Abstract
Nitrogen starvation (NS) induces sexual development when mating partners are available or enter into quiescent state (G0) in heterothallic background in fission yeast. However, little is known whether the two processes share common signaling molecules or cells defective in the two processes share common transcriptional signatures. To address these questions, we first assessed 77 kinase-deletion strains for NS-induced G0-arrest phenotypes. Our result indicated that 10 out of 77 kinase-deletion strains exhibited defect in G0-arrest, only 3 of which were defective in sexual development based on a previous study, suggesting that the two processes hardly share common signaling components. We subsequently performed transcriptional profiling analysis. Our result indicated that NS-induced transcriptional change was so robust that it prevailed the alteration by individual kinase-deletion alleles. Based on comparison between kinase-deletion strains proficient and deficient in sexual development or G0-arrest, we identified subsets of genes that were associated with sexual development-deficient or G0-arrest-deficient kinase-deletion strains. Multiple pairing analyses allowed grouping of functional related kinases. Furthermore, we showed that Pka1-mediated pathways were required for upregulation of NS-induced genes upon NS and downregulation of the same set of genes under the N-replete conditions. Taken together, our analyses indicate that sexual development and NS-induced G0-arrest are unrelated; and sexual development-deficient and G0-arrest-deficient kinase-deletion strains possess distinct transcriptional signatures. We propose that Pka1 is a key regulator of nitrogen metabolic pathways and Pka1-mediated signaling pathways play roles in regulation of NS-induced genes under both N-depleted and N-replete conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Protein kinase-mediated signaling pathways play an important role in regulation of various biological processes (Hunter and Plowman 1997). Approximately 106 protein kinases are found in the Schizosaccharomyces pombe genome (Bimbo et al. 2005; Wood et al. 2002), 16 of which are essential for viability (Bimbo et al. 2005; Han et al. 2010; Kim et al. 2010). At least half of the dispensable kinases are required for growth fitness under various stress conditions (Bimbo et al. 2005). It has been shown that many kinases are required for sexual development based on iodine vapor staining (Bimbo et al. 2005). Nitrogen starvation (NS) not only induces sexual development of homothallic fission yeast strains or heterothallic strains when mating partners are available, but also induces G0-arrest in a heterothallic background (Yanagida 2009; Yanagida et al. 2011; Yamamoto et al. 1997). It will be interesting to see if the two NS-induced processes share common signaling molecules.
The MAP kinase cascade Wis4/Win1-Wis1-Sty1 plays a pivotal role in regulation of cellular responses to oxidative stresses (Pearce and Humphrey 2001; Shiozaki and Russell 1995). A number of transcription factors (TFs) such as Atf1, Pcr1, and Pap1 are regulated by this signaling pathway (Degols and Russell 1997). Atf1 and Pcr1 are known to form a functional heterodimer (Kanoh et al. 1996; Wahls and Smith 1994). Cells defective for these kinases exhibit hypersensitive phenotypes and fail in transcriptional response to oxidative stresses (Chen et al. 2003; Eshaghi et al. 2010). Besides its role in response to oxidative stresses, Sty1 is thought to be involved in general cell growth through interaction with the protein biosynthesis machinery (Asp et al. 2008). On the other hand, the MAP kinase cascade Byr2-Byr1-Spk1 regulates the onset of sexual development in response to nutrient limitation and pheromone (Neiman 1993).
The major cAMP-dependent protein kinase in fission yeast consists of the catalytic subunit Pka1 and the regulatory subunit Cgs2 (Byrne and Hoffman 1993). Under the nutrient-replete condition, the adenylate cyclase Cyr2 is activated for cAMP synthesis by the G-protein subunit Gpa2 (Byrne and Hoffman 1993). Elevated cAMP concentration disrupts the repression of Pka1 by Cgs2 (Byrne and Hoffman 1993). Pka1 is known to negatively regulate transcription factor Rst2 that regulates a number of mating-specific genes including Ste11 (Gupta et al. 2011; Higuchi et al. 2002). Pka1 is essential in glucose metabolism that ultimately generates ATP to supply energy for cell growth (Byrne and Hoffman 1993).
Transcriptional profiling of fission yeast cells undergoing the onset of sexual development reveals a number of gene clusters with various expression patterns (Mata et al. 2002) and their dependence on various transcription factors (Mata et al. 2007). Alteration of genome-wide transcriptional profiles occurs in cells when challenged with various stress factors (Chen et al. 2003; Gasch et al. 2000). Similarly, transcriptional profile also changes in cells when a gene is deleted, which constitutes a stress to cell (Hughes et al. 2000). A compendium of transcriptional profiles of deletion strains under various growth conditions allows functional discovery (Hughes et al. 2000).
In this study, we want to address if the NS-induced G0-arrest process shares common protein kinases with the sexual development process, given that both processes are induced by NS. In addition, we want to know if kinases defective in NS-induced G0-arrest share common transcriptional signatures with those defective in sexual development. For this reason, we have performed phenotypic assessment for NS-induced G0-arrest and transcriptional profiling of various kinase-deletion strains prior to and after nitrogen starvation (NS) using genome-wide S. pombe expression DNA microarrays (Peng et al. 2005). Our analyses reveal that the NS-induced G0-arrest and sexual development processes hardly share common components and kinase-deletion strains defective in these two processes display distinctive transcriptional signatures. Notably, transcriptional profiling analysis prior to and after NS indicates that the cAMP-dependent Pka1-mediated signaling pathway plays a major role in regulation of the NS-induced genes under both the N-replete and N-depleted conditions.
