Abstract
Transient receptor potential canonical (TRPC) 4 channels are calcium-permeable, nonselective cation channels and are widely expressed in mammalian tissue, especially in the GI tract and brain. TRPC4 channels are known to be involved in neurogenic contraction of ileal smooth muscle cells via generating cationic current after muscarinic stimulation (muscarinic cationic current (mIcat)). Polyamines exist in numerous tissues and are believed to be involved in cell proliferation, differentiation, scar formation, wound healing, and carcinogenesis. Besides, physiological polyamines are essential to maintain inward rectification of cardiac potassium channels (Kir2.1). At membrane potentials more positive than equilibrium potential, intracellular polyamines plug the cytosolic surface of the Kir2.1 so that potassium ions cannot pass through the pore. Recently, it was reported that polyamines inhibit not only cardiac potassium channels but also nonselective cation channels that mediate the generation of mIcat. Here, we report that TRPC4, a definite mIcat mediator, is inhibited by intracellular spermine with great extent. The inhibition was specific to TRPC4 and TRPC5 channels but was not effective to TRPC1/4, TRPC1/5, and TRPC3 channels. For this inhibition to occur, we found that glutamates at 728th and 729th position of TRPC4 channels are essential whereby we conclude that spermine blocks the TRPC4 channel with electrostatic interaction between negative amino acids at the C-terminus of the channel.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Transient receptor potential canonical (TRPC) channel is a ubiquitously expressed, nonselective cation channel in a mammalian cell and is known to be involved in numerous physiological functions such as GI tract motility, vascular smooth muscle contraction/relaxation, endothelial permeability, salivary gland secretion, fear control, and many others [2, 3, 39]. The TRPC family is comprised of seven channels, i.e., TRPC1 to TRPC7 where TRPC1, 4, and 5 are classified as one subgroup while TRPC3, 6, and 7 are classified as the other subgroup based on their amino acid sequence homology.
While various TRPC channels conduct dynamic physiological functions, one of the most prominent cases is the role of TRPC4 and TRPC6 in gastrointestinal (GI) physiology, especially in ileum myocytes [16, 30, 36]. Muscarinic cationic current (mIcat), a cationic current evoked by muscarinic stimulation, in murine ileal myocyte had been heavily studied to identify its molecular candidates. At present, the molecular nature of channels carrying mIcat has been well defined. We showed that mIcat was not recorded in TRPC4 knockout mice [16]. Importantly, the activation of TRPC4 requires PTX-sensitive G proteins and an increase in [Ca2+]i [11, 27]. These properties are similar to those of mIcat in native ileal smooth muscle cells [11, 16]. Recent studies presented conclusive evidence that TRPC4 and TRPC6 function as two separate channels responsible for mIcat [32, 38]. Their findings suggest that TRPC4 and TRPC6 couple muscarinic receptors to the depolarization of intestinal smooth muscle cells, voltage-activated Ca2+ influx and muscle contraction, and consequently regulating small intestinal motility in vivo. Tsvilovskyy et al. studied mIcat activation and smooth muscle function in mice lacking TRPC4 alone, TRPC6 alone, or both TRPC4 and TRPC6. Their data showed that, in intestinal smooth muscle cells, TRPC4 forms a 55-pS cation channel whose currents contribute >80 % to mIcat and TRPC6 currents contribute the remaining <20 %. There appeared to be no overlap of or compensation between the currents mediated by TRPC4 and TRPC6 in the single TRPC knockouts, whereas mIcat was completely eliminated in the TRPC4/TRPC6 double knockouts. These findings demonstrated that TRPC4 and TRPC6 constitute the muscarinic receptor-activated channels in gastrointestinal smooth muscle cells and thereby critically regulate smooth muscle contraction.
Polyamines are known to be involved in proliferation, differentiation, wound healing, scar formation, and carcinogenesis [5]. Although they are in charge of various cellular functions, the importance of intracellular spermine in inward rectification of potassium channels is critical. Studied by Kir2.1 channels, it has been reported that intracellular spermine plugs the inner vestibule and pore of the Kir2.1 channel whereby it prohibits further outflow of potassium ions. Without physiological concentration of intracellular spermine, Kir2.1 lost its inward rectification and showed ohmic I-V relationship. Electrophysiological recording in Kir2.1-expressing Xenopus oocytes further revealed that binding sites for spermine is D172, E224, and E299. These site-directed mutagenesis studies showed that switching these sites to neutral amino acids knocks out the inward-rectifying property of Kir2.1 channels [6, 7, 17, 21]. Clinically, loss of inward rectification in Kir2.1 channel due to inherited mutation at one of the spermine-binding site (D172) caused short QT syndrome which may lead to atrial fibrillation, polymorphic ventricular tachycardia mainly due to increased vulnerability to reentry [13, 31]. Even more, congenital short QT syndrome could cause sudden infant death syndrome (SIDS) [13].
Meanwhile, it has been reported that extracellular polyamines, especially spermine, potently inhibit mIcat [39]. mIcat measured from isolated guinea pig ileal myocytes was strongly inhibited by micromolar extracellular spermine while the I-V shape of dampened mIcat remained constant: double-rectifying. Tsvilovskyy et al. showed that not only spermine but smaller polyamines such as spermidine and putrescine were also able to inhibit mIcat in guinea pig ileal myocytes. These findings are concordant with prior discoveries [25, 28] which demonstrated that extracellular polyamines relaxed smooth muscles by decreasing their electrical excitability by decreasing cytosolic Ca2+.
