Abstract
Purpose
This study investigated the role of sarcopenia in the long-term outcomes of patients with early-stage intrahepatic recurrent hepatocellular carcinoma (HCC).
Methods
The study included 136 patients with intrahepatic recurrent Barcelona Clinic Liver Cancer (BCLC) stage 0/A HCC following liver resection diagnosed between 2006 and 2020 and underwent surgery, radiofrequency ablation (RFA), or transcatheter arterial chemoembolization (TACE). Sarcopenia was defined based on the skeletal muscle index using computed tomography at the time of recurrence, and its association with long-term outcomes was evaluated. Tumor-infiltrating lymphocytes (CD4 + , CD8 + , and CD45RO + T cells) were assayed using immunohistochemistry on specimens obtained from repeat hepatectomies, and their association with sarcopenia was evaluated.
Results
The overall survival (OS) and recurrence-free survival (RFS) rates after initial recurrence of patients with sarcopenia were significantly lower than those without sarcopenia (p < 0.001 and p < 0.001, respectively). Multivariate analysis identified sarcopenia as an independent prognostic factor for RFS (p < 0.001). In patients without sarcopenia, surgery resulted in better RFS than RFA or TACE. Contrastingly, in patients with sarcopenia, the RFS was extremely poor regardless of the treatment type: surgery, RFA, or TACE (median RFS, 11.7, 12.7, and 10.1 months). Significantly low levels of tumor-infiltrating CD4 + , CD8 + , and CD45RO + lymphocytes were observed in patients with sarcopenia (p = 0.001, p = 0.001, and p = 0.001, respectively).
Conclusions
This study suggests that patients with sarcopenia have poor RFS regardless of the treatment type for early-stage intrahepatic recurrent HCC. Impaired host immunity might be one of the underlying mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sarcopenia is defined as the progressive loss of skeletal muscle mass, and is associated with survival outcomes and recurrence in patients with various cancers [1,2,3]. In hepatocellular carcinoma (HCC), sarcopenia occurs early and is an independent predictor of mortality [4]. Moreover, previous studies have reported that sarcopenia is associated with prognosis and recurrence in patients with HCC following various treatments, such as liver resection, radiofrequency ablation (RFA), and transcatheter arterial chemoembolization (TACE) [5,6,7,8]. However, no study has evaluated the role of sarcopenia on long-term outcomes in patients with intrahepatic recurrent HCC.
HCC is one of the most common malignancies and a leading cause of cancer-related mortality worldwide [9, 10]. Liver resection is the primary curative treatment modality for patients with HCC. However, the risk of recurrence following resection is up to 70% at 5 years [11, 12], and intrahepatic recurrence accounts for more than 90% [13, 14]. There are various treatment options for intrahepatic recurrence, including repeat liver resection, RFA, TACE, and liver transplantation [15,16,17,18]. A prospective cohort study on early-stage intrahepatic recurrent HCC showed that the patients who underwent repeat hepatectomy had better survival outcome [19]; however, the appropriate treatment is still controversial. Owing to the high recurrence rate of HCC, treatment after recurrence is very important. Thus, the identification of an indicator for determining the appropriate treatment is essential to improve the prognosis of patients with HCC.
In the local immune microenvironment, tumor-infiltrating lymphocytes (TILs) were earlier thought to host immune responses to tumors [20]. TILs play a significant role in the development, treatment, and prognosis of HCC [20,21,22]. In particular, CD4 + , CD8 + , and CD45RO + T cells have been reported to be lowered in the tumor tissue and linked with poor prognosis in HCC. CD8 + T lymphocyte is the most prevalent T cell subset, and CD4 + T lymphocyte promotes the immune response against tumor by secreting cytokines and activating CD8 + T cells. In addition, CD45RO + T lymphocyte is related to anti-tumor responses as a memory T cell [23,24,25,26]. Recently, several studies reported that sarcopenia may be associated with TILs and affect prognosis in cancer patients [27, 28]. However, no study has evaluated the association between sarcopenia and TILs in intrahepatic recurrent HCC.
Therefore, in this study, we aimed to investigate the impact of sarcopenia on long-term outcomes and its association with host immunity in patients with early-stage intrahepatic recurrent HCC categorized based on the treatment type.