Materials and methods
Strains and culture manipulation
All protein kinase deletion strains were constructed in our laboratory previously (Bimbo et al. 2005). Deletion strains cyr1Δ, cgs1Δ, and rst2Δ were constructed using the same protocol as described by Bahler et al. (1998). Primers used in deletion strain construction and validation are summarized in Table S1 (Supplemental materials). All strains used in this study are listed in Table 1. Cells were cultivated in Edinburgh Minimal Medium with nitrogen supplement (MM + N) (http://www-bcf.usc.edu/~forsburg/media.html). A fraction of log-phase growing cells (OD ~ 0.5–0.7) was harvested by centrifugation at 2,000 rcf; and the cell pellet was rapidly chilled in liquid nitrogen ready for RNA extraction and microarray profiling analysis as under the nitrogen-replete condition. Another fraction was shifted to the minimal medium without nitrogen supplement (MM-N) after harvested and washed with MM-N. For DNA content analysis, cells were harvested at 24 h after growing in MM-N; for DNA microarray analysis, cells were collected at 60 min after growing in MM-N. We designated a strain as G0-arrest deficient if the population size of 2C-DNA content-containing cells was equal or greater than that of 1C-DNA content-containing cells after NS for 24 h, provided that the 2C-DNA content was not a result of two unseparated daughter cells with 1C-DNA content, that is, percent of septation cells should be less than 10 %.
Fluorescence-activated cell sorting (FACS) and fluorescence microscope analyses
To determine DNA content, cells were fixed in ice-cold ethanol and subsequently digested with RNase A in sodium citrate buffer overnight. After propidium iodide staining, fluorescence intensity of individual cells was measured by the flow cytometer BD FACScan (BD Biosciences, Franklin Lakes, NJ, USA). To monitor physical separation of daughter cells after mitosis, formaldehyde-fixed cells were stained with calcofluor white for cell wall and septum and DAPI for nucleus. Stained cells were examined using the Leica DMRXE fluorescence microscope (Leica, Wetzlar, Germany) equipped with the CoolSNAP HQ2 CCD camera (Roper Scientific, Gottingen, Germany).
RNA extraction and labeling with fluorescent dyes
Total RNA was extracted using the acid-phenol protocol. In brief, acid phenol was added to frozen cell pellets and mixed for 15 min at 65 °C. RNA in aqueous phase was phenol: chloroform extracted and isopropanol precipitated. The resulting total RNA was used as template for cDNA synthesis in presence of Cy3- or Cy5-coupled dUTP (Invitrogen Corporation, Carlsbad, CA, USA). Fluorescence-labeled cDNA was hybridized with the spotted S. pombe oligonucleotide-based microarray (Peng et al. 2005). Microarray slides were scanned using a GenePix scanner (Axon Instruments, Union City, CA, USA) at 635 and 532 nm wavelengths at a resolution of 10 μm using GenePix Pro software (Axon Instruments). High quality data were acquired and LOWESS normalized.
Expression microarray data analysis
Microarrays were selected for analysis only when missing features were <5 % of total features (i.e., 4,929 genes) except for bc119.07_60mor after NS (whose missing features was ~6 %). To this end, transcriptional profiles of the 68 kinase-deletion strains prior to and after NS were obtained for correlation analysis. To determine correlation coefficient between profiles (i.e., kinase-deletion strains prior to or after NS), 500 genes (i.e., 10 % of the genome) with the top-ranked standard deviation across all profiles were selected. Hierarchical clustering analysis for best neighbors was performed using Cluster analysis and visualization software (Eisen et al. 1998) or Gene Spring GX version 10 (Agilent Technologies, Inc., Santa Clara, CA) with uncentered similarity metrics and average linkage. In correlation analysis with multiple-pairing visualization, Pearson’s correlation coefficient was used. It was calculated using the following equation:
Correlation between profiles of two kinase-deletion strains was considered when the correlation coefficient was 0.7 or greater. Cytoscape software (Shannon et al. 2003) was applied for visualizing correlations between profiles of kinase-deletion strains. In Cytoscape, a profile of kinase-deletion strains prior to or after NS represents a node; and correlation (i.e., r > 0.7) defines an edge. According to numbers of edges, anode can link as many other nodes. We defined a group of kinase-deletion strains based on their profiles when it consisted of at least five nodes, each of which should contain at least three edges connecting to other nodes within the same group.
To identify subset of genes specifically associated with the sexual development-deficient or G0-arrest-deficient kinase-strains, kinase-deletion strains defective in sexual development or G0-arrest were grouped. Deletion strains lacking assessment data on one of the two phenotypes (i.e., I2 vapor staining and G0-arrest) were excluded in this analysis. Kinase-deletion strains proficient in both sexual development and G0-arrest were grouped as the proficient kinase group.
Statistical analyses
The t test is used in standard analysis of microarray (SAM) (Tusher et al. 2001) and assays for differences between profiles of kinase-deletion strains prior to and after NS. Binomial test is used in assays for non-random distribution such as enrichment of gene-sets or gene functions (i.e., GO-term association).
Microarray datasets
Original 148 microarray datasets used in this study can be obtained from the GEO database with the accession number GSE 47544.
