Abstract
Background
Pancreatic ductal adenocarcinoma (PDAC) is a fatal tumor with grave prognosis. Pyroptosis, a programmed cell death, is involved in tumorigenesis. However, a few studies have elucidated the functions of pyroptosis in PDAC.
Methods
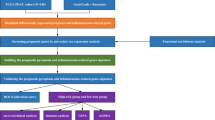
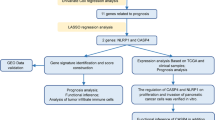
The mRNA expression profiles were downloaded from the TCGA and GEO databases. Univariate and LASSO Cox regression analyses were used to screen out differentially expressed genes (DEGs) and construct the pyroptosis-related genes (PRGs) risk model. The efficiency of model was examined by Kaplan–Meier curve, ROC curve, and nomogram. Univariate and multivariate Cox regression analyses were utilized to assess whether the risk model could be used as an independent prognostic factor. The biological function was analyzed by GO, KEGG, and GSEA enrichment analysis. qRT-PCR and immunohistochemical staining detected gene expression.
Results
Totally 9 PRGs with differential expression were identified between normal and PDAC tissues. Then, according to PRGs, we filtered out three key DEGs and constructed the prognostic risk model. Kaplan–Meier curve, ROC curve, and nomogram indicated that the prognostic risk model had high survival prediction efficiency. Meanwhile, the risk model had also shown to be an independent prognostic factor. Further functional enrichment analysis showed that cell adhesion, PI3K–AKT signaling pathway, and dysregulated immune status may be associated with PDAC development. External validation of the model was carried out in the GEO cohort, and the results were similar to that in the TCGA cohort. Finally, the expression of three genes was verified by qRT-PCR and immunohistochemical staining.
Conclusion
The prognostic risk model established in this study can give a good prediction of the prognosis of PDAC patients, which might provide insights into clinical treatments and prognostic prediction of PDAC.










Similar content being viewed by others
Availability of data and materials
The original data presented in this study are included in the article/Supplementary Material, and further inquiries are available from the corresponding author.
References
Aglietti RA, Estevez A, Gupta A, Ramirez MG, Liu PS, Kayagaki N, Ciferri C, Dixit VM, Dueber EC (2016) GsdmD p30 elicited by caspase-11 during pyroptosis forms pores in membranes. Proc Natl Acad Sci USA 113:7858–7863. https://doi.org/10.1073/pnas.1607769113
Aubert DF, Xu H, Yang J, Shi X, Gao W, Li L, Bisaro F, Chen S, Valvano MA, Shao F (2016) A burkholderia type VI effector deamidates Rho GTPases to activate the pyrin inflammasome and trigger inflammation. Cell Host Microbe 19:664–674. https://doi.org/10.1016/j.chom.2016.04.004
Baker PJ, Boucher D, Bierschenk D, Tebartz C, Whitney PG, D’Silva DB, Tanzer MC, Monteleone M, Robertson AA, Cooper MA, Alvarez-Diaz S, Herold MJ, Bedoui S, Schroder K, Masters SL (2015) NLRP3 inflammasome activation downstream of cytoplasmic LPS recognition by both caspase-4 and caspase-5. Eur J Immunol 45:2918–2926. https://doi.org/10.1002/eji.201545655
Bock FJ, Tait SWG (2020) Mitochondria as multifaceted regulators of cell death. Nat Rev Mol Cell Biol 21:85–100. https://doi.org/10.1038/s41580-019-0173-8
Brokatzky D, Dorflinger B, Haimovici A, Weber A, Kirschnek S, Vier J, Metz A, Henschel J, Steinfeldt T, Gentle IE, Hacker G (2019) A non-death function of the mitochondrial apoptosis apparatus in immunity. EMBO J. https://doi.org/10.15252/embj.2018100907
