Abstract
The anatomical and histological features of the foveae in a gull, Larus michahellis, were investigated combining immunofluorescence and transmission electron microscopy. In an area of higher acuity, which extends in a band across the field of view, a central convexiclivate deep fovea was observed near of the pecten oculi. Moreover, a shallow fovea was present in the dorsonasal region. The central deep fovea was characterized by a remarkably and well-excavated pit containing extremely thin cones and a radial displacement of the cone axons and inner retinal layers. Immunofluorescence demonstrated the absence of rods and the presence of violet and green/red sensitive cone opsins. Ultrastructural analyses confirmed the lack of double cones, and specialized Müller cell processes forming a network that stabilizes the foveal structure. This deep central fovea, which provides the highest resolution and colour discrimination, may be used for monocular sideways vision of distant objects and a binocular fixation. The dorsonasal fovea was shallower and all retinal layers were present at the pit. This fovea showed the same composition of photoreceptors than central fovea and could be implied in magnification of a wide monocular part of the retinal image. The bifoveate condition of yellow-legged gull retina would allow the formation of three separate and distinct visual fields: two wide lateral monocular fields mediated by the dorsonasal and the central foveae, and a small binocular field mediated by the central deep fovea depending on ocular movements.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most birds use the vision as main sense to obtain information about their environment. Vision provides spatial information to navigating, evading predators, finding food and matchmaking (Brach 1977; Hart 2001; Fernández-Juricic et al. 2004; Jones et al. 2007; Martin 2007; Baumhardt et al. 2014). In birds, visual sensory demands vary substantially across species and the eyes and the retina of different species are adapted to best support their unique visoecological lifestyles (Walls 1942; Hughes 1977; Tyrrell et al. 2013; Moore et al. 2017a; Baden and Osorio 2019). The information received about the habitat will define the characteristics of the retina and different types, numbers and positions of retinal specializations coinciding with areas of greater visual acuity that allow to obtain a higher information about the visual space (Collin 1999b; Moore et al. 2017a). These specializations are areas, visual streak, radial anisotropy, area gigantocellularis and foveae (Moore et al. 2017a).
Areas are regions of the retina where a thickening occurs due the increased density of retinal neurons. They allow greater visual acuity and depending on the species they can be found in different retinal regions, although they are usually located at the centre of the retina (Slonaker 1897; Walls 1942; Hughes 1977; Collin 1999b; Rahman et al. 2006; Tyrrell and Fernández-Juricic 2015). The visual streak is described as a band-like thickening across the retina due to the increase in the density of ganglion cells. Visual streak serves to increase spatial resolution across the horizon and is generally found in species that live in open habitats dominated by a skyline (Slonaker 1897; Pumphrey 1948a; Hughes 1977; Hayes et al. 1991; Collin and Partridge 1996; Fernández-Juricic et al. 2011; Lisney et al. 2013). Radial anisotropy is a retinal specialization where neural cell density is configured opposite to the area, producing a concentric increase toward the periphery (Dunlop and Beazley 1984). This arrangement appears to be that it improves acuity at the periphery of the visual field, as well as being involved in motion detection. It is found in many aquatic species but it has not been described in birds (Dunlop and Beazley 1981, 1984; Collin and Partridge 1996). The area gigantocellularis is a specialized region consisting of a high proportion of giant retinal ganglion cells. These cells have large receptive fields which could increase sensitivity at the expense of spatial resolution, and are involved in motion detection (Hayes et al. 1991; Inzunza et al. 1991; Reuter and Peichl 2008; Coimbra et al. 2009). Hayes et al. (1991) hypothesized that the area gigantocellularis in procellariiform seabirds is used in movement detection and is usually assumed to be the same as that of the giant Y-type retinal ganglion cells in mammals (i.e., motion detection). Moreover, it has been reported in passerines (Coimbra et al. 2009) and fishes (Collin et al. 1998; Muguruma et al. 2013).
In birds, improved visual acuity may be correlated with the presence of well-developed area and fovea (one or two). Foveae are always found in an area, although this does not imply that there is a fovea in all areas. Foveae consist of an invagination of the retinal tissue caused by a radial displacement of the innermost layers of the retinal area resulting in a shallow, saucer-shaped or deep funnel-shaped (convexiclivate) cavity filled with vitreous humor (Meyer 1977; Moore et al. 2017a). It is characterized by presenting a peak of cone density and in most species rods could be absent (Bruhn and Cepko 1996; Querubin et al. 2009; Coimbra et al. 2015; Mitkus et al. 2017). Martin and Grünert (1999) and Wikler and Rakic (1990) reported that not blue-sensitive cones but only green and red-sensitivity cones were present in primate fovea. Then, fovea improves the optical, chromatic and spatial processing of the retina, resulting in a higher visual acuity (Pumphrey 1948b; Mitkus et al. 2017; Bringmann 2019). In a recent study in raptor foveae, Mitkus et al. (2017) had reported a lack of double cones in the center of the central fovea, except in the Eurasian sparrowhawk, and the presence of violet and green-sensitive cone opsine cells, which suggest a high-resolution tetrachromatic vision in the central fovea of raptors.
Foveal depths vary in different birds and can be classified into two types depending on the depth of the pit: deep fovea or shallow fovea (Slonaker 1897; Walls 1937; Pumphrey 1948b; Fite and Rosenfield-Wessels 1975). The deep fovea or convexiclivate fovea is usually located in the centre of the retina and has a deep invagination. It is characteristic of fish, reptiles and birds (Walls 1937, 1942; Pumphrey 1948b; Fite and Rosenfield-Wessels 1975; Makaretz and Levine 1980; Locket 1992; Collin 1999a), and is especially deeper in raptors and other birds which catch small or fast-moving objects (Wood 1917; Snyder and Miller 1978; Reymond 1985, 1987). In turn, the deep fovea can be classified into three subtypes depending on the arrangement of the inner retinal layers in the foveal pit (Collin 1999a; Moore et al. 2017a): type I, deep or convexiclivate fovea without a lateral displacement of the inner retinal layers (Schwassmann 1968; Lisney et al. 2015; Moore et al. 2016). Type II is also a deep fovea with inner retinal layers displaced laterally (Moore et al. 2016), which allows light to strike the photoreceptors without passing through inner retinal layers. Type III fovea is similar to type II but also possesses a thick and dense layer in the bottom of the foveal pit with radial inner processes of Müller cells (Locket 1992; Zueva et al. 2014). Finally, the shallow or concaviclivate fovea is an invagination that exhibits no lateral displacement or radial fibres (Collin 1999a). It is relatively flat and is found in anthropoids (Bringmann et al. 2018) and in the temporal retina of some birds (Ruggeri et al. 2010; González-Martín-Moro et al. 2017; Mitkus et al. 2017).
Birds can possess one or two foveae (Slonaker 1897; Walls 1942; Pumphrey 1948b; Meyer 1977) and the variation of their position in the retina may be related to each bird lifestyle and its possible functions in its vision. Monofoveal birds have a deep fovea in the central retina (Pumphrey 1948b; Fite and Rosenfield-Wessels 1975; Locket 1992; Coimbra et al. 2006, 2014a; Moore et al. 2016; Mitkus et al. 2017; Potier et al. 2017; Bringmann 2019) while bifoveal birds also have a second shallow fovea in the temporal retina (O’Day 1940; Moroney and Pettigrew 1987; Inzunza et al. 1989; Collin 1999a; Tucker 2000; Baumhardt et al. 2014; Mitkus et al. 2017, 2018; Moore et al. 2017b; Potier et al. 2017; Bringmann 2019). The central fovea possesses the highest density of cone photoreceptors of the entire retina, improving the visual acuity and colour discrimination (Slonaker 1897; Walls 1937; Pumphrey 1948b; Fite and Rosenfield-Wessels 1975; Meyer 1977; Reymond 1985, 1987; Collin 1999a; Moore et al. 2012; Zueva et al. 2014; Lisney et al. 2015; Mitkus et al. 2017; Bringmann 2019). It is used for monocular sideway vision of distant objects, whereas the temporal shallower fovea serves to facilitate frontal binocular vision to guide the bill at close distances (Querubin et al. 2009).