Results
Kinases involved in sexual development are not necessarily required for NS-induced G0-arrest
We previously showed that 23 out of 77 protein kinase deletion-strains exhibited defects in sexual development based on the iodine vapor staining, indicating that these kinases are involved in regulation of mating, conjugation, meiosis, and/or sporulation (see Table 1; Bimbo et al. 2005). Nitrogen starvation (or NS) induces sexual development in homothallic strains or heterothallic strains when mating partners are available (Yanagida et al. 2011). On the other hand, NS induces entry into a dormant state or at G0-phase in heterothallic strains when mating partners are not available (Yanagida et al. 2011). To investigate whether the sexual development-defective kinase-deletion strains would also exhibit defect in NS-induced G0-arrest (hereafter G0-arrest), all kinase-deletion strains were subjected to assays for competence of G0-arrest. To this end, log-phase growing cells in growth medium supplemented with nitrogen were transferred into medium without nitrogen for 24 h. Cells prior to (i.e., +N) and after (i.e., −N) NS for 24 h were analyzed for DNA content using a fluorescence-activated cell sorter (FACS). All of the actively growing wild-type cells in N-replete medium exhibited a 2C-DNA content (or two copies of the genome) (Fig. 1a). On the other hand, majority (i.e., >70 %) of the wild-type cells displayed a 1C-DNA content after NS for 24 h, indicating a G0-arrest.
Phenotypic assessment of kinase deletion strains for defect in NS-induced G0-arrest. Profiles of DNA contents prior to and after NS (+N vs. −N) are shown at the top of each panel. Fluorescence images of cells stained with DAPI (for DNA) and Calcofluor white (for cell wall) plus a scale bar of 10 μm are shown at the bottom. Arrow indicates the G1-cell population (with 1C-DNA content) that is smaller than the G2-cell population (with 2C-DNA content) under the N-depleted condition. Arrowhead indicates the G1-cell population that is larger than the G2-cell population. a Characteristic of the wild-type strain. b–k DNA-content profile and cell morphology of the individual kinase deletion strains are shown. Individual strains are indicated on top of the panel. l Venn diagram showing the relationship between kinase-deletion strains with defect in sexual development (or I2 staining) and G0-arrest
DNA-content analysis indicated that 10 out of 77 kinase-deletion strains showed that less than half of the NS-induced cells exhibiting 1C-DNA contents, suggesting a defect in G0-arrest compared to that of wild type (Fig. 1b–k, see Table S2 in Supplemental materials for all strains tested). Reduced number of 1C-DNA content-containing cells could also be attributed to defects in physical separation of daughter cells after mitosis. To ascertain that cells with 2C-DNA contents after NS were not a result of accumulation (e.g., >10 %) of two unseparated daughter cells or septum-containing cells, we visually examined cells under fluorescence microscope after staining with DAPI (for DNA) and calcofluor white (for cell wall) (see Fig. 1). No apparent accumulation of the septum-containing cells was found in the NS-induced deletion strains, indicating that most of the 2C-DNA content-containing cells were bona fide G2 cells. Hence, the 10 protein kinase-deletion strains were truly defective of NS-induced G0-arrest.
Of 77 kinase-deletion strains assessed in both the iodine vapor staining and copy numbers of DNA content, three strains byr1Δ, ssp1Δ, and sty1Δ exhibited defects in both sexual development and NS-induced G0-arrest (Fig. 1m). This result suggested that signaling pathways involved in regulation of sexual development were not necessarily required for the NS-induced G0-arrest, or vice versa.
Transcriptional profile of pka1Δ cells is hardly altered by NS compared to other kinase-deletion strains
A compendium of transcriptional profiles or signatures of gene-deletion strains under various growth conditions is useful for function discovery (Hughes et al. 2000). Hence, we wanted to investigate whether there was a unique transcriptional signature in kinase-deletion strains defective in iodine vapor staining or sexual development (hereafter sexual development) and G0 arrest. To address this question, we performed transcriptional profiling analyses of cells prior to (i.e., 0 min) and after NS for 60 min (i.e., 60 min) using the spotted S. pombe genome-wide 50-mer oligonucleotide-based DNA microarray containing 10 thousand features (Peng et al. 2005). A common reference (i.e., pooled wild-type cultures under the N-replete condition) was used in all microarray analyses.
To assess correlations between transcriptional profiles of the kinase-deletion strains prior to and after NS, 500 top-ranked highly variable genes (i.e., 10 % of the genome) based on the standard deviation across all 138 microarrays (i.e., 68 kinase strains and a wild type under N-replete and N-depleted conditions) were selected (Table S3 in Supplemental materials). Pearson correlation coefficients between profiles of the kinase-deletion strains prior to NS were moderate (i.e., median r = 0.34) (Fig. 2a). On the other hand, very high correlation coefficients (e.g., median r = 0.95) were found between profiles (i.e., based on a common reference) of the kinase-deletion strains after NS (Fig. 2b). Given that these profiles were based on a common reference, high correlation coefficients between NS-induced profiles would suggest that NS-induced transcriptional response were so robust that it overwrote the alteration induced by a kinase deletion allele in cells. Profiles of all kinase-deletion strains prior to NS hardly showed any correlation with profiles of the same strains after NS (i.e., median r = 0.12) except for that of pka1Δ (i.e., r = 0.71) (Fig. 2c). These results indicate that transcriptional profile of pka1Δ is hardly changed by NS when compared to the other kinase-deletion strains.
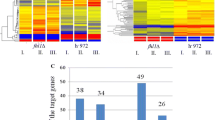
Correlation analysis of transcriptional profiles of 68 protein kinase-deletion strains prior to and after NS. a Histogram of correlations between kinase-deletion strains prior to NS. X-axis shows the binned correlation coefficients; Y-axis shows the number of kinase-deletion strain (kin) pairs (self pairs are not included). Numbers of pairs in each bin are indicated on top of the bar. b Histogram of correlations between kinase strains after NS. The display is identical to (a). c Histogram of correlations between kinase-deletion strains prior to and after NS. The display is identical to (a). d The Venn diagram shows the NS-induced genes (at 60 m) in this study enriched for NS and pheromone induced genes. NS-induced genes are based on the analysis of wild-type cells using SAM method
When comparative analysis of the NS-induced profiles of the kinase-deletion strains was performed based on the normalization by the self-0 min profile (i.e., the profile of same strains prior to NS) but not the common reference profile, correlation coefficients between the self-0 min-normalized NS-induced profiles of all kinase-deletion strains were greater than 0.7 except for that between pka1Δ and byr1Δ (Figure S1 in Supplemental materials), consistent with the correlation analysis based on a common reference.
