Abstract
Purpose
The effect of immuno-chemotherapy on patients with advanced non-small-cell lung cancer (NSCLC) harboring oncogenic mutations remains poorly understood. This study aimed to characterize the efficacy of immuno-chemotherapy and determine the optimal treatment strategy for such patients.
Methods
We conducted this retrospective cohort study on patients with NSCLC harboring oncogenic driver alterations and treated with an immune checkpoint inhibitor combined with chemotherapy at five institutions. The clinical characteristics and outcomes of immuno-chemotherapy for NSCLC with oncogenic mutations in a real-world setting were analyzed.
Results
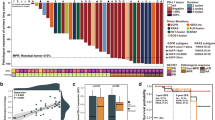
Among 846 patients diagnosed with advanced or recurrent NSCLC between April 2017 and April 2021, 43 patients with oncogenic mutations were treated with immuno-chemotherapy. The median age of patients was 68 (range 44–78) years; 42% of patients never smoked, and adenocarcinoma was the most common histology (95%). In patients with KRAS mutations (n = 10) or PD-L1 expression of 50% or greater (n = 10), the disease control rate was 100%. The median progression-free survival (PFS) was 5.4, 6.3, and 8.9 months in patients harboring mutations in EGFR, KRAS, and other genes, respectively (P = 0.22). Patients with PD-L1 expression of 50% or greater had significantly longer median PFS than patients with PD-L1 expression of less than 50% (16.4 vs. 5.1 months; P = 0.001). Two patients experienced grade 3 immuno-related adverse events.
Conclusion
Immuno-chemotherapy has a clinical benefit and is safe for patients with oncogenic mutations. Notably, patients with PD-L1 expression of 50% or more experience greater benefit from immuno-chemotherapy than those with PD-L1 expression of less than 50%.




Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Addeo A, Passaro A, Malapelle U, Luigi Banna G, Subbiah V, Friedlaender A (2021) Immunotherapy in non-small cell lung cancer harbouring driver mutations. Cancer Treat Rev 96:102179. https://doi.org/10.1016/j.ctrv.2021.102179
Arrieta O, Barrón F, Ramírez-Tirado LA et al (2020) Efficacy and safety of pembrolizumab plus docetaxel vs docetaxel alone in patients with previously treated advanced non-small cell lung cancer: the PROLUNG phase 2 randomized clinical trial. JAMA Oncol 6(6):856–864. https://doi.org/10.1001/jamaoncol.2020.0409
Dafni U, Tsourti Z, Vervita K, Peters S (2019) Immune checkpoint inhibitors, alone or in combination with chemotherapy, as first-line treatment for advanced non-small cell lung cancer. A systematic review and network meta-analysis. Lung Cancer 134:127–140. https://doi.org/10.1016/j.lungcan.2019.05.029
Gadgeel S, Rodriguez-Abreu D, Felip E et al (2019) LBA5 - KRAS mutational status and efficacy in KEYNOTE-189: pembrolizumab (pembro) plus chemotherapy (chemo) vs placebo plus chemo as first-line therapy for metastatic non-squamous NSCLC. Ann Oncol. https://doi.org/10.1093/annonc/mdz453.002
Gandhi L, Rodríguez-Abreu D, Gadgeel S et al (2018) Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med 378(22):2078–2092. https://doi.org/10.1056/NEJMoa1801005
Guisier F, Dubos-Arvis C, Viñas F et al (2020) Efficacy and safety of anti-PD-1 immunotherapy in patients with advanced NSCLC with BRAF, HER2, or MET mutations or RET translocation: GFPC 01–2018. J Thorac Oncol 15(4):628–636. https://doi.org/10.1016/j.jtho.2019.12.129
Harada D, Takata K, Mori S et al (2019) Previous immune checkpoint inhibitor treatment to increase the efficacy of docetaxel and ramucirumab combination chemotherapy. Anticancer Res 39(9):4987–4993. https://doi.org/10.21873/anticanres.13688
Korpanty GJ, Graham DM, Vincent MD, Leighl NB (2014) Biomarkers that currently affect clinical practice in lung cancer: EGFR, ALK, MET, ROS-1, and KRAS. Front Oncol 4:204. https://doi.org/10.3389/fonc.2014.00204
Long Y, Xiong Q, Song Q et al (2022) Immunotherapy plus chemotherapy showed superior clinical benefit to chemotherapy alone in advanced NSCLC patients after progression on osimertinib. Thorac Cancer 13(3):394–403. https://doi.org/10.1111/1759-7714.14271
Masuda K, Horinouchi H, Tanaka M et al (2021) Efficacy of anti-PD-1 antibodies in NSCLC patients with an EGFR mutation and high PD-L1 expression. J Cancer Res Clin Oncol 147(1):245–251. https://doi.org/10.1007/s00432-020-03329-0
Mazieres J, Drilon A, Lusque A et al (2019) Immune checkpoint inhibitors for patients with advanced lung cancer and oncogenic driver alterations: results from the immunotarget registry. Ann Oncol 30(8):1321–1328. https://doi.org/10.1093/annonc/mdz167