In the present study, we investigated whether intracellular spermine inhibits TRPC4 or not. Furthermore, we tried to find the regions of TRPC4 channel to which spermine binds.
Materials and methods
Cell culture and transient transfection
Human embryonic kidney (HEK293) cells (ATCC, Manassas, VA) were maintained according to the supplier’s recommendations. For the transient transfection, the cells were seeded in 12-well plates. The following day, 0.5 ∼ 2.5 μg/well of complementary DNA (cDNA) was transfected using the transfection reagent FuGENE 6 (Roche Molecular Biochemicals), as detailed in the manufacturer’s protocol. For the co-expression of TRPC1 channels and TRPC4 or TRPC5 channels, 1.0 μg of TRPC1 cDNA, 1.0 μg of TRPC4 or TRPC5 cDNA, and 0.5 μg of muscarinic receptor cDNA were loaded in corresponding wells. Detailed protocol for co-expression experiment can be found elsewhere [15]. After 30–40 h, the cells were trypsinized and transferred to a small recording chamber for whole-cell recording.
Cloning and mutagenesis
All mutagenesis was conducted using QuikChange site-directed mutagenesis kit (Stratagene). Human TRPC1 isoform long (GenBank ID: U31110, UniProt ID: P48995-1) inserted in pcDNA3.1 vector was kindly donated by Dr. Michael Mederos. Mouse TRPC4 isoform short cDNA (GenBank ID: U50921.1, UniProt ID: Q9QUQ5-2) was kindly donated by Dr. V. Flockerzi and Dr. M. Schaefer. DNA sequences encoding the channel was truncated by BglIII and SalI restriction enzymes and inserted into pEGFP-N1 vector. Mouse TRPC5 cDNA (GenBank ID: AF029983, UniProt ID: Q9QX29) inserted in pEGFP-N1 vector was kindly donated by Dr. S. Kaneko and Dr. Y. Mori. Human muscarinic acetylcholine receptor types 2 and 3 in pcDNA3.1+ vector were purchased from Missouri S&T cDNA resource center (Missouri, USA).
Electrophysiology
The cells were transferred to a small chamber on the stage of an inverted microscope (Eclipse Ti, Nikon, Japan) and attached to coverslips in the small chamber for 10 min prior to the patch recording. Glass microelectrodes with 2–2.5 MΩ resistance were used to obtain gigaohm seals. The bath solutions were constantly perfused with an extracellular solution at a rate of 1–2 ml/min. The currents were recorded using an Axopatch 200B patch clamp amplifier (Axon Instruments, USA). The current was recorded for 550-ms duration ramps from +100 to 120 mV with a time resolution of 0.4 mV/ms and with a holding membrane potential of −60 mV. pCLAMP software v.10.2 and a Digidata 1440A (Axon Instruments) were used for data acquisition and application of the command pulses. The data were filtered at 5 kHz and displayed on a computer monitor. The data were analyzed using pCLAMP v.10.2 and Origin Pro 8 software (OriginLab, MA, USA). For recordings of the TRPC channels, we used normal Tyrode solution (NT) unless otherwise mentioned and occasionally Cs+-rich external solution. The normal Tyrode solution contained 135 mM NaCl, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, 10 mM glucose, and 10 mM HEPES with a pH of 7.4 adjusted with NaOH. The Cs+-rich external solution contained equimolar CsCl rather than NaCl and KCl. pH was adjusted to 7.4 using CsOH. For receptor stimulation, 100 μM carbachol (carbamoylcholine chloride) was added to each extracellular bath solution. The internal solution contained 140 mM CsCl, 10 mM HEPES, 0.2 mM Tris-GTP (Tris-guanosine 5’-triphospate), 0.5 mM EGTA, and 3 mM Mg-ATP (adenosine 5′-triphosphate) with a pH of 7.3 adjusted with CsOH. All reagents were purchased from Sigma (Sigma Aldrich, USA).
Statistics
Results are expressed as means ± SEM. Results were compared using Student’s t test between two groups. P < 0.05 was considered statistically significant. All statistical analysis was done with Origin Pro 8 software (OriginLab, MA, USA).
Results
Intracellular spermine block TRPC4 channel regardless of activation mechanism
In order to investigate the effect of polyamines to TRPC4 channel, we expressed mouse TRPC4β tagged with EGFP at the C-terminus (mTRPC4β-EGFP) in HEK293 cells. After a whole-cell configuration was made, Cs+-rich solution ([Cs+]o = 140 mM) was applied to magnify the activity of TRPC4 channel [15]. Since TRPC4 channel can be activated by either infusion of intracellular GTPγS or stimulation of G protein-coupled receptor [15], we used these activation mechanisms to open the TRPC4 channels. For G protein-coupled receptors and their agonists, muscarinic acetylcholine receptor subtype 2 (mAChR2, hM2R) or 3 (mAChR3, hM3R) and 100 μM of extracellular carbachol (CCh) were used.