To investigate whether the 500 highly variable genes based on the standard deviation across all 138 microarrays (i.e., 68 kinase deletion strains and a wild type under N-replete and N-depleted conditions) enriched for differentially transcribed genes upon NS stress, significant analysis of microarray (SAM) (Tusher et al. 2001) was performed by using triplicates of transcriptional profiles of wild-type cells prior to and after NS. To this end, 98 induced and 124 repressed genes in response to NS were identified according to a cutoff of fold change >4 and p value <0.001 (Table S4 in Supplemental materials). We noted that all NS-responsive genes were found in the 500 highly variable genes except for 4 (i.e., p value <2.2E–16), indicating that highly variable genes enrich for NS-responsive genes.
Gene ontology analysis indicated that response to stress (GO:0006950) and nitrogen compound transport (GO:0071705) were significantly enriched in the NS-induced genes (i.e., p value <2E−04). On the other hand, translation (GO:0006412) was greatly enriched in the NS-repressed genes (i.e., p value <2.2E−16). In addition, the NS-induced genes enriched for pheromone-induced “continuous” and “transient” genes (i.e., p value <2.2E−16) based on the study by Mata et al. (2002) (Fig. 2d). These results indicated that the NS-induced transcriptional profiling is of good quality and consistent with the previous report.
Genes associated with the sexual development-deficient kinase-deletion strains are different from those associated with the G0-arrest-deficient ones
Hierarchical clustering analysis (Eisen et al. 1998) is a common method for finding best neighbors from a given set of samples based on similarities between transcriptional profiles using a set of 500 highly variable genes (Fig. 3a). We defined that a cluster of kinase-deletion strains would contain at least five conditions (i.e., kinase-deletion strains prior to or after NS) with a minimal (average) correlation coefficient of 0.7 (see “Materials and methods”). As a result, five clusters of kinase-deletion strains were identified. We noted that no cluster was found to enrich for profiles of the sexual development-deficient or G0-arrest-deficient kinase-deletion strains, suggesting that the sexual development-deficient or G0-arrest-deficient kinase-deletion strains share no apparent transcriptional signatures.
Distinct subsets of genes associated with the sexual development-deficient and G0-arrest-deficient kinase-deletion strains. a Hierarchical clustering analysis of transcriptional profiles of 86 kinase deletion strains prior to and after NS. Dendrogram of the clustering analysis is shown on the left. Five clusters with minimal correlation of 0.7 are indicated. Transcription profiles are shown in the middle. Genes are shown by columns and kinase strains are shown by rows. Red, black, green indicate the increased, unchanged, and decreased transcription levels compared to a common reference, respectively. Kinase strains prior to (0 m) and after (60 m) NS are shown on the right. A color key is shown at the bottom. b The heatmap plot shows correlation coefficients between transcriptional profiles of kinase strains. The dendrogram on the left is identical to a. Correlation coefficient is indicated in red when it is 0.7 or greater or in black when it is less than 0.7. Hierarchical clusters are indicated by white dash lined-box. c A magnified view of heatmap plot in (a) shows correlations between pka1 and other kinases after NS. Kinase-deletion strains are indicated when its correlation coefficient with pka1∆ is less than 0.7. Red indicates kinases defective in sexual development based on the Iodine staining (Bimbo et al. 2005). d Profile of genes associated with the sexual development-deficient kinase-deletion strains (or I2 genes). Boxplot shows the profile of I2 genes in the sexual development-deficient (deficient) and -proficient (proficient) kinase-deletion strains. Triple asterisks indicate p value <0.001. e Profile of genes associated with the G0-arrest-deficient kinase-deletion strains (or G0 genes). Boxplot shows that profile of G0 genes in the G0-arrest-deficient (deficient) and -proficient (proficient) kinase-deletion strains. Top and bottom panels show the upregulated and downregulated genes upon NS based on the wild-type profile
Multiple-pairing analysis using correlation matrix (i.e., Pearson’s correlation coefficient cutoff = 0.7) revealed detailed relationships between profiles of kinase-deletion strains prior to and after NS (Fig. 3b) (see “Materials and methods”). It was clear that the NS-induced profiles of all strains tested were highly correlated with one another except for pka1Δ. Eleven out of 86 kinase-deletion strains whose NS-induced profiles showed no correlation (i.e., r < 0.7) with that of pka1Δ (Fig. 3c). Notably, 9 out of 11 pka1Δ profile-unrelated kinase-deletion strains were defective in sexual development, which was 2.5-fold higher than that of background (i.e., 0.82 vs. 0.32; p value <0.001). This result implied that the sexual development-deficient kinase-deletion strains possessed a unique subset of genes whose transcriptional levels differed from those of sexual development-proficient ones.
