Abstract
Purpose
Gastroenteropancreatic Neuroendocrine Carcinoma (GEP-NEC) in children is an exceptionally rare and aggressive form of cancer. We aimed to conduct a population-based cohort study to predict overall survival (OS) in pediatric patients with GEP-NEC.
Methods
The Surveillance, Epidemiology, and End Results (SEER) database was employed to identify all pediatric patients with GEP-NEC diagnosed between 2000 and 2019. To create survival curves based on various criteria, Kaplane-Meier estimations were utilized. The log-rank test was used to compare survival curves. The variables associated with OS were determined using Cox proportional-hazards regression. Furthermore, we developed a nomogram to predict overall survival in pediatric GEP-NEC patients.
Results
A total of 103 pediatric GEP-NEC patients were identified. The tumors primarily affected females (62.2%). The majority of GEP-NEC was found in the appendix (63.1%), followed by the pancreas (23.3%) and the intestinal tract (13.6%). The highest rates of localized stage (76.9%) and surgery (98.5%) were found in the NEC of appendix origin. However, pancreatic origins had the largest proportion of distant disease (66.7%) but the lowest percentage of surgery (37.5%). Overall 1-year, 3-year, and 5-year survival rates for all patients were 94.4%, 85.4%, and 85.4%, respectively. Tumors of pancreatic origin had the worst survival compared with those of the appendix and intestinal tract. The Cox proportional hazard regression revealed that only site was an important independent predictor of survival.
Conclusions
Our study revealed that only the primary site was found to be the most important predictor of the OS in pediatric GEP-NEC. It’s important to work closely with a multidisciplinary team, including oncologists, surgeons, and other specialists, to determine the most appropriate treatment plan for pediatric GEP-NEC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Epithelial neoplasms with a predominance of neuroendocrine differentiation are known as neuroendocrine neoplasms (NENs) (Rindi et al. 2022). The prevalence of NENs is roughly 6 cases per 100,000 in adults and 2.8 cases per million in children, respectively (Dasari et al. 2017; Navalkele et al. 2011). NENs derived from embryological gut neuroendocrine cells are known as gastroenteropancreatic neuroendocrine neoplasms (GEP-NENs) (Fang et al. 2022). They can occur in both adults and children, although they are more commonly diagnosed in adults. GEP-NENs are extremely rare in children, with the maximum occurrence occurring after the fifth decade of life (Delle Fave et al. 2012). Gastroenteropancreatic Neuroendocrine Carcinoma (GEP-NEC) is a poorly differentiated neuroendocrine neoplasms (Mollazadegan et al. 2021). GEP-NEC in children is an exceptionally rare and aggressive form of cancer, that is characterized by early metastasis and a tendency to be diagnosed at an advanced stage (Khanna et al. 2008).
Little is known regarding the survival rates of pediatric GEP-NEC due to its lower overall prevalence (Gaiani et al. 2019; Fukuda et al. 2023). Therefore, it is crucial to explore the factors that affect pediatric GEP-NEC survival rates separately. In the current study, we examined the largest population size obtained from the SEER database from 2000 to 2019 and hypothesized that our findings may present an objective viewpoint on the characteristics and prognosis of pediatric GEP-NEC.
Methods
Study population
From 2000 to 2019, the SEER database was utilized to identify all malignant instances of pediatric GEP-NEC. Patients with a GEP-NEC diagnosis before their 20th year of age (0–19 years) were chosen. We used the Third Edition (ICD-O-3) morphology code for GEP-NEC (8246/3: Neuroendocrine carcinoma). The study sample contained no duplicate cases. Because the patients’ private information could not be identified, informed consent or an ethical review were not required.
The database was used to extract patient demographics and clinical features. Based on the SEER staging criteria, tumors were classified as localized, regional, or distant. Localized disease was limited to the site of origin, regional stage included nearby structures and/or regional lymph nodes, while distant disease was tumor metastases. The race was divided into white and others because other races make up a relatively minor percentage. The age ranges at diagnosis were split into two groups: 0–14 years old and 15–19 years old. Primary sites include the appendix, intestinal tract, and pancreas. Treatment options include no treatment, surgery alone, and chemotherapy alone. Overall survival (OS) was the major outcome measure. The survival period was calculated from the date of diagnosis to the most recent follow-up date, or until death.
