (Dani et al. 2010). These two genes had similar expression pattern that they were continuously down-regulated form NEW to foragers, indicating that they may play a vital role in binding to some volatiles at the NEW stage. CSP4 was found to be largely restricted to olfactory tissues (antennae, integument and legs), with the highest expression level in the antennae (Forêt et al. 2007). Accordingly, CSP4 seems to be related to the chemoreceptive process. CSP3 was proved to bind specifically to large fatty acids and ester derivatives, which are brood pheromone components, indicating that it was involved in the perception of the brood pheromone (Briand et al. 2002). In this study, CSP3 was heavily expressed in nurses, suggesting that it is crucial in the perception of the brood pheromone during the nursing behaviors; whereas it has much higher expression at NEW and foragers, indicating that CSP3 may play a distinctly different physiological role in NEW and foragers.
Notably, all Ors we detected had no differential expression between nurses and foragers, presumably owing to two reasons. Firstly, it is well-known that odorants were transported by OBPs/CSPs through the sensillum lymph, and then interact with ORs (Leal 2013). Honeybee antennae perceive complex chemical signals with sensilla in the same way. As many odorants can interact with same odorant receptor, the changes in the volatiles in its environment did not significantly affect the expression levels of corresponding odorant receptors. Secondly, only trace amounts of odorants activate corresponding Ors, which cause changes in the expression levels of corresponding odorant receptors, but with no significant difference.
Ten Grs were identified in A. mellifera genome (Robertson and Wanner 2006). In this study, the expression profiles of Gr1, Gr2 and Gr3 were continuously decreased from NEW to foragers (Table S7). In Drosophila, three Grs (Gr63F1, Gr10B1 and Gr21D1) were specifically detected in the antenna, presumably function as olfactory receptors (Scott et al. 2001). Similarly, the three Grs may also be associated with olfactory perception in A. mellifera at the antennae of different developments. More interestingly, the expression of Gr10 was firstly increased from NEW to nurse, and then lightly decreased from nurses to foragers, which coincided with the previous result that Gr10 was involved in nursing behavior (nursing or brood care) in A. mellifera (Paerhati et al. 2015).
Some P450 genes have been proven to be strongly expressed in the antennae and are located in sensilla trichodea, which are responsible for odorant detection (Maïbèche-Coisne et al. 2005). In our study, CYP4G11, CYP9Q1 and CYP6BD1 were expressed with significant differences along with age-related task performance, with expression levels increasing progressively from low detection in NEW, to significantly higher in nurses and highest in foragers, which was consistence with previous results that task-related genes (CYP4G11 and CYP9Q1) were continuously up-regulated from NEW to foragers at olfactory organ (antennae and legs) (Mao et al. 2015). It is well-known that NEW and nurses working in the hive are exposed to few phytochemicals; on the contrary, foragers collect pollen and nectar outside, thereby encountering numerous phytochemicals and pesticides. The three genes with the highest expression in foragers might rapidly deactivate odorant signals, prevent the accumulation of stimulants and allow the honeybee to rapidly respond to changes in the volatiles in the environment.