To define the subset of genes specific to the sexual development-deficient kinase-deletion strains, we performed comparative analysis between transcriptional profiles (i.e., after NS) of the sexual development-deficient and -proficient kinase-deletion strains (i.e., 20 vs. 37 strains) (see “Materials and methods”). In this case, 3 out of 20 sexual development-deficient kinase-deletion strains were defective in NS-induced G0-arrest. On the other hand, all 37 sexual development-proficient deletion strains showed no deficiency in G0-arrest. As a result, 45 out of 500 highly variable genes whose levels in the sexual development-deficient kinase-deletion strains were significantly different from those in the sexual development-proficient kinase-deletion strains based on a cutoff of fold change >20 % and t test p value <0.05 (Table 2). We noted that 39 of the 45 sexual development-deficient kinase-deletion strain-specific genes were upregulated upon NS in wild-type cells. It was clear that in the sexual development-deficient kinase-deletion strains, the subset of 39 genes failed to reach the fully upregulated level as seen in sexual development-proficient kinase-deletion strains (Fig. 3d) (i.e., median level 1.40 vs. 1.76 in log2 scale, p value = 4.79E−14). Many of the genes showed to be involved in carbohydrate metabolism, sexual differentiation, and stress-response, suggesting that the upregulation of these genes to the wild-type level is likely to be associated with the normal sexual development upon NS (see Table 2).
By using the same approach, we identified a subset of 190 genes whose transcription levels in the G0-arrest-deficient kinase-deletion strains were significantly different from those in the G0-arrest-proficient kinase-deletion strains using the cutoff of fold-change >20 % and p value <0.05 (i.e., 7 vs. 37 kinase-deletion strains). We noted that only 14 out of 190 genes were present in the subset of 45 genes specific to the sexual development-deficient kinase-deletion strains, indicating that the 2 sets of genes are unrelated (i.e., binomial test p value >0.05).
Of the 190 genes, 52 were upregulated and 138 were downregulated according to the profile of wild-type cells after NS. We found that these genes were either not fully upregulated (Fig. 3e, upper panel) or not completely downregulated (Fig. 3e, lower panel) in the G0-arrest-deficient kinase-deletion strains when compared to those of G0-arrest-proficient kinase-deletion strains (i.e., for upregulated genes: median level 1.68 vs. 2 in log2 scale, p value = 1.33E−12; for downregulated genes: median level −1.46 vs. −2.1 in log2 scale, p value <2.2E−16). Many genes defective in upregulation upon NS were mainly involved in carbohydrate metabolism, nucleic acid metabolism, response to stress, and transport. On the other hand, genes defective in downregulation after NS showed to be primarily involved in amino acid metabolism and protein biosynthesis (Table 3). These results are consistent with the notion that in the NS-induced G0 phase, protein biosynthesis is generally diminished and carbohydrate metabolism and stress-response dominate (Marguerat et al. 2012; Yanagida 2009).
Genes associated with multiple-pairing groups of the kinase-deletion strains
The multiple-pairing analysis using the Cytoscape (Shannon et al. 2003) was performed to connect the correlated profiles (i.e., correlation coefficient r ≥ 0.7) of kinase-deletion strains prior to and after NS (see “Materials and methods”). To this end, NS-induced profiles of all kinase-deletion strains were linked with one another except for pka1∆ (Fig. 4a). We noted that profiles of 11 kinase-deletion strains, 9 of which were defective in sexual development, were not linked to pka1Δ (i.e., p value <0.001) (see Fig. 3c). On the other hand, the profile of pka1Δ prior to NS was connected through the profile of pka1Δ after NS to profiles of all kinase-deletion strains after NS including that of wild type, consistent with the result that profile of pka1Δ is hardly altered after NS compared to profile changes of all other kinase-deletion strains (see Fig. 2c). Hence, all kinase-deletion strains after NS in cluster 1 plus pka1Δ prior to NS formed the group A (see Fig. 4a).
Specific subsets of genes associated with individual multiple-paring groups of kinase-deletion strains. a Correlation map of kinase-deletion strains. A line (or an edge) connects two kinase-deletion strains when their correlation coefficient is 0.7 or greater. Gene groups A, B, C, and D are circled with dash lines as indicated. b–d Profile of genes associated to the kinase-deletion strains in group B (B), C (C), and D (D). The display of the boxplot is identical to Fig. 3d
By using the same approach, three out of four remaining clusters derived from the hierarchical clustering analysis (see Fig. 3a) were found to form individual groups (i.e., a group was consistent of at least five nodes, each of which contained three edges or more with other nodes in the same group) (see Fig. 4a). Group B was comprised of most of the kinase-deletion strains in cluster 2 and ac890.03Δ in cluster 4. We noted that the group B enriched for kinase-deletion strains required for growth fitness under brefeldin A (BFA) stress (Bimbo et al. 2005) (i.e., 3.75-fold above background; p value = 0.033). While group C consisted of all kinase-deletion strains in cluster 3, group D contained only a subset of kinase-deletion strains in cluster 4. Kinase-deletion strains in cluster 5 were not qualified for a group.
We next examined subsets of genes whose transcription levels in the individual multiple-pairing groups B, C, or D of kinase-deletion strains were significantly different from the background (i.e., the non-grouped kinase-deletion strains) using a cutoff of fold change >25 % and t test p value <0.05. To this end, 79, 31, and 29 genes out of the 500 highly variable genes were found to be unique to the kinase-deletion strains in groups B, C, and D, respectively.
It appeared that genes specifically associated with the group B kinase-deletion strains displayed a slightly lower level than that in the non-grouped kinase-deletion strains (i.e., median −0.3 vs. −0.15, p value <0.001) (Fig. 4b). We found that genes associated with group B were involved in amino acid metabolism, carbohydrate metabolism, cell wall biogenesis, proteolysis, protein biosynthesis, stress-response, and transport (Table 4). It has been shown that genes with compromised function in intracellular transport, membrane biogenesis and/or cell wall biosynthesis may lead to brefeldin A sensitivity (Murén et al. 2001). Hence, it is possible that reduced expression levels of genes involved in transport and cell wall biogenesis may be associated with the brefeldin A sensitivity in this group B kinase-deletion strains.
