Abstract
To compare phase contrast microscopy (PCM) of unstained slides for the detection of Cryptosporidium spp. oocysts with a commercially available enzyme immunoassay (EIA) for the detection of cryptosporidial antigen in human stool samples, we prospectively analysed by both methods 463 fresh human stool samples obtained from diarrhoeic patients between July and October 2014. Compared with the EIA, the sensitivity, specificity, positive and negative predictive value of PCM were 88.9 % (95 % confidence interval (CI), 66.0–98.1 %), 100 % (95 % CI, 99.0–100 %), 100 % (95 % CI, 77.3–100 %) and 99.6 % (95 % CI, 98.3–100 %), respectively. Additionally, we retrospectively examined with PCM 65 fixed stool samples that had been collected in 2010 from mostly asymptomatic Rwandan children <5 years of age; 14 of these samples had previously yielded positive results with a highly sensitive real-time (RT)-PCR. PCM detected cryptosporidia in 5/14 RT-PCR-positive samples, and notably, also in one of 51 RT-PCR-negative samples, which was subsequently confirmed by acid-fast staining. Positive and negative percent agreement of PCM with RT-PCR were 35.7 % (95 % CI, 16.2–61.4 %) and 98.0 % (95 % CI, 88.7–100 %), respectively. Positive PCM results were associated with higher RT-PCR cycle threshold values (p = 0.044). In conclusion, PCM offers a highly specific, undemanding and inexpensive method for the laboratory diagnosis of acute human cryptosporidiosis independent of the causative Cryptosporidium species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cryptosporidia are world-wide spread and may cause potentially severe gastroenteritis in animals and man. Cryptosporidium parvum and Cryptosporidium hominis are the most prevalent pathogenic species in humans; C. hominis is mainly transmitted between humans by the faecal-oral route or more rarely acquired by inhalation while infections with C. parvum are considered zoonotic with bovines constituting the main reservoir (Shirley et al. 2012). Since outbreaks have been reported (and Cryptosporidium spp. caused more outbreaks in 2004–2010 than other protozoan parasites; Baldursson and Karanis 2011), cryptosporidiosis is a notifiable disease in many industrialized countries. In the USA, 10,000–12,000 cases are seen annually (Yoder and Beach 2010) while around three cases of cryptosporidiosis per 100,000 population have been reported for European countries in 2012 (European Centre for Disease Prevention and Control 2014).
Cryptosporidiosis is a particular problem in malnourished children in developing countries where watery diarrhoea may last longer than 2 weeks in many children (Checkley et al. 2015). In Sub-Saharan Africa (SSA), C. hominis was the prevailing species in patients from Uganda (Tumwine et al. 2003), Malawi (Morse et al. 2007), Kenya (Gatei et al. 2006; Wanyiri et al. 2014), Nigeria (Molloy et al. 2010), South Africa (Abu Samra et al. 2013), São Tomé and Principe (Lobo et al. 2014) and Ghana (Eibach et al. 2015), while C. parvum was more frequently detected than C. hominis in patients from Equatorial Guinea (Blanco et al. 2009) and Ethiopia (Adamu et al. 2014). Prevalences of >20 % have been reported for SSA in children aged <5 years with diarrhoea, and numbers are even higher for HIV-positive children. Under these conditions, almost every second child experiences cryptosporidiosis before the age of 2 years (Valentiner-Branth et al. 2003). The disease is associated with malnutrition and impaired physical fitness in late childhood, and an independent and significant predictor of childhood death in SSA (Mor and Tzipori 2008). A recent prospective, case–control study involving data of more than 20,000 children aged 0–59 months identified Cryptosporidium spp. as one of four pathogens responsible for moderate-to-severe diarrhoea and associated with increased risk of death in toddlers aged 12–23 months (Kotloff et al. 2013). Notably, Cryptosporidium spp. are part of the WHO Neglected Disease Initiative since 2004 (Savioli et al. 2006).