In addition to improve visual acuity, several functions have been attributed to the fovea. A local image magnification and detection of the focus plane (Walls 1937; Pumphrey 1948b; Snyder and Miller 1978; Collin 1999a; Bringmann et al. 2018; Syrbe et al. 2018; Bringmann 2019), so that incoming light is centrifugally refracted through Müller cells towards the foveal pit (Walls 1937, 1942; Pumphrey 1948b; Snyder and Miller 1978; Bringmann et al. 2018; Bringmann 2019). The role of Müller cells as light-guiding elements has recently been proposed (Franze et al. 2007; Agte et al. 2011; Zueva et al. 2014; Bringmann et al. 2018; Syrbe et al. 2018; Bringmann 2019). They have at least two optical functions in the foveal pit: to increase the image in the centre of the fovea and to direct light from the internal limiting membrane (ILM) to the photoreceptors acting as “light fibres” (Franze et al. 2007; Omri et al. 2010; Agte et al. 2011, 2018; MacDonald et al. 2015; Syrbe et al. 2018). Moreover, processes of Müller cells could give structural support of the tissue, especially in the region of the fovea (Franze et al. 2007), acting like a belt which prevent a collapse of the foveal pit (Bringmann 2019). In this way, the inner surface of type III fovea presents a high density of inner processes of Müller cells forming dense aggregates as stellate rays in the internal part of the foveal pit (Snyder and Miller 1978; Locket 1992; Zueva et al. 2014; Bringmann et al. 2018; Bringmann 2019). On the other hand, Kreithen and Keeton (1974) attributed a navigation role of the fovea based in the detection of polarized light in Columba livia. They believe that the sloping walls of the fovea might reflect light so that it strikes the opposite side of the fovea at an angle different depending on planes polarized light. Finally, Pumphrey (1948a, b) proposed that the deep bird fovea serves motion detection, and Bloch and Martinoya (1982) suggested that the central fovea is ideal for detecting fast-moving objects, whereas the afoveal dorsal area is best suited for detecting slow moving and static objects.
There are several studies that describe the structure of the fovea in birds, such as raptors, passerines, hummingbirds, cockatoos, albatross and others (Wood 1917; O’Day 1940; Pumphrey 1948b; Fite and Rosenfield-Wessels 1975; Meyer 1977; Snyder and Miller 1978; Reymond 1985, 1987; Khokhlova et al. 2000; Zueva et al. 2003, 2014; Coimbra et al. 2006, 2014b; Ruggeri et al. 2010; Lisney et al. 2015; Bringmann 2019), although they are scarce in seabirds (Mitkus et al. 2016). There are few studies in which the fovea in the family Laridae has been described. Slonaker (1897) reported that both Larus canus and Larus ridibundus had two foveae, deep and shallow. Kajikawa (1923) described a central deep fovea and a temporal shallow fovea in Larus argentatus, and a streak-like and a central fovea in Larus canus.
The aim of this study was to analyse the morphology, photoreceptor composition and ultrastructural characteristics of the foveae in a seabird, Larus michahellis, combining immunochemistry and transmission electron microscopy. We provide data concerning several anatomical aspects of the two foveae found in Larus michahellis and discuss the possible role of each one.
Materials and methods
Animals and tissue preservation
For this study, ten ocular globes from five adults’ specimens and two ocular globes of juvenile gulls, L. michahellis, were obtained from Santa Faz Wildlife Recovery Centre (Alicante). Animals were euthanised, for reasons not connected with this work, by means of an intravenous overdose of sodium pentobarbital (EutanaxR) according to the European Union and the Spanish government regulations and standards (Council Directive 86/609/EEC).
Eyes were enucleated and hemisected carefully under normal light room 1–2 h after death. Following the enucleation, eyes were dissected and their anterior portion (including cornea, lens and iris) was cut away. Three right retinas were processed for transmission electron microscopy. Samples were immersed in a fixing solution consisted of 1% paraformaldehyde, 1.6% glutaraldehyde, 0.15 mM CaCl2 in 0.1 M phosphate buffer adjusted at pH 7.4, were they remained overnight at 4 °C as described by Segovia et al. (2016). On the other hand, seven retinas (five left retinas embedded in paraffin and two right retinas for wholemount technique) were processed for immunolabeling and fixed in 4% paraformaldehyde for 2 h.
Processing for transmission electron microscopy
After fixation, retinas were excised from the eyecup. Foveae were located, photographed, and then, cut carefully. Small pieces (2 × 2 mm) including foveae, central and peripheral retina were postfixed in 1% OsO4 in 0.1 M phosphate buffer for 1 h, pH 7.4 and dehydrated in ascending graded of ethanol solutions and propylene oxide. They were embedded in Epon 812 epoxy resin (EMS). Blocks were oriented and semi-thin sections of 1 µm were cut serially in a Leica LKB-III ultramicrotome. Sections were mounted on gelatinised slides, stained with 0.5% toluidine blue, and examined under a Leica DMRB light microscope. Photomicrographs were taken with a Lumenera Infinity microscope camera. Ultrathin sections were double-contrasted with uranyl acetate (0.5%) and lead citrate (0.25%) and examined under a JEOL JEM-1400 Plus transmission electron microscope at 120 kV (Tokyo, Japan) equipped with a Gatan Orius digital camera (Pleasanton, USA) for image acquisition.
Retinal immunofluorescence
To evaluate photoreceptor composition and Müller cells arrangement in foveae, immunohistochemical identification was performed. Different cone opsins were appropriately identified: anti-L/M-opsin (red/green cone sensitivity opsin, AB5405; Chemicon®), and anti-S-opsin polyclonal antibodies (AB5407) which have been shown to be ultraviolet/violet cone-specific in birds (Nießner et al. 2011; Mitkus et al. 2017). Rods’ outer segments were labelled with a mouse-monoclonal anti-rhosopsin antibody (clone RET-P1, MAB5316, Chemicon®), which reacts with a protein of 39 kDa identified as rhodopsin (opsin), and allows to label rod outer segments in a wide range of vertebrate species including rodents, fishes, reptiles and some birds (BarnsTable 1980; Silver et al. 1988; Querubin et al. 2009; Bhattacharyya et al. 2017; Schweikert and Grace 2018). Finally, to identify and quantify Müller cells, we used a rabbit-polyclonal anti-glutamine synthetase antibody (GS, G2781, Sigma Aldrich Co.). GS is present in the brain mainly in astrocytes and in Müller glial cells supporting retinal neurons using glutamate as a neurotransmitter (Martinez-Hernandez et al. 1977; Riepe and Norenberg 1978; Norenberg and Martinez-Hernandez 1979; Lewis et al. 1988; Newman and Reichenbach 1996). All primary antibodies used in this work have been previously used in other studies and have been well characterized by other authors (Riepe and Norenberg 1978; Kentroti et al. 1991; Fischer et al. 2007; Vlahos et al. 2014; Coimbra et al. 2015; Mitkus et al. 2017).
Sections and wholemount retinas were immunostained at room temperature overnight or for 2 days, respectively, using antibodies targeting distinct molecular markers (summarized in Table 1). The dilutions in PBS 0.1 M with 1% Triton X-100 are indicated in Table 1. Afterwards, secondary antibodies were added (at a dilution as it is indicated in Table 1) for 2 h for sections and overnight for wholemount retinas. Sections were rinsed in PBS and coverslipped with Vectashield H-1000 with 4ʹ6-diamidino-2-phenylindole (DAPI) (Vector laboratories, Burlingame CA, USA) used as a nuclear marker. Finally, wholemount retinas were washed in PBS and mounted (photoreceptor side up) on nongelatinized glass slides. To allow them to lay down flat, radial cuts were made and the pecten oculi was cut out but used to conserve its orientation. Immunohistochemical controls were carried out by omitting the primary antibodies. The resulting images were processed with a confocal microscope (Zeiss LSM-800) and microphotographs were taken with an AxioCam 506 digital camera. Image adjustments were made with the ZEN blue imaging software (Zeiss), maintaining the intensity and the exposure times in all the samples.