Material and methods
Patients and their clinical data
The study included consecutive patients with initial recurrent HCC following liver resection treated at Nara Medical University Hospital between January 2006 and December 2020. Initial recurrent HCC patients were defined as the patients who occurred initial recurrence after resection for initial HCC. The inclusion criteria of this study were the patients with intrahepatic recurrent HCC within Barcelona Clinic Liver Cancer (BCLC) stage 0/A (single or ≤ 3 nodules ≤ 3 cm). The patients with intrahepatic recurrent HCC beyond BCLC stage 0/A were excluded because they were not indicated surgery or RFA. The study protocol was approved by the local ethics committee of Nara Medical University (no. 3474), and the study was conducted in accordance with the Declaration of Helsinki. This retrospective study waived the requirement for individual informed consent owing to the “opt-out” principle. Patients were allowed to “optout” of the database if they wished.
Baseline patient characteristics, including recurrence and initial resection status, were obtained from their medical records. The albumin-bilirubin (ALBI) score was calculated as [− 0.085 × albumin (g/dl)] + [0.66 × log10 total bilirubin (mg/dl)]. Furthermore, the ALBI score was classified into three grades: grade 1: ≤ − 2.6; grade 2: > − 2.6, ≤ − 1.39; and grade 3: > − 1.39. The neutrophil-to-lymphocyte ratio (NLR) was calculated using neutrophil count (/μL) divided by lymphocyte count (/μL), and the platelet-to-lymphocyte ratio (PLR) was measured as platelet count (/μL) divided by lymphocyte count (/μL). Prognostic nutritional index (PNI) was calculated as [10 × serum albumin concentration (g/dl) + 0.005 × lymphocyte count (/μL)].
Measurement of skeletal muscle area
Computed tomography at the time of initial recurrence was used to assess the patients’ total cross-sectional transverse areas of the skeletal muscles at the third lumbar vertebra. Images were analyzed using the volume analyzer SYNAPSE VINCENT software (Fujifilm Company, Tokyo, Japan). A threshold range of − 29 to + 150 Hounsfield units was used to define the muscle mass. The total muscle area (cm2) was normalized to height in square meters (m2) and reported as the skeletal muscle index (SMI). The cutoff values of SMI for sarcopenia were set at < 42 cm2/m2 for men and < 38 cm2/m2 for women from the Working Group for the Creation of Sarcopenia Assessment Criteria of the Japan Society of Hepatology [29].
Immunohistochemistry and evaluation
Immunohistochemistry was performed on specimens obtained from repeat hepatectomies for intrahepatic recurrent HCC. A 4-μm section was prepared from the representative formalin-fixed paraffin-embedded tissue slices. The sections were deparaffinized in xylene substitute (Clear Plus®, FALMA, Japan) and rehydrated in a graded series of ethanol. For antigen retrieval, the sections were heated at 121 °C for 20 min in the Target Retrieval Solution, pH9 (Dako Cytomation, Denmark). After blocking the endogenous peroxidases with 3% hydrogen peroxide, the sections were incubated with 2.5% normal horse serum (Vector Laboratories, USA) at room temperature for 60 min to prevent non-specific reactions. The primary antibodies used were monoclonal mouse anti-human CD4 (4B12, Dako Cytomation, 1:40), CD8 (C8/144B, Dako Cytomation, 1:100), and CD45RO (55618S, Cell Signaling Technology, USA, 1:400) which were added to the sections. Incubation with primary antibodies was performed for 120 min. The sections were then incubated with anti-mouse/rabbit IgG (MP-7500, Vector Laboratories) for 30 min. Next, the sections were subjected to 3,3-diaminobenzidine solution (SK-4105, Vector Laboratories). After counterstaining with hematoxylin, dehydration in a series of ethanol solutions, and clearing in xylene, the sections were embedded in a mounting material. The sections were washed in phosphate-buffered saline as needed between each step.
Immunohistochemistry for CD4 + , CD8 + , and CD45RO + T cells was performed under 200 × magnification by counting the TILs in five different areas with the most abundant TILs for each target molecule.