A variety of novel and highly sensitive molecular assays for the detection of specific antigens or parasitic DNA have been developed (Checkley et al. 2015), and the same assays also can be used to identify animals with cryptosporidiosis (Helmy et al. 2014). Yet, their relatively high costs and the dependency of their performance on further equipment hamper the introduction of these methods in resource-poor areas where, however, cryptosporidiosis is a major health problem. When parasites are suspected most laboratories in these areas may perform light microscopy of wet mounts, but this does not allow the detection of Cryptosporidium oocysts due to their small size. While most institutions also keep acid-fast staining (AFS; which also enables the detection of Cryptosporidium oocysts) methods available for TB diagnosis, a simpler and faster method might facilitate the diagnosis of cryptosporidiosis under field conditions.
Heine (1982) has developed an undemanding microscopic method by spreading stool mixed with carbol fuchsin on glass slides, covering of air-dried slides with immersion oil and a cover slip, and immediate phase contrast microscopy (PCM). Subsequently, this method (without the use of carbol fuchsin) has been applied to stool samples enriched by centrifugation (Kimmig and Hartmann 1986). Here, we prospectively compared PCM under routine conditions with an enzyme immunoassay (EIA) regarding the detection of Cryptosporidium oocysts or their antigens, respectively, in human stool samples. In addition, we analysed retrospectively 65 fixed samples collected from Rwandan children at < 5 years of age; 14 of these samples had yielded positive results with a highly sensitive real-time (RT-)PCR.
Materials and methods
Stool samples
Between July and October 2014, 463 stool samples collected from diarrhoeic patients were submitted to our laboratory in Stuttgart for the detection of Cryptosporidium oocysts and included in the first part of the study. When more than one sample per patient was submitted, only the first sample was included.
In a second part, we included 65 merthiolate-iodine-formaldehyde (MIF)-fixed stool samples collected from mainly asymptomatic Rwandan community children <5 years of age; non-fixed portions of these samples had been analysed before by RT-PCR (Verweij et al. 2004). Light microscopic examination of these 65 samples before the present study had revealed the following microorganisms: 32× Giardia duodenalis, 18× Ascaris lumbricoides, 13× Entamoeba coli, 7× Entamoeba histolytica/dispar, 4× Blastocystis hominis, 3× Iodamoeba buetschlii, 2× Trichuris trichiura, 2× Endolimax nana, 1× Chilomastix mesnili and 1× Pentatrichomonas hominis.
For the present study, all samples were evaluated by PCM by two examiners who were not aware of the previous RT-PCR results. Fourteen of these samples had yielded positive RT-PCR results although most of them with high cycle threshold (Ct) values indicating relatively low amounts of target DNA, which correspond to oocyst numbers (Table 2). The other 51 RT-PCR-negative samples were from age- and sex-matched children living in the same areas in Rwanda as the RT-PCR-positive children. Details of the previous study including the ethics statement and information regarding the RT-PCR assay used have been published before (Ignatius et al. 2012).
Microscopy
Slides were prepared according to a modified protocol of the method originally published by Heine (1982). Briefly, fresh or fixed stool material was thinly spread onto dry glass slides and air-dried for 5–10 min. Slides were directly overlayed with a drop of immersion oil, covered with a cover slip (18 × 18 mm), and the entire field covered by the cover slip scanned with PCM (magnification, ×400). As a result, Cryptosporidium oocysts appear as bright organisms of 4 to 6 μm with a characteristic refraction of the microscope light (Fig. 1; Kimmig and Hartmann 1986). From all EIA- or RT-PCR-positive samples that initially yielded negative PCM results, additional slides were prepared and examined by PCM and also following AFS, which was preformed according to standard laboratory procedures.
Enzyme-linked immunoassay
All 463 fresh stool specimens collected in Stuttgart within the first part of the study were additionally examined by using a commercially available EIA (Ridascreen Cryptosporidium; R-Biopharm, Darmstadt, Germany), which was performed according to the manufacturer’s instructions.
