Abstract
The current study is the first to use gross anatomy, light, and scanning electron microscopy to describe the oropharyngeal cavity of a bogue (Boops boops Linnaeus, 1758) fish. Fourteen 17–18 cm long bogue fish were used. The lower jaw held the lower lip, labial teeth, tongue, and pharyngeal teeth, and the oral cavity floor appeared u-shaped. The lower jaw was small and narrow in the anterior and wide in the posterior as it approached the esophagus. The lower lip carried different lengths of cilia-like projections that looked like the cactus plant arranged in transverse layers, and its apex appeared as an open rose. These cilia were encircled by ridges that looked like sea waves. The labial teeth were 15 in a row, with long roots placed beneath a pocket-like structure. Long, medium, and short pharyngeal papilliform teeth emerged on the oropharyngeal cavity floor at the level of the fourth gill arch in a small V-shaped arrangement. The tongue was smooth and small, with an apex, body, root, and lateral edges that were all clearly defined. The tongue’s apex was pointed and triangular, with a thin, transparent apical pouch around it. There was a median longitudinal fold on the dorsum of the tongue body and root. On the tongue surface, taste bud types I and II emerged. The bogue fish’s oropharyngeal cavity had unique anatomical characteristics, indicating that it was omnivorous.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bogue, Boops boops (Linnaeus, 1958), is a teleost marine fish belonging to the Sparidae family that is found primarily in the Eastern Atlantic, and it is a very dominant species in large quantities along the Mediterranean Sea coast (El-Okda 2008; FAO 2019; Khemiri et al. 2005).
The bogue fish is a gregarious, demersal, semi-pelagic species found as deep as 350 m over different substrata, including sand, mud, rocks, and seaweed, where it is more common at depths of less than 150 m and sometimes in coastal waters. This species moves in aggregations, ascending to the surface mainly at night (Frimodt 1995; Pollard et al. 2014).
The bogue fish is omnivorous and feeds on benthic (Crustacea, Mollusca, Annelida, Sipuncula, Plantae) and pelagic prey (Siphonophorae, Copepoda, eggs) (Derbal and Kara 2008; Frimodt 1995). Individuals can reach a height of 36 cm, but an average of 20 cm (Dahel et al. 2019).
Some researchers have investigated the mechanism of fish feeding, the oral cavity morphology, and the types of food consumed in the marine ecosystem (Beukers-Stewart and Jones 2004; Matich et al. 2011). The oropharyngeal cavity of fishes differs significantly in both morphologic and histologic descriptions, which may be due to differences in feeding habits, body format, and environmental conditions (Bone and Moore 2008; Khalaf-Allah 2009).
The adaptation of fishes to their environment is due to their feeding ability and the oropharyngeal cavity, especially the tongue, which plays a significant role. The morphology of the oropharyngeal cavity in fish has been the subject of numerous research (Abbate et al. 2006, 2017, 2011, 2012; Alsafy et al. 2022, 2018; El Bakary 2014; Elgendy et al. 2016; Gamal et al. 2012; Guerrera et al. 2015; Levanti et al. 2017; Mahmoud et al. 2016; Sayed et al. 2019; Yashpal et al. 2006, 2009); and teeth morphology (Aljalaud et al. 2017; Alsafy et al. 2021; Germain and Meunier 2020).
The biology of bogue fish in the Mediterranean Sea has attracted the attention of many researchers prompting some studies in Egypt (Allam 2003; El-Okda 2008; Hassan 1990; Mehanna 2014), Turkey (Kara and Bayhan 2015; Soykan et al. 2015), Greece (Stergiou and Moutopoulos 2001), Tunisia (Anato and Ktari 1983; Khemiri et al. 2005), and Algeria (Dahel et al. 2019; Kherraz et al. 2016; Rachid et al. 2014).
There is a scarcity of morphological data on bogue (Boops boops) fishes, particularly the oropharyngeal cavity. The goal of this study is to look at the morphological structures of the floor of bogue (Boops boops) fish using gross morphology, scanning electron microscopy, and morphometric analysis.
Materials and methods
Samples
Fourteen bogue (Boops boops Linnaeus, 1758) fish, 17–18 cm in length, were collected from Matrouh Governorate in the Mediterranean Sea. The fish’s oropharyngeal cavities were first washed with normal saline and examined to be free from any oral injuries or abnormalities. They have then preserved in formalin 10% solution and directly transported to the dissecting laboratory.