Statistical analyses
Continuous variables were expressed as medians with ranges, and compared using the Mann–Whitney U test. Categorical variables are presented as numbers and percentages, and compared using the chi-square test or Fisher’s exact test. Overall survival (OS) was defined as the period from the date of the initial recurrence to the date of death or final follow-up. Recurrence-free survival (RFS) was defined as the duration from the date of the initial recurrence to the date of the second recurrence, death, or final follow-up. Survival curves were generated using the Kaplan–Meier method, and the differences between the curves were analyzed using the log-rank test. Cox regression analysis was used to determine the hazard ratios (HRs). Variables with a p value < 0.05 on univariate analysis and each treatment type (surgery, RFA, and TACE) were further assessed using multivariate analysis. ROC curve analysis was performed to determine cut-off values for continuous variables, including NLR, PLR, and PNI at the time of recurrence. Optimal cut-off values were determined using the Youden Index [30]. P values of < 0.05 were considered statistically significant, and 95% confidence intervals (CIs) were calculated. All statistical analyses were performed using SPSS (version 22.0; SPSS, Chicago, IL, USA).
Results
Baseline characteristics of the patients in sarcopenia and non-sarcopenia groups
Figure 1 shows the flow diagram of the present study. During the study period, 238 consecutive patients were diagnosed with initial recurrent HCC at the Nara Medical University Hospital. Among them, 51 patients with extrahepatic recurrent HCC, 44 with more than four nodules, and seven with two or three nodules > 3 cm were excluded. Finally, 136 patients with intrahepatic recurrent HCC within BCLC stage 0/A were included in the study. For intrahepatic recurrence, 55 (40.4%), 27 (19.9%), and 54 (39.7%) patients underwent surgery, RFA, or TACE, respectively. No patients underwent liver transplantation and immunotherapy during the study period. Treatment selection was based on the surgeon’s judgment and the patient’s condition.
Based on the criteria of the Japan Society of Hepatology [29], study participants were classified into sarcopenia and non-sarcopenia groups. Among the included patients, 37 (27.2%) had sarcopenia. Table 1 shows the baseline characteristics of the patients with and without sarcopenia. Patients with sarcopenia had a significantly higher age (p = 0.034), lower PNI (p = 0.031), and a higher rate of early recurrence within 1 year of initial resection (p < 0.001). Initial resection status, including immunonutrition and tumor status, was not significantly different between the two groups.
Relationship between sarcopenia and long-term outcomes
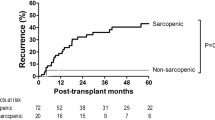
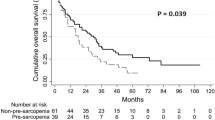
The OS and RFS rates after initial recurrence of patients in the sarcopenia group were significantly lower than those in the non-sarcopenia group (p < 0.001 and p < 0.001, respectively) (Fig. 2A, B). Table 2 summarizes the univariate and multivariate analyses of RFS. Univariate analysis showed that the presence of sarcopenia (p < 0.001), having undergone surgery (p < 0.001) or TACE (p < 0.001), an ALBI score ≥ 2 (p = 0.006), PNI < 52 (p = 0.049), two or three numbers of tumors (p < 0.001), and early recurrence within 1 year of initial resection (p < 0.001) were significantly associated with RFS. Multivariate analysis showed that the presence of sarcopenia, as well as having undergone surgery or TACE, and the presence of multiple tumors, was a strong independent prognostic factor for RFS (HR = 3.380, p < 0.001). Univariate analysis showed that the presence of sarcopenia (p < 0.001) was significantly associated with OS; however, multivariate analysis of OS did not show that sarcopenia was an independent prognostic factor (data not shown).
Survival rates based on the treatment type in patients with or without sarcopenia
In patients in the non-sarcopenia group, surgery resulted in better RFS than RFA or TACE. In contrast, patients with sarcopenia had extremely poor RFS, regardless of treatment options such as surgery, RFA, or TACE. The median RFS after surgery, RFA, and TACE were only 11.7, 12.7, and 10.1 months, respectively (Fig. 3A–C). The OS rates were significantly lower in patients with sarcopenia in the surgery and RFA groups (p = 0.003 and p = 0.002, respectively), but not in the TACE group (Figures S1A-S1C). The baseline characteristics of the patients in the non-sarcopenia and sarcopenia groups categorized based on the treatment type are summarized in Tables S1 and S2, respectively. Among both groups, patients who underwent TACE were significantly more likely to have multiple tumors and worse ALBI scores.