Statistics
Data were statistically analysed by using GraphPad Prism version 6.0a for Mac OS X. Differences were considered statistically significant for p < 0.05. The Mann–Whitney test was used to compare Ct values; medians plus 25 and 75 % percentiles are presented. The EIA detects antigens expressed by various Cryptosporidium spp. and could therefore be considered gold standard in the first, prospective part of the study. Accordingly, we calculated sensitivity, specificity, positive (PPV) and negative predictive value (NPV) of PCM with 95 % confidence intervals (95 % CI). Since the RT-PCR assay applied does not detect all Cryptosporidium spp. (while PCM is species-independent only considering size, shape, and brightness of the microorganisms), we calculated the positive and negative percent agreement (PPA and NPA, respectively) and 95 % CIs of PCM with RT-PCR.
Results
Prospective study
Fresh stool samples from 463 diarrhoeic patients (median age, 3 years; range, <1–88 years) were investigated prospectively by PCM and EIA for the presence of Cryptosporidium oocysts. PCM was positive for 16 samples (3.5 %; 95 % CI, 2.1–5.6 %) while 447 samples were negative. By EIA, the 16 PCM-positive plus two additional samples yielded a positive result (3.9 %; 95 % CI, 2.4–6.1 %). Thirteen of the 18 EIA-positive patients were male (72.2 %; 95 % CI, 48.8–87.8 %) and five female (27.8 %; 95 % CI, 12.2–51.2 %; p = 0.018, Fisher’s exact test); the median age was 4 years (range, 1–46 years).
Upon microscopic examination of additional slides of the two previously negative but EIA-positive samples, some single oocysts could be found in both specimens, thereby excluding false-positive EIA results. Therefore, we considered the two discrepant samples false-negatives in PCM. Based on this data (Table 1), the sensitivity and specificity of PCM compared with the EIA were 88.9 % (95 % CI, 65.0–98.1 %) and 100 % (95 % CI, 99.0–100 %), and PPV and NPV were 100 % (95 % CI, 77.3–100 %) and 99.6 % (95 % CI, 98.3–100 %), respectively.
Retrospective study
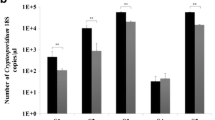
Subsequently, we examined 65 fixed stool samples from Rwandan community children <5 years of age. All samples had previously been examined by RT-PCR, and 14 of these had yielded positive RT-PCR results. The majority of these 14 children were asymptomatic; the median age was 26 months (range, 9–57 months; Table 2). PCM detected Cryptosporidium oocysts in five RT-PCR-positive samples (PPA; 35.7 %, 95 % CI, 16.2–61.4 %; Table 2). The other nine RT-PCR-positive samples were initially negative by PCM. The Ct values of microscopically positive samples were lower than those of negative samples (Fig. 2; p = 0.044).
Upon examination of additional slides, single oocysts could be detected by PCM in two of the RT-PCR-positive samples with very low oocyst numbers (and correspondingly high Ct values; Table 2). While six of the seven repeatedly negative samples had Ct values ≥30, the corresponding value for sample 714a was 26.0 indicating relatively high amounts of target DNA in this sample. Still this sample repeatedly yielded negative results when examined by PCM or following AFS.
Notably, one of the 51 RT-PCR negative samples was positive by PCM demonstrating oocysts in moderate numbers. The presence of Cryptosporidium oocysts in this sample was verified by AFS. The specimen had been collected from a 16-month-old child with fever (38.5 °C), cough, and diarrhoea all of which could have been caused by cryptosporidia. Furthermore, none of the other parasites present in the 65 Rwandan samples (for details, see “Materials and methods”) yielded a positive PCM result. NPA of PCM with RT-PCR was 98.0 % (95 % CI, 88.7–100 %).
Discussion
In the present study, PCM (in the absence of any staining solution) proved to be a highly specific and undemanding method for the detection of Cryptosporidium oocysts in human stool samples. As compared with a commercially available EIA, also its sensitivity was good.