When 1 mM of spermine was added to internal solution and waited at least 3 min for diffusive dialysis [39], TRPC4 current was largely decreased (Fig. 1a, b, c, and d). This inhibition was consistent regardless of activation mechanism. Although 1 mM of intracellular spermine severely inhibited TRPC4 current, the same concentration of other polyamines was not as potent as spermine. One millimolar of intracellular spermidine and putrescine slightly decreased or showed no effect to TRPC4 current (Supplementary Fig. 1a and b).
Intracellular spermine inhibits TRPC4 channels regardless of activation mechanisms. From (a) to (c), all left panels indicate current traces and middle panels indicate corresponding I-V curves at noted points (1, 2, and 3). The right panel summarizes current densities of each group. a Whole-cell current measured from HEK293 cells expressing mTRPC4β-EGFP. One millimolar of spermine (SPM) was added in pipette solution together with 0.2 mM of GTPγS. Whole-cell TRPC4 current was greatly reduced, especially outward currents. b, c Whole-cell current of TRPC4 channels activated by either type 3 muscarinic acetylcholine receptor (hM3R) or type 2 muscarinic acetylcholine receptor (hM2R)
These results suggest that intracellular spermine inhibits TRPC4 channel efficiently, and the potency of it is strongest among physiological polyamines.
Blocking action of intracellular spermine is specific to TRPC4 and TRPC5 channels
In mammalian tissues, seven TRPC channels are known to exist [8]. Among them, TRPC1, 4, and 5 are classified as one subgroup and TRPC3 and 6 are classified as the other subgroup based on amino acid sequence homology [8]. Since the molecular behavior of these channels could be unique on their own, we examined the action of intracellular spermine on various TRPC channels.
As a result, intracellular spermine inhibited TRPC5 channels but could not block TRPC1α/4 or TRPC1α/5 channels (Fig. 2a, b, and c). For the activation of TRPC1α/4 and TRPC1α/5 channels, muscarinic acetylcholine receptor subtype 3 and 100 μM of extracellular carbachol was used. After waiting at least 3 min for diffusive dialysis of spermine in pipette solution, carbachol was treated. As the I-V curves and current trace show, extracellular carbachol induced outward-rectifying TRPC1α/4 and TRPC1α/5 currents (Fig. 2b and c). We also tested whether spermine inhibits TRPC3 channels. TRPC3 was activated by muscarinic stimulation since GTPγS was not effective to activate TRPC3 unlike TRPC4 or TPRC5 channels. Spermine inhibited both inward and outward current equally in case of TRPC3 (Supplementary Fig. 2).
Intracellular spermine inhibits only TRPC4 and TRPC5 but cannot inhibit TRPC1α/4 or TRPC1α/5 channels. From (a) to (c), all left panels indicate current traces and middle panels indicate corresponding I-V curves at noted points (1, 2, and 3). The right panel summarizes current densities of each group. a Whole-cell current measured from HEK293 cells expressing TRPC5. A concentration of 0.2 mM of GTPγS was infused in pipette solution. Current-voltage relationship showed typical double-rectifying shape. b, c Whole-cell current measured from HEK293 cells expressing TRPC1α/4 or TRPC1α/5 channels. Since TRPC1α/4 or TRPC1α/5 channels cannot be activated by intracellular GTPγS [15], muscarinic receptor and extracellular carbachol was used. Intracellular spermine could not inhibit either of the channels
These results suggest that the blocking action of intracellular spermine is specific to TRPC4 and TRPC5 channels.
Intracellular block of spermine is dose-dependent and voltage-dependent
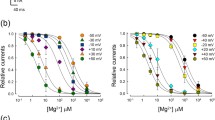
While the inhibitory action of intracellular spermine to TRPC4 channel was clear, the magnitude of inhibition was different along the membrane potential. In other words, outward current was more inhibited than inward current which can be easily seen in I-V curves (Fig. 1a, b, c, and d). In order to see if the action of intracellular spermine to TRPC4 channel is dose-dependent or voltage-dependent, we first titrated intracellular spermine concentration. As the concentration of intracellular spermine increased from 0.01 mM to 1 mM, the blocking action was gradually increased (Fig. 3a). Since the steady-state current could be obtained at each membrane potential, half-inhibitory concentration (IC50) was calculated (Fig. 3b and c). For each steady-state current, Hill plot was used to fit the data.
Dose-dependent and voltage-dependent inhibition of intracellular spermine onto TRPC4 current. a TRPC4 currents with different concentrations of intracellular spermine (0, 0.1, 0.32, and 1 mM, respectively). As the concentration of intracellular spermine increases, the magnitude of inhibition was also increased. b, c Hill-plot and parametric coefficients for currents measured in (a). From −100 mV ∼ +100 mV, IC50 was in a range of 0.12 mM ∼ 0.22 mM with dynamic fluctuation along membrane potential. Hill-coefficient (n) also showed dynamic fluctuation along membrane potential
Although the blocking action of spermine seen by whole-cell current was more prominent at positive membrane potential, IC50 at +100 mV (0.207 ± 0.006 mM) was higher than IC50 at −100 mV (0.155 ± 0.001 mM). Overall, IC50 was in a range of 0.12 mM ∼ 0.22 mM. Hill coefficient (n) at +100 mV (2.76 ± 0.15) was lower than Hill coefficient (n) at −100 mV (5.52 ± 0.01). At membrane potentials from 100 mV to +100 mV, IC50 and Hill coefficient (n) showed dynamic fluctuation (Fig. 3c).