Relationship between sarcopenia and TILs
Finally, to understand the mechanisms underlying poor RFS in patients with sarcopenia, we investigated local immunity using immunohistochemical analysis of TILs in specimens obtained from repeat hepatectomies. Significantly decreased levels of tumor-infiltrating CD4 + , CD8 + , and CD45RO + lymphocytes were observed in patients in the sarcopenia group when compared to the non-sarcopenia group (p = 0.001, p = 0.001, and p = 0.001, respectively) (Fig. 4). In addition, RFS rates were compared between the two groups divided according to the median values of CD4 + , CD8 + , and CD45RO + . RFS was significantly lower in the CD4 + , CD8 + , and CD45RO + low groups than in the high groups (p < 0.001, p = 0.018, and p = 0.015, respectively) (Figures S2A-S2C). These findings suggested that local immune response prevents recurrence.
Expression of CD4, CD8, and CD45RO on tumor-infiltrating lymphocytes. A, B, C CD4, CD8, and CD45RO in the non-sarcopenia group; D, E, F CD4, CD8, and CD45RO in the sarcopenia group; G, H, I Comparison of infiltration of CD4, CD8, and CD45RO positive cells in the non-sarcopenia and sarcopenia groups. *Statistically significant difference at p < 0.05
Discussion
This study investigated the role of sarcopenia on the long-term outcomes in patients with early-stage intrahepatic recurrent HCC who were classified based on the treatment received. To the best of our knowledge, this is the first study to evaluate the clinical impact of sarcopenia on early-stage intrahepatic recurrent HCC. Although there are diverse treatment strategies for intrahepatic recurrence of HCC, many studies reported that repeat hepatectomy achieved better long-term outcomes [12, 18]. Wang et al. indicated that repeat hepatectomy had better survival outcomes even in early-stage intrahepatic recurrent HCC within the Milan criteria and reported the following prognostic factors: more invasive primary tumors, underlying cirrhosis, recurrent tumor number, time to recurrence, and treatment for recurrence [19]. However, the evaluation of the patient’s nutritional and functional statuses was not performed. Sarcopenia is characterized by the loss of muscle strength, mass, and functional ability [1]. Recently, sarcopenia has been reported to be associated with prognosis and recurrence in primary HCC [4,5,6,7,8]; however, its association with recurrent HCC remains unclear. Interestingly, the present study demonstrated that sarcopenia was a strong independent prognostic factor for RFS; furthermore, patients with sarcopenia had poor RFS, regardless of treatment type. In particular, the RFS of patients with sarcopenia who underwent surgery was as poor as those who underwent TACE, although those in the surgery group were likely to have better baseline characteristics, including better liver function and a single tumor. Regarding OS, in this study, patients with sarcopenia had a significantly poorer prognosis; however, sarcopenia was not an independent prognostic factor in multivariate analysis. The reason may be due to our study population. We included only patients with early-stage intrahepatic recurrence, and the number of patients was relatively small. In addition, aggressive therapeutic intervention after further recurrence may have mitigated the obvious differences in the OS. However, our data suggests that patients with sarcopenia are at extremely high risk of recurrence; therefore, the indication for treatment should be considered carefully.
Moreover, to clarify the mechanism underlying poor outcomes in patients with sarcopenia, we evaluated host immunity. Previous research on the relationship between sarcopenia and host immunity suggested that impaired activity of anti-inflammatory immune responses increases the secretion of inflammatory factors such as IL-6, which promote the development of sarcopenia [31,32,33]. Huang et al. reported that SMI was negatively correlated with CD4 + CD28null T lymphocytes in patients with sarcopenia [34]. Additionally, nutrition and exercise improved the level of CD8 + lymphocytes in mice [35, 36]. Furthermore, sarcopenia might be associated with TILs and affect prognosis in cancer patients [27, 28]. The present study showed that TIL expression was significantly decreased in patients with sarcopenia than in those without sarcopenia and that the TIL-low group had worse RFS than the TIL-high group. Moreover, we also showed that patients with sarcopenia had a significantly lower PNI and a higher rate of early recurrence within 1 year of the initial resection. Since PNI is an established indicator of systemic nutritional and immune status [37], sarcopenia might be associated with impaired local and systemic immune systems and early recurrence of HCC. Additionally, cancer immunotherapies are currently being evaluated [38]. Immune checkpoint inhibitors reverse T cell suppression; therefore, they might effectively improve antitumor immunity and prevent early recurrence in patients with sarcopenia. On the other hand, somatic mutation–derived neoantigens have also been reported to be involved in TILS and induce strong immune responses [39, 40]. However, we focused on host immunity; therefore, tumor-specific factors were not evaluated. In addition, no patients have received immunotherapy for HCC in this study. Thus, further studies, including basic research, are warranted to clarify the comprehensive mechanisms.