We and others have reported equivalent or even higher sensitivity of a distinct light microscopic method, i.e., AFS, compared with Cryptosporidium oocyst-specific EIAs (Ignatius et al. 1997; Johnston et al. 2003; Kehl et al. 1995; Martin-Ampudia et al. 2012; Parisi and Tierno 1995). In contrast, others have observed a lower sensitivity of AFS in comparison with EIAs (Chalmers et al. 2011; Kaushik et al. 2008; Khurana et al. 2012). These discrepancies may be due to a dissimilarity of the patients included in the studies (e.g., regarding HIV status, severeness of the disease, co-infections etc.) and thereby differences in oocyst numbers shed with the faeces. In fact, AFS detected in a recent Egyptian study Cryptosporidium oocysts in 18 of 38 RT-PCR-positive samples from diarrhoeic children (EIA, 22/38 samples), but in none of 18 RT-PCR-positive samples from non-diarrhoeic children (EIA, 9/18) who most likely shed less oocysts than diarrhoeic children as further discussed below (Fathy et al. 2014). Furthermore, EIAs may differ regarding their sensitivity (Ignatius et al. 1997). and new generations of EIAs may be more sensitive than previous products.
The data obtained for PCM in comparison with RT-PCR was less satisfactory than that observed in the first part of our study, but this second set of samples had mainly been obtained from non-diarrhoeic children. Moreover, RT-PCR is generally accepted to be the most sensitive laboratory assay for the detection of many pathogens including Cryptosporidium spp., and its increased sensitivity in comparison with AFS has been reported before (Fathy et al. 2014; Kaushik et al. 2008; Khurana et al. 2012; Martin-Ampudia et al. 2012; Morgan et al. 1998; Zaidah et al. 2008). Interestingly, we had obtained comparable results for the detection of G. duodenalis by screening stool samples of the same group of Rwandan children with RT-PCR vs. light microscopy (Ignatius et al. 2012).
The sensitivity of PCM was significantly associated with the RT-PCR Ct values, and thus with the numbers of excreted oocysts. Therefore, its performance likely correlates with the severity of the clinical symptoms of infected individuals because oocysts numbers in human and experimental animal cryptosporidiosis decline with overcoming the infection and cessation of diarrhoea (Baxby et al. 1985; Bukhari and Smith 1997). In fact, for a 100 % detection rate by AFS, 50 times more oocysts per gramme stool are necessary in formed than in watery stool samples (Weber et al. 1991). Possibly for this reason, Cryptosporidium oocysts were detected microscopically in a large German study exclusively in diarrhoeic individuals and not in non-diarrhoeic controls while the opposite was found for G. duodenalis (Schuster et al. 1991). Alternatively and in contrast to the situation observed by us in Rwanda, European children may not continue to shed Cryptosporidium spp. after cessation of diarrhoea and therefore not constitute a relevant reservoir for this parasite. This is supported by recent findings by Sagebiel et al. (2009) who applied a highly sensitive antigen detection assay and detected G. duodenalis but not Cryptosporidium oocysts in stool samples from non-diarrhoeic German kindergarten children. Thus, PCM (as AFS as discussed above; Fathy et al. 2014) may be more suited to detect the pathogens in diarrhoeic patients (as those patients included in the first part of our study) than in rather asymptomatic carriers like the mainly randomly collected and mostly asymptomatic community Rwandan children from the second part of our study who nevertheless constitute reservoirs for new infections. Still, Cryptosporidium spp. have recently been detected in 7.8 % of 880 symptomatic Rwandan children <5 years demonstrating that these parasites also in that area are important causative agents of acute childhood gastroenteritis (Kabayiza et al. 2014).
Like AFS, PCM is species independent and can therefore be applied in areas where various Cryptosporidium spp. are prevalent. In our study, it detected Cryptosporidium oocysts in one RT-PCR negative sample. Both C. hominis and Cryptosporidium meleagridis, which also is present in SSA (Mor and Tzipori 2008) might be detected by the RT-PCR assay applied by us, but it most likely does not cross-react with Cryptosporidium andersoni, Cryptosporidium felis and Cryptosporidium muris all of which have been described in some individuals in SSA (Fontaine and Guillot 2002).
Additional advantages of PCM are (i) its extremely low costs of less than 0.1 € and (ii) its independence from all further equipment (except a microscope, but including staining solutions and even water to rinse the slides). In addition, it can be performed from fresh or fixed stool specimens. While AFS is slightly more expensive (0.2 €) and requires staining solutions and water supply, costs for the implementation, maintenance, and performance of molecular methods are considerably higher (5–7 €, excluding costs for the equipment) than those of PCM.