These results suggest that the blocking action of intracellular spermine is dose-dependent and voltage-dependent.
Cluster of negative amino acids at the C-terminus (720–740) is responsible for the blocking of outward current
Structural topology of TRPC4 channels has been continuously developed throughout decades by efforts. Although definite structural information is missing due to lack of crystallography, it is generally perceived that TRPC4 channel has six transmembrane domains and two cytosolic terminals, i.e., one N-terminus and one C-terminus [9]. Among those, publications showed that numerous interactions are occurring between the C-terminus of TRPC4 channel and various biomolecules such as calmodulin [37, 43], inositol triphosphate receptor (IP3R) [22, 35], α-spectrin II [27], phosphatidylinositol 4,5-bisphosphate (PIP2) [23, 29], α subunit of inhibitory heterotrimeric G protein (Gαi) [12], and many others [9].
Meanwhile, the discovery that polyamine blocks the outward current of cation channels is not the first. The importance of polyamines to maintain inward rectification in inwardly rectifying potassium channels is widely known [33, 34]. In these channels, spermine binds to and blocks the selectivity filter and lower cavity with Coulomb interaction, i.e., negative residues (D172, E224, and E299 of Kir2.1) at those sites mediate ionic interaction between the channel and polyamines [10, 33]. Since the pKa value of each amine group is higher than physiological pH [30], it could be presumed that ionic valence (z) of spermine at intracellular milieu is +4.
In order to locate the interaction site between spermine, we first examined negative amino acids at the C-terminus of TRPC4 channel. Among 270 amino acids at the C-terminus, a total 43 of negative amino acids were found (Supplementary Fig. 3a). Although setting the window width to 40 amino acids showed sporadic distribution of negative amino acids, setting the cut-off amino acid at 740th residue yielded clear stratification. In other words, 29 negative amino acids were located after 740th residue while only 14 negative amino acids were located before 740th residue (Supplementary Fig. 3b). Moreover, residues from 720 to 740 had 6 negative amino acids out of 14. Therefore, we prepared two truncated TRPC4 channels; one contained amino acids from 1 to 720 and the other contained 1 to 740 (Fig. 4a and b).
Topological diagram and truncation mutants of TRPC4 channel targeting for possible spermine binding sites. a Schematic diagram illustrating basic topology and significant domains of TRPC4 channels. TRPC4 channel is comprised of cytosolic N-terminus, six transmembrane domains and cytosolic C-terminus. N-terminus consists of four ankyrin-repeat domains, a coiled-coil domain and a calmodulin (CaM)-binding site. There are many binding sites in the C-terminus including PH-like domain, CaM and IP3R-binding domain (CIRB1), αII-spectrin-binding domain, and others. There are 14 negative amino acids before 740th residue and 29 negative amino acids after 740th residue (see Supplementary Fig. 3). b Schematic diagram for truncation mutants, i.e., TRPC4Δ(741–890) and TRPC4Δ(721–890). c, d I-V curves for truncation mutants. TRPC4Δ(741–890) showed little recovery while TRPC4Δ(721–890) showed significant recovery from spermine block
After expressed to HEK293 cells, these mutated channels were activated by 0.2 mM of intracellular GTPγS and its activity was magnified by using Cs+-rich extracellular solution ([Cs+]o = 140 mM). As a result, 1 mM of spermine blocked Δ(741 ∼ 890) as wild type but the blocking was relieved in Δ(721 ∼ 890) mutant (Fig. 4c). In Δ(721 ∼ 890)-expressing cells, spermine blocking was recovered up to 50 % at +100 mV whereas Δ(741 ∼ 890)-expressing cells showed little relief (Fig. 4c and d).
Since the blocking action of spermine was relieved when negative amino acids after 720th residue were deleted, we now truncated amino acids from position 720 to position 740, i.e., we prepared Δ(720–740) mutant of TRPC4 channel (Fig. 5a). Intracellular spermine could not block the inward current of Δ(720–740) mutant and it blocked only half of the outward current (Fig. 5b). The whole-cell current in response to intracellular spermine titration could not be fitted into Hill’s equation since the gradual change was too minute (Fig. 5b).
Negative amino acid clusters in 720–740 residue are responsible for spermine binding. a Schematic representation of truncation mutant TRPC4Δ(720–740). b Left panel, I-V relationship from whole-cell current measured from HEK293 cells expressing TRPC4Δ(720–740) channels with (open circle) and without (closed circle) intracellular spermine infusion. Although spermine had been infused, TRPC4Δ(720–740) mutant showed indifferent inward currents and outward currents were also significantly recovered (p < 0.05). Right panel, since spermine could not inhibit the channel properly, normalized currents could not be fitted into Hill-plot. Only a minute decrease in current along increased spermine concentration was observed
These results suggest that the outward current block of spermine to TRPC4 channel is mediated by negative amino acids at 720 ∼ 740 residue at the C-terminus.