Retinal measurements
Foveal depth and width were measured in 20 serial cuts of semi-thin sections of the foveal pit and settled on the one with the greatest depth. In this way, we were able to confirm the most central part of the fovea. These measurements were made in four central and one dorsonasal fovea with the FOVEA software (Moore et al. 2016). We confirmed the results with imageJ software. We also measured the thickness of the retinal layers in the foveal pit and the parafovea, the photoreceptors diameter, the inclination of the columns of bipolar cells, the ratio photoreceptors-bipolar cells-ganglion cells, and the rod-free zone surface in wholemount retinas with ImageJ software. The ratio photoreceptors-bipolar cells-ganglion cells was measured along the width of the foveae. Criteria to identify and counting different types of cells located in inner nuclear layer are based on Vidal et al. (2018).
To know the position of the foveae in the retina, we calculated the angle formed when drawing an imaginary line between the central fovea (α) and the dorsonasal fovea (β) from the optic nerve entrance with respect to the equatorial line.
To estimate the angle of light of sight (LOS) of L. michahellis, we analysed the images taken from directly above gull’s head while the animal was alive. Once dead, we made images of the skull showing the position of the orbits. On the other hand, images of the eyeballs were made to know their position in the skull through the optic nerve. Images were overlaid in Adobe Photoshop 2020 and drawn. Knowing the exact position of both foveae on the retina, an imaginary line was drawn from each fovea passing through the centre of the lens. From a reference line located at the beak of the animal (which we consider the central axis of the head), we calculated the estimated angle that the LOS of each fovea formed with respect to it. Angle measurements were calculated with Adobe Photoshop 2020.
Results
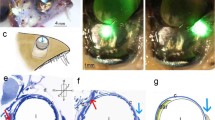
Located in a band-like area which extends horizontally in the retina, L. michahellis has two different circular foveae: one deep fovea located in the central region and one shallow fovea in the dorsonasal region (Fig. 1). The central fovea was easily recognizable macroscopically in retinal wholemounts after dissection as a dark point surrounded by a thickened round area. It is nearly located in the central region of the retina, slightly above and toward the nasal side of the optic nerve (ON) entrance and the pecten oculi. It was located 2.285 ± 0.163 mm from the ON entrance and forms an angle of 28° (α) with respect to the equatorial line. The dorsonasal fovea is more difficult to observe, so it is necessary to carefully remove the highest amount of vitreous humour. It is located 8.259 ± 0.048 mm from the central fovea and forms an angle of 18° (β) from the ON entrance with respect to the horizon. The pecten oculi forms an angle of 67° with respect to the equatorial line (Fig. 1b).
a Position of the deep fovea or central fovea (Df) and shallow dorsonasal fovea (Sf) in the retina of L. michahellis. b Angle that forms the deep fovea (α) an the shallow fovea (β) from the entrance of the optic nerve with respect to the position of the equatorial line. c Appearance of the retina showing position of both foveas after removing the anterior part of the eye. In both cases a right retina is shown. Pecten oculi (PO)
Having two foveas implied that yellow-legged gull had two different lines of sight (LOS), one corresponding to the deep fovea and another characteristic of the shallow fovea. LOS of central fovea makes an angle of 62.5° with the reference line (located at the bill), whereas LOS of dorsonasal fovea makes an angle of 89°. The deep fovea, had its LOS closer to the reference line, therefore was involved in both monocular and binocular vision, while the shallower fovea participated in monocular vision (Fig. 2).
Foveal morphology
Light microscopy analyses revealed that the central fovea showed a radial displacement of the outer and inner retinal layers resulting in a deep funnel-shaped (convexiclivate) cavity and thickened sloping sides (Fig. 3a). Then, the foveal pit presented a reduced number of cells in outer and inner nuclear layers, and ganglion cell layer. The retina was 150 µm in thickness within the foveal pit (a), increasing to 342 µm in the parafoveal region (b) (Fig. 3a). In contrast, dorsonasal fovea had a shallower pit where the retina has 205 µm in thickness (aʹ) and 263 µm in the parafovea (bʹ) (Fig. 3b). The depth of the pit in the central fovea was 226 µm (d) while in the dorsonasal fovea was 49 µm (dʹ). The width, which was measured from the points of maximum thickness, was 598 µm (w) in the central fovea and 273 µm (wʹ) in the dorsonasal fovea (Fig. 3a). In dorsonasal fovea, there are also reduced cell numbers in the central outer and inner nuclear layers.
Histological features of the central deep fovea
Vertical sections of the central deep fovea showed that photoreceptors become thinner and longer when approaching to the pit, and their elongated processes run obliquely through the outer nuclear layer (Fig. 4a). The diameter of internal segments was 1.7 ± 0.19 µm (Fig. 4b). On the other hand, it is conspicuous of the spindle shape of the photoreceptor cell nuclei.
Photomicrographs of semi-thin (a, c, e) and ultrathin sections (b, d, f) showing different regions of the central fovea of L. michahellis. a, b Vertical sections of the central deep fovea showed the photoreceptors. It observes a decrease in the relative thickness of the layers in the pit. c Note that bipolar and amacrine cells are aligned in rows and they are obliquely disposed. d A ganglion cell surrounded by processes of Müller cells. e, f Ganglion cells in parafoveal region are arranged in columns of up to 4 ganglion cells. OS outer segments, IS inner segments, ONL outer nuclear layer, INL inner nuclear layer, IPL inner plexiform layer, GCL ganglion cell layer, NFL nerve fibre layer, GC ganglion cell, VH vitreous humour, Bp bipolar cells, Am amacrine cells. Asterisk: Müller cells processes, arrows: OLM (outer limiting membrane); black arrowhead: mitochondria/ellipsoid; white arrowhead: Müller cells endfeets
The decrease in the relative thickness of the layers in the pit with respect to the maximum thicknesses of the inner retinal layers in the parafovea implied a radial displacement of cells and their processes away from the foveal pit (Fig. 3a). Bipolar cells were aligned in rows and obliquely disposed, with a maximum inclination of 65° from the direction of light that reach the retina. Amacrine cells were also arranged in columns but in the opposite direction to the bipolar rows (Fig. 4c). In the centre of the fovea, ganglion cells were scarce or practically non-existent (Fig. 4d) while in the parafovea, they were arranged in columns of up to 4 ganglion cells (Fig. 4e, f). The ratio of photoreceptors-bipolar cells to ganglion cells in the central fovea was 2:4:1. From the fovea to central and peripheral regions of the retina, the number of ganglion cells is reduced from about four cells thick to about one cell thick.
The radial alignment of inner retinal layers reflected the arrangement of Müller cells processes which framed the rows of bipolar, amacrine and ganglion cells (Fig. 4). Ultrastructural analyses demonstrated the presence of Müller cells processes in the bottom of the foveal pit and the walls of the slope (Fig. 5a). The main inner processes of Müller cells are radially oriented, whereas the thinner side processes appeared as a tangled and very electrodense aggregation (Fig. 5b). This characteristic arrangement was also observed in the outer plexiform layer (OPL), between photoreceptors somata and their terminal axons (Fig. 5c). In this layer, there are more or less regular stacks of thin lamellar Müller cell processes. Müller cells somata were usually located in the inner part of the INL between bipolar cells nuclei and amacrine cells (Fig. 5d). In the rest of the retina, Müller cells run vertically from the ILM to the outer limiting membrane (OLM). Due to the increased density of ganglion cells in the parafovea, their axons formed vertical columns in the nerve fibre layer (NFL) surrounded by Müller cells inner processes (Fig. 5e). In peripheral retina ganglion cells, axons were randomly located between inner processes of Müller cells. Its endfeet were very electrodense forming the ILM (Fig. 5f).