References
Ament SA, Corona M, Pollock HS, Robinson GE (2008) Insulin signaling is involved in the regulation of worker division of labor in honey bee colonies. Proc Natl Acad Sci USA 105:4226–4231
Anders S, Huber W (2010) Differential expression analysis for sequence count data. Genome Biol 11:1–13
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B 57:289–300
Braks MAH, Meijerink J, Takken W (2001) The response of the malaria mosquito, Anopheles gambiae, to two components of human sweat, ammonia and L-lactic acid, in an olfactometer. Physiol Entomol 26:142–148
Briand L, Swasdipan N, Nespoulous C, Bézirard V, Blon F, Huet J, Ebert P, Penollet J (2002) Characterization of a chemosensory protein (ASP3c) from honeybee (Apis mellifera L.) as a brood pheromone carrier. Eur J Biochem 269:4586–4596
Brockmann A, Dietz D, Spaethe J, Tautz J (2006) Beyond 9-ODA: sex pheromone communication in the European honey bee Apis mellifera L. J Chem Ecol 32:657–667
Chertemps T, Younus F, Steiner C, Durand N, Coppin CW, Pandey G, Oakeshott JG, Maïbèche M (2015) An antennal carboxylesterase from Drosophila melanogaster, esterase 6, is a candidate odorant-degrading enzyme toward food odorants. Front Physiol 6:315
Conte YL, Arnold G, Trouiller J, Masson C, Chappe B (1990) Identification of a brood pheromone in honeybees. Naturwissenschaften 77:334–336
Dani FR, Iovinella I, Felicioli A, Niccolini A, Calvello MA, Carucci MG, Qiao H, Pieraccini G, Turillazzi S, Moneti G (2010) Mapping the expression of soluble olfactory proteins in the honeybee. J Proteome Res 9:1822–1833
Dissous C (2015) Venus kinase receptors at the crossroads of insulin signaling: their role in reproduction for helminths and insects. Front Endocrinol 6:118
Esslen J (1976) Zahl und verteilung antennaler sensillen bei der honigbiene (Apis mellifera L.). Zoomorphology 83:227–251
Fang Y, Song F, Zhang L, Aleku DW, Han B, Feng M, Li J (2012) Differential antennal proteome comparison of adult honeybee drone, worker and queen (Apis mellifera L.). J Proteom 75:756–773
Feng M, Song F, Aleku DW, Han B, Fang Y, Li J (2011) Antennal proteome comparison of sexually mature drone and forager honeybees. J Proteome Res 10:3246–3260
Forêt S, Maleszka R (2006) Function and evolution of a gene family encoding odorant binding-like proteins in a social insect, the honey bee (Apis mellifera). Genome res 16:1404–1413
Forêt S, Wanner KW, Maleszka R (2007) Chemosensory proteins in the honey bee: Insights from the annotated genome, comparative analyses and expressional profiling. Insect Biochem Mol Biol 37:19–28
Galizia CG, Szyszka P (2008) Olfactory coding in the insect brain: molecular receptive ranges, spatial and temporal coding. Entomol Exp Appl 128:81–92
Gary NE (1962) Chemical mating attractants in the queen honey bee. Science 136:773–774
Geier M, Bosch OJ, Boeckh J (1999) Ammonia as an attractive component of host odour for the yellow fever mosquito, Aedes aegypti. Chem Senses 24:647–653
He XJ, Zhang XC, Jiang WJ, Barron AB, Zhang JH, Zeng ZJ (2016) Starving honey bee (Apis mellifera) larvae signal pheromonally to worker bees. Sci Rep 6:22359
Hojo MK, Ishii K, Sakura M, Yamaguchi K, Shigenobu S, Ozaki M (2015) Antennal RNA-sequencing analysis reveals evolutionary aspects of chemosensory proteins in the carpenter ant, Camponotus japonicus. Sci Rep 5:13541
Huang ZY, Robinson GE, Tobe SS, Yagi KJ, Strambi C, Strambi A, Stay B (1991) Hormonal regulation of behavioural development in the honey bee is based on changes in the rate of juvenile hormone biosynthesis. J Insect Physiol 37:733–741
Ishida Y, Leal WS (2005) Rapid inactivation of a moth pheromone. Proc Natl Acad Sci USA 102:14075–14079
Ishida Y, Leal WS (2008) Chiral discrimination of the Japanese beetle sex pheromone and a behavioral antagonist by a pheromone-degrading enzyme. Proc Natl Acad Sci USA 105:9076–9080