Glutamate at 728th and 729th position is responsible for the spermine-mediated outward current block of TRPC4 channels
After localizing putative region where spermine and the C-terminus of TRPC4 interact, we investigated which amino acids among 720–740 regions are responsible for spermine binding. We mutated D or E to alanine (A) and made six single mutants, i.e., D724A, E728A, E729A, E733A, E734A, and E738A mutants. The current amplitudes of the mutants were smaller than wild-type TRPC4 channel (Fig. 6a). Among the six mutants, E728A and E729A showed less inhibition by spermine without any effect on the inward current of TRPC4 channel (Fig. 6a). Based on single mutation results, we also made double mutants, i.e., E728A/E729A, E728A/E734A, and E734A/E738A (Fig. 6a). The latter two mutants were made because E734A mutant showed large current while maintaining inhibitory action by spermine. The magnitude of inhibitory action of spermine to each mutants, which was speculated by current ratio between outward currents measured without and with spermine at +100 mV (I spermine/I mock), was relieved at double mutant E728A/E729A and E728A/E734A, but not at E734A/E738A (Fig. 6b). In addition, I spermine/I mock analysis for all of the currents measured above showed clear tendency that E728 and E729 are responsible for the spermine-mediated outward current block of TRPC4 channels (Fig. 6b). All single and double mutants with noticeable current showed typical double-rectifying I-V curves which are similar to wild-type TRPC4 channel, E728A and E729A (Fig. 6c).
Glutamates at 728th and 729th position are crucial for spermine binding to TRPC4 channels. a Summarized current density of single and double mutants for glutamates at 720–740 region. Spermine showed little effect to E728A and E728A single mutants and E728/E729A and E728/E734A double mutants. Many of the mutants showed smaller current density compared to wild-type channels. b In order to quantify the magnitude of spermine block at positive driving force (hence positive membrane potential since E rev ∼ 0 mV), current ratios at +100 mV before and after spermine infusion was calculated (I spermine/I mock). Mutants containing E728A or E729A showed significant recovery from spermine block. c I-V relationship of E728A and E729A mutants showed typical double-rectifying shape which clearly demonstrates that these mutants keep the characteristics of TRPC4 channel intact
Overall, these results suggest that the outward current block of spermine to TRPC4 channel is mediated by negative amino acids (glutamates) at 728th and 729th residue at the C-terminus.
Discussion
In this study, we showed that intracellular spermine (∼1 mM) inhibited TRPC4 channel regardless of the activation mechanism of the channels. In other words, 1 mM spermine inhibited TRPC4 channel whether it was opened by intracellularly infused GTPγS or by stimulation of muscarinic receptors via extracellularly applied carbachol (CCh, 100 μM). In addition, the subtype of muscarinic receptor was not relevant, i.e., spermine inhibited TRPC4 channel activated by either mAChR3 or mAChR2. Other physiological polyamines, i.e., spermidine and putrescine, could not effectively inhibit TRPC4 channels while they slowed the activation kinetics of the channels. Smaller polyamines may compete with the local Ca2+ and slow down the activation process. Meanwhile, the potency of inhibitory action of spermine to TRPC4 channel was both dose-dependent and membrane potential-dependent. With the same concentration of intracellular spermine, outward current was more potently inhibited. Since reversal potential was fixed constantly throughout measurement, it could be said that the potency of the spermine at a given concentration is dependent on a membrane potential. Likewise, at a given membrane potential the inhibition of the current was dependent on the concentration of the intracellular spermine. The inhibitory action of spermine to TRPC channels was specific to TRPC4 and TRPC5 channels, at least among TRPC1, 4, 5 and their heterotetrameric combinations. Spermine inhibited TRPC4 and TRPC5 channels but could not block TRPC1α/4 and TRPC1α/5 channels. The nature of molecular interaction between polycationic spermine and TRPC4 channel was ionic-ionic interaction, i.e., negative amino acids (glutamates) at the C-terminus of TRPC4 were responsible for spermine block. In specific, E728 and E729 were crucial for spermine to block the channel.
In our previous study, we showed that TRPC4 is a molecular candidate for mIcat in GI smooth muscle cells [18]. After that, Tsvilovskyy et al. showed that TRPC6 is also a molecular candidate for mIcat as well as TRPC4. mIcat has an intrinsic voltage gating starting from below −120 mV and saturating at −40 mV [14, 42] where intracellular GTPγS shifts the G-V curve to the left side and increases mIcat even at the resting membrane potential. Besides to N-shaped conductance curve, another unique electrophysiological feature of TRPC channels is the flat region from 0 mV to +40 mV in I-V relationship. Obukhov et al. showed that D633 and D636 of TRPC5 mediate Mg2+ block related to the flat region of TRPC5 [26]. Mg2+ directly blocks the pathway of ions across the cytoplasmic pores or modulates gating through TRP domain to induce channel closing. In this study, we have found that 1 mM of intracellular spermine extended the flat region to around 80 mV. Recently discovered structure of TRPV1 showed that there is a large space at the cytoplasmic C-terminal region although the TRPV1 crystal did not contain all C-terminal areas [4, 19]. The reason why there is a flat region seems that there is a large space at the C-terminus. With Mg2+ and polyamine, mainly spermine at the concentration of around 300 nM, cation can permeate only at the more depolarized region like more than 50 mV. In our hands, 10 mM Mg2+ did not extend the flat region to more than 50 mV (Supplementary Fig. 4). Considering physiological concentration in normal tissue, spermine seems more important than Mg2+ because 1 mM spermine extended the flat region up to 80 mV.
