Abstract
Background
Tamoxifen (Tam) is an effective treatment for estrogen receptor (ER) positive breast cancer. However, a significant proportion of patients develop resistance under treatment, presenting a therapeutic challenge. The study aims to determine the role of early growth response protein (EGR) 3 in tamoxifen resistance (TamR) and elucidate its molecular mechanism.
Methods
TamR cell models were established and NGS was used to screening signaling alternation. Western blot and qRT-PCR were used to analysis the expression of ERα, EGR3, MCL1 and factors associated with apoptosis. CCK8, colony formation and apoptosis assay were used to analysis resistance to Tam. Immunofluorescence, chromatin immunoprecipitation, and dual luciferase assays were used to investigate mechanism of regulation.
Results
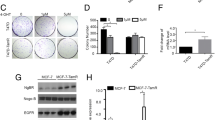
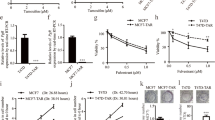
We observed that EGR3, a deeply rooted ERα response factor, showed increased upregulation in response to both estrone (E1) and Tam in TamR cells with elevated level of E1 and ERα expression, indicating a potential connection between EGR3 and TamR. Mechanically, manipulating EGR3 expression revealed that it imparted resistance to Tam through increased expression of the downstream molecule MCL1 (apoptosis suppressor gene) that it regulated. Mechanismly, EGR3 directly binds to the promoter of the anti-apoptotic factor MCL1 gene, facilitating its transcription. Furthermore, apoptosis assays revealed that E1 reduces Tam induced apoptosis by upregulating EGR3 expression. Importantly, clinical public database confirmed the high expression of EGR3 in breast cancer tissue and in Tam-treated patients.
Conclusions
These findings shed light on the novel estrogen/EGR3/MCL1 axis and its role in inducing TamR in ER positive breast cancer. EGR3 emerges as a promising target to overcome TamR. The elucidation of this mechanism holds potential for the development of new therapeutic modalities to overcome endocrine therapy resistance in clinical settings.





Similar content being viewed by others
Data availability
Data will be made available on request.
References
Alcon C, Gomez Tejeda Zanudo J, Albert R et al (2021) ER+ Breast cancer strongly depends on MCL-1 and BCL-xL anti-apoptotic proteins. Cells. https://doi.org/10.3390/cells10071659
Campbell KJ, Mason SM, Winder ML et al (2021) Breast cancer dependence on MCL-1 is due to its canonical anti-apoptotic function. Cell Death Differ 28(9):2589–2600. https://doi.org/10.1038/s41418-021-00773-4
Castro-Mondragon JA, Riudavets-Puig R, Rauluseviciute I et al (2022) JASPAR 2022: the 9th release of the open-access database of transcription factor binding profiles. Nucleic Acids Res 50(D1):D165–D173. https://doi.org/10.1093/nar/gkab1113
Curtis C, Shah SP, Chin SF et al (2012) The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups. Nature 486(7403):346–352. https://doi.org/10.1038/nature10983
Early Breast Cancer Trialists’ Collaborative, G (2015) Aromatase inhibitors versus tamoxifen in early breast cancer: patient-level meta-analysis of the randomised trials. Lancet 386(10001):1341–1352. https://doi.org/10.1016/S0140-6736(15)61074-1
Engeln M, Mitra S, Chandra R et al (2020) Sex-Specific Role for Egr3 in Nucleus Accumbens D2-Medium Spiny Neurons Following Long-Term Abstinence From Cocaine Self-administration. Biol Psychiatry 87(11):992–1000. https://doi.org/10.1016/j.biopsych.2019.10.019
Ghandi M, Huang FW, Jane-Valbuena J et al (2019) Next-generation characterization of the Cancer Cell Line Encyclopedia. Nature 569(7757):503–508. https://doi.org/10.1038/s41586-019-1186-3