Jin X, Brandazza A, Navarrini A, Ban L, Zhang S, Steinbrecht RA, Zhang L, Pelosi P (2005) Expression and immunolocalisation of odorant-binding and chemosensory proteins in locusts. Cell Mol Life Sci 62:1156–1166
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S, Okuda S, Tokimatsu T (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36:D480-D484
Kim D, Pertea G, Trapnell C, Pimentel H, Kelley R, Salzberg SL (2013) TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol 14:1
Lazard D, Zupko K, Poria Y, Net P, Lazarovits J, Horn S, Khen M, Lancet D (1991) Odorant signal termination by olfactory UDP glucuronosyl transferase. Nature 349:790–793
Leal WS (2013) Odorant reception in insects: roles of receptors, binding proteins, and degrading enzymes. Annu Rev Entomol 58:373–391
Libyarlay H, Rittschof CC, Massey JH, Pittendrigh BR, Robinson GE (2014) Socially responsive effects of brain oxidative metabolism on aggression. Proc Natl Acad Sci USA 111:12533–12537
Liu Y, Gu S, Zhang Y, Guo Y, Wang G (2012) Candidate olfaction genes identified within the Helicoverpa armigera antennal transcriptome. PLoS One 7:e48260
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−∆∆CT method. Methods 25:402–408
Maïbèche-Coisne M, Merlin C, François M-C, Porcheron P, Jacquin-Joly E (2005) P450 and P450 reductase cDNAs from the moth Mamestra brassicae: cloning and expression patterns in male antennae. Gene 346:195–203
Mao X, Cai T, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the KEGG orthology (KO) as a controlled vocabulary. Bioinformatics 21:3787–3793
Mao W, Schuler M, Berenbaum M (2015) Task-related differential expression of four cytochrome P450 genes in honeybee appendages. Insect Mol Biol 24:582–588
McQuillan HJ, Barron AB, Mercer AR (2012) Age-and behaviour-related changes in the expression of biogenic amine receptor genes in the antennae of honey bees (Apis mellifera). J Comp Physiol A 198:753–761
Menuz K, Larter NK, Park J, Carlson JR (2014) An RNA-seq screen of the Drosophila antenna identifies a transporter necessary for ammonia detection. PLoS Genet 10:e1004810
Moritz RFA, Bürgin H (1987) Group response to alarm pheromones in social wasps and the honeybee. Ethology 76:15–26
Nie H, Liu C, Cheng T, Li Q, Wu Y, Zhou M, Zhang Y, Xia Q (2014) Transcriptome analysis of integument differentially expressed genes in the pigment mutant (quail) during molting of silkworm, Bombyx mori. PLoS One 9:e94185
Ohashi K, Natori S, Kubo T (1999) Expression of amylase and glucose oxidase in the hypopharyngeal gland with an age-dependent role change of the worker honeybee (Apismellifera L.). Eur J Biochem 265:127–133
Ozaki M, Wada-Katsumata A, Fujikawa K, Iwasaki M, Yokohari F, Satoji Y, Nisimura T, Yamaoka R (2005) Ant nestmate and non-nestmate discrimination by a chemosensory sensillum. Science 309:311–314
Paerhati Y, Ishiguro S, Ueda-Matsuo R, Yang P, Yamashita T, Ito K, Maekawa H, Tani H, Suzuki K (2015) Expression of amgr10 of the gustatory receptor family in honey bee is correlated with nursing behavior. PLoS One 10:e0142917
Pitts RJ, Derryberry SL Jr, Pulous FE, Zwiebel LJ (2014) Antennal-expressed ammonium transporters in the malaria vector mosquito anopheles gambiae. PLoS One 9:e111858
Robertson HM, Wanner KW (2006) The chemoreceptor superfamily in the honey bee, Apis mellifera: expansion of the odorant, but not gustatory, receptor family. Genome res 16:1395–1403
Robinson G (1992) Regulation of division of labor in insect societies. Annu Rev Entomol 37:637
Rogers ME, Jani MK, Vogt RG (1999) An olfactory-specific glutathione-S-transferase in the sphinx moth Manduca sexta. J Exp Biol 202:1625–1637
Sandoz JC, Deisig N, Sanchez MGDB, Giurfa M (2007) Understanding the logics of pheromone processing in the honeybee brain: from labeled-lines to across-fiber patterns. Front Behav Neurosci 1:5
