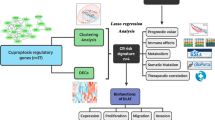
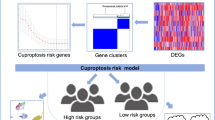
Abstract
Background
Copper homeostasis imbalance has been implicated in tumor progression, aggressiveness, and treatment response. However, the precise roles of cuproptosis-related genes (CRGs) in hepatocellular carcinoma (HCC) remain poorly understood.
Methods
In this study, we employed a consensus clustering algorithm to identify distinct molecular subtypes. We then performed Kaplan–Meier analysis and univariate Cox regression analysis to identify prognostic differentially expressed genes. The expression of these genes was subsequently validated using qPCR on fresh-frozen tissues obtained from HCC patients. Moreover, leveraging the TCGA-HCC cohort, we constructed a CRGs-related risk prediction model using the LASSO and multivariate Cox regression analysis.
Results
By analyzing the data, we successfully established a CRGs risk prognostic model for HCC patients, comprising five differential genes (CAD, SGCB, TXNRD1, KDR, and MTND4P20). Cox regression analysis revealed that the CRGs risk score could serve as an independent prognostic factor for overall survival (hazard ratio [HR] = 1.308, 95% confidence interval [CI] = 1.200 − 1.426, P < 0.001). The area under the curve (AUC) values of the CRGs-score for predicting 1-year, 3-year, and 5-year survival rates were 0.785, 0.724, and 0.723, respectively. Notably, the expression levels of immune checkpoints (including PD-1, PD-L1, and CTLA4) significantly differed between the low- and high-risk score groups. Furthermore, the low-risk score group displayed increased sensitivity to sorafenib, cisplatin, cyclopamine, nilotinib, salubrinal, and gemcitabine, whereas the high-risk score group exhibited heightened sensitivity to lapatinib, erlotinib, and gefitinib.
Conclusions
Our findings highlight the potential of the CRGs risk score as an independent and promising biomarker for clinical prognosis and immunotherapy sensitivity in HCC patients.







Similar content being viewed by others
Availability of data and materials
The raw data used in this article were retrieved from public databases (https://portal.gdc.cancer.gov/repository). All analyzed data are included in this published article and its supplementary information file, which are available from the corresponding author upon reasonable request.
References
Aggett PJ, Fairweather-Tait S (1998) Adaptation to high and low copper intakes: its relevance to estimated safe and adequate daily dietary intakes. Am J Clin Nutr 67(5 Suppl):1061S-S1063. https://doi.org/10.1093/ajcn/67.5.1061
Aubert L, Nandagopal N, Steinhart Z, Lavoie G, Nourreddine S, Berman J et al (2020) Copper bioavailability is a KRAS-specific vulnerability in colorectal cancer. Nat Commun 11(1):3701. https://doi.org/10.1038/s41467-020-17549-y
Brady DC, Crowe MS, Greenberg DN, Counter CM (2017) Copper chelation inhibits BRAF(V600E)-driven melanomagenesis and counters resistance to BRAF(V600E) and MEK1/2 inhibitors. Cancer Res 77(22):6240–6252. https://doi.org/10.1158/0008-5472.CAN-16-1190
Chew EH, Nagle AA, Zhang Y, Scarmagnani S, Palaniappan P, Bradshaw TD et al (2010) Cinnamaldehydes inhibit thioredoxin reductase and induce Nrf2: potential candidates for cancer therapy and chemoprevention. Free Radic Biol Med 48(1):98–111. https://doi.org/10.1016/j.freeradbiomed.2009.10.028
Daniel KG, Harbach RH, Guida WC, Dou QP (2004) Copper storage diseases: Menkes, Wilsons, and cancer. Front Biosci 9:2652–2662. https://doi.org/10.2741/1424
Dong J, Wang X, Xu C, Gao M, Wang S, Zhang J et al (2021) Inhibiting NLRP3 inflammasome activation prevents copper-induced neuropathology in a murine model of Wilson’s disease. Cell Death Dis 12(1):87. https://doi.org/10.1038/s41419-021-03397-1
Ebara M, Fukuda H, Hatano R, Saisho H, Nagato Y, Suzuki K et al (2000) Relationship between copper, zinc and metallothionein in hepatocellular carcinoma and its surrounding liver parenchyma. J Hepatol 33(3):415–422. https://doi.org/10.1016/s0168-8278(00)80277-9
Geeleher P, Cox N, Huang RS (2014) pRRophetic: an R package for prediction of clinical chemotherapeutic response from tumor gene expression levels. PLoS ONE 9(9):e107468. https://doi.org/10.1371/journal.pone.0107468