Gross morphology
Five fresh fishes were examined grossly. The five fish's jaws were separated by cutting horizontally through the mouth to the esophagus. The floor structures of those five fishes were photographed by a digital camera.
Scanning electron microscopy (SEM)
Three fish were used. Samples were taken from all parts of the oropharyngeal cavity floor and fixed in a buffered solution (2% formaldehyde, 1.25% glutaraldehyde in 0.1 M sodium cacodylate buffer, pH 7.2) for 48 h at 4 °C. Once fixed, the samples were washed in 0.1 M sodium cacodylate containing 5% sucrose, processed through tannic acid, and dehydrated in increasing grades of ethanol (15 min each in 50, 70, 80, 90, 95, and 100% ethanol). Then the samples were at a critical point, dried in CO2, attached to stubs with colloidal carbon, sputter-coated with gold, and investigated by a JEOL JSM-IT200 scanning electron microscope (Goldstein et al. 2017).
Light microscopy (LM)
Two fishes were used. Small samples of the tongue were taken and fixed with 10% formalin. After a few days of fixation, the samples were thoroughly washed in 70% alcohol (three times in 24 h) to remove the fixative before proceeding with the tissue processing. In addition, the tissue samples were dehydrated in a graded series of ethanol (80, 95, and absolute), cleared in xylene, impregnated, and embedded in paraffin wax. The paraffin blocks were sliced to a 6 μm thick slice and stained with Harris hematoxylin and eosin stain (H&E) (Bancroft and Gamble 2008).
Morphometric analysis
The obtained SEM images were processed by the ImageJ software to determine the different measured lengths of the different types of teeth, and the mean and standard error were done by Microsoft excel.
Results
External features
The bogue fish had an elongated body with different colored lines, including silver, yellow, and whitish lines, and a clear lateral line running the length of its body. Furthermore, the fish had large, prominent eyes and an oblique mouth (Fig. 1A). The bogue fish had several fins that varied from single to double fins. The single fins here included the dorsal, anal, and caudal fins. The pelvic and pectoral fins were double, whereas the tail fin was a fork shape.
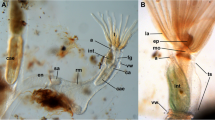
Overview images of (A) lateral view of formalized bogue fish; B magnification of the head region; C the floor of the fresh bogue fish’s oral cavity; and D the floor of the formalized bogue fish’s oral cavity. Pectoral fin (PF), pelvic fin (PeF), lateral line (LL), dorsal fin (DF), anal fin (AF), caudal fin (CdF), vent (V), operculum (OP), eye (E), upper jaw (UJ), lower jaw (LJ), upper lip (UL), lower lip (LL), lower teeth (LT), sublingual floor (SF), tongue (T), interbranchial septum (IbS), 1,2,3 and 4 refer to the four gill arches, lower pharyngeal teeth (LPT)
Mouth
Overall, the bogue had a small, oblique mouth opening with upper and lower attached jaws, wherein the upper jaw attached the lips, called the premaxillary, appeared thin and weak. The two lips are attached laterally at the mouth angle in the opposing lower jaw, known as the dentary lip (Fig. 1B). The results showed that the oral cavity floor looked like a U-shaped structure and included the lower lip, lower jaw, lower teeth, tongue, and lower pharyngeal teeth.
By SEM, it was found that the floor was composed of the lower lip with its striation, the arrangement of the lower teeth and shape, and a thin apical pouch covered the tongue apex, the interbranchial septum, and the lower pharyngeal teeth (Fig. 2).
Overview image A and SEM images B, C, D, E, and F of the first half of the floor of the oral cavity from the lower lip to the tongue. Labial teeth (LT), apical pouch (AP), lower lip (LL), the apex of the tongue A, the body of the tongue B, sublingual floor (SF), 1, 2, 3, 4 refer to the four separated areas in the enamel part of the labial teeth, the central part of the apical pouch (CAP) shows the ridges (R) which look like the tortuous bulged pipe, dome-shape elevation (D) on the lateral side of the apical pouch (LAP). The purple area under LT in image B refers to enlarged central labial teeth, which is well explained in image C, and the purple area under LL refers to enlarged lateral labial teeth, which is well explained in image D. The purple area under CAP is the enlarged central portion of the apical pouch, as shown in image E. The purple area under LAP is referred to as the enlarged lateral part of the apical pouch, which is clearly illustrated in image E
Lower jaw
Overall, the lower jaw appeared to be shorter and thinner than the upper one. A flexible short ligament on the right and left angles of the mouth also connected it to the upper jaw (Fig. 1B, C).