Nogami N, Barlesi F, Socinski MA et al (2022) IMpower150 final exploratory analyses for atezolizumab plus bevacizumab and chemotherapy in key NSCLC patient subgroups with EGFR mutations or metastases in the liver or brain. J Thorac Oncol 17(2):309–323. https://doi.org/10.1016/j.jtho.2021.09.014
Offin M, Guo R, Wu SL et al (2019) Immunophenotype and response to immunotherapy of RET-rearranged lung cancers. JCO Precis Oncol. https://doi.org/10.1200/PO.18.00386
Reck M, Mok TSK, Nishio M et al (2019) Atezolizumab plus bevacizumab and chemotherapy in non-small-cell lung cancer (IMpower150): key subgroup analyses of patients with EGFR mutations or baseline liver metastases in a randomised, open-label phase 3 trial. Lancet Respir Med 7(5):387–401. https://doi.org/10.1016/S2213-2600(19)30084-0
Rodríguez-Antona C, Taron M (2015) Pharmacogenomic biomarkers for personalized cancer treatment. J Intern Med 277(2):201–217. https://doi.org/10.1111/joim.12321
Saalfeld FC, Wenzel C, Christopoulos P et al (2021) Efficacy of immune checkpoint inhibitors alone or in combination with chemotherapy in NSCLC harboring ERBB2 mutations. J Thorac Oncol 16(11):1952–1958. https://doi.org/10.1016/j.jtho.2021.06.025
Sabari JK, Leonardi GC, Shu CA et al (2018) PD-L1 expression, tumor mutational burden, and response to immunotherapy in patients with MET exon 14 altered lung cancers. Ann Oncol 29(10):2085–2091. https://doi.org/10.1093/annonc/mdy334
Sholl LM, Aisner DL, Varella-Garcia M et al (2015) Multi-institutional oncogenic driver mutation analysis in lung adenocarcinoma: the lung cancer mutation consortium experience. J Thorac Oncol 10(5):768–777. https://doi.org/10.1097/JTO.0000000000000516
Socinski MA, Nishio M, Jotte RM et al (2021) IMpower150 final overall survival analyses for Atezolizumab plus bevacizumab and chemotherapy in first-line metastatic nonsquamous NSCLC. J Thorac Oncol 16(11):1909–1924. https://doi.org/10.1016/j.jtho.2021.07.009
Sun L, Hsu M, Cohen RB, Langer CJ, Mamtani R, Aggarwal C (2021) Association between KRAS variant status and outcomes with first-line immune checkpoint inhibitor-based therapy in patients with advanced non-small-cell lung cancer. JAMA Oncol 7(6):937–939. https://doi.org/10.1001/jamaoncol.2021.0546
Yoda S, Dagogo-Jack I, Hata AN (2019) Targeting oncogenic drivers in lung cancer: recent progress, current challenges and future opportunities. Pharmacol Ther 193:20–30. https://doi.org/10.1016/j.pharmthera.2018.08.007
Yoshimura A, Yamada T, Okuma Y et al (2019) Retrospective analysis of docetaxel in combination with ramucirumab for previously treated non-small cell lung cancer patients. Transl Lung Cancer Res 8(4):450–460. https://doi.org/10.21037/tlcr.2019.08.07
Zhao S, Xian X, Tian P, Li W, Wang K, Li Y (2021) Efficacy of combination chemo-immunotherapy as a first-line treatment for advanced non-small-cell lung cancer patients with HER2 alterations: A case series. Front Oncol 11:633522. https://doi.org/10.3389/fonc.2021.633522
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
TH: conceptualization, data curation, investigation, writing, and editing. CS: project administration, investigation, writing—review. KH: formal analysis and methodology. HK, MI, TN, YT, NT, SS, TT, TY, TY, MT: investigation. YN: supervision.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Yamada received honoraria from Eli Lilly Japan K.K. and research funding from ONO Pharmaceutical Co., Ltd., Janssen Pharmaceutical K.K., CHUGAI Pharmaceutical Co., Ltd., Takeda Pharmaceutical Co., Ltd., and Pfizer Inc. All other authors declare no conflicts of interest.
Ethical approval
This study was approved by the institutional review committee of each participating hospital. (the Institutional Review Board of Rakuwakai Otowa Hospital, the Medical Ethics Committee of Kurashiki Central Hospital, the Ethics Review Board of the Kyoto Prefectural University of Medicine, the Ethics Review Committee of Japanese Red Cross Kyoto Daini Hospital, and the Ethics Review Committee of Japanese Red Cross Kyoto Daiichi Hospital).
Consent for publication
This manuscript contains no individual’s personal data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hata, T., Sakaguchi, C., Hirano, K. et al. Efficacy and safety of immuno-chemotherapy in patients with advanced non-small-cell lung cancer harboring oncogenic mutations: a multicenter retrospective study. J Cancer Res Clin Oncol 149, 2475–2482 (2023). https://doi.org/10.1007/s00432-022-04125-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04125-8