As Zholos suggested [41], the role of TRPC4 and TRPC5 in electrogenesis is of special interest since it is predominantly expressed in neurons. In this scenario, the N-shaped conductance curve of TRPC4/5 appears to be perfectly shaped to trigger action potential discharge, but not to interfere with the shape and amplitude of the action potential [41]. Thus, when TRPC4/5 opens its conductance peaks at normal membrane resting potential level ensuring efficiency of membrane depolarization towards the action potential threshold, but rapid conductance decline occurs over the region of action potential upstroke [41]. If not for this peculiar internal Mg2+ or spermine channel block, one would envisage an efficient “clamp” of membrane potentials near 0 mV, whereby K+ efflux begins to dominate in NSCC.
The level of intracellular spermine regulates the excitability in neurons, the degree of which depends on the level of the spermine concentration. Snyder-Robinson syndrome occurs due to the alteration in spermine synthase gene (SMS) and has the following symptoms: X-linked intellectual disability syndrome, speech abnormalities, osteoporosis, kyphoscoliosis, and asthenic habitus with diminished muscle mass, ambulatory difficulties, facial dysmorphism, mild short stature, and high incidence of seizures. Recently, CNV of TRPC5 was shown one of the causes related with X-linked ID syndrome [24]. Considering the same location at the X-chromosome of TRPC5 and SMS, both genes can induce X-linked ID syndrome together.
The level of spermine also determines the cell proliferation in cancer cells. Recently, Akbulut et al. showed the unexpected possibility for the rapid and selective killing of renal cancer cells (RCCs) through activation of calcium-permeable nonselective transient receptor potential canonical (TRPC) calcium channels by the sesquiterpene (−)-englerin A [1]. This compound was found to be a highly efficient, fast-acting, potent, selective, and direct stimulator of TRPC4 and TRPC5 channels. Provided, it may be said that TRPC4 and TRPC5 channels are hazardous to some cancer cells. In anticancer medicine, it is generally perceived that cancer cells somewhat evolve to downregulate proteins whose activity is hazardous to proliferation or survival of themselves. Thus, in specific cancer cell, it may seek biological pathways that downregulate TRPC4 and TRPC5 channels. Interestingly, many of cancer cells and hyperproliferative diseases show high concentration of intracellular polyamines [20, 40]. If this high concentration of polyamines are defense mechanism of cancer cells, anti-polyamine agents such as 2-difluoromethylornithine (DFMO)—an ornithine decarboxylase (ODC) inhibitor—can be used as anticancer therapeutics for cancer cells with high TRPC4/5 expression profile, e.g., RCC.
Carbachol, (2-hydroxyethyl)trimethylammonium chloride carbamate; SPM, spermine tetrahydrochloride, N, N′-Bis(3-aminopropyl)-1,4-butanediamine tetrahydrochloride; TRPC, transient receptor potential canonical.
References
Akbulut Y, Gaunt HJ, Muraki K, Ludlow MJ, Amer MS, Bruns A, Vasudev NS, Radtke L, Willot M, Hahn S, Seitz T, Ziegler S, Christmann M, Beech DJ, Waldmann H (2015) Englerin A is a potent and selective activator of TRPC4 and TRPC5 calcium channels. Angew Chem Int Ed Engl 54(12):3787–3791
Beech DJ (2005) TRPC1: store-operated channel and more. Pflugers Arch - Eur J Physiol 451:53–60
Beech DJ (2007) Canonical transient receptor potential 5. Handb Exp Pharmacol 179:109–123
Cao E, Liao M, Cheng Y, Julius D (2013) TRPV1 structures in distinct conformations reveal activation mechanisms. Nature 504(7478):113–118
Casero RA, Marton LJ (2007) Targeting polyamine metabolism and function in cancer and other hyperproliferative diseases. Nat Rev Drug Discov 6:373–390
Fakller B, Brandle U, Glowatzki E, Weidemann S, Zenner HP, Ruppersberg JP (1995) Strong voltage-dependent inward rectification of inward rectifier K+ channels is caused by intracellular spermine. Cell 80:149–154
Ficker E, Taglialatela M, Wible BA, Henley CM, Brown AM (1994) Spermine and spermidine as gating molecules for inward rectifier K+ channels. Science 266:1068–1072
Flockerzi V, Nilius B (2014) TRPs: truly remarkable proteins. Handb Exp Pharmacol 222:1–12
Freichel M, Tsvilovskyy V, Camacho-Londono JE (2014) TRPC4- and TRPC4-containing channels. Handb Exp Phamacol 222:85–128
Huang CW, Kuo CC (2014) The bundle crossing region is responsible for the inwardly rectifying internal spermine block of the Kir2.1 channel. Pflugers Arch - Eur J Physiol 466(2):275–293
Inoue R, Isenberg G (1990) Acetylcholine activates nonselective cation channels in guinea pig ileum through a G protein. Am J Physiol 258:C1173–C1178
Jeon JP, Hong C, Park EJ, Jeon JH, Cho NH, Kim IG, Choe H, Muallem S, Kim HJ, So I (2012) Selective Gαi subunits as novel direct activators of transient receptor potential canonical (TRPC)4 and TRPC5 channels. J Biol Chem 287(21):17029–17039