By SEM, it was found that the lower jaw consisted mainly of a lower lip that appeared well. It was very close to the lower teeth surrounding them. It was narrow anteriorly and increased in width posteriorly towards the esophagus (Fig. 2D).
Lower lip
Overall, the lower lip was attached to its corresponding lower jaw; it seemed shorter than the upper one (Fig. 1B).
By SEM, it was found that the lower lip appeared striated; it has several irregular layers arranged behind each other (Fig. 3A). The lower lip carried different lengths of cilia-like structures, which were seen with their broad bases implanted in the underlying layer with an apex directed forward with prominent ridges surrounding them (Fig. 3B). These cilia-like structures looked like the cactus plant arranged in transverse layers, and thorn-like structures covered them from their broad base to their rose-shaped apex (Fig. 3C). Moreover, the ridges surrounding these cilia appeared like the sea waves had thin serrated edges because they had thorn-like structures on their whole surfaces (Fig. 3C). The apex of the cilia appeared as an open rose (Fig. 3D).
SEM images of the lower lip. Lower teeth (LT), lower lip (LL), the location of dislocated teeth with its pyramidal shape shows the pulp cavity (pc), dentin (D), in view B the striated lower lip shows different cilia (C), longitudinal ridges (R), folds (F), view (C) shows the cactus-like cilia (C), with wavy-like ridges (R) that carry thorn-like structures (T), and view (D) shows the cilia apex that looks like a rose and thin serrated edge (S). The purple area under LL in image A refers to as an enlarged lower lip, which is well explained in image B. The purple area under C in image B refers to the enlarged lateral surface of the cactus-like cilia that was well explained in image C. The purple area at the tips of cactus-like cilia and the surrounding ridge in image C, which is well explained in image D
Labial teeth
Overall, the labial teeth are arranged in the denteries as a single row of teeth. They are distinguished by their small, tiny teeth with long roots and broad bases. They were 15 to 16 in number and only found in the anterior part of the mouth (Fig. 2A).
By SEM, it was found that the labial teeth were 15 incisive-like teeth arranged in 1 row, and the large teeth were anterior (Fig. 2B). The labial teeth's roots were long and implanted in the underlying layer, with a pocket-like structure surrounding the tooth roots (Fig. 2D). The labial teeth were not visible along the entire length of the lower jaw.
The mean length of the labial teeth was 700 mm and their mean width was about 570 mm, as well as measuring the width of the teeth at their distal part, which showed a difference of about 240 mm, we found that the area of the upper cutting part of the teeth was about 21,376 mm (Table 1).
Tongue
Overall, the tongue is smooth and small, about 1 cm in length, and presents along most of the oral cavity floor. The tongue apex, body, root, and lateral margins were all visible. The tongue apex is pointed and triangular, with a thin and transparent apical pouch surrounding it (Figs.2A, 4A, 5A, 6A). The body and root of the tongue are broader than the apex, and they possess a median longitudinal fold (Fig. 5A, 6A).
Overview image A and SEM images B, C, D, E, and F of the apex of the tongue. Lower teeth (LT), apical pouch (AP), the apex of the tongue (A), lower teeth (LT), the body of the tongue (B), sublingual floor (SF), view (C) represents types of taste buds, type I (I), type II (II), view D shows taste buds type II (II), and view E shows taste buds type I (I). The purple area at the apex of the tongue is enlarged in image B to explain type I and II taste buds in image C. The purple area of type I and II taste buds is enlarged in the image C to explain the macro-structure of type I and II (D and E)
Overview image A and SEM images B, C, D, E, and F of the body of the tongue. The lower teeth (LT), apical pouch (AP), the apex of the tongue (A), the body of the tongue (B), the root of the tongue (R), median longitudinal fold (MLF), left lateral buccal fold (LLbf), right lateral buccal fold (RLbf), the two lateral borders of the tongue (LB), the small yellow dots indicate the lateral borders of the MLF. View (C and D) shows luminous structures on the body of the tongue as dome papillae (dp) and cylindrical papillae (cp) with smooth epithelium (Se) in the background. View E shows the lateral border of the MLF, which consists of longitudinal ridges (RI) with numerous luminous structures. View F—detail of the ridges (RI) on the MLF’s lateral border reveals corrugated epithelium (Ce), dome papillae (dp), and taste buds type I (I). In image B, the purple area at the median longitudinal fold at the body of the tongue is enlarged in image C. The purple area at the lateral border of the median longitudinal fold at the body of the tongue is enlarged in image E. In image E, the purple area at the lateral border of the median longitudinal fold at the body of the tongue is enlarged more in image F