Gu W, Dong N, Wang P et al (2017) Tamoxifen resistance and metastasis of human breast cancer cells were mediated by the membrane-associated estrogen receptor ER-alpha36 signaling in vitro. Cell Biol Toxicol 33(2):183–195. https://doi.org/10.1007/s10565-016-9365-6
Hanker AB, Sudhan DR, Arteaga CL (2020) Overcoming endocrine resistance in breast cancer. Cancer Cell 37(4):496–513. https://doi.org/10.1016/j.ccell.2020.03.009
Holohan C, Van Schaeybroeck S, Longley DB et al (2013) Cancer drug resistance: an evolving paradigm. Nat Rev Cancer 13(10):714–726. https://doi.org/10.1038/nrc3599
Hong SE, Kim EK, Jin HO et al (2013) S6K1 inhibition enhances tamoxifen-induced cell death in MCF-7 cells through translational inhibition of Mcl-1 and survivin. Cell Biol Toxicol 29(4):273–282. https://doi.org/10.1007/s10565-013-9253-2
Inoue A, Omoto Y, Yamaguchi Y et al (2004) Transcription factor EGR3 is involved in the estrogen-signaling pathway in breast cancer cells. J Mol Endocrinol 32(3):649–661. https://doi.org/10.1677/jme.0.0320649
Jeffreys SA, Powter B, Balakrishnar B et al (2020) Endocrine resistance in breast cancer: the role of estrogen receptor stability. Cells. https://doi.org/10.3390/cells9092077
Jordan VC (2003) Tamoxifen: a most unlikely pioneering medicine. Nat Rev Drug Discov 2(3):205–213. https://doi.org/10.1038/nrd1031
Kwon Y, Kim M, Kim Y et al (2021) EGR3-HDAC6-IL-27 axis mediates allergic inflammation and is necessary for tumorigenic potential of cancer cells enhanced by allergic inflammation-promoted cellular interactions. Front Immunol 12:680441. https://doi.org/10.3389/fimmu.2021.680441
Li XZ, Tu YJ, Zhou T et al (2021) MicroRNA-483-5p predicts poor prognosis and promotes cancer metastasis by targeting EGR3 in nasopharyngeal carcinoma. Front Oncol 11:720835. https://doi.org/10.3389/fonc.2021.720835
Liang J, Shang Y (2013) Estrogen and cancer. Annu Rev Physiol 75:225–240. https://doi.org/10.1146/annurev-physiol-030212-183708
Liao F, Ji MY, Shen L et al (2013) Decreased EGR3 expression is related to poor prognosis in patients with gastric cancer. J Mol Histol 44(4):463–468. https://doi.org/10.1007/s10735-013-9493-8
Mandlekar S, Kong AN (2001) Mechanisms of tamoxifen-induced apoptosis. Apoptosis 6(6):469–477. https://doi.org/10.1023/a:1012437607881
Merino D, Lok SW, Visvader JE et al (2016) Targeting BCL-2 to enhance vulnerability to therapy in estrogen receptor-positive breast cancer. Oncogene 35(15):1877–1887. https://doi.org/10.1038/onc.2015.287
Morita K, Okamura T, Sumitomo S et al (2016) Emerging roles of Egr2 and Egr3 in the control of systemic autoimmunity. Rheumatology (oxford) 55(suppl 2):ii76–ii81. https://doi.org/10.1093/rheumatology/kew342
Nabieva N, Fasching PA (2021) Endocrine Treatment for breast cancer patients revisited-history, standard of care, and possibilities of improvement. Cancers (basel). https://doi.org/10.3390/cancers13225643
Nie F, Zhang Q, Ma J et al (2021) Schizophrenia risk candidate EGR3 is a novel transcriptional regulator of RELN and regulates neurite outgrowth via the Reelin signal pathway in vitro. J Neurochem 157(6):1745–1758. https://doi.org/10.1111/jnc.15225
Obrero M, Yu DV, Shapiro DJ (2002) Estrogen receptor-dependent and estrogen receptor-independent pathways for tamoxifen and 4-hydroxytamoxifen-induced programmed cell death. J Biol Chem 277(47):45695–45703. https://doi.org/10.1074/jbc.M208092200
Parkinson RM, Collins SL, Horton MR et al (2014) Egr3 induces a Th17 response by promoting the development of gammadelta T cells. PLoS ONE 9(1):e87265. https://doi.org/10.1371/journal.pone.0087265