Ultrathin vertical sections of the retina of L. michahellis. a–c Conformation of Müller cells processes in the central fovea. d Electron-dense soma of a Müller cell. Disposition of Müller cells processes in the NFL of a central region of the retina (e) and a peripheral one (f). Asterisk: Müller cells inner processes; white arrowhead: Müller cells endfeet, arrows: Müller cells outer processes. PR photoreceptors, ST synaptic terminal, M Müller cell nucleus, GC Ganglion cell, Ax axons of the NFL
Histological features of the dorsonasal fovea
In the dorsonasal fovea, all retinal layers were present in the foveal pit. However, inner retinal layers were thinner than in the parafovea. The diameter of internal segments in dorsonasal fovea was 2.1 ± 0.31 µm (Fig. 6a), thicker than the internal segments of the central fovea. Bipolar cells columns were slightly curved, but they did not reach the inclination observed in the central fovea (Fig. 6b). As occurs in the central deep fovea, in the center of the dorsonasal fovea in adults, ganglion cells were scarce whereas in the parafovea, they were arranged in columns of up to 4 ganglion cells. Ganglion cells were separated by Müller cells processes. In juvenile retinae, only one layer of ganglion cells was found in the pit of dorsonasal fovea (Fig. 6c). The ratio of photoreceptors-bipolar cells to ganglion cells in the dorsonasal fovea was 3.5:7:1.
Photoreceptor composition in the foveal pit
Anti-Rhosopsin immunolabeling in wholemount retinas shows a central rod-free zone corresponding with the central fovea. This rod-free region was relativity circular in shape (0.122 ± 0.01 mm2) and occupied about 0.02% of the total retinal area (5.342 ± 0.411 cm2) (Fig. 7a). In radial sections of both foveae, we confirmed that this rod-free zone corresponds with the foveal pit (Fig. 7b, c). These results indicate that both foveae of the yellow-legged gull lack rods. In both wholemount and vertical sections, a weakly labelling is detected in the rod-free zone that suggests a RET-P1 antibody cross reaction with other cone-photoreceptors situated in the foveal region.
Distribution of rods outer segments in the deep central fovea of a flattened retina (a) and vertical sections (b) of L. michahellis. Wholemount (a) and vertical section of deep central fovea (b) and dorsonasal nasal fovea (c) labelled with antibodies directed to rhodopsin (Ret-P1; green). DAPI used to counter-stain nuclei are shown in blue
In regard to cone composition, we observed a positive labelling when using anti-S opsin and anti-L/M-opsin antibodies (Fig. 8). In fact, the cone labelling becomes more intense in the foveal pit due to increased cone density. This pattern indicated that both foveae of L. michahellis contained red, green and UV-sensitive cones.
Müller cells immunofluorescence
Glutamine-Synthetase expression in the yellow-legged gull retina demonstrated that Müller cells were ranged from the ILM to the OLM (Fig. 9). In vertical sections of central deep fovea, stalks and distal processes of Müller cells run obliquely from GCL to the bottom of the OPL, with an opposite direction to the foveal pit. In the INL, bipolar cells were stacked in columnar rows and intersected with Müller cell processes displaying an x-shaped arrangement (Fig. 9a′). Once in the OPL, there was a change in the direction of their distal processes. Two horizontal rows of Müller cell processes were highly labelled in the OPL and in the GCL in the foveal pit (Fig. 9a, a′). An intense labelling was observed in central regions in the endfoot of the Müller cells (Fig. 9b, b′). A weak labelling of IPL in central and peripheral regions (Fig. 9c, c′) was observed.
In dorsonasal fovea, the Glutamine-Synthetase expression did not show the x-shaped arrangement of Müller cell processes.
Discussion
In recent years, considerable amount of literature has been published on structure and function of fovea in birds (Bringmann 2019), especially in raptors (Lisney et al. 2013; Velasco Gallego 2015; Potier et al. 2016, 2017, 2020; Mitkus et al. 2018). However, there have been relatively little reports on fovea of seabirds’ retina (Mitkus et al. 2016). Although recent studies have only focused on morphological characteristics of the retina and the pecten oculi of yellow-legged gull (Vidal et al. 2018; Segovia et al. 2019), the few studies about the fovea in gulls are very ancient (Slonaker 1897; Kajikawa 1923). In the present study, we have reported the presence of two foveae in the retina of L. michahellis, a central deep fovea and an unexpected shallower dorsonasal fovea. Both have been observed in the wholemount retinas analysed under a steromicroscope.
This study has been performed using adult and juvenile individuals of different sex and different eyes, and no significant differences in the foveal morphology were observed. These data support the results of Potier et al. (2020) in Milvus migrans where no difference between individuals of different age or sex was found, although significantly differences exist in the size of the eye and structure of the fovea during development.
Anatomical features of two foveae
Histological analysis of cross retinal sections established that the central fovea of L. michahellis had a deeper pit and steeper walls compared to the dorsonasal fovea where the invagination was shallower. This shallow depression is compatible with the “band-like fovea” described by Slonaker (1897) and Kajikawa (1923) in the retina of L. canus and L. ridibundus. In addition, in other seabird as the tern, Sterna hirundo, a linear area with enclosed central area (and fovea) and separate lateral area (and fovea) has been described (Krause 1894; Slonaker 1897). In this sense, wholemount retinas of L. michahellis show a horizontal linear band across the field of view containing a deep central fovea and a dorsonasal fovea. The existence of a central band projecting upwards and forwards to locate targets both above and below the water is compatible with the results of previous studies that found that the central region of L. michahellis retina was thicker than the rest of the retina (Vidal et al. 2018).
These results are consistent with the considerable variation reported regarding morphological characteristics of the fovea (Walls 1942; Fite and Rosenfield-Wessels 1975; Reymond 1985, 1987; Moroney and Pettigrew 1987; Inzunza et al. 1991; Locket 1992; Tucker 2000; Ruggeri et al. 2010; Potier et al. 2016). The variation in both depth and width of the foveal pit is according to the lifestyle and habitat (Fite and Rosenfield-Wessels 1975; Coimbra et al. 2014b; Potier et al. 2017). Birds that capture their prey in the air need to have a higher motion sensitivity compared to those that feed on passive prey on the ground, so they usually present larger eyes, with a thicker retina and have a deeper central fovea and a shallow temporal fovea (Fite and Rosenfield-Wessels 1975; Inzunza et al. 1991; Baumhardt et al. 2014; Moore et al. 2015; Potier et al. 2017). Many other bird species also possess a fovea, and two foveae have been reported in other pursuit-hunting species, such as swallows, martins, kingfishers, and terns (Wood 1917; Walls 1942; Meyer 1977). Our results demonstrated that the yellow-legged gull is a bifoveate seabird similar to other birds that hunting their prey. The central fovea is comparable to deep central fovea in raptors, and the dorsonasal fovea is similar to temporal fovea of these birds.
Although foveae have been classified in four types (Collin 1999a; Collin and Shand 2003), in a recent review study Bringmann (2019) established that there are two ranges of foveal depth among different birds’ groups. On the one hand, the central foveae of the accipitriform, falconiform, coraciiform and passeriform have values between 190 and 240 µm and, on the other hand, the central foveae of psittaciform birds and domestic pigeons, as well as the temporal foveae of accipitriform, falconiform, coraciiform and strigiform birds with a depth range between 80 and 120 µm. The deep central fovea of L. michahellis (226 µm in depth) could be classified within the first group but the shallower dorsonasal fovea (49 µm in depth) could be classified as shallow.
According to Bringmann (2019), the higher thickness of the foveal centre in the shallow foveae may result in greater image magnification than in central foveae. This may compensate the lower receptor and ganglion cell densities found in the shallower fovea than in the deeper central fovea of bifoveal birds. In this way, the dorsonasal fovea in yellow-legged gull, where the photoreceptor-bipolar-ganglion ratio is 3.5:7:1, suggests an image magnification and a higher sensitivity, whereas the central fovea, with a 2:4:1 may be related with a high visual acuity plus better motion sensitivity.
Foveal position and implications in the visual field
In the present study, we have reported the presence of two foveae in the retina of L. michahellis, a central deep fovea and a shallower dorsonasal fovea. There are similarities between these results and those reported in genus Larus. Thus, in an ancient research, Slonaker (1897), showed that both Larus canus and Larus ridibundus present two foveae, a deep and a shallow fovea. Later, Kajikawa (1923) described that Larus argentatus had a central deep fovea and a temporal shallow fovea. Furthermore, Larus canus presented a streak-like central fovea, but a second fovea had not been found in the six eyes studied (Kajikawa 1923), despite having been described in Chievitz’s analysis (Slonaker 1897).