Similarly, genes associated with the group C kinase-deletion strains showed a reduced level compared to that of the non-grouped kinases (Fig. 4c). On the other hand, genes associated with the group D kinase-deletion strains exhibited an elevated level compared to the background (Fig. 4d). It appeared that individual kinase-deletion groups were associated with genes of diverse function including amino acid metabolism, carbohydrate metabolism, proteolysis, stress-response, translation, and transport. Hence, a group of kinase-deletion strains based on multiple-paring analysis may not necessarily exhibit a readily scored phenotype.
cAMP-dependent Pka1-mediated signaling pathway plays a major role in transcriptional regulation of the NS-induced genes prior to and after NS
To investigate whether other factors involved in cAMP-dependent Pka1-mediated pathway would play a role in transcriptional regulation of the NS-induced genes, we examined the levels of the 97 NS-induced genes in strains containing a deletion allele of cyr1 (encoding adenylate cyclase), cgs1 (encoding a negative regulatory subunit of Pka1), pka1, and rst2 (encoding a zinc-finger transcription factor negatively regulated by Pka1). In this analysis, we also included strains containing a deletion allele of genes involved in ROS-activated MAP kinase signaling pathway and in pheromone-activated MAP kinase signaling pathway (Fig. 5a).
Transcriptional profiling of strains containing a deleted component involved in various signaling pathway. a Schematic drawing of the cAMP-dependent Pka1-mediated signaling pathway, the ROS-activated MAPK Sty1-mediated signaling pathway, and the NS/pheromone-activated MAPK Spk1 signaling pathway. Circle, square, and diamond indicate kinase, other factor, and transcription factor, respectively. Arrow and bar indicate activation and repression, respectively. b Hierarchical clustering analysis of the 97 NS-induced genes. Dendrogram of the cluster is shown on the top, where two clusters α and β are indicated. To contrast the level of cluster α genes in cyr1∆ and pka1∆ to that of other strains, the color keys for phasogram before and after NS are difference are indicated. c Expression of the NS-induced cluster α genes under both N-replete and N-depleted conditions is Pka1-dependent. Box plot shows the transcriptional profiles of the cluster α genes in various deletion strains indicated. Median level difference (∆M) to that of wild type is shown on the right. Significantly upregulated and downregulated profiles are indicated in orange and blue colors, respectively. d Expression of the cluster β genes is Pka1-independent. The display is identical to (c)
Microarray analyses of the deletion strains prior to and after NS were performed as those of kinase-deletion strains using a common reference (see “Materials and methods”). Profiles prior to NS were normalized using the wild-type-0 min profile for analysis of gene-level changes in various deletion strains under the N-replete condition. On the other hand, profiles after NS were normalized using the self-0 min profile for analysis of transcription-level alteration in individual deletion strains under N-depleted condition. Hierarchical clustering analysis of the 97 NS-upregulated genes revealed 2 gene clusters, the clusters α and β (i.e., correlation coefficient >0.7, gene number >10) (Fig. 5b). The cluster α contained 83 genes, most of which showed elevated and reduced levels in profiles of cyr1Δ and pka1Δ prior to and after NS, respectively (Fig. 5B, see arrows). However, this pattern was absent in the Cluster β.
Among the strains containing a deletion allele of components involved in the cAMP-dependent Pka1-mediated pathway, it was clear that the level (i.e., the median level) of the cluster α genes in cyr1∆ and pka1∆ but not cgs1∆ or rst2∆ were elevated under the N-replete condition compared to that of wild type (i.e., fold change >0.2 in log2 scale, p value <0.05) (Fig. 5c). Whereas the same set of genes whose levels were reduced in cyr1∆ and pka1∆ but not cgs1∆ or rst2∆ upon NS compared to that of wild type (i.e., fold change >0.2 in log2 scale, p value <0.05). These results indicated that a high level of cAMP is essential for activation of Pka1 that repressed the NS-induced genes under N-replete conditions. Hence, the cAMP-dependent Pka1-mediated signaling pathway is not only involved in the repression of the Cluster α genes under the N-replete condition but also participated in upregulation of these genes under the N-depleted condition. We noted that many genes in the cluster α were involved in amino acid metabolism, carbohydrate metabolism, nucleic acid metabolism, sexual development, stress-response, and transport (Table 5).
With the same cutoff, we found that levels of the cluster α genes were reduced in sty1Δ compared to that of wild type under the N-replete condition, suggesting that a component involved in the ROS-activated signaling pathway plays a role in maintaining the wild-type transcription level of the cluster α genes under the N-replete condition. This is consistent with the notion that Sty1 is involved in regulation of protein biosynthesis for cell growth (Asp et al. 2008). On the other hand, levels of the cluster α gene in byr1∆ were not fully upregulated upon NS compared to that of wild type, indicating that a component of the pheromone-activated signaling pathway plays a role in regulation of the cluster α genes upon NS, but not in cells prior to NS.