Hanzelmann S, Castelo R, Guinney J (2013) GSVA: gene set variation analysis for microarray and RNA-seq data. BMC Bioinformatics 14:7. https://doi.org/10.1186/1471-2105-14-7
Huster D (2014) Structural and metabolic changes in Atp7b-/-mouse liver and potential for new interventions in Wilson’s disease. Ann N Y Acad Sci 1315:37–44. https://doi.org/10.1111/nyas.12337
Huster D, Purnat TD, Burkhead JL, Ralle M, Fiehn O, Stuckert F et al (2007) High copper selectively alters lipid metabolism and cell cycle machinery in the mouse model of Wilson disease. J Biol Chem 282(11):8343–8355. https://doi.org/10.1074/jbc.M607496200
Iasonos A, Schrag D, Raj GV, Panageas KS (2008) How to build and interpret a nomogram for cancer prognosis. J Clin Oncol 26(8):1364–1370. https://doi.org/10.1200/JCO.2007.12.9791
Iverson SV, Eriksson S, Xu J, Prigge JR, Talago EA, Meade TA et al (2013) A Txnrd1-dependent metabolic switch alters hepatic lipogenesis, glycogen storage, and detoxification. Free Radic Biol Med 63:369–380. https://doi.org/10.1016/j.freeradbiomed.2013.05.028
Jie J, Hao S, Hongxiu Y, Huiying Y, Jun M, Chenji W et al (2007) Evaluation of Cu in hepatocellular carcinoma by particle induced X-ray emission. J Trace Elem Med Biol 21(4):255–260. https://doi.org/10.1016/j.jtemb.2007.06.004
Llovet JM, Zucman-Rossi J, Pikarsky E, Sangro B, Schwartz M, Sherman M (2016) Hepatocellular carcinoma. Nat Rev Dis Primers 2:16018. https://doi.org/10.1038/nrdp.2016.18
Llovet JM, Kelley RK, Villanueva A, Singal AG, Pikarsky E, Roayaie S et al (2021) Hepatocellular carcinoma. Nat Rev Dis Primers 7(1):6. https://doi.org/10.1038/s41572-020-00240-3
Maillet A, Pervaiz S (2012) Redox regulation of p53, redox effectors regulated by p53: a subtle balance. Antioxid Redox Signal 16(11):1285–1294. https://doi.org/10.1089/ars.2011.4434
Miura H, Miyazaki T, Kuroda M, Oka T, Machinami R, Kodama T et al (1997) Increased expression of vascular endothelial growth factor in human hepatocellular carcinoma. J Hepatol 27(5):854–861. https://doi.org/10.1016/s0168-8278(97)80323-6
Morgan RL, Baack B, Smith BD, Yartel A, Pitasi M, Falck-Ytter Y et al (2013) Eradication of hepatitis C virus infection and the development of hepatocellular carcinoma: a meta-analysis of observational studies. Ann Intern Med 158(5 Pt 1):329–337. https://doi.org/10.7326/0003-4819-158-5-201303050-00005
Nakagawa H, Fujita M, Fujimoto A (2019) Genome sequencing analysis of liver cancer for precision medicine. Semin Cancer Biol 55:120–127. https://doi.org/10.1016/j.semcancer.2018.03.004
Pfeiffenberger J, Mogler C, Gotthardt DN, Schulze-Bergkamen H, Litwin T, Reuner U et al (2015) Hepatobiliary malignancies in Wilson disease. Liver Int 35(5):1615–1622. https://doi.org/10.1111/liv.12727
Polishchuk EV, Merolla A, Lichtmannegger J, Romano A, Indrieri A, Ilyechova EY et al (2019) Activation of autophagy, observed in liver tissues from patients with Wilson disease and from ATP7B-deficient animals, protects hepatocytes from copper-induced apoptosis. Gastroenterology 156(4):1173–1189. https://doi.org/10.1053/j.gastro.2018.11.032
Poo JL, Rosas-Romero R, Montemayor AC, Isoard F, Uribe M (2003) Diagnostic value of the copper/zinc ratio in hepatocellular carcinoma: a case control study. J Gastroenterol 38(1):45–51. https://doi.org/10.1007/s005350300005
Que EL, Domaille DW, Chang CJ (2008) Metals in neurobiology: probing their chemistry and biology with molecular imaging. Chem Rev 108(5):1517–1549. https://doi.org/10.1021/cr078203u
Rahmanto AS, Davies MJ (2012) Selenium-containing amino acids as direct and indirect antioxidants. IUBMB Life 64(11):863–871. https://doi.org/10.1002/iub.1084
Rapisarda A, Melillo G (2012) Role of the VEGF/VEGFR axis in cancer biology and therapy. Adv Cancer Res 114:237–267. https://doi.org/10.1016/B978-0-12-386503-8.00006-5
Ren X, Li Y, Zhou Y, Hu W, Yang C, Jing Q et al (2021) Overcoming the compensatory elevation of NRF2 renders hepatocellular carcinoma cells more vulnerable to disulfiram/copper-induced ferroptosis. Redox Biol 46:102122. https://doi.org/10.1016/j.redox.2021.102122