Presence of two foveae implies that the yellow-legged gull has two different lines of sight. Each foveal LOS points forward and lateral forming an angle with the reference line located at the peak. In the deep fovea it was approximately 62.5° and 89° in the shallower fovea. In comparison, Tucker (2000) proposed that the ideal visual system of a falcon would have the LOS of the deep fovea at about 45° from the bill and the shallow fovea, in temporal region, points more forward at an angle of 15°. Despite having practically the same angle between the two LOS, these differences could due to the fact that the falcon has its eyes more forward than the gull.
In L. michahellis, the central fovea, is pointing slightly below and towards the edges of the bill and may be involved in monocular sideways vision of distant objects and binocular vision. We have observed that the yellow-legged gull is able to move the eyes inside the orbits, which would align the center of acute vision to objects of interest and to increase the amount of the binocular field. This fovea matches with an elongated strip of high photoreceptor density which is found in the equator of the retina (nonpublished data). The location of the strip, which had been described in other seabirds (Hayes et al. 1991), would project out towards the horizon when the gull is in flight, and the central fovea, concerned primarily with high spatial resolution, suggests a function in sideways vision of distant objects and guiding the procurement of small prey in the open sea. A central deep fovea implied in monocular sideways vision of distant objects had been described in raptors, passerines, martins, terns, kingfishers, and some other birds (Khokhlova et al. 2000; Zueva et al. 2003).
The dorsonasal fovea, has its LOS projecting further back, which added to the lateral placed of the orbits, increases its monocular field of vision. This is opposite to the possible role of the temporal shallow fovea in bifoveate birds, which could participate in binocular vision to guide the bill at close distances (Reymond 1985, 1987; Tucker 2000). The particular location of the shallower fovea in the dorsonasal region could be explained by the high laterality of the eyes in the skull comparing with other birds, including raptors (Tucker 2000). This shallow fovea would be related to monocular vision improving the visual exploration downwards when searching for food (Moore et al. 2015).
All data suggest that in L. michahellis, the foveal position, central and dorsonasal, would permit to see both the horizon and a vast monocular vision field creating a superb mechanism to detect objects. Moreover, small ocular movements in combination with the head movements would permit both eyes to focus the objects on their foveae.
Histological features in the fovea
Analyses of vertical sections revealed that in the deep central fovea of yellow-legged gull, all inner retinal layers were shifted laterally and the centre of the fovea and displayed a strongly reduced numbers of neurons and photoreceptors. In fact, the pit of central fovea practically lacks of ganglion cells. However, Kajikawa (1923) reported the presence of ganglion cells at the bottom of the central foveal pit in retina of Larus canus, which support the great variation in foveal morphology. Contrarily, in shallower dorsonasal fovea, all neural layers were present and a light centrifugal displacement was observed.
In regard to the histological features, a considerably variability has been reported. Whereas American goldfinches, doves, cockatoos, gulls, hummingbirds, pigeons, terns, tyrant flycatchers and sparrows, present all at the retinal layers in the deep fovea, (Kajikawa 1923; Coimbra et al. 2006, 2014b; Querubin et al. 2009; Baumhardt et al. 2014; Lisney et al. 2015; Moore et al. 2016), raptors, some passerines, petrels, kingfishers, albatrosses, eastern meadowlarks, house sparrows, and great kiskadees, do not present ganglion cells in the foveal pit (Kajikawa 1923; O’Day 1940; Pumphrey 1948b; Reymond 1985; Moroney and Pettigrew 1987; Coimbra et al. 2006; Tyrrell et al. 2013; Mitkus et al. 2016, 2017; Moore et al. 2016). Once again, deep fovea of the yellow-legged gull is like other birds that hunting their prey, including seabirds as albatrosses and kingfishers. However, the morphology of shallower dorsonasal fovea is similar to temporal fovea of birds which have two foveae.
As it was confirmed by immunolabelling with RET-P1, the foveae of the yellow-legged gull do not present rods. As rods cannot operate under bright-light conditions, they are absent in the foveae of many diurnal species, such as: primates—including humans—(Provis et al. 2005, 2013; O’Brien 2008; Bringmann et al. 2018) and most birds (Querubin et al. 2009; Coimbra et al. 2015; Mitkus et al. 2017). In regard to cones, immunofluorescence showed the presence of UV-sensitive cones and LWS- and MWS-sensitive cones.
Previous studies demonstrated the presence of UVS opsin in two families of seabirds, Laridae and Sternidae (Ödeen et al. 2010; Machovsky Capuska et al. 2011; Ödeen and Håstad 2013). While short wavelengths sensitive cones could produce chromatic aberrations that would harm the image quality obtained in the fovea (Mitkus et al. 2017), Varela et al. (1993) proposed that UV wavelengths could serve a signalling function and could also play a role in aerial navigation as an adaptation to the coloration of an unclouded sky. We think the presence of UV pigments in yellow-legged gull may assist navigation when the sun is hidden by clouds. Another reason why UV-sensitive photoreceptors are present is that many prey animals like reptiles, insects, and birds are visible in UV light because they send out UV rays.
Although immunolabelling with anti-L/M-opsin antibody has not allowed us to differentiate between LWS, MWS-sensitive cones or double cones, ultrastructural analyses demonstrated the absence of double cones in the foveal pit region, coinciding with studies that confirm the absence of double cones in the raptor fovea (Mitkus et al. 2017). Once more, the anatomy, histology and photoreceptor composition of central fovea in L. michahellis, allow us to compare it with a diurnal raptor.
Light and ultrastructural micrographs suggested a highest density of cones in the central fovea of L. michahellis that results in thinner and slender inner segments, as occurs in many other birds species (O’Day 1940; Pumphrey 1948b; Fite and Rosenfield-Wessels 1975; Tucker 2000; Mitkus et al. 2017). Then, the high density of cones correlated with a high density of bipolar and ganglion cells which are arranged in columns that run obliquely towards the foveal pit. This special arrangement is mechanically stabilized by Müller cells as immunofluorescence had demonstrated. In this way, Bringmann (2019) had suggested that Müller cells in fovea could act like a belt which prevent a collapse of the foveal pit.
Ultrastructural analyses of central fovea demonstrated that Müller cells processes significantly increase its surface area. Müller cells inner processes form dense aggregates as stellate rays in the internal part of the foveal pit. Moreover, the ganglion cell columns were separated by the Müller cells inner processes. This arrangement had been reported previously in bird foveae (Snyder and Miller 1978; Collin 1999a; Zueva et al. 2014). In birds of prey, it has been described that these aggregates are packaged by intermediate filaments (Snyder and Miller 1978). Specialized layers of Müller cells processes have been found too in the inner surface of the foveal centre in teleost, lizards, primates and other birds (Snyder and Miller 1978; Makaretz and Levine 1980; Locket 1992; Zueva et al. 2014; Syrbe et al. 2018; Bringmann 2019).
The importance of Müller cells in the retina has been studied in multiple species, such as primates, caimans, zebrafish, birds, guinea pig (Inoue et al. 1980; Agte et al. 2011, 2018; Zueva et al. 2014; MacDonald et al. 2015; Bringmann et al. 2018; Karl et al. 2018; Syrbe et al. 2018). Besides providing structural support, Müller cells extend perpendicularly its endfeeds that form the ILM, guiding the light to the outer processes that form the OLM, at the level of the photoreceptor’s inner segments (Walls 1942; Reichenbach 1989; Newman and Reichenbach 1996; Omri et al. 2010; Reichenbach and Bringmann 2013; Zueva et al. 2014; Agte et al. 2018; Karl et al. 2018). However, in the retina of many birds, Müller cells inner processes are divided into numerous thin branches, which would not facilitate the light guidance (Reichenbach and Bringmann 2010). This could justify the presence of a second fovea in birds, so the internal processes are more aligned as occurs in dorsonasal fovea in L. michahellis, acting as living optical fibre that guide light through the retina (Franze et al. 2007; Agte et al. 2011; Zueva et al. 2014). Therefore, Müller cells would become an optical extension of cones (Zueva et al. 2014) that would link the inner retina with the photoreceptors like a “light cable” (Franze et al. 2007) of the foveal pit, suggesting that the conical shape of the pit is an advantage for images formation (Zueva et al. 2014).