John RM, Stevenson WG (2015) Ventricular arrhythmias. In. Kasper DL (ed) Harrison's principle of internal medicine, 19th edn. McGraw Hill, New York, pp 1489–1500
Kang TM, Kim YC, Sim JH, Rhee JC, Kim SJ, Uhm DY, So I, Kim KW (2001) The properties of carbachol-activated nonselective cation channels at the single channel level in guinea pig gastric myocytes. Jpn J Pharmacol 85(3):291–298
Kim J, Kwak M, Jeon JP, Myeong J, Wie J, Hong C, Kim SY, Jeon JH, Kim HJ, So I (2014) Isoform- and receptor-specific channel property of canonical transient receptor potential (TRPC)1/4 channels. Pflugers Arch - Eur J Physiol 466:491–504
Kim YC, Kim SJ, Sim JH, Cho CH, Juhnn YS, Suh SH, So I, Kim KW (1998) Suppression of the carbachol-activated nonselective cationic current by antibody against alpha subunit of Go protein in guinea-pig gastric myocytes. Pflugers Arch - Eur J Physiol 436(3):494–496
Kubo Y, Murata Y (2001) Control of rectification and permeation by two distinct sites after the second transmembrane region in Kir2.1 K+ channel. J Physiol 531(3):645–660
Lee KP, Jun JY, Chang IY, Suh SH, So I, Kim KW (2005) TRPC4 is an essential component of the nonselective cation channel activated by muscarinic stimulation in mouse visceral smooth muscle cells. Mol Cell 20(3):435–441
Liao M, Cao E, Julius D, Cheng Y (2013) Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature 504(7478):107–112
Linsalata M, Orlando A, Russo F (2014) Pharmacological and dietary agents for colorectal cancer chemoprevention: effects on polyamine metabolism (review). Int J Oncol 45(5):1802–1812
Lopatin AN, Makhina EN, Nichols CG (1994) Potassium channel block by cytoplasmic polyamines of potassium channels: “long-pore plugging” by cytoplasmic polyamines. J Gen Physiol 106:923–955
Mery L, Magnino F, Schmidt K, Krause KH, Dufour JF (2001) Alternative splice variants of hTrp4 differentially interact with the C-terminal portion of the inositol 1,4,5-triphosphate receptors. FEBS Lett 487(3):377–383
Miehe S, Bieberstein A, Arnould I, Ihdene O, Rutten H, Strubing C (2010) The phospholipid-binding protein SESTD1 is a novel regulator of the transient receptor potential channels TRPC4 and TRPC5. J Biol Chem 285(16):12426–12434
Mignon-Ravix C, Cacciagli P, Choucair N, Popovici C, Missirian C, Milh M, Megarbane A, Busa T, Julia S, Girard N, Badens C, Siguady S, Philip N, Villard L (2014) Intragenic rearrangements in X-linked intellectual deficiency: results of a-CGH in a series of 54 patients and identification of TRPC5 and KLHL15 as potential XLID genes. Am J Med Genet A 164A(8):1991–1997
Nilsson BO, Hellstrand P (1993) Effects of polyamines on intracellular calcium and mechanical activity in smooth muscle of guinea-pig taenia coli. Acta Physiol Scand 148:37–43
Obukhov AG, Nowycky MC (2005) A cytosolic residue mediates Mg2+ block and regulates inward current amplitude of a transient receptor potential channel. J Neurosci 25(5):1234–1239
Odell AF, Van Helden DF, Scott JL (2008) The spectrin cytoskeleton influences the surface expression and activation of human transient receptor potential channel 4 channels. J Biol Chem 283(7):4395–4407
Onodera K, Unemoto T, Miyaki K, Hayashi M (1968) Pharmacological studies on polyamines. I. Relaxing effect of spermine and spermidine on smooth muscle of guinea pig ileum contracted by 5-hydroxytryptamine and nicotine. Arch Int Pharmacodyn Ther 174:491–494
Otsuguro K, Tang J, Tang Y, Xiao R, Freichel M, Tsvilovskyy V, Ito S, Flockerzi V, Zhu MX, Zholos AV (2008) Isoform-specific inhibition of TRPC4 channel by phosphatidylinositol 4,5-bisphoshate. J Biol Chem 283(15):10026–10036
Pegg AE (2014) The function of spermine. IUBMB Life 66(1):8–18
Priori SG, Pandit SV, Rivolta I, Berenfeld O, Ronchetti E, Dhamoon A, Napolitano C, Anumonwo J, Barletta MR, Gudapakkam S, Bosi G, Badiale MS, Jalife J (2005) A novel form of short QT syndrome (SQT3) is caused by a mutation in the KCNJ2 gene. Circulation 96:800–807
Sakamoto T, Unno T, Kitazawa T, Taneike T, Yamada M, Wess J, Nishimura M, Komori S (2007) Three distinct muscarinic signaling pathways for cationic channel activation in mouse gut smooth muscle cells. J Physiol 582:41–61
So I, Ashmole I, Soh H, Park CS, Spencer PJ, Leyland M, Stanfield PR (2003) Intrinsic gating in inward rectifier potassium channels (Kir2.1) with low polyamine affinity generated by site directed mutagenesis. Korean J Physiol Pharmacol 7:131–142
Stanfield PR, Sutcliffe MJ (2003) Spermine is fit to block inward rectifier (Kir) channels. J Gen Physiol 122(5):481–484
Tang J, Lin Y, Zhang Z, Tikunova S, Birnbaumer L, Zhu MX (2001) Identification of common binding sites for calmodulin and inositol 1,4,5-triphosphate receptors on the carboxyl termini of trp channels. J Biol Chem 276(24):21303–21310