Ribas A, Wolchok JD (2018) Cancer immunotherapy using checkpoint blockade. Science 359(6382):1350–1355. https://doi.org/10.1126/science.aar4060
Roayaie S, Blume IN, Thung SN, Guido M, Fiel MI, Hiotis S et al (2009) A system of classifying microvascular invasion to predict outcome after resection in patients with hepatocellular carcinoma. Gastroenterology 137(3):850–855. https://doi.org/10.1053/j.gastro.2009.06.003
Sangro B, Sarobe P, Hervas-Stubbs S, Melero I (2021) Advances in immunotherapy for hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 18(8):525–543. https://doi.org/10.1038/s41575-021-00438-0
Smith NR, Baker D, James NH, Ratcliffe K, Jenkins M, Ashton SE et al (2010) Vascular endothelial growth factor receptors VEGFR-2 and VEGFR-3 are localized primarily to the vasculature in human primary solid cancers. Clin Cancer Res 16(14):3548–3561. https://doi.org/10.1158/1078-0432.CCR-09-2797
Tan HW, Mo HY, Lau ATY, Xu YM (2018) Selenium species: current status and potentials in cancer prevention and therapy. Int J Mol Sci. https://doi.org/10.3390/ijms20010075
Topalian SL, Taube JM, Pardoll DM (2020) Neoadjuvant checkpoint blockade for cancer immunotherapy. Science. https://doi.org/10.1126/science.aax0182
Trachootham D, Alexandre J, Huang P (2009) Targeting cancer cells by ROS-mediated mechanisms: a radical therapeutic approach? Nat Rev Drug Discov 8(7):579–591. https://doi.org/10.1038/nrd2803
Tsvetkov P, Coy S, Petrova B, Dreishpoon M, Verma A, Abdusamad M et al (2022) Copper induces cell death by targeting lipoylated TCA cycle proteins. Science 375(6586):1254–1261. https://doi.org/10.1126/science.abf0529
Villanueva A (2019) Hepatocellular carcinoma. N Engl J Med 380(15):1450–1462. https://doi.org/10.1056/NEJMra1713263
Wilkerson MD, Hayes DN (2010) ConsensusClusterPlus: a class discovery tool with confidence assessments and item tracking. Bioinformatics 26(12):1572–1573. https://doi.org/10.1093/bioinformatics/btq170
Wu X, Gu Z, Chen Y, Chen B, Chen W, Weng L et al (2019) Application of PD-1 blockade in cancer immunotherapy. Comput Struct Biotechnol J 17:661–674. https://doi.org/10.1016/j.csbj.2019.03.006
Zhang J, Yao J, Peng S, Li X, Fang J (2017) Securinine disturbs redox homeostasis and elicits oxidative stress-mediated apoptosis via targeting thioredoxin reductase. Biochim Biophys Acta Mol Basis Dis 1863(1):129–138. https://doi.org/10.1016/j.bbadis.2016.10.019
Zou P, Xia Y, Ji J, Chen W, Zhang J, Chen X (2016) Piperlongumine as a direct TrxR1 inhibitor with suppressive activity against gastric cancer. Cancer Lett 375(1):114–126. https://doi.org/10.1016/j.canlet.2016.02.058
Zucman-Rossi J, Villanueva A, Nault JC, Llovet JM (2015) Genetic landscape and biomarkers of hepatocellular carcinoma. Gastroenterology 149(5):1226–1239. https://doi.org/10.1053/j.gastro.2015.05.061. (e4)
Funding
This work was supported by the National Natural Science Foundation of China (81772276) and Hubei Provincial Natural Science Fund for Creative Research Groups (2019CFA018).
Author information
Authors and Affiliations
Contributions
SX, WC, and S-ML conceived and drafted the original manuscript; SX and YY conducted data analysis and bioinformatics analysis; KD, and RG performed the experiments. S-ML, YZ, and CL revised the manuscript for intellectual content. S-ML conduced project administration. All authors reviewed and approved the final version prior to its submission.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval and consent to participate
The processing of clinical tissue samples is in strict compliance with the ethical standards of the Declaration of Helsinki. The present study was approved by the ethics committee of Zhongnan Hospital of Wuhan University (Approval number: 2017058). Written informed consent was obtained from all the participants.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, S., Dong, K., Gao, R. et al. Cuproptosis-related signature for clinical prognosis and immunotherapy sensitivity in hepatocellular carcinoma. J Cancer Res Clin Oncol 149, 12249–12263 (2023). https://doi.org/10.1007/s00432-023-05099-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05099-x