In conclusion, PCM is the least expensive and undemanding, but highly specific diagnostic method for species-independent detection of Cryptosporidium oocysts. It may therefore be well-suited for field studies. Since its sensitivity depends on the number of oocysts excreted, it may preferentially identify acutely infected patients while more sensitive PCR assays may be required to also identify asymptomatic carriers. Implementation of such assays, however, is demanding in resource-poor settings and expensive.
References
Abu Samra N, Thompson PN, Jori F, Frean J, Poonsamy B, du Plessis D, Mogoye B, Xiao L (2013) Genetic characterization of Cryptosporidium spp. in diarrhoeic children from four provinces in South Africa. Zoonoses Public Health 60(2):154–159
Adamu H, Petros B, Zhang G, Kassa H, Amer S, Ye J, Feng Y, Xiao L (2014) Distribution and clinical manifestations of Cryptosporidium species and subtypes in HIV/AIDS patients in Ethiopia. PLoS Negl Trop Dis 8(4):e2831
Baldursson S, Karanis P (2011) Waterborne transmission of protozoan parasites: review of worldwide outbreaks—an update 2004–2010. Water Res 45(29):6603–6614
Baxby D, Hart CA, Blundell N (1985) Shedding of oocysts by immunocompetent individuals with cryptosporidiosis. J Hyg (Lond) 95(3):703–709
Blanco MA, Iborra A, Vargas A, Nsie E, Mbá L, Fuentes I (2009) Molecular characterization of Cryptosporidium isolates from humans in Equatorial Guinea. Trans R Soc Trop Med Hyg 103(12):1282–1284
Bukhari Z, Smith HV (1997) Cryptosporidium parvum: oocyst excretion and viability patterns in experimentally infected lambs. Epidemiol Infect 119(1):105–108
Chalmers RM, Campbell BM, Crouch N, Charlett A, Davies AP (2011) Comparison of diagnostic sensitivity and specificity of seven Cryptosporidium assays used in the UK. J Med Microbiol 60(Pt 11):1598–1604
Checkley W, White AC Jr, Jaganath D, Arrowood MJ, Chalmers RM, Chen XM, Fayer R, Griffiths JK, Guerrant RL, Hedstrom L, Huston CD, Kotloff KL, Kang G, Mead JR, Miller M, Petri WA Jr, Priest JW, Roos DS, Striepen B, Thompson RC, Ward HD, Van Voorhis WA, Xiao L, Zhu G, Houpt ER (2015) A review of the global burden, novel diagnostics, therapeutics, and vaccine targets for cryptosporidium. Lancet Infect Dis 15(1):85–94
Eibach D, Krumkamp R, Al-Emran HM, Sarpong N, Hagen RM, Adu-Sarkodie Y, Tannich E, May J (2015) Molecular characterization of Cryptosporidium spp. among children in rural Ghana. PLoS Negl Trop Dis 9(3):e0003551
European Centre for Disease Prevention and Control (2014) Annual epidemiological report 2014—food- and waterborne diseases and zoonoses. ECDC, Stockholm
Fathy MM, Abdelrazek NM, Hassan FA, El-Badry AA (2014) Molecular copro-prevalence of Cryptosporidium in Egyptian children and evaluation of three diagnostic methods. Indian Pediatr 51(9):727–729
Fontaine M, Guillot E (2002) Development of a TaqMan quantitative PCR assay specific for Cryptosporidium parvum. FEMS Microbiol Lett 214(1):13–17
Gatei W, Wamae CN, Mbae C, Waruru A, Mulinge E, Waithera T, Gatika SM, Kamwati SK, Revathi G, Hart CA (2006) Cryptosporidiosis: prevalence, genotype analysis, and symptoms associated with infections in children in Kenya. Am J Trop Med Hyg 75(1):78–82
Heine J (1982) A simple technic for the demonstration of cryptosporidia in feces. Zentralbl Veterinarmed B 29(4):324–327
Helmy YA, Krücken J, Nöckler K, von Samson-Himmelstjerna G, Zessin KH (2014) Comparison between two commercially available serological tests and polymerase chain reaction in the diagnosis of Cryptosporidium in animals and diarrhoeic children. Parasitol Res 113(1):211–216