Conclusion
In summary, our study confirms that foveal morphology varies greatly among species according to its visual ecology. Retinae of L. michahellis show many features usually associated with a diurnal raptor, as the presence of a deep central fovea probably of high-acuity resolution and related to the monocular and binocular vision. Moreover, cone composition is similar to those of raptor. As diurnal raptor, it has a shallower second fovea located in dorsonasal region which may be related to the increase of ability to follow moving objects. It would be related to increasing its monocular visual field improving the visual exploration downwards when searching for food. Although a linear area with a central fovea has been described in many ground feeders, swimmers, divers and waders, a shallow dorsonasal fovea has not previously been described in raptors neither Laridae nor other seabirds. The particular location of the shallower fovea in the dorsonasal region could be explained by the high laterality of the eyes in the skull comparing with raptors. Ocular and head movements may increase the binocular vision. Moreover, this work has added numerous details emphasizing the high acuity of the central fovea in this gull: (1) No rods and double cones are present, but an increased density of cones, at least, red, green and UV-sensitive cones may confer a good chromatic vision. (2) A ratio photoreceptor-ganglion cells 2:1 which suggest a good visual acuity. (3) The particular arrangement of Müller cell processes could suggest a role as guiding the light to the photoreceptors in dorsonasal fovea and a mechanical role in deep central fovea.
References
Agte S, Junek S, Matthias S et al (2011) Müller glial cell-provided cellular light guidance through the vital guinea-pig retina. Biophys J 101:2611–2619. https://doi.org/10.1016/j.bpj.2011.09.062
Agte S, Savvinov A, Karl A et al (2018) Müller glial cells contribute to dim light vision in the spectacled caiman (Caiman crocodilus fuscus): analysis of retinal light transmission. Exp Eye Res 173:91–108. https://doi.org/10.1016/j.exer.2018.05.009
Baden T, Osorio D (2019) The retinal basis of vertebrate color vision. Annu Rev Vis Sci 5:177–200. https://doi.org/10.1146/annurev-vision-091718-014926
Barnstable CJ (1980) Monoclonal antibodies which recognize different cell types in the rat retina. Nature 286:231–235. https://doi.org/10.1038/286231a0
Baumhardt PE, Moore BA, Doppler M, Fernández-Juricic E (2014) Do American goldfinches see their world like passive prey foragers? A study on visual fields, retinal topography, and sensitivity of photoreceptors. Brain Behav Evol 83:181–198. https://doi.org/10.1159/000357750
Bhattacharyya N, Darren B, Schott RK et al (2017) Cone-like rhodopsin expressed in the all-cone retina of the colubrid pine snake as a potential adaptation to diurnality. J Exp Biol 220:2418–2425. https://doi.org/10.1242/jeb.156430
Bloch S, Martinoya C (1982) Comparing frontal and lateral viewing in the pigeon. I. Tachistoscopic visual acuity as a function of distance. Behav Brain Res 5(3):231–244
Brach V (1977) The functional significance of the avian pecten: a review. Condor 79:321–327. https://doi.org/10.2307/1368009
Bringmann A (2019) Structure and function of the bird fovea. Anat Histol Embryol. https://doi.org/10.1111/ahe.12432
Bringmann A, Syrbe S, Görner K et al (2018) The primate fovea: structure, function and development. Prog Retin Eye Res. https://doi.org/10.1016/j.preteyeres.2018.03.006
Bruhn SL, Cepko CL (1996) Development of the pattern of photoreceptors in the chick retina. J Neurosci 16:1430–1439. https://doi.org/10.1523/jneurosci.16-04-01430.1996
Coimbra JP, Videira Marceliano ML, Da Silveira Andrade-Da-Costa BL, Yamada ES (2006) The retina of tyrant flycatchers: topographic organization of neuronal density and size in the ganglion cell layer of the great kiskadee Pitangus sulphuratus and the rusty margined flycatcher Myiozetetes cayanensis (Aves: Tyrannidae). Brain Behav Evol 68:15–25. https://doi.org/10.1159/000092310
Coimbra JP, Trévia N, Marceliano MLV et al (2009) Number and distribution of neurons in the retinal ganglion cell layer in relation to foraging behaviors of tyrant flycatchers. J Comp Neurol 514:66–73. https://doi.org/10.1002/cne.21992
Coimbra JP, Collin SP, Hart NS (2014a) Topographic specializations in the retinal ganglion cell layer of Australian passerines. J Comp Neurol 522:3609–3628. https://doi.org/10.1002/cne.23624
Coimbra JP, Collin SP, Hart NS (2014b) Topographic specializations in the retinal ganglion cell layer correlate with lateralized visual behavior, ecology, and evolution in cockatoos. J Comp Neurol 522:3363–3385. https://doi.org/10.1002/cne.23637
Coimbra JP, Collin SP, Hart NS (2015) Variations in retinal photoreceptor topography and the organization of the rod-free zone reflect behavioral diversity in Australian passerines. J Comp Neurol 523:1073–1094. https://doi.org/10.1002/cne.23718
Collin SP (1999a) The foveal photoreceptor mosaic in the pipefish, Corythoichthyes paxtoni (Syngnathidae, Teleostei). Histol Histopathol 14:369–382
Collin SP (1999b) Behavioural ecology and retinal cell topography. In: Archer S, Djamgoz MB, Loew E, Partridge JC, Vallerga S (eds) Adaptive mechanisms in the ecology of vision. Kluwer Academic, Dordrecht, pp 509–535
Collin SP, Partridge JC (1996) Retinal specializations in the eyes of deep-sea teleosts. J Fish Biol 49:157–174. https://doi.org/10.1006/jfbi.1996.0143
Collin SP, Shand J (2003) Retinal sampling and the visual field in fishes. In: Collin SP, Marshall NJ (eds) Sensory processing in aquatic environments. Springer, New York, pp 139–169
Collin SP, Hoskins RV, Partridge JC (1998) Seven retinal specializations in the tubular eye of the deep-sea pearleye, Scopelarchus michaelsarsi: a case study in visual optimization. Brain Behav Evol 51:291–314. https://doi.org/10.1159/000006544
Dunlop SA, Beazley LD (1981) Changing retinal ganglion cell distribution in the frog Heleioporus eyrei. J Comp Neurol 202:221–236. https://doi.org/10.1002/cne.902020208
Dunlop SA, Beazley LD (1984) A morphometric study of the retinal ganglion cell layer and optic nerve from metamorphosis in Xenopus laevis. Vis Res 24:417–427. https://doi.org/10.1016/0042-6989(84)90040-3
Fernández-Juricic E, Erichsen JT, Kacelnik A (2004) Visual perception and social foraging in birds. Trends Ecol Evol 19:25–31
Fernández-Juricic E, Moore BA, Doppler M et al (2011) Testing the terrain hypothesis: Canada geese see their world laterally and obliquely. Brain Behav Evol 77:147–158. https://doi.org/10.1159/000326053
Fischer AJ, Stanke JJ, Aloisio G et al (2007) Heterogeneity of horizontal cells in the chicken retina. J Comp Neurol 500:1154–1171. https://doi.org/10.1002/cne.21236
Fite KV, Rosenfield-Wessels S (1975) A comparative study of deep avian fovea. Brain Behav Evol 12:97–115
Franze K, Grosche J, Skatchkov SN et al (2007) Müller cells are living optical fibers in the vertebrate retina. Proc Natl Acad Sci USA 104:8287–8292. https://doi.org/10.1073/pnas.0611180104
González-Martín-Moro J, Hernández-Verdejo JL, Clement-Corral A (2017) El sistema visual de las rapaces diurnas: revisión actualizada. Arch Soc Esp Oftalmol 92:225–232. https://doi.org/10.1016/j.oftal.2016.11.019
Hart NS (2001) The visual ecology of avian photoreceptors. Prog Retin Eye Res 20:675–703