Overview image A and SEM images B, C, D, E, and F of the root of the tongue. The lower teeth (LT), apical pouch (AP), the apex of the tongue (A), the body of the tongue (B), the root of the tongue (R), median longitudinal fold (MLF), left lateral buccal fold (LLbf), right lateral buccal fold (RLbf), the lateral borders of the tongue (LB), interbranchial septum (Ibs), gill arch (Ga), longitudinal ridge (Lr), taste buds type I (I), taste buds type II (II), and conical papillae (CP). View D shows the interbranchial septum with its prominent longitudinal ridge (Lr), view E shows the prominent longitudinal ridge (Lr) borders, and the left side of the interbranchial septum (LIbs), and view F—detail of the lateral border of the root of the tongue shows multiple irregular ridges (R). In image B, the purple area in the middle of the root is magnified in image C, the purple area in the middle of the interbranchial septum area is enlarged in image D. The purple area on the lateral part of the root is an enlarged image F. In image D, the purple area on the lateral side of the interbranchial septum is enlarged in image E
By SEM, it was found that the apical pouch enveloped the tongue apex. It had irregular longitudinal ridges as a crooked tube on its central part, while its lateral side had dome-shaped elevations (Fig. 2B, E, F). The tongue apex, body, root, and lateral margins were distinguished clearly (Fig. 4B). The tongue body and root showed a median longitudinal fold with cylindrical and small conical papillae with smooth epithelium. The lateral border of the median longitudinal fold had longitudinal folds with corrugated epithelium that carried dome-shaped papillae and taste buds of type I. The root appeared to be a broad, striated part with clear lines at its middle and longitudinal ridges on both sides (Figs. 5B, F and 6B, C). On the median part of the tongue root, a large number of numerous cone-like papillae and luminous taste buds of types I and II appeared (Fig. 6C, F). The lateral side of the tongue root was striated with multiple longitudinal ridges (Fig. 6F).
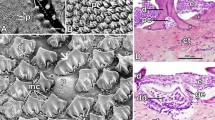
The different types of taste buds were visible as luminous structures on the tongue (Fig. 4C). Type I taste buds were raised from the epithelium around them and had a volcano-like appearance. They had several folds in their base, several transverse folds in their body, and taste pores in their apex (Figs. 4E, 5F, 6C). Type II taste buds were raised from the epithelium around them and had taste pores in the center (Figs. 4E, 6C). By LM, the tongue was covered by stratified squamous epithelium and had filiform or conical mechanical papillae at the apex, in addition to the many fungiform papillae with taste buds (Figs. 7A, B).
Light photomicrograph of the tongue of the bogue fish. View (A) (H & E, X40) shows the filiform (FP) or conical mechanical papillae on the epithelial surface (CP). View (B) (H &E, X400) represents the photomicrograph of the tip of the tongue at higher magnification which shows fungiform papilla (arrow), sensory cells (Se), supporting cells (Su), basal cells (Bc), and nerves (Ne). Dense irregular connective tissue (CT) and white adipose connective tissue (ACT) with the signet-ring appearance of white adipocytes (arrowhead)
Pharyngeal teeth
Overall, the pharyngeal teeth appear in a small V-shaped structure on the floor of the oropharyngeal cavity after the end of the fourth-gill arch. They are small and have less sharpness. The pharyngeal teeth have a needle-like body and are not closely fitted to each other; they end at the beginning of the esophagus (Fig. 8A).
Overview image (A) and SEM image (B) of the floor of the oral cavity from the interbranchial septum to the lower pharyngeal teeth. Interbranchial septum (Ibs), gill arch (Ga), 1, 2, 3, 4 (the four gill arches), lower pharyngeal teeth (LPHT), A (caudal row of the pharyngeal teeth), B (rostral row of the pharyngeal teeth), left operculum (LO), long papilliform teeth (LPT), medium papilliform teeth (MPT), short papilliform teeth (SPT), esophagus (ESO)
By SEM, it was found that the lower pharyngeal teeth appeared papilliform. They were arranged in a small V-shaped arrangement on the oropharyngeal cavity floor at a level after the end of the interbranchial septum. They appeared in three rows of varying sizes; long, medium, and short papilliform (Fig. 8B, C).
The long papilliform tooth length and base diameters were 723 mm and 180 mm, the medium papilliform was 448 mm and 128 mm, and the small papilliform was 313 mm and 72 mm (Table 2).