de Vasconcelos NM, Van Opdenbosch N, Van Gorp H, Parthoens E, Lamkanfi M (2019) Single-cell analysis of pyroptosis dynamics reveals conserved GSDMD-mediated subcellular events that precede plasma membrane rupture. Cell Death Differ 26:146–161. https://doi.org/10.1038/s41418-018-0106-7
Delitto D, Vertes-George E, Hughes SJ, Behrns KE, Trevino JG (2014) c-Met signaling in the development of tumorigenesis and chemoresistance: potential applications in pancreatic cancer. World J Gastroenterol 20:8458–8470. https://doi.org/10.3748/wjg.v20.i26.8458
Egawa S, Toma H, Ohigashi H, Okusaka T, Nakao A, Hatori T, Maguchi H, Yanagisawa A, Tanaka M (2012) Japan Pancreatic Cancer Registry; 30th year anniversary: Japan Pancreas Society. Pancreas 41:985–992. https://doi.org/10.1097/MPA.0b013e318258055c
Faria SS, Costantini S, de Lima VCC, de Andrade VP, Rialland M, Cedric R, Budillon A, Magalhães KG (2021) NLRP3 inflammasome-mediated cytokine production and pyroptosis cell death in breast cancer. J Biomed Sci 28:26. https://doi.org/10.1186/s12929-021-00724-8
Gao J, Qiu X, Xi G, Liu H, Zhang F, Lv T, Song Y (2018) Downregulation of GSDMD attenuates tumor proliferation via the intrinsic mitochondrial apoptotic pathway and inhibition of EGFR/Akt signaling and predicts a good prognosis in non-small cell lung cancer. Oncol Rep 40:1971–1984. https://doi.org/10.3892/or.2018.6634
Heinrich S, Lang H (2017) Neoadjuvant therapy of pancreatic cancer: definitions and benefits. Int J Mol Sci. https://doi.org/10.3390/ijms18081622
Jia C, Zhang Z, Tang J, Cai MC, Zang J, Shi K, Sun Y, Wu J, Shi H, Shi W, Ma P, Zhao X, Yu Z, Fu Y, Zhuang G (2021) Epithelial-mesenchymal transition induces GSDME transcriptional activation for inflammatory pyroptosis. Front Cell Dev Biol 9:781365. https://doi.org/10.3389/fcell.2021.781365
Jin Y, Zhang Z, Zou S, Li F, Chen H, Peng C, Deng X, Wen C, Shen B, Zhan Q (2021) A novel c-MET-targeting antibody-drug conjugate for pancreatic cancer. Front Oncol 11:634881. https://doi.org/10.3389/fonc.2021.634881
Kowalewski B, Lamanna WC, Lawrence R, Damme M, Stroobants S, Padva M, Kalus I, Frese MA, Lübke T, Lüllmann-Rauch R, D’Hooge R, Esko JD, Dierks T (2012) Arylsulfatase G inactivation causes loss of heparan sulfate 3-O-sulfatase activity and mucopolysaccharidosis in mice. Proc Natl Acad Sci U S A 109:10310–10315. https://doi.org/10.1073/pnas.1202071109
Li J, Xia F, Reithmeier RA (2014) N-glycosylation and topology of the human SLC26 family of anion transport membrane proteins. Am J Physiol Cell Physiol 306:C943–C960. https://doi.org/10.1152/ajpcell.00030.2014
Li C, Qiu J, Xue Y (2021a) Low-dose Diosbulbin-B (DB) activates tumor-intrinsic PD-L1/NLRP3 signaling pathway mediated pyroptotic cell death to increase cisplatin-sensitivity in gastric cancer (GC). Cell Biosci 11:38. https://doi.org/10.1186/s13578-021-00548-x
Li E, Huang X, Zhang G, Liang T (2021b) Combinational blockade of MET and PD-L1 improves pancreatic cancer immunotherapeutic efficacy. J Exp Clin Cancer Res 40:279. https://doi.org/10.1186/s13046-021-02055-w
Liang C, Fan J, Liang C, Guo J (2021) Identification and validation of a pyroptosis-related prognostic model for gastric cancer. Front Genet 12:699503. https://doi.org/10.3389/fgene.2021.699503
Liao P, Huang WH, Cao L, Wang T, Chen LM (2022) Low expression of FOXP2 predicts poor survival and targets caspase-1 to inhibit cell pyroptosis in colorectal cancer. J Cancer 13:1181–1192. https://doi.org/10.7150/jca.62433
Luo J, Lai J (2022) Pyroptosis-related molecular classification and immune microenvironment infiltration in breast cancer: a novel therapeutic target. J Cell Mol Med 26:2259–2272. https://doi.org/10.1111/jcmm.17247