Hayes B, Martin GR, de L. Brooke M (1991) Novel area serving binocular vision in the retinae of procellariiform seabirds. Brain Behav Evol 37:79–84. https://doi.org/10.1159/000114348
Hughes A (1977) The topography of vision in mammals of contrasting life style: comparative optics and retinal organization. In: Crescitelli F (ed) Handbook of sensory physiology, vol 5. Springer, Berlin, pp 613–756
Inoue Y, Sugihara Y, Nishimura Y, Shimai K (1980) Atypical neural sheaths formed by Müller cells in chicken retina. Okajimas Folia Anat Jpn 57:79–87
Inzunza O, Bravo H, Smith RL (1989) Foveal regions of bird retinas correlate with the aster of the inner nuclear layer. Anat Rec 223:342–346. https://doi.org/10.1002/ar.1092230313
Inzunza O, Bravo H, Smith RL, Angel M (1991) Topography and morphology of retinal ganglion cells in Falconiforms: a study on predatory and carrion-eating birds. Anat Rec 229:271–277. https://doi.org/10.1002/ar.1092290214
Jones MP, Pierce KE, Ward D (2007) Avian vision: a review of form and function with special consideration to birds of prey. J Exot Pet Med 16:69–87. https://doi.org/10.1053/j.jepm.2007.03.012
Kajikawa J (1923) Beiträge zur Anatomie und Physiologie des Vogelauges. Graefes Arch Ophtalmologiy 112:260–346
Karl A, Agte S, Zayas-Santiago A et al (2018) Retinal adaptation to dim light vision in spectacled caimans (Caiman crocodilus fuscus): analysis of retinal ultrastructure. Exp Eye Res 173:160–178. https://doi.org/10.1016/j.exer.2018.05.006
Kentroti S, Baker R, Lee K et al (1991) Platelet-activating factor increases glutamine synthetase activity in early and late passage C-6 glioma cells. J Neurosci Res 28:497–506. https://doi.org/10.1002/jnr.490280406
Khokhlova TV, Zueva LV, Golubeva TB (2000) Postnatal developmental stages in the retinal photoreceptor cells of Ficedula hypoleuca. Zhurnal Evolyutsionnoi Biokhimii i Fiziol 36:354–361
Krause W (1894) Die Retina. Anat Physiol 11:1–66
Kreithen ML, Keeton WT (1974) Detection of polarized light by the homing pigeon. Columba livia. J Comp Physiol 89(1):83–92
Lewis GP, Kaska DD, Vaughan DK, Fisher SK (1988) An immunocytochemical study of cat retinal Müller cells in culture. Exp Eye Res 47:855–868. https://doi.org/10.1016/0014-4835(88)90068-1
Lisney TJ, Stecyk K, Kolominsky J et al (2013) Ecomorphology of eye shape and retinal topography in waterfowl (Aves: Anseriformes: Anatidae) with different foraging modes. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 199:385–402. https://doi.org/10.1007/s00359-013-0802-1
Lisney TJ, Wylie DR, Kolominsky J, Iwaniuk AN (2015) Eye morphology and retinal topography in hummingbirds (Trochilidae: Aves). Brain Behav Evol 86:176–190. https://doi.org/10.1159/000441834
Locket NA (1992) Problems of deep foveas. Aust N Z J Ophthalmol 20:281–295
MacDonald RB, Randlett O, Oswald J et al (2015) Müller glia provide essential tensile strength to the developing retina. J Cell Biol 210:1075–1083. https://doi.org/10.1083/jcb.201503115
Machovsky Capuska GE, Huynen L, Lambert D, Raubenheimer D (2011) UVS is rare in seabirds. Vis Res 51:1333–1337. https://doi.org/10.1016/j.visres.2011.04.008
Makaretz M, Levine RL (1980) A light microscopic study of the bifoveate retina in the lizard Anolis carolinensis: general observations and convergence ratios. Vis Res 20:679–686. https://doi.org/10.1016/0042-6989(80)90092-9
Martin GR (2007) Visual fields and their functions in birds. J Ornithol. https://doi.org/10.1007/s10336-007-0213-6
Martin PR, Grünert U (1999) Analysis of the short wavelength-sensitive (“blue”) cone mosaic in the primate retina: Comparison of New World and Old World monkeys. J Comp Neurol 406(1):1–14
Martinez-Hernandez A, Bell KP, Norenberg MD (1977) Glutamine synthetase: glial localization in brain. Science (80-) 195:1356–1358. https://doi.org/10.1126/science.14400
Meyer DB (1977) The avian eye and its adaptations. In: Crescitelli F (ed) Handbook of sensory physiology, vol 5. Springer, New York, pp 550–611
Mitkus M, Nevitt GA, Danielsen J, Kelber A (2016) Vision on the high seas: spatial resolution and optical sensitivity in two procellariiform seabirds with different foraging strategies. J Exp Biol 219:3329–3338. https://doi.org/10.1242/jeb.140905
Mitkus M, Olsson P, Toomey MB et al (2017) Specialized photoreceptor composition in the raptor fovea. J Comp Neurol 525:2152–2163. https://doi.org/10.1002/cne.24190
Mitkus M, Potier S, Martin GR et al (2018) Raptor vision, vol 1. In: Oxford Research Encyclopedia of Neuroscience. https://doi.org/10.1093/acrefore/9780190264086.013.232
Moore BA, Kamilar JM, Collin SP et al (2012) A novel method for comparative analysis of retinal specialization traits from topographic maps. J Vis 12:1–24. https://doi.org/10.1167/12.12.13
Moore BA, Pita D, Tyrrell LP, Fernandez-Juricic E (2015) Vision in avian emberizid foragers: maximizing both binocular vision and fronto-lateral visual acuity. J Exp Biol 218:1347–1358. https://doi.org/10.1242/jeb.108613
Moore BA, Yoo I, Tyrrell LP et al (2016) FOVEA: a new program to standardize the measurement of foveal pit morphology. PeerJ 4:e1785. https://doi.org/10.7717/peerj.1785
Moore BA, Tyrrell LP, Kamilar JM et al (2017a) Structure and function of regional specializations in the vertebrate retina. In: Kaas JH, Striedter G (eds) Evolution of nervous systems: second edition, 2nd edn. Elsevier, Amsterdam, pp 149–172
Moore BA, Tyrrell LP, Pita D et al (2017b) Does retinal configuration make the head and eyes of foveate birds move? Sci Rep 7(1):1–7. https://doi.org/10.1038/srep38406
Moroney MK, Pettigrew JD (1987) Some observations on the visual optics of kingfishers (Aves, Coraciformes, Alcedinidae). J Comp Physiol A 160:137–149. https://doi.org/10.1007/BF00609721
Muguruma K, Takei S, Yamamoto N (2013) Retinal ganglion cell distribution and spatial resolving power in the Japanese catshark Scyliorhinus torazame. Zool Sci 30:42–52. https://doi.org/10.2108/zsj.30.42
Newman E, Reichenbach A (1996) The Muller cell: a functional element of the retina. Trends Neurosci 19:307–312. https://doi.org/10.1016/0166-2236(96)10040-0
Nießner C, Denzau S, Gross JC et al (2011) Avian Ultraviolet/violet cones identified as probable magnetoreceptors. PLoS ONE 6:1–8. https://doi.org/10.1371/journal.pone.0020091
Norenberg MD, Martinez-Hernandez A (1979) Fine structural localization of glutamine synthetase in astrocytes of rat brain. Brain Res 161:303–310. https://doi.org/10.1016/0006-8993(79)90071-4
O’Brien KMB (2008) Development of the foveal specialization. In: Tombran-Tink J, Barnstable CJ (eds) Visual transduction and non-visual light perception. Humana Press, Totowa, pp 17–33
O’Day K (1940) The fundus and fovea centralis of the albatross (Diomedea cauta cauta gould). Br J Ophthalmol 24:201–207. https://doi.org/10.1136/bjo.24.5.201
Ödeen A, Håstad O (2013) The phylogenetic distribution of ultraviolet sensitivity in birds. BMC Evol Biol. https://doi.org/10.1186/1471-2148-13-36
Ödeen A, Håstad O, Alström P (2010) Evolution of ultraviolet vision in shorebirds (Charadriiformes). Biol Lett 6:370–374. https://doi.org/10.1098/rsbl.2009.0877
Omri S, Omri B, Savoldelli M et al (2010) The outer limiting membrane (OLM) revisited: clinical implications. Clin Ophthalmol 4:183–195. https://doi.org/10.2147/opth.s5901
Potier S, Bonadonna F, Kelber A et al (2016) Visual abilities in two raptors with different ecology. J Exp Biol 219:2639–2649. https://doi.org/10.1242/jeb.142083