Ignatius R, Eisenblatter M, Regnath T, Mansmann U, Futh U, Hahn H, Wagner J (1997) Efficacy of different methods for detection of low Cryptosporidium parvum oocyst numbers or antigen concentrations in stool specimens. Eur J Clin Microbiol Infect Dis 16(10):732–736
Ignatius R, Gahutu JB, Klotz C, Steininger C, Shyirambere C, Lyng M, Musemakweri A, Aebischer T, Martus P, Harms G, Mockenhaupt FP (2012) High prevalence of Giardia duodenalis assemblage B infection and association with underweight in Rwandan children. PLoS Negl Trop Dis 6(6):e1677
Johnston SP, Ballard MM, Beach MJ, Causer L, Wilkins PP (2003) Evaluation of three commercial assays for detection of Giardia and Cryptosporidium organisms in fecal specimens. J Clin Microbiol 41(2):623–626
Kabayiza JC, Andersson ME, Nilsson S, Baribwira C, Muhirwa G, Bergstrom T, Lindh M (2014) Diarrhoeagenic microbes by real-time PCR in Rwandan children under 5 years of age with acute gastroenteritis. Clin Microbiol Infect 20(12):O1128–O1135
Kaushik K, Khurana S, Wanchu A, Malla N (2008) Evaluation of staining techniques, antigen detection and nested PCR for the diagnosis of cryptosporidiosis in HIV seropositive and seronegative patients. Acta Trop 107(1):1–7
Kehl KS, Cicirello H, Havens PL (1995) Comparison of four different methods for detection of Cryptosporidium species. J Clin Microbiol 33(2):416–418
Khurana S, Sharma P, Sharma A, Malla N (2012) Evaluation of Ziehl-Neelsen staining, auramine phenol staining, antigen detection enzyme linked immunosorbent assay and polymerase chain reaction, for the diagnosis of intestinal cryptosporidiosis. Trop Parasitol 2(1):20–23
Kimmig P, Hartmann S (1986) Ein einfacher Kryptosporidiennachweis im Rahmen der parasitologischen Stuhl-Routinediagnostik. Lab Med 10:285–286
Kotloff KL, Nataro JP, Blackwelder WC, Nasrin D, Farag TH, Panchalingam S, Wu Y, Sow SO, Sur D, Breiman RF, Faruque AS, Zaidi AK, Saha D, Alonso PL, Tamboura B, Sanogo D, Onwuchekwa U, Manna B, Ramamurthy T, Kanungo S, Ochieng JB, Omore R, Oundo JO, Hossain A, Das SK, Ahmed S, Qureshi S, Quadri F, Adegbola RA, Antonio M, Hossain MJ, Akinsola A, Mandomando I, Nhampossa T, Acacio S, Biswas K, O'Reilly CE, Mintz ED, Berkeley LY, Muhsen K, Sommerfelt H, Robins-Browne RM, Levine MM (2013) Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the Global Enteric Multicenter Study, GEMS): a prospective, case–control study. Lancet 382(9888):209–222
Lobo ML, Augusto J, Antunes F, Ceita J, Xiao L, Codices V, Matos O (2014) Cryptosporidium spp., Giardia duodenalis, Enterocytozoon bieneusi and other intestinal parasites in young children in Lobata province, democratic republic of São Tomé and Principe. PLoS One 9(5):e97708
Martin-Ampudia M, Mariscal A, Lopez-Gigosos RM, Mora L, Fernandez-Crehuet J (2012) Under-notification of cryptosporidiosis by routine clinical and laboratory practices among non-hospitalised children with acute diarrhoea in Southern Spain. Infection 40(2):113–119
Molloy SF, Smith HV, Kirwan P, Nichols RA, Asaolu SO, Connelly L, Holland CV (2010) Identification of a high diversity of Cryptosporidium species genotypes and subtypes in a pediatric population in Nigeria. Am J Trop Med Hyg 82(4):608–613
Mor SM, Tzipori S (2008) Cryptosporidiosis in children in Sub-Saharan Africa: a lingering challenge. Clin Infect Dis 47(7):915–921