Schulz DJ, Robinson GE (2001) Octopamine influences division of labor in honey bee colonies. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 187:53–61
Scott K, Brady R, Cravchik A, Morozov P, Rzhetsky A, Zuker C, Axel R (2001) A chemosensory gene family encoding candidate gustatory and olfactory receptors in Drosophila. Cell 104:661–673
Slessor KN, Winston ML, Le Conte Y (2005) Pheromone communication in the honeybee (Apis mellifera L.). J Chem Ecol 31:2731–2745
Stabentheiner A, Kovac H (2016) Honeybee economics: optimisation of foraging in a variable world. Sci Rep 6:28339
Sun W, Shen YH, Yang WJ, Cao YF, Xiang ZH, Zhang Z (2012) Expansion of the silkworm GMC oxidoreductase genes is associated with immunity. Insect Biochem Mol Biol 42:935–945
Swanson JAI, Torto B, Kells SA, Mesce KA, Tumlinson JH, Spivak M (2009) Odorants that induce hygienic behavior in honeybees: identification of volatile compounds in chalkbrood-infected honeybee larvae. J Chem Ecol 35:1108–1116
Taylor AJ, Cook DJ, Scott DJ (2008) Role of odorant binding proteins: comparing hypothetical mechanisms with experimental data. Chemosens Percept 1:153–162
Trapnell C, Pachter L, Salzberg SL (2009) TopHat: discovering splice junctions with RNA-SEq. Bioinformatics 25:1105–1111
Trhlin M, Rajchard J (2011) Chemical communication in the honeybee (Apis mellifera L.): a review. Vet Med 56:265–273
Vanderstraete M, Gouignard N, Ahier A, Morel M, Vicogne J, Dissous C (2013) The venus kinase receptor (VKR) family: structure and evolution. BMC Genom 14:1–13
Vogt RG (2005) Molecular basis of pheromone detection in insects. In: Gilbert LI, Iatrou K, Gill SS (eds) Comprehensive molecular insect science. Pergamon, Oxford, pp 753–804
Wickramasinghe VO, Gonzàlezporta M, Perera D, Bartolozzi AR, Sibley CR, Hallegger M, Ule J, Marioni JC, Venkitaraman AR (2015) Regulation of constitutive and alternative mRNA splicing across the human transcriptome by PRPF8 is determined by 5′ splice site strength. Genome Biol 16:201
Winston ML (1987) The biology of the honey bee. Harvard University Press, Cambridge
Woltedji D, Song F, Zhang L, Gala A, Han B, Feng M, Fang Y, Li J (2012) Western honeybee drones and workers (Apis mellifera ligustica) have different olfactory mechanisms than eastern honeybees (Apis cerana cerana). J Proteome Res 11:4526–4540
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol 11:R14
Younus F, Fraser NJ, Coppin CW, Liu JW, Correy GJ, Chertemps T, Pandey G, Maïbèche M, Jackson CJ, Oakeshott JG (2017) Molecular basis for the behavioral effects of the odorant degrading enzyme Esterase 6 in Drosophila. Sci Rep 7:46188
Zhang N, Zhang HJ, Zhao B, Sun QQ, Cao YY, Li R, Wu XX, Weeda S, Li L, Ren S (2014) The RNA-seq approach to discriminate gene expression profiles in response to melatonin on cucumber lateral root formation. J Pineal Res 56:39–50
Acknowledgements
We thank Prof. Shaokang Huang of College of Bee Science, Fujian Agriculture and Forestry University, for helpful suggestions and improvement on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was funded by Educational and scientific research program for young and middle-aged instructor of Fujian province (No. JAT160161) and the earmarked fund for Modern Agro-industry Technology Research System (No. CARS-45-KXJ3).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Data availability
The raw data presented in this article have been deposited in NCBI Short Read Archive (http://www.ncbi.nlm.nih.gov/sra/) and are accessible through SRA accession number: SRP081079.
Additional information
Communicated by S. Hohmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nie, H., Xu, S., Xie, C. et al. Comparative transcriptome analysis of Apis mellifera antennae of workers performing different tasks. Mol Genet Genomics 293, 237–248 (2018). https://doi.org/10.1007/s00438-017-1382-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-017-1382-5