Luo Y, Li J, Yu P, Sun J, Hu Y, Meng X, Xiang L (2022) Targeting lncRNAs in programmed cell death as a therapeutic strategy for non-small cell lung cancer. Cell Death Discov 8:159. https://doi.org/10.1038/s41420-022-00982-x
Lux A, Kahlert C, Grützmann R, Pilarsky C (2019) c-Met and PD-L1 on circulating exosomes as diagnostic and prognostic markers for pancreatic cancer. Int J Mol Sci. https://doi.org/10.3390/ijms20133305
Mizrahi JD, Surana R, Valle JW, Shroff RT (2020) Pancreatic cancer. Lancet 395:2008–2020. https://doi.org/10.1016/s0140-6736(20)30974-0
Neoptolemos JP, Palmer DH, Ghaneh P, Psarelli EE, Valle JW, Halloran CM, Faluyi O, O’Reilly DA, Cunningham D, Wadsley J, Darby S, Meyer T, Gillmore R, Anthoney A, Lind P, Glimelius B, Falk S, Izbicki JR, Middleton GW, Cummins S, Ross PJ, Wasan H, McDonald A, Crosby T, Ma YT, Patel K, Sherriff D, Soomal R, Borg D, Sothi S, Hammel P, Hackert T, Jackson R, Büchler MW (2017) Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): a multicentre, open-label, randomised, phase 3 trial. The Lancet 389:1011–1024. https://doi.org/10.1016/s0140-6736(16)32409-6
Siegel RL, Miller KD, Jemal A (2017) Cancer statistics, 2017. CA Cancer J Clin 67:7–30. https://doi.org/10.3322/caac.21387
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70:7–30. https://doi.org/10.3322/caac.21590
Siegel RL, Miller KD, Fuchs HE, Jemal A (2021) Cancer statistics, 2021. CA Cancer J Clin 71:7–33. https://doi.org/10.3322/caac.21654
Tan YF, Wang M, Chen ZY, Wang L, Liu XH (2020) Inhibition of BRD4 prevents proliferation and epithelial-mesenchymal transition in renal cell carcinoma via NLRP3 inflammasome-induced pyroptosis. Cell Death Dis 11:239. https://doi.org/10.1038/s41419-020-2431-2
Tempero MA (2019) NCCN guidelines updates: pancreatic cancer. J Natl Compr Canc Netw 17:603–605. https://doi.org/10.6004/jnccn.2019.5007
Van Opdenbosch N, Gurung P, Vande Walle L, Fossoul A, Kanneganti TD, Lamkanfi M (2014) Activation of the NLRP1b inflammasome independently of ASC-mediated caspase-1 autoproteolysis and speck formation. Nat Commun 5:3209. https://doi.org/10.1038/ncomms4209
Wei W, Zeng H, Zheng R, Zhang S, Lan An Ru, Chen SW, Sun K, Matsuda T, Bray F, He J (2020) Cancer registration in China and its role in cancer prevention and control. Lancet Oncol 21:e342–e349. https://doi.org/10.1016/s1470-2045(20)30073-5
Xu S, Wang J, Jiang J, Song J, Zhu W, Zhang F, Shao M, Xu H, Ma X, Lyu F (2020) TLR4 promotes microglial pyroptosis via lncRNA-F630028O10Rik by activating PI3K/AKT pathway after spinal cord injury. Cell Death Dis 11:693. https://doi.org/10.1038/s41419-020-02824-z
Yan Z, Da Q, Li Z, Lin Q, Yi J, Su Y, Yu G, Ren Q, Liu X, Lin Z, Qu J, Yin W, Liu J (2022) Inhibition of NEK7 suppressed hepatocellular carcinoma progression by mediating cancer cell pyroptosis. Front Oncol 12:812655. https://doi.org/10.3389/fonc.2022.812655
Acknowledgements
The authors acknowledge the GEO and TCGA database for providing the platform for uploading meaningful datasets.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
HX and JX processed data and wrote this main manuscript. HX conducted the experiments. QZ designed the study and guided writing manuscript. The final manuscript was read and confirmed by all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that there are no conflicts of interest.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xie, H., Xu, J. & Zhao, Q. Identification of a potential prognostic model combining pyroptosis-related gene with immune microenvironment for pancreatic ductal adenocarcinoma. J Cancer Res Clin Oncol 149, 17175–17187 (2023). https://doi.org/10.1007/s00432-023-05436-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05436-0