Given the poor RFS of patients with sarcopenia, improvement in their nutritional status is of great importance. In particular, malnutrition in patients with advanced liver disease is a significant problem. Viral hepatitis might contribute to the progression of sarcopenia, but the interaction has not been clarified [41]. In our data, viral hepatitis was not significantly associated with sarcopenia and prognosis. Previously, studies reported that nutritional interventions could improve sarcopenia and long-term survival in patients with HCC [42, 43]. In particular, a multidisciplinary approach combining nutritional supplementation with physical exercise has been attempted in patients with HCC. A recent study reported that cancer rehabilitation not only increased muscle mass but also prolonged long-term outcomes in patients with HCC who underwent TACE [44]. However, the impact of these nutritional interventions on the long-term outcomes of patients with HCC remains unclear and warrants further large-scale trials. Nevertheless, SMI might be useful to screen patients who require nutritional intervention and evaluate the effects of nutritional intervention.
This study had some limitations. First, it was a retrospective study conducted at a single center with a relatively small population. Second, TILs were evaluated only in specimens obtained from repeat hepatectomies. Thirdly, the mechanisms underlying the association between sarcopenia and TILs remain unclear. Lastly, the tumor-specific factors associated with TILs, such as the number of somatic mutations and neoantigens, were not evaluated. Therefore, further investigations, including in vivo studies, are warranted. Despite these limitations, this study provides useful information regarding the relationship between sarcopenia, recurrence, and TILs in patients with early-stage intrahepatic recurrent HCC.
Conclusion
This study suggests that patients with sarcopenia have poor RFS regardless of the treatment type in early-stage intrahepatic recurrent HCC. Impaired host immunity might be one of the underlying mechanisms.
Data availability
The datasets analysed during the current study are available from the corresponding author on reasonable request.
References
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinková E, Vandewoude M, Zamboni M (2010) Sarcopenia: European consensus on definition and diagnosis: Report of the European working group on sarcopenia in older people. Age Ageing 39(4):412–423. https://doi.org/10.1093/ageing/afq034
Kamitani N, Migita K, Matsumoto S, Wakatsuki K, Kunishige T, Nakade H, Miyao S, Sho M (2019) Association of skeletal muscle loss with the long-term outcomes of esophageal cancer patients treated with neoadjuvant chemotherapy. Surg Today 49(12):1022–1028. https://doi.org/10.1007/s00595-019-01846-1
Pamoukdjian F, Bouillet T, Lévy V, Soussan M, Zelek L, Paillaud E (2018) Prevalence and predictive value of pre-therapeutic sarcopenia in cancer patients: a systematic review. Clin Nutr 37(4):1101–1113. https://doi.org/10.1016/j.clnu.2017.07.010
Perisetti A, Goyal H, Yendala R, Chandan S, Tharian B, Thandassery RB (2022) Sarcopenia in hepatocellular carcinoma: current knowledge and future directions. World J Gastroenterol 28(4):432–448. https://doi.org/10.3748/wjg.v28.i4.432
Yang J, Chen K, Zheng C, Chen K, Lin J, Meng Q, Chen Z, Deng L, Yu H, Deng T, Bo Z, He Q, Wang Y, Chen G (2022) Impact of sarcopenia on outcomes of patients undergoing liver resection for hepatocellular carcinoma. J Cachexia Sarcopenia Muscle 13(5):2383–2392. https://doi.org/10.1002/jcsm.13040
Zhang H, Zhang L, Wang Y, Zou J, Zhang T (2022) Sarcopenia predicts prognosis of patients undergoing liver resection for hepatocellular carcinoma: a systematic review and meta-analysis. Nutr Cancer 1–9. https://doi.org/10.1080/01635581.2022.2159046
Fujiwara N, Nakagawa H, Kudo Y, Tateishi R, Taguri M