Statistical analysis
SPSS software (version 22.0, SPSS Inc., Chicago, IL, USA) was used for all statistical analyses. For each variable, descriptive statistics were calculated. The chi-square test was used to determine correlations between category variables. The Student t test was used to compare data with a normal distribution, while the Mann–Whitney U test was used to examine nonparametric data. To find predictors of survival, a forward, stepwise logistic regression was applied. The log-rank test was employed to determine differences in univariate analysis. Multivariate Cox proportional hazards regression models with hazard ratios (HR) were used to examine independent prediction performance. To predict OS, we constructed a nomogram based on the multivariate Cox regression model. A two-tailed P-value of 0.05 was used to evaluate statistical significance.
Results
Patient characteristics
There were 103 eligible cases of GEP-NEC in children or adolescents who had received histological confirmation. Table 1 provides a summary of the clinical factors and patient demographics. Of the 103 patients, 34 (33%) and 69 (67%) were 14 years or younger and older than 14 years of age, respectively. The median follow-up was 51 months. The tumors primarily affected females (62.2%). Most of the patients in our study had a localized stage (59.2%), and 77.7% of them underwent surgery.
A list of the 103 patients with GEP-NEC from various origins can be found in Table 2. The highest rates of localized stage (76.9%) and surgery (98.5%) were found in the NEC of appendix origin. Additionally, the prevalence of older children (> 14 years old) (92.9%), lymph nodes removed (92.9%), and no treatment (14.3%) were highest in NEC with intestinal tract origins. In contrast to appendix and intestinal tract origins, pancreatic origins had the largest proportion of distant disease (66.7%) but the lowest percentage of the localized stage (25%), and surgery (37.5%).
Survival and prognosis analysis
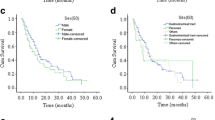
All patients had overall 1-year, 3-year, and 5-year survival rates of 94.4%, 85.4%, and 85.4%, respectively (Table 3) (Fig. 1A). There were no significant differences in OS based on gender, race, or age at diagnosis (P = 0.465, P = 0.772, and P = 0.664, respectively) (Table 4). Also, there was no statistically significant change in OS for any patient who had lymph nodes removed (P = 0.947). Children with NEC of the appendix and intestinal tract had similar and noticeably higher overall survival rates than patients with NEC of the pancreas origin (P = 0.001) (Fig. 1B). In comparison to individuals with distant disease, patients with localized stage had a much greater survival rate (3-year OS, 97.7% versus 52.7%, P < 0.001) (Fig. 1C). In terms of treatment, children who did not undergo chemotherapy had a greater chance of surviving (P < 0.001). When compared to no surgery, surgery had better survival outcomes (P < 0.001) (Fig. 1D).
Table 4 shows the results of a Cox proportional hazards model analysis involving 103 NEC patients. Primary site, SEER stage, and treatment received were included in the multivariate analysis to investigate their impact on the risk of mortality. According to the results of Cox proportional hazard regression, only the primary site was an important independent predictor of OS in this model. Patients with pancreas-originating NEC showed a greater risk of mortality (hazard ratio (HR) 12.5, 95% confidence interval (CI) 1.2–127; P = 0.033). A nomogram was developed to predict OS in pediatric GEP-NEC patients. The model takes into account age, race, gender, primary sites, SEER stage, surgery, lymph node removal, and chemotherapy. The probability of death increased in patients with pancreatic origins, as seen in Fig. 2. In addition, the higher the SEER, the greater the chance of death. Following that, we did ROC analysis on the OS nomogram to validate its sensitivity and specificity, as shown in Fig. 3. The 1-, 3-, and 5-year AUCs are 0.914, 0.879, and 0.869, respectively, indicating that the model has good sensitivity and specificity.
Discussion
In children, GEP-NENs are extremely rare and present unique challenges in terms of diagnosis and management (Johnson 2014). GEP-NENs are classified as grade 1 well-differentiated neuroendocrine tumors (WD NETs), grade 2 WD NETs, and poorly differentiated neuroendocrine carcinomas (PD NECs), according to the 2019 WHO grading and categorization (Ahadi et al. 2021). GEP-NEC is an extremely aggressive malignancy with a poor prognosis and rapid disease progression (Rosa and Sessa 2014). We found significant differences in epidemiological factors and investigated the impact of each factor on the patient’s overall survival rate. In this study, only the primary site was a statistically significant factor associated with overall survival, both in the univariable and multivariable analyses.