Potier S, Mitkus M, Bonadonna F et al (2017) Eye size, fovea, and foraging ecology in accipitriform raptors. Brain Behav Evol 90:232–242. https://doi.org/10.1159/000479783
Potier S, Mitkus M, Lisney TJ et al (2020) Inter-individual differences in foveal shape in a scavenging raptor, the black kite Milvus migrans. Sci Rep 10:1–12. https://doi.org/10.1038/s41598-020-63039-y
Provis JM, Penfold PL, Cornish EE et al (2005) Anatomy and development of the macula: specialisation and the vulnerability to macular degeneration. Clin Exp Optom 88:269–281
Provis JM, Dubis AM, Maddess T, Carroll J (2013) Adaptation of the central retina for high acuity vision: cones, the fovea and the a vascular zone. Prog Retin Eye Res 35:63–81
Pumphrey R (1948a) The sense organs of birds. 90(2):171–199
Pumphrey RJ (1948b) The theory of the fovea. J Exp Biol 25:299–312
Querubin A, Hie RL, Provis JM, Bumsted O’Brien KM (2009) Photoreceptor and ganglion cell topographies correlate with information convergence and high acuity regions in the adult pigeon (Columba livia) retina. J Comp Neurol 517:711–722. https://doi.org/10.1002/cne.22178
Rahman ML, Sugita S, Aoyama M, Sugita S (2006) Number, distribution and size of retinal ganglion cells in the jungle crow (Corvus macrorhynchos). Anat Sci Int 81:253–259. https://doi.org/10.1111/j.1447-073X.2006.00151.x
Reichenbach A (1989) Attempt to classify glial cells by means of their process specialization using the rabbit retinal Müller cell as an example of cytotopographic specialization of glial cells. Glia 2:250–259
Reichenbach A, Bringmann A (2010) Müller cells in the healthy retina. In: Müller Cells in the Healthy and Diseased Retina. Springer, New York, pp 35–214. https://doi.org/10.1007/978-1-4419-1672-3
Reichenbach A, Bringmann A (2013) New functions of Müller cells. Glia 61:651–678. https://doi.org/10.1002/glia.22477
Reuter T, Peichl L (2008) Structure and function of the retina in Aquatic Tetrapods University. In: Thewissen JGM, Nummela S (eds) Sensory evolution on the threshold: adaptations in secondarily aquatic vertebrates. University of California Press, Berkeley, pp 1–41
Reymond L (1985) Spatial visual acuity of the eagle Aquila audax: a behavioural, optical and anatomical investigation. Vis Res 25:1477–1491. https://doi.org/10.1007/BF01588452
Reymond L (1987) Spatial visual acuity of the falcon, Falco berigora: a behavioural, optical and anatomical investigation. Vis Res 27:1859–1874. https://doi.org/10.1016/0042-6989(87)90114-3
Riepe RE, Norenberg MD (1978) Glutamine synthetase in the developing rat retina: an immunohistochemical study. Exp Eye Res 27:435–444. https://doi.org/10.1016/0014-4835(78)90022-2
Ruggeri M, Major JC, McKeown C et al (2010) Retinal structure of birds of prey revealed by ultra-high resolution spectral-domain optical coherence tomography. Investig Ophthalmol Vis Sci 51:5789–5795. https://doi.org/10.1167/iovs.10-5633
Schwassmann HO (1968) Visual projection upon the optic tectum in foveate marine teleosts. Vis Res 8:1337–1348. https://doi.org/10.1016/0042-6989(68)90054-0
Schweikert LE, Grace MS (2018) Altered environmental light drives retinal change in the Atlantic Tarpon (Megalops atlanticus) over timescales relevant to marine environmental disturbance. BMC Ecol 18:1. https://doi.org/10.1186/s12898-018-0157-0
Segovia Y, García M, Gómez-Torres MJ Mengual R (2016) Ultrastructural study of retinal development in the turtle Trachemys scripta elegans. Zoomorphology 135(2):205–216
Segovia Y, Victory N, Navarro-Sempere A et al (2019) A comparative ultrastructural study of the pecten oculi in adult, juvenile, and nestling yellow-legged gulls, Larus michahellis (Naumann, 1840). Vet Ophthalmol. https://doi.org/10.1111/vop.12695
Silver R, Witkovsky P, Horvath P et al (1988) Coexpression of opsin- and VIP-like-immunoreactivity in CSF-contacting neurons of the avian brain. Cell Tissue Res 253:189–198. https://doi.org/10.1007/bf00221754
Slonaker JR (1897) A comparative study of the area of acute vision in vertebrates. J Morphol 13:445–502
Snyder A, Miller W (1978) Telephoto lens system of falconiform eyes. Nature 275:127–129
Syrbe S, Kuhrt H, Gärtner U et al (2018) Müller glial cells of the primate foveola: an electron microscopical study. Exp Eye Res 167:110–117. https://doi.org/10.1016/j.exer.2017.12.004
Tucker VA (2000) The deep fovea, sideways vision and spiral flight paths in raptors. J Exp Biol 203:3745–3754. https://doi.org/10.1016/S0169-5347(01)02393-X
Tyrrell LP, Fernández-Juricic E (2015) Sensory systems and escape behavior. In: Cooper WE Jr, Blumstein DT (eds) Escaping from predators: an integrative view of escape decisions. Cambridge University Press, Cambridge, pp 322–342
Tyrrell LP, Moore BA, Loftis C, Fernandez-Juricic E (2013) Looking above the prairie: localized and upward acute vision in a native grassland bird. Sci Rep 3:1–6. https://doi.org/10.1038/srep03231
Varela FJ, Palacios AG, Goldsmith TH (1993) Color vision of birds. In: Zeigler HP, Bischoff HJ (eds) Vision, brain, and behavior in birds. MIT Press, Cambridge, pp 77–98
Velasco Gallego ML (2015) Imaging of physiological retinal structures in various raptor species using Optical Coherence Tomography (OCT) (Doctoral dissertation, lmu)
Vidal MN, Segovia Y, Victory N et al (2018) Light microscopy study of the retina of the Yellow-legged Gull, Larus michahellis, and the relationship between environment and behaviour. Avian Biol Res 11(4):231–237
Vlahos LM, Knott B, Valter K, Hemmi JM (2014) Photoreceptor topography and spectral sensitivity in the common brushtail possum (Trichosurus vulpecula). J Comp Neurol 522:3423–3436. https://doi.org/10.1002/cne.23610
Walls GL (1937) Significance of the foveal depression. Arch Ophtalmol 18:912–919
Walls GL (1942) The vertebrate eye and its adaptive radiation. Cranbrook Institute of Science, Bloomfield Hills
Wikler KC, Rakic P (1990) Distribution of photoreceptor subtypes in the retina of diurnal and nocturnal primates. J Neurosci 10(10):3390–3401
Wood CA (1917) The fundus oculi of birds, especially as viewed by the ophthalmoscope a study in comparative anatomy and physiology. Lakeside Press, Chicago
Zueva LV, Golubeva TB, Kerov VS, Zuev AV (2003) Heterochronic retina development in pied flycatcher Ficedula hypoleuca. Zh Evol Biokhim Fiziol 39:586–591
Zueva L, Makarov V, Zayas-Santiago A et al (2014) Müller cell alignment in bird fovea: possible role in vision. J Neurosci Neuroeng 3:85–91. https://doi.org/10.1166/jnsne.2014.1104
Acknowledgements
This research was supported by the University of Alicante VIGROB-186. We thank Drs. P. María-Mojica and A. Izquierdo of the Santa Faz Wildlife Recovery Centre for helping to obtain the samples used in this study and Vanessa Pinilla for technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Collection of specimens was conducted in accordance with national guidelines and permits.
Informed consent
This article does not contain any studies with human participants performed by any of the authors.
Consent for publication
All authors agreed with the content, and to submit this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Victory, N., Segovia, Y. & García, M. Foveal shape, ultrastructure and photoreceptor composition in yellow-legged gull, Larus michahellis (Naumann, 1840). Zoomorphology 140, 151–167 (2021). https://doi.org/10.1007/s00435-020-00512-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00435-020-00512-2