We noted that the cluster β, a small subset (i.e., 13 out of 97 genes) of the NS-induced genes, were upregulated under the N-replete condition in all deletion strains tested except for atf1Δ compared to that of wild type, suggesting that genes in this subset were sensitive to or induced by a deletion allele in a Atf1-dependent manner (Fig. 5d). Upon NS, this subset of genes in all deletion strains except for wis4∆ and atf1∆ displayed a noticeable upregulation (i.e., >0 in logs scale). Though the level of upregulation was not as high as that of wild type, the difference was not statistically significant (i.e., p value >0.05). These results suggested that Atf1 and Wis4 are most likely involved in the upregulation of these genes under the N-depleted condition. It was noted that the cluster β consisted of all genes involved in stress-response besides the function-unknown genes (see Table 5). It was possible that these genes are induced by not only the external stress such as NS but also an internal stress such as a gene deletion in an Atf1-dependent manner.
Discussion
Protein kinases are signaling molecules that play key roles in mediating signals that regulates many biological processes (Hunter and Plowman 1997). Many protein kinases are required for growth fitness under stress conditions (Bimbo et al. 2005). NS-stress is known to induce sexual development in yeast when mating partners avail. On the other hand, it also G0-arrest in heterothallic background when mating partners are not available (Yamamoto et al. 1997; Yanagida 2009; Yanagida et al. 2011). In this study, we show that the two NS-induced processes hardly share common signaling molecules (see Fig. 1), implying that cells are equipped with two independent mechanisms for survival in case of nutrient deprivation.
Transcriptional profiling is a powerful tool for quantitative assessment of phenotypes in various mutant strains (Hughes et al. 2000). We show that NS-induced alteration of transcriptional profiles is highly robust that prevails change of profiles induced by a gene deletion (see Fig. 2). Dramatic changes of transcriptional profiles may reflect the life cycle change of cells (i.e., from mitotic cycle to meiotic cycle) and its accompanied morphology change. This makes NS-induced profiles undistinguishable between different kinase-deletion mutants and therefore unsuitable for phenotypic assessment of mutants (see Fig. 4a, group a). On the other hand, profiles of individual kinase-deletion strains prior to NS are unique (i.e., with low levels of correlation). By using the profiles of kinase-deletion strains prior to NS, it is possible to discern difference between signatures of protein kinases defective in sexual development and G0-arrest (see Fig. 3d, e), supporting the notion that regulatory components of the two pathways are hardly shared.
Hierarchical clustering is one of the most widely used tools in analysis of transcriptional profiles (Eisen et al. 1998). This method allows identification of best neighbors to one cluster of conditions (i.e., deletion strains with a common defect) but not two or more. Given that many protein kinases-deletion strains exhibit multiple defects (Bimbo et al. 2005) or they may well belong to multiple conditions, the best neighbor approach becomes hard to reflect this fact. By using the multiple-pairing approach (see “Materials and methods”) visualized by Cytoscape (Shannon et al. 2003), we are able to show not only various kinase-deletion strains within individual groups but also the ones connecting with different groups (see Fig. 4a). Due to the current limited information on phenotypes of kinase-deletion strains, not all groups of kinase-deletion strains can be linked to common phenotypes (see Fig. 4a). Nevertheless, further phenotypic assessment of these deletion strains should provide a clue in the future.
Pka1-mediated signaling pathways are known to play a major role in regulation of glucose metabolism and active cell growth (Byrne and Hoffman 1993). In this study, we show that transcriptional profile of the pka1-deletion strain prior to NS displays the most deviated pattern from that of all other kinase-deletion strains (see Figs. 3a, 4a). Notably, profile of the pka1-deletion strain experiences the least change when compared to that of all other kinase-deletion strains (see Figure S1), implying that Pka1-mediated signaling pathways plays a role in maintaining a transcriptional profile for active growth. Consistent with this, NS-induced genes fail to be upregulated in cells without Cyr1 or Pka1 after NS. On the other hand, the same set of NS-induced genes is unable to be repressed in cyr1-deletion or pka1-deletion cells prior to NS.
In conclusion, our phenotypic assessment indicates that the NS-induced G0-arrest process hardly share common protein kinases with the sexual development process. Furthermore, our transcriptional profiling analysis of many kinase-deletion strains reveals that the distinct subsets of genes are associated with the sexual development-deficient and G0-arrest-deficient kinase-deletion strains, suggesting that these genes play a role in regulation of sexual development or G0-arrest. Moreover, our profiling analysis of the deletion strains with disrupted components involved in various signaling pathways demonstrates that the cAMP-dependent Pka1-mediated signaling pathway plays a role in activation of the NS-induced genes upon NS stress and repression of the same set of genes under the N-replete condition. Our data provide insight into roles of individual protein kinases played in regulation of NS-induced transcriptional response in fission yeast.