Discussion
There have been no previous studies on the morphology of the bogue fish's oropharyngeal cavity. As a result, this is the first study of its kind on the oropharyngeal floor. Gross anatomy, light, and scanning electron microscopy were used to study the anatomical parts of the oropharyngeal cavity in this work.
The bogue, Boops boops, belongs to the Sparidae fish family and is one of the most widespread sparid fish species in the Mediterranean Sea and Atlantic coast (Bauchot 1990). Bogue in Libya is a nearly omnivorous species that feed on a diverse range of prey types, including crustaceans (49.0%), Porifera (22.7%), Coelenterate (10.2%), Seagrasses (9.2%), Mollusca (8.1%), and Protozoa (0.9%) (El-Maremie and El-Mor 2015; Motaref 2013).
The obtained results of the bogue oropharyngeal cavity indicate the adaptation of fishes to their environment concerning their feeding ability, like another seabream (Elgendy et al. 2016). Teeth are found in almost all vertebrates and are associated with the oral cavity. The teeth are exoskeleton structures (Shimada et al. 2013). Teeth were discovered in a variety of locations in fish, including the mouth and pharynx (Debiais‐Thibaud et al. 2007).
This study confirmed the presence of labial and pharyngeal teeth in bogue fish. They have labial teeth, similar to incisor teeth, that are presented in one row and used for grasping prey and cutting hard structures, as well as papilliform teeth with different levels in the pharyngeal region that may allow additional cutting of food. The presence of papilliform and molariform teeth in the buccal cavity of omnivorous fish, such as brown-spotted grouper and Clarias gariepinus, is associated with prey seizing, grasping, holding, crushing, and grinding (Gamal et al. 2012; Hassan 2013). Unlike in carnivores fish, gilt-head seabream had mainly molar teeth in addition to canine and papilliform teeth in many rows (Elgendy et al. 2016). The bogue fish's pharyngeal papilliform teeth were classified into three types. These tooth types have also been discovered in Bagrus bayad but in a different configuration, where they emerge into two triangular areas rostral to the esophagus (Alsafy et al. 2018).
According to our findings, the tongue of the bogue fish was relatively small, with a distinct apex, body, root, and two lateral margins. Many teleosts, however, had a thick tongue (Ostrander and Hopkins 2000), pointed in Diplodus sargus (Guerrera et al. 2015; Levanti et al. 2017), rounded in Trachelyopterus striatulus (Santos et al. 2015), pyramidal in blackspot seabream (Abbate et al. 2022), and cylinder shaped in zebrafish (Abbate et al. 2006).
A transparent apical pouch wrapped the bogue tongue apex. The most important characteristic feature of the seabream mouth floor is the presence of an apical pouch, which probably has a protective role for the apex (Abbate et al. 2022). On the tongue’s lateral sides, the epithelium formed an irregular structure that blew up in the center point of the pouch, similar to investigations in Diplodus sargus fish (Guerrera et al. 2015; Levanti et al. 2017) and blackspot seabream (Abbate et al. 2022).
There were no ridges on the apex of bogue's tongue. The body had a median longitudinal fold, and the root surface exhibited several cone-like papillae. Also, seabream fishes have different types of lingual papillae (Elgendy et al. 2016). Diplodus and white grouper had a median raphe as a characteristic feature (Alsafy et al. 2022; Levanti et al. 2017). In common solea, the tongue had a rounded apex with several epithelial protrusions, flagella, and microridges on the body (El Bakary 2014). The root surface of Diplodus sargus had numerous cone-like papillae (Levanti et al. 2017).
The current investigation of the bogue fish observed that the taste bud type II on the tongue indicates the tasting and selective habits of the food of this fish. The taste bud pores that appeared at its center raised somewhat from the surrounding epithelium. The taste buds type I formed a volcano shape that appeared raised from the surrounding epithelium and had taste pores in the seabream, which has scattered and numerous taste buds, and the sea bass, which has a typical pear–onion shape with small and numerous taste pores (Abbate et al. 2022; Elgendy et al. 2016) and (Abbate et al. 2012). In white grouper, the lower lip carried some taste buds of types II and III (Alsafy et al. 2022). The tongues of the white grouper and Diretmus species do not have any taste buds (Meyer-Rochow 1981). There are many taste buds on the dorsal lingual surface in some carnivorous fish such as (Yashpal et al. 2006, 2009) in carp, (Elgendy et al. 2016) in gilt-head seabream, (Alsafy et al. 2018), and in Bagrus Bayad; however, Meyer-Rochow 1981 reported the presence of a couple of taste buds on the dorsal lingual surface of Carapus mourlani.
