Abstract
Purpose
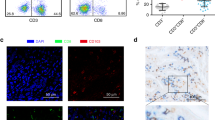
Defining the phenotypic characteristics of CD8+ T cell subsets in gastric cancer (GC) can help remodel the immune microenvironment of the tumor, thereby improving patient prognosis. CD226 has recently been shown to regulate the activity of CD8+ T cell in several malignancies. However, the clinical relevance of CD226+CD8+ T cells in GC remains unclear.
Methods
Fudan University Shanghai Cancer Center (FUSCC) cohort (n = 316), The Cancer Genome Atlas (TCGA) cohort (n = 407), KUGH/KUCM cohort (n = 202), and Asian Cancer Research Group (ACRG) cohort (n = 300) were included in prognosis and response to adjuvant chemotherapy (ACT) analyses. Flow cytometry and multiplex immunostaining were used to characterize CD226+CD8+ T cells.
Results
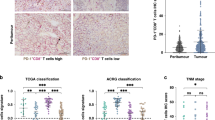
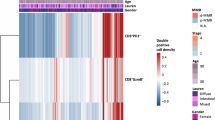
CD226+CD8+ T cells predicted favorable outcomes in patients undergoing curative resection for GC. GC patients with high CD226+CD8+ T cell infiltration benefitted more from adjuvant chemotherapy. CD155 is upregulated in GC tissues and is associated with decreased intra-tumoral CD226+CD8+ T cell infiltration. The combination of intra-tumoral CD226+CD8+ T cells and CD155 is a strong prognostic predictor in patients with GC.
Conclusion
CD226+CD8+ T cells may represent a novel therapeutic target and a useful marker of prognosis and therapeutic response in patients with GC.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ACT:
-
Adjuvant chemotherapy
- GC:
-
Gastric cancer
- mIHF:
-
Multiple immunofluorescence
References
Braun M, Aguilera AR, Sundarrajan A et al (2020) CD155 on Tumor cells drives resistance to immunotherapy by inducing the degradation of the activating receptor CD226 in CD8(+) T cells. Immunity 53:805–823. https://doi.org/10.1016/j.immuni.2020.09.010
Danisch S, Qiu Q, Seth S et al (2013) (2012) CD226 interaction with CD155 impacts on retention and negative selection of CD8 positive thymocytes as well as T cell differentiation to follicular helper cells in Peyer’s Patches. Immunobiology 218:152–158. https://doi.org/10.1016/j.imbio.02.010
Gaud G, Roncagalli R, Chaoui K et al (2018) The costimulatory molecule CD226 signals through VAV1 to amplify TCR signals and promote IL-17 production by CD4(+) T cells. Sci Signal. https://doi.org/10.1126/scisignal.aar3083
Gilfillan S, Chan CJ, Cella M et al (2008) DNAM-1 promotes activation of cytotoxic lymphocytes by nonprofessional antigen-presenting cells and tumors. J Exp Med 205:2965–2973. https://doi.org/10.1084/jem.20081752
Han J, Khatwani N, Searles TG et al (2020) Memory CD8(+) T cell responses to cancer. Semin Immunol 49:101435. https://doi.org/10.1016/j.smim.2020.101435
Hirota T, Irie K, Okamoto R et al (2005) Transcriptional activation of the mouse Necl-5/Tage4/PVR/CD155 gene by fibroblast growth factor or oncogenic Ras through the Raf-MEK-ERK-AP-1 pathway. Oncogene 24:2229–2235. https://doi.org/10.1038/sj.onc.1208409
Koyama M, Kuns RD, Olver SD et al (2013) Promoting regulation via the inhibition of DNAM-1 after transplantation. Blood 121:3511–3520. https://doi.org/10.1182/blood-2012-07-444026
Lees JR (2020) CD8+ T cells: The past and future of immune regulation. Cell Immunol 357:104212. https://doi.org/10.1016/j.cellimm.2020.104212
Niederlova V, Tsyklauri O, Chadimova T et al (2021) CD8(+) Tregs revisited: a heterogeneous population with different phenotypes and properties. Eur J Immunol 51:512–530. https://doi.org/10.1002/eji.202048614
O’Donnell JS, Madore J, Li XY et al (2020) Tumor intrinsic and extrinsic immune functions of CD155. Semin Cancer Biol 65:189–196. https://doi.org/10.1016/j.semcancer.2019.11.013
Shibuya A, Campbell D, Hannum C et al (1996) DNAM-1, a novel adhesion molecule involved in the cytolytic function of T lymphocytes. Immunity 4:573–581. https://doi.org/10.1016/s1074-7613(00)70060-4
Stamm H, Wellbrock J, Fiedler W (2018) Interaction of PVR/PVRL2 with TIGIT/DNAM-1 as a novel immune checkpoint axis and therapeutic target in cancer. Mamm Genome 29:694–702. https://doi.org/10.1007/s00335-018-9770-7
Tsuburaya A, Guan J, Yoshida K et al (2021) Clinical biomarkers in adjuvant chemotherapy for gastric cancer after D2 dissection by a pooled analysis of individual patient data from large randomized controlled trials. Gastric Cancer 24:1184–1193. https://doi.org/10.1007/s10120-021-01228-y
Weulersse M, Asrir A, Pichler AC et al (2020) Eomes-dependent loss of the Co-activating receptor CD226 restrains CD8(+) T cell anti-tumor functions and limits the efficacy of cancer immunotherapy. Immunity 53:824–839. https://doi.org/10.1016/j.immuni.2020.09.006
Yamashita K, Hosoda K, Niihara M et al (2021) History and emerging trends in chemotherapy for gastric cancer. Ann Gastroenterol Surg 5:446–456. https://doi.org/10.1002/ags3.12439
Acknowledgements
We thank Dr. Zhen Xiang and Dr. Siyuan Liang for their helpful discussion and technical assistance.
Funding
National Natural Science Foundation of China (81902392); Clinical Research Plan of SHDC (No. SHDC2020CR3033B).
Author information
Authors and Affiliations
Contributions
YZ: performed the experiments; YZ: analyzed the data and prepared the manuscript; YZ, ZZ, JG and HH: provided the samples. YZ and HH: wrote the paper; YZ, ZZ, JG, YH and HH: commented on the study and revised the paper; HH: supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
The study was approved by the ethics committee of the FUSCC.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
All authors have consented to publication of the results presented in this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Zhao, Zx., Gao, Jp. et al. Tumor-infiltrating CD226+CD8+ T cells are associated with postoperative prognosis and adjuvant chemotherapeutic benefits in gastric cancer patients. J Cancer Res Clin Oncol 149, 4381–4389 (2023). https://doi.org/10.1007/s00432-022-04346-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04346-x