We used one of the biggest datasets to investigate the role of surgical resection and chemotherapy treatment in pediatric GEP-NEC. Our findings confirmed what had previously been reported, namely that the bulk of them were discovered in the appendix (Boxberger et al. 2013). Despite the fact that girls are more prone than boys to develop GEP-NEC, our study observed no difference in survival rates between the sexes. Except for appendix NEC, which appears in 98.5% of cases as loco-regional stage, the majority of these tumors develop at the distant disease. Our research found that tumor stage is a major predictive predictor. Localized tumors had a 5-year survival rate of 97.7%, whereas distant tumors had a 5-year survival rate of 52.7%. The location of the tumor was found to be a significant predictor of survival in this study, with pancreatic origins of NEC having a much lower 5-year survival rate of 56% compared to appendix and intestinal tract origins, which is consistent with previously published reports on GEP-NEC (Xu et al. 2021).
Surgery and chemotherapy are two important treatment modalities used in the management of GEP-NEC. It's important to note that the specific treatment plan varies based on factors such as the tumor's location, stage, and the patient's overall health. Surgery plays a critical role in the treatment of GEP-NEC when the tumor is localized and resectable (Abdalla et al. 2023). The goal of surgery is to remove the primary tumor along with any affected nearby lymph nodes or tissues. In some cases, depending on the tumor's location, surgery might involve removing part of the affected organ (partial resection) or, in more extensive cases, removing the entire organ (total resection). For example, for pancreatic GEP-NEC, a Whipple procedure might be performed to remove the head of the pancreas, part of the small intestine, and other adjacent structures (Scott and Howe 2019). Surgical resection can help alleviate symptoms and potentially slow down the progression of the disease. However, high-risk characteristics, including a sizable tumor and severe illness, are contraindications to surgery. Systemic therapy for palliative relief should be investigated for those patients instead. It should be noted that, because of selection bias, surgery was primarily undertaken in cases of early stage, even though our study showed that surgery can lead to a longer survival time than no surgery. As first-line therapy for advanced NEC, platinum-based chemotherapy with cisplatin/etoposide or carboplatin/etoposide is suggested (Pavel et al. 2020; Garcia-Carbonero et al. 2016). The idea of using platinum-based chemotherapy to treat NEC came from the experience of treating small-cell lung cancer, which has many biological similarities with NEC (Evans et al. 1985). Chemotherapy, according to our findings, was associated with significantly poorer overall survival. This could be related to the increased use of chemotherapy in palliative care, as well as the inability of patients with advanced GEP-NEC to undergo surgery.
There are some potential limitations to the current study that should be considered. To begin, much detailed information, such as genomic data and Ki-67 levels, was not provided. This is crucial since it is well established that TP53, KRAS, PIK3CA/PTEN, and BRAF mutations play a substantial role in the development and progression of malignancies (Mafficini and Scarpa 2019; McNamara et al. 2020). In addition, patients with Ki-67 < 55% were less susceptible to platinum-based chemotherapy but lived longer (Sorbye et al. 2013). Second, while information on the recipient of surgical resection was available, specifics on the degree of surgical resection were not. Third, no validation cohort was provided due to the small number of cases, which may have affected the prediction model. Finally, no record of metastatic sites or surgical approaches that would affect survival was provided. Therefore, additional research is required to verify the results.
In conclusion, we assessed the clinical characteristics and prognosis of pediatric GEP-NEC using a population-based dataset. The most important predictor of survival was revealed to be the primary site. A multidisciplinary team of medical experts, including oncologists, surgeons, and other specialists, is essential to creating a customized treatment plan due to the rarity and complexity of GEP-NEC in children.
Availability of data and materials
The dataset used and analyzed during the current study are available from the corresponding author upon reasonable request.