Abbreviations
- NS:
-
Nitrogen starvation
References
Asp E, Nilsson D, Sunnerhagen P (2008) Fission yeast mitogen-activated protein kinase Sty1 interacts with translation factors. Eukaryot Cell 7:328–338
Bahler J, Wu JQ, Longtine MS, Shah NG, McKenzie A 3rd et al (1998) Heterologous modules for efficient and versatile PCR-based gene targeting in Schizosaccharomyces pombe. Yeast 14:943–951
Bimbo A, Jia Y, Poh SL, Karuturi RK, den Elzen N et al (2005) Systematic deletion analysis of fission yeast protein kinases. Eukaryot Cell 4:799–813
Byrne SM, Hoffman CS (1993) Six git genes encode a glucose-induced adenylate cyclase activation pathway in the fission yeast Schizosaccharomyces pombe. J Cell Sci 105(Pt 4):1095–1100
Chen D, Toone WM, Mata J, Lyne R, Burns G et al (2003) Global transcriptional responses of fission yeast to environmental stress. Mol Biol Cell 14:214–229
Degols G, Russell P (1997) Discrete roles of the Spc1 kinase and the Atf1 transcription factor in the UV response of Schizosaccharomyces pombe. Mol Cell Biol 17:3356–3363
Eisen MB, Spellman PT, Brown PO, Botstein D (1998) Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci USA 95:14863–14868
Eshaghi M, Lee JH, Zhu L, Poon SY, Li J et al (2010) Genomic binding profiling of the fission yeast stress-activated MAPK Sty1 and the bZIP transcriptional activator Atf1 in response to H2O2. PLoS One 5:e11620
Gasch AP, Spellman PT, Kao CM, Carmel-Harel O, Eisen MB et al (2000) Genomic expression programs in the response of yeast cells to environmental changes. Mol Biol Cell 11:4241–4257
Gupta DR, Paul SK, Oowatari Y, Matsuo Y, Kawamukai M (2011) Multistep regulation of protein kinase A in its localization, phosphorylation and binding with a regulatory subunit in fission yeast. Curr Genet 57:353–365
Han TX, Xu XY, Zhang MJ, Peng X, Du LL (2010) Global fitness profiling of fission yeast deletion strains by barcode sequencing. Genome Biol 11:R60
Higuchi T, Watanabe Y, Yamamoto M (2002) Protein kinase A regulates sexual development and gluconeogenesis through phosphorylation of the Zn finger transcriptional activator Rst2p in fission yeast. Mol Cell Biol 22:1–11
Hughes TR, Marton MJ, Jones AR, Roberts CJ, Stoughton R et al (2000) Functional discovery via a compendium of expression profiles. Cell 102:109–126
Hunter T, Plowman GD (1997) The protein kinases of budding yeast: six score and more. Trends Biochem Sci 22:18–22
Kanoh J, Watanabe Y, Ohsugi M, Iino Y, Yamamoto M (1996) Schizosaccharomyces pombe gad7+ encodes a phosphoprotein with a bZIP domain, which is required for proper G1 arrest and gene expression under nitrogen starvation. Genes Cells 1:391–408
Kim DU, Hayles J, Kim D, Wood V, Park HO et al (2010) Analysis of a genome-wide set of gene deletions in the fission yeast Schizosaccharomyces pombe. Nat Biotechnol 28:617–623
Marguerat S, Schmidt A, Codlin S, Chen W, Aebersold R et al (2012) Quantitative analysis of fission yeast transcriptomes and proteomes in proliferating and quiescent cells. Cell 151:671–683
Mata J, Lyne R, Burns G, Bahler J (2002) The transcriptional program of meiosis and sporulation in fission yeast. Nat Genet 32:143–147
Mata J, Wilbrey A, Bahler J (2007) Transcriptional regulatory network for sexual differentiation in fission yeast. Genome Biol 8:R217
Murén E, Oyen M, Barmark G, Ronne H (2001) Identification of yeast deletion strains that are hypersensitive to brefeldin A or monensin, two drugs that affect intracellular transport. Yeast 18:163–172
Neiman AM (1993) Conservation and reiteration of a kinase cascade. Trends Genet 9:390–394
Pearce AK, Humphrey TC (2001) Integrating stress-response and cell-cycle checkpoint pathways. Trends Cell Biol 11:426–433
Peng X, Karuturi RK, Miller LD, Lin K, Jia Y et al (2005) Identification of cell cycle-regulated genes in fission yeast. Mol Biol Cell 16:1026–1042
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT et al (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504
Shiozaki K, Russell P (1995) Cell-cycle control linked to extracellular environment by MAP kinase pathway in fission yeast. Nature 378:739–743
Tusher VG, Tibshirani R, Chu G (2001) Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci USA 98:5116–5121
Wahls WP, Smith GR (1994) A heteromeric protein that binds to a meiotic homologous recombination hot spot: correlation of binding and hot spot activity. Genes Dev 8:1693–1702
Wood V, Gwilliam R, Rajandream MA, Lyne M, Lyne R et al (2002) The genome sequence of Schizosaccharomyces pombe. Nature 415:871–880
Yamamoto M, Imai Y, Watanabe Y (1997) Mating and sporulation in Schizosaccharomyces pombe. In: Pringle JR, Broach JB, Jones EW (eds) Molecular and cellular biology of the yeast Saccharomyces. Cold Spring Harbor Laboratory Press, New York, pp 1037–1106
Yanagida M (2009) Cellular quiescence: are controlling genes conserved? Trends Cell Biol 19:705–715
Yanagida M, Ikai N, Shimanuki M, Sajiki K (2011) Nutrient limitations alter cell division control and chromosome segregation through growth-related kinases and phosphatases. Philos Trans R Soc Lond B Biol Sci 366:3508–3520
Acknowledgments
The authors would like to thank X. Peng and R. Krishnan for manufacturing the S. pombe 50-mer oligonucleotide-based DNA microarrays and L. Zhu and R. K. M. Karuturi for assistance in bioinformatics analysis. This work is initiated in GIS, Singapore and completed in the Zhejiang University, Zhejiang, China.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by T. Ito.
Electronic supplementary material
Below is the link to the electronic supplementary material.
438_2014_966_MOESM5_ESM.pdf
Correlation analysis of the self-0min normalized profiles of various kinase-deletion strains after NS (pdf-format) (PDF 118 kb)
Rights and permissions
About this article
Cite this article
Liu, J., Jia, Y., Li, J. et al. Transcriptional profiling analysis of individual kinase-deletion strains of fission yeast in response to nitrogen starvation. Mol Genet Genomics 290, 1067–1083 (2015). https://doi.org/10.1007/s00438-014-0966-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-014-0966-6