By light microscopy, mechanical and gustatory papillae were discovered on the bogue fish tongue. The taste pores and taste buds were visible in gilt-head seabream's fungiform and filiform-like papillae (Abbate et al. 2022). While the epithelium of European seabass fish had a fungiform or conical shape, goblet cells and taste buds were present on the entire dorsal surface of the tongue (Abbate et al. 2012). The presence of conical papillae with taste buds and mechanosensitive tasks allows fish to choose and evaluate their food (Kubitza and Lovshin 1997).
The distribution of teeth in the upper roof and the lower floor differs among carnivorous teleostean fish. Anatomically, the current study noted the presence of teeth in the lower jaw and on the tongue related to fixing the prey and preventing its escape, which agrees with what was stated by Alsafy et al. 2021 and El Bakary 2012.
The presence of papillae and taste buds on the bogue fish's tongue demonstrates the evident interplay between the mechanics of food taste and swallowing.
Conclusion
The bogue fish’s oropharyngeal cavity had distinct structural features that were evident in its omnivorous behavior. Bogue fish has one row of incisive-like teeth, varying sizes of papilliform pharyngeal teeth and papillae, taste buds on the tongue, and an apical pouch.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abbate F, Germana G, De Carlos F, Montalbano G, Laura R, Levanti M, Germana A (2006) The oral cavity of the adult zebrafish (Danio rerio). Anat Histol Embryol 35(5):299–304
Abbate F, Guerrera MC, Montalbano G, De Carlos F, Suárez AÁ, Ciriaco E, Germanà A (2011) Morphology of the European sea bass (Dicentrarchus labrax) tongue. Microsc Res Tech 75(5):643–649
Abbate F, Guerrera MC, Montalbano G, De Carlos F, Suárez AÁ, Ciriaco E, Germanà A (2012) Morphology of the European sea bass (Dicentrarchus labrax) tongue. Microsc Res Tech 75(5):643–649
Abbate F, Guerrera MC, Cavallaro M, Montalbano G, Germanà A, Levanti M (2017) LM and SEM study on the swordfish (Xiphias gladius) tongue. Tissue Cell 49(6):633–637
Abbate F, Guerrera MC, Levanti M, Laurà R, Aragona M, Mhalhel K, Montalbano G, Germanà A (2022) Morphological characteristics of the blackspot seabream (Pagellus bogaraveo) tongue: A structural and immunohistochemical study. Anat Histol Embryol 51(1):103–111
Aljalaud N, Abou-Zaid DFA, Salem SB (2017) Morphological specializations of the teeth of Red Sea fish in relation to feeding habit. Egypt J Exp Biol 13(2):329–337
Allam S (2003) Growth, mortality and yield per recruit of Bogue, Boops boops (L.), from the Egyptian Mediterranean waters off Alexandria. Mediter Mar Sci 4(1):87–96
Alsafy MA, Bassuoni NF, Hanafy BG (2018) Gross morphology and scanning electron microscopy of the Bagrus bayad (Forskal, 1775) oropharyngeal cavity with emphasis to teeth-food adaptation. Microsc Res Tech 81(8):878–886
Alsafy M, Madkour NF, El-Bakary R, Karkoura A, El-Gendy S, Zaki MA, Tanekhy M, Abumandour MM (2021) Age-related ultrastructural characterizations of the teeth of the white grouper (Epinephelus aeneus) in the different three age-stages. Microsc Res Tech 84(6):1115–1134
Alsafy M, Madkour NF, El-Bakary R, Karkoura A, El-Gendy S, Abumandour MM (2022) Ultrastructural comparison between the oral cavity floor of the juvenile and adult Epinephelus aeneus: New vision of aging development and its carnivorous adaptation. Microsc Res Tech 85(2):767–780