References
Abdalla TSA, Klinkhammer-Schalke M, Zeissig SR et al (2023) Prognostic factors after resection of locally advanced non-functional pancreatic neuroendocrine neoplasm: an analysis from the German Cancer Registry Group of the Society of German Tumor Centers. J Cancer Res Clin Oncol 149(11):8535–8543
Ahadi M, Sokolova A, Brown I et al (2021) The 2019 World health organization classification of appendiceal, colorectal and anal canal tumours: an update and critical assessment. Pathology 53(4):454–461
Boxberger N, Redlich A, Böger C et al (2013) Neuroendocrine tumors of the appendix in children and adolescents. Pediatr Blood Cancer 60:65–70
Dasari A, Shen C, Halperin D et al (2017) Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol 3:1335–1342
DelleFave G, Kwekkeboom DJ, Van Cutsem E et al (2012) ENETs consensus guidelines for the management of patients with gastroduodenal neoplasms. Neuroendocrinology 95:74–87
Evans WK, Shepherd FA, Feld R et al (1985) VP-16 and cisplatin as first-line therapy for small-cell lung cancer. J Clin Oncol 3(11):1471–1477
Fang JM, Li J, Shi J (2022) An update on the diagnosis of gastroenteropancreatic neuroendocrine neoplasms. World J Gastroenterol 28(10):1009–1023
Fukuda S, Suzuki M, Minowa K et al (2023) pediatric pancreatic endocrine tumor presenting as acute pancreatitis: a case report. Children (Basel) 10(5):900
Gaiani F, de Angelis N, Minelli R et al (2019) Pediatric gastroenteropancreatic neuroendocrine tumor: a case report and review of the literature. Medicine (Baltimore). 98(37):17154
Garcia-Carbonero R, Sorbye H, Baudin E et al (2016) ENETS consensus guidelines for high-grade gastroenteropancreatic neuroendocrine tumors and neuroendocrine carcinomas. Neuroendocrinology 103(2):186–194
Johnson PR (2014) Gastroenteropancreatic neuroendocrine (carcinoid) tumors in children. Semin Pediatr Surg 23(2):91–95
Khanna G, O’Dorisio SM, Menda Y et al (2008) Gastroenteropancreatic neuroendocrine tumors in children and young adults. Pediatr Radiol. 38(3):251–9 (Quiz 358-9)
La Rosa S, Sessa F (2014) High-grade poorly differentiated neuroendocrine carcinomas of the gastroenteropancreatic system: from morphology to proliferation and back. Endocr Pathol 25(2):193–198
Mafficini A, Scarpa A (2019) Genetics and epigenetics of gastroenteropancreatic neuroendocrine neoplasms. Endocr Rev 40(2):506–536
McNamara MG, Scoazec JY, Walter T (2020) Extrapulmonary poorly differentiated NECs, including molecular and immune aspects. Endocr Relat Cancer 27(7):R219–R238
Mollazadegan K, Welin S, Crona J (2021) Systemic treatment of gastroenteropancreatic neuroendocrine carcinoma. Curr Treat Options Oncol 22(8):68
Navalkele P, O’Dorisio MS, O’Dorisio TM et al (2011) Incidence, survival, and prevalence of neuroendocrine tumors versus neuroblastoma in children and young adults: nine standard SEER registries, 1975–2006. Pediatr Blood Cancer 56:50–57
Pavel M, Öberg K, Falconi M et al (2020) Gastroenteropancreatic neuroendocrine neoplasms: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 31(7):844–860
Rindi G, Mete O, Uccella S et al (2022) Overview of the 2022 WHO classification of neuroendocrine neoplasms. Endocr Pathol 33(1):115–154
Scott AT, Howe JR (2019) Evaluation and management of neuroendocrine tumors of the pancreas. Surg Clin North Am 99(4):793–814
Sorbye H, Welin S, Langer SW et al (2013) Predictive and prognostic factors for treatment and survival in 305 patients with advanced gastrointestinal neuroendocrine carcinoma (WHO G3): the NORDIC NEC study. Ann Oncol 24(1):152–160
Xu Z, Wang L, Dai S et al (2021) Epidemiologic trends of and factors associated with overall survival for patients with gastroenteropancreatic neuroendocrine tumors in the United States. JAMA Netw Open 4(9):e2124750
Funding
None.
Author information
Authors and Affiliations
Contributions
WL: conception and design of study, acquisition of data, revising the manuscript. ZZ: conception and design of study, acquisition of data, and drafting the manuscript. YS: conception and design of study, acquisition of data, analysis and/or interpretation of data, drafting the manuscript. All authors read and approved the final manuscript as submitted.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
None.
Consent to participate
None.
Consent for publication
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, W., Zhu, Z. & Shang, Y. Gastroenteropancreatic neuroendocrine carcinoma in children and adolescents: a population-based study. J Cancer Res Clin Oncol 150, 4 (2024). https://doi.org/10.1007/s00432-023-05568-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00432-023-05568-3