Morgan UM, Pallant L, Dwyer BW, Forbes DA, Rich G, Thompson RC (1998) Comparison of PCR and microscopy for detection of Cryptosporidium parvum in human fecal specimens: clinical trial. J Clin Microbiol 36(4):995–998
Morse TD, Nichols RA, Grimason AM, Campbell BM, Tembo KC, Smith HV (2007) Incidence of cryptosporidiosis species in paediatric patients in Malawi. Epidemiol Infect 135(8):1307–1315
Parisi MT, Tierno PM Jr (1995) Evaluation of new rapid commercial enzyme immunoassay for detection of Cryptosporidium oocysts in untreated stool specimens. J Clin Microbiol 33(7):1963–1965
Sagebiel D, Weitzel T, Stark K, Leitmeyer K (2009) Giardiasis in kindergartens: prevalence study in Berlin, Germany, 2006. Parasitol Res 105(3):681–687
Savioli L, Smith H, Thompson A (2006) Giardia and Cryptosporidium join the ‘neglected diseases initiative’. Trends Parasitol 22(5):203–208
Schuster W, Fischer R, Alsleben S, Schuster B (1991) Cryptosporidium sp. in stool specimens from diarrhoeic and asymptomatic individuals in the Magdeburg area (East Germany). Angew Parasitol 32(4):193–197
Shirley DA, Moonah SN, Kotloff KL (2012) Burden of disease from cryptosporidiosis. Curr Op Infect Dis 25(5):555–563
Tumwine JK, Kekitiinwa A, Nabukeera N, Akiyoshi DE, Rich SM, Widmer G, Feng X, Tzipori S (2003) Cryptosporidium parvum in children with diarrhea in Mulago Hospital, Kampala, Uganda. Am J Trop Med Hyg 68(6):710–715
Valentiner-Branth P, Steinsland H, Fischer TK, Perch M, Scheutz F, Dias F, Aaby P, Molbak K, Sommerfelt H (2003) Cohort study of Guinean children: incidence, pathogenicity, conferred protection, and attributable risk for enteropathogens during the first 2 years of life. J Clin Microbiol 41(9):4238–4245
Verweij JJ, Blange RA, Templeton K, Schinkel J, Brienen EA, van Rooyen MA, van Lieshout L, Polderman AM (2004) Simultaneous detection of Entamoeba histolytica, Giardia lamblia, and Cryptosporidium parvum in fecal samples by using multiplex real-time PCR. J Clin Microbiol 42(3):1220–1223
Wanyiri JW, Kanyi H, Maina S, Wang DE, Steen A, Ngugi P, Kamau T, Waithera T, O'Connor R, Gachuhi K, Wamae CN, Mwamburi M, Ward HD (2014) Cryptosporidiosis in HIV/AIDS patients in Kenya: clinical features, epidemiology, molecular characterization and antibody responses. Am J Trop Med Hyg 91(2):319–328
Weber R, Bryan RT, Bishop HS, Wahlquist SP, Sullivan JJ, Juranek DD (1991) Threshold of detection of Cryptosporidium oocysts in human stool specimens: evidence for low sensitivity of current diagnostic methods. J Clin Microbiol 29(7):1323–1327
Yoder JS, Beach MJ (2010) Cryptosporidium surveillance and risk factors in the United States. Exp Parasitol 124(1):31–39
Zaidah AR et al (2008) Detection of Cryptosporidium parvum in HIV-infected patients in Malaysia using a molecular approach. Southeast Asian J Trop Med Public Health 39(3):511–516
Acknowledgements
This study was in part supported by the German Federal Ministry for Economic Cooperation and Development through the ESTHER programme (Ensemble pour une Solidarité Thérapeutique Hospitaliére En Réseau) and by the German Federal Ministry of Education and Research (grant 01DG13006A, MOPACUR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ignatius, R., Klemm, T., Zander, S. et al. Highly specific detection of Cryptosporidium spp. oocysts in human stool samples by undemanding and inexpensive phase contrast microscopy. Parasitol Res 115, 1229–1234 (2016). https://doi.org/10.1007/s00436-015-4859-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4859-3