Thompson PA, Wertheim BC, Zell JA, Chen WP, McLaren CE, Lafleur BJ, Meyskens FL, Gerner EW (2010) Levels of rectal mucosal polyamines and prostaglandin E2 predict ability of DFMO and sulindac to prevent colorectal adenoma. Gastroenterology 139:797–805
Trost C, Bergs C, Himmerkus N, Flockerzi V (2001) The transient receptor potential, TRP4, cation channel is a novel member of the family of calmodulin binding proteins. Biochem J 335:663–670
Tsvilovskyy VV, Zholos AV, Aberle T, Philipp SE, Dietrich A, Zhu MX, Birnbaumer L, Freichel M, Flockerzi V (2009) Deletion of TRPC4 and TRPC6 in mice imparis smooth muscle contraction and intestinal motility in vivo. Gastroenterology 137:1415–1424
Tsvilovskyy VV, Zholos AV, Bolton TB (2004) Effects of polyamines on the muscarinic receptor-operated cation current in guinea-pig ileal smooth muscle myocytes. Br J Pharmacol 143:968–975
Wang W, Liu LQ, Higuchi CM (1996) Mucosal polyamine measurements and colorectal cancer risk. J Cell Biochem 63(2):252–257
Zholos AV (2014) TRPC5. Handb Exp Phamacol 222:129–156
Zholos AV, Zholos AA, Bolton TB (2004) G-protein-gated TRP-like cationic channel activated by muscarinic receptors: effect of potential on single-channel gating. J Gen Physiol 123(5):581–598
Zhu MX (2005) Multiple roles of calmodulin and other Ca2 + −binding proteins in the functional regulation of TRP channels. Pflugers Arch - Eur J Physiol 451:105–115
Acknowledgments
We thank Dr. Michael Mederos and Dr. Thomas Gudermann for kindly donating human TRPC1 isoform long (hTRPC1α) construct. TRPC5 were kindly donated by Dr S. Kaneko and Dr Y. Mori. TRPC4 were kindly donated by Dr V. Flockerzi and Dr. M. Schaefer.
This study was supported by grants from the National Research Foundation of Korea, which is funded by the Ministry of Science, ICT (Information & Communication Technology) and Future Planning (MSIP) of the Korean Government (MSIP) (2013R1A1A1010783) (K.P. Lee), (2012R1A2A1A01003073) (I. So). Y. S. was supported by the BK21 plus program from the MSIP. This work was supported by the Education and Research Encouragement Fund of Seoul National University Hospital (I. So).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Jinsung Kim and Sang Hui Moon contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
Effect of intracellular spermidine and putrescine to TRPC4 channel. (a), (b) all left panels indicate current traces and right panels indicate corresponding I-V curves at noted points (1, 2 and 3). 1 mM of spermidine and putrescine could not inhibit TRPC4 channel as strong as spermine. There was no asymmetry between outward current and inward current as was in spermine. Both polyamines, however, somewhat slowed down the activation process. (PPTX 202 kb)
Supplementary Figure 2
Intracellular spermine inhibit TRPC3 with nonspecific manner. (a) Normalized current density of TRPC3 channels. White circle indicates currents without spermine and black circle indicates current with intracellular spermine. Two currents showed indifferent I-V shape. (b) Current density of TRPC3 current with and without intracellular spermine. Intracellular spermine reduced both inward and outward current density of TRPC3 channels. (c) Current trace for TRPC3 recording. Muscarinic acetylcholine receptor 3 and extracellular carbachol (CCh, 100 μM) was used for activation of TRPC3 channels. In addition, divalent-free solution (DVF) was used in the activation process. (PPTX 106 kb)
Supplementary Figure 3
Distribution of negative amino acids in C-terminus of TRPC4 channel. (a) Amino acid sequence of C-terminus of TRPC4 channel. Red capitals indicate negative amino acids (D and E), green capitals indicate TRP box sequence (EWKFAR) and gray capitals indicate alternative splicing site for TRPC4β. (b) Left panel, From 621st amino acid to 890th amino acid in TRPC4β, negative amino acids showed sporadic distribution when window width was set to 20 amino acids. When 740th amino acid was set as cut-off number, however, distribution of negative amino acids showed clear stratification. (PPTX 65 kb)
Supplementary Figure 4
Effect of high magnesium ([Mg2+]i = 10 mM) on TRPC4 channels. (a) I-V curves for whole-cell TRPC4 current with 10 mM of intracellular Mg2+. Augmented intracellular Mg2+ could not alter the shape of I-V curves. (b) Representative current race of TRPC4 current under 10 mM intracellular Mg2+. Activation kinetics were similar to physiological condition ([Mg2+]i ∼ 3 mM). (PPTX 390 kb)
Rights and permissions
About this article
Cite this article
Kim, J., Moon, S.H., Shin, YC. et al. Intracellular spermine blocks TRPC4 channel via electrostatic interaction with C-terminal negative amino acids. Pflugers Arch - Eur J Physiol 468, 551–561 (2016). https://doi.org/10.1007/s00424-015-1753-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-015-1753-x