Anato C, Ktari M (1983) Reproduction de Boops boops (Linné, 1758) et de Sarpa salpa (Linné, 1758), poissons téléostéens, sparidés du golfe de Tunis
Bancroft JD, Gamble M (2008) Theory and practice of histological techniques. Elsevier Health Sciences, Amsterdam
Bauchot M (1990) Check-list of the fishes of the eastern tropical Atlantic (CLOFETA). Sparidae In: JC Quero, JC Hureau, C Karrer, A Post and L Saldanha JNICT-Portugal, SEI-France. Publication
Beukers-Stewart B, Jones G (2004) The influence of prey abundance on the feeding ecology of two piscivorous species of coral reef fish. J Exp Mar Biol Ecol 299(2):155–184
Bone Q, Moore R (2008) Biology of fishes. Taylor & Francis
Dahel T, Rachedi M, Tahri M, Benchikh N, Diaf A, Djebar A (2019) Fisheries status of the bogue Boops boops (Linnaeus, 1758) in Algerian East Coast (Western Mediterranean Sea). Egypt J Aquat Biol Fish 23(4):577–589
Debiais-Thibaud M, Borday-Birraux V, Germon I, Bourrat F, Metcalfe CJ, Casane D, Laurenti P (2007) Development of oral and pharyngeal teeth in the medaka (Oryzias latipes): comparison of morphology and expression of eve1 gene. J Exp Zool B Mol Dev Evol 308(6):693–708
Derbal F, Kara MH (2008) Composition du régime alimentaire du bogue Boops boops (Sparidae) dans le golfe d’Annaba (Algérie). Cybium 32(4):325–333
El Bakary NESR (2012) Morphology of the buccal cavity of the sea bream (Sparus aurata) and its relation to the type of feeding using scanning electron microscopy. Global Veterinaria 9(6):779–784
El Bakary NESR (2014) Morphological study of the asymmetrical buccal cavity of the flatfish common solea (Solea solea) and its relation to the type of feeding. Asian Pac J Trop Biomed 4(1):13–17
Elgendy SAA, Alsafy MAM, Tanekhy M (2016) Morphological characterization of the oral cavity of the gilthead seabream (Sparus aurata) with emphasis on the teeth-age adaptation. Microscopy Res Technique 79(3):227–236
El-Maremie H, El-Mor M (2015) Feeding habits of the bogue, Boops boops (Linnaeus, 1758) (Teleostei: Sparidae) in Benghazi coast, eastern Libya. J Life Sci 9(5):189–196
El-Okda NI (2008) Age and growth of Boops boops (L) from Egyptian Mediterranean waters off Alexandria. Egypt J Aquat Biol Fisheries 12(1):13–23
FAO (2019) Species Fact Sheets Boops boops (Linnaeus, 1758).
Frimodt C (1995) Multilingual illustrated guide to the world’s commercial warmwater fish. Fishing News Books Ltd, Oxford
Gamal AM, Elsheikh EH, Nasr ES (2012) Morphological adaptation of the buccal cavity in relation to feeding habits of the omnivorous fish Clarias gariepinus: a scanning electron microscopic study. J Basic Appl Zool 65(3):191–198
Germain D, Meunier FJ (2020) A tomographic study of the histological structure of teeth in the gilthead sea bream, Sparus aurata (Teleostei, Perciformes, Sparidae). J Fish Biol 97(1):273–278
Goldstein JI, Newbury DE, Michael JR, Ritchie NW, Scott JH, Joy DC (2017) Scanning electron microscopy and X-ray microanalysis. Springer
Guerrera MC, Montalbano G, Germana A, Maricchiolo G, Ciriaco E, Abbate F (2015) Morphology of the tongue dorsal surface in white sea bream (Diplodus sargus sargus). Acta Zoologica 96(2):236–241
Hassan A (2013) Anatomy and histology of the digestive system of the carnivorous fish, the brown-spotted grouper, Epinephelus chlorostigma (Pisces; Serranidae) from the Red Sea. Life Sci J 10(2):1–16
Hassan MWA (1990) Comparative biological studies between two species of family Sparidae, Boops boops and Boops salpa in Egyptian Mediterranean waters. M Sc. Alexandria Univ, Alexandria.