Patel HK, Bihani T (2018) Selective estrogen receptor modulators (SERMs) and selective estrogen receptor degraders (SERDs) in cancer treatment. Pharmacol Ther 186:1–24. https://doi.org/10.1016/j.pharmthera.2017.12.012
Pio R, Jia Z, Baron VT et al (2013) Early growth response 3 (Egr3) is highly over-expressed in non-relapsing prostate cancer but not in relapsing prostate cancer. PLoS ONE 8(1):e54096. https://doi.org/10.1371/journal.pone.0054096
Qureshi R, Picon-Ruiz M, Aurrekoetxea-Rodriguez I et al (2020) The major pre- and postmenopausal estrogens play opposing roles in obesity-driven mammary inflammation and breast cancer development. Cell Metab 31(6):1154-1172 e1159. https://doi.org/10.1016/j.cmet.2020.05.008
Shahbandi A, Rao SG, Anderson AY et al (2020) BH3 mimetics selectively eliminate chemotherapy-induced senescent cells and improve response in TP53 wild-type breast cancer. Cell Death Differ 27(11):3097–3116. https://doi.org/10.1038/s41418-020-0564-6
Shin SH, Kim I, Lee JE et al (2020) Loss of EGR3 is an independent risk factor for metastatic progression in prostate cancer. Oncogene 39(36):5839–5854. https://doi.org/10.1038/s41388-020-01418-5
Tang J, Luo Y, Long G et al (2021a) MINDY1 promotes breast cancer cell proliferation by stabilizing estrogen receptor alpha. Cell Death Dis 12(10):937. https://doi.org/10.1038/s41419-021-04244-z
Tang J, Wu Z, Tian Z et al (2021b) OTUD7B stabilizes estrogen receptor alpha and promotes breast cancer cell proliferation. Cell Death Dis 12(6):534. https://doi.org/10.1038/s41419-021-03785-7
Tang Z, Li C, Kang B et al (2017) GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res 45(W1):W98–W102. https://doi.org/10.1093/nar/gkx247
Turner NC, Neven P, Loibl S et al (2017) Advances in the treatment of advanced oestrogen-receptor-positive breast cancer. Lancet 389(10087):2403–2414. https://doi.org/10.1016/S0140-6736(16)32419-9
Vareslija D, McBryan J, Fagan A et al (2016) Adaptation to AI therapy in breast cancer can induce dynamic alterations in ER activity resulting in estrogen-independent metastatic tumors. Clin Cancer Res 22(11):2765–2777. https://doi.org/10.1158/1078-0432.CCR-15-1583
Viedma-Rodriguez R, Baiza-Gutman LA, Garcia-Carranca A et al (2013) Suppression of the death gene BIK is a critical factor for resistance to tamoxifen in MCF-7 breast cancer cells. Int J Oncol 43(6):1777–1786. https://doi.org/10.3892/ijo.2013.2127
Vincze B, Kapuvari B, Udvarhelyi N et al (2015) Serum estrone concentration, estrone sulfate/estrone ratio and BMI are associated with human epidermal growth factor receptor 2 and progesterone receptor status in postmenopausal primary breast cancer patients suffering invasive ductal carcinoma. Springerplus 4:387. https://doi.org/10.1186/s40064-015-1171-8
Zhang P, Yang X, Wang L et al (2019) Overexpressing miR335 inhibits DU145 cell proliferation by targeting early growth response 3 in prostate cancer. Int J Oncol 54(6):1981–1994. https://doi.org/10.3892/ijo.2019.4778
Funding
This study was supported by the Natural Science Foundation of China (nos. 31770968 and 31800661), the Science Fund Project of Tianjin (nos. 21JCYBJC00240 and 20JCYBJC01130).
Author information
Authors and Affiliations
Contributions
YX: investigation, data curation, conceptualization, original draft. XH: cell model establishment. JY: experiment assistance. MY & YY & JQ & LL: review and editing. YW: project administration, funding acquisition, review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xie, Y., Han, X., Yu, J. et al. EGR3 and estrone are involved in the tamoxifen resistance and progression of breast cancer. J Cancer Res Clin Oncol 149, 18103–18117 (2023). https://doi.org/10.1007/s00432-023-05503-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05503-6