Kara A, Bayhan B (2015) Age and growth of Boops boops (L innaeus, 1758) in Izmir Bay, Aegean Sea. Turkey J of Appl Ichthyol 31(4):620–626
Khalaf-Allah H (2009) Biological studies on some Mediterranean Sea fish species with special reference to their feeding habits, growth and reproduction. Ph. D. Thesis, Zool. Dep. Fac. Sci., Al-Azhar Univ., Egypt., Pp 432. Accessed
Khemiri S, Gaamour A, Zylberberg L, Meunier F, Romdahane MS (2005) Age and growth of bogue, Boops boops, in Tunisian waters. Acta Adriat Int J Mar Sci 46(2):159–175
Kherraz A, Kherraz A, Boutiba Z (2016) Interrelationship age and growth of Boops boops (Linnaeus, 1758) in Western Mediterranean coasts of Algeria. Adv Environ Biol 10(4):140–146
Kubitza F, Lovshin LL (1997) The use of freeze-dried krill to feed train largemouth bass (Micropterus salmoides): feeds and training strategies. Aquaculture 148(4):299–312
Levanti M, Germanà A, Montalbano G, Guerrera MC, Cavallaro M, Abbate F (2017) The tongue dorsal surface in fish: A comparison among three farmed species. Anat Histol Embryol 46(2):103–109
Mahmoud UM, Essa F, Sayed AE-DH (2016) Surface architecture of the oropharyngeal cavity and the digestive tract of Mulloidichthys flavolineatus from the red sea, Egypt: a scanning electron microscope study. Tissue Cell 48(6):624–633
Matich P, Heithaus MR, Layman CA (2011) Contrasting patterns of individual specialization and trophic coupling in two marine apex predators. J Anim Ecol 80(1):294–305
Mehanna SF (2014) Stock assessment of bogue, Boops boops (Linnaeus, 1758) from the Egyptian Mediterranean waters. Vulnerability of agriculture, water and fisheries to climate change. Springer, Berlin, pp 313–322
Meyer-Rochow VB (1981) Fish tongues—surface fine structures and ecological considerations. Zool J Linn Soc 71(4):413–426
Motaref S (2013) Characterization and biological study on some species of family Sparidae in Ain El-Ghazala Gulf of eastern Libya. M. Sc. Thesis, Zoology Dep. Faculty of science. Univ. of Omar Al Mukhtar. Accessed.
Ostrander GK, Hopkins J (2000) The laboratory fish. Elsevier
Pollard D, Carpenter KE, Russell B (2014) Boops boops. The IUCN Red List of Threatened Species
Rachid B, Salim M, Zitouni B (2014) Estimation of the exploitable biomass and the reference biological point, F0. 1, of bogue Boops boops L., in the bay of Bou-Ismail, centre Algerian. J Biodivers Environ Sci (JBES) 5:420–427
Santos MLd, Arantes FP, Pessali TC, Santos JEd (2015) Morphological, histological and histochemical analysis of the digestive tract of Trachelyopterusstriatulus (Siluriformes: Auchenipteridae). Zoologia (curitiba) 32:296–305
Sayed AEDH, Mahmoud UM, Essa F (2019) The microstructure of buccal cavity and alimentary canal of Siganus rivulatus: Scanning electron microscope study. Microsc Res Tech 82(4):443–451
Shimada A, Kawanishi T, Kaneko T, Yoshihara H, Yano T, Inohaya K, Kinoshita M, Kamei Y, Tamura K, Takeda H (2013) Trunk exoskeleton in teleosts is mesodermal in origin. Nat Commun 4(1):1–8
Soykan O, İlkyaz AT, Metin G, Kinacigil HT (2015) Growth and reproduction of Boops boops, Dentex macrophthalmus, Diplodus vulgaris, and Pagellus acarne (Actinopterygii: Perciformes: Sparidae) from east-central Aegean Sea. Turkey Acta Ichthyologica Et Piscatoria 45:1
Stergiou KI, Moutopoulos DK (2001) A review of length-weight relationships of fishes from Greek marine waters.
Yashpal M, Kumari U, Mittal S, Kumar MA (2006) Surface architecture of the mouth cavity of a carnivorous fish Rita rita (Hamilton, 1822) (Siluriformes, Bagridae). Belg J Zool 136(2):155
Yashpal M, Kumari U, Mittal S, Mittal AK (2009) Morphological specializations of the buccal cavity in relation to the food and feeding habit of a carp Cirrhinus mrigala: A scanning electron microscopic investigation. J Morphol 270(6):714–728
Acknowledgements
We thank the Alexandria and Matrouh Universities for their help to complete this work.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
MA, MS, and SE wrote the main manuscript text. MA, MS, and SE prepared the samples and made the SEM, and light microscopy. MS, MA, MEB and MED made gross morphology and morphometry. MA and MS prepared figures. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
None of the authors have any financial or personal relationships that could inappropriately influence or bias the content of the paper.
Ethical approval
This study was approved by the local Animal Welfare and Ethics Committee, Faculties of Veterinary Medicine, Alexandria, and Matrouh Universities, Egypt.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alsafy, M.A.M., Seif, M.A., El-Gendy, S.A.A. et al. Micro-morphological characteristics of the oropharyngeal cavity floor of bogue (Boops boops) (Linnaeus, 1758) fish: gross morphology, light, and scanning electron microscopic imaging. Zoomorphology 142, 99–109 (2023). https://doi.org/10.1007/s00435-022-00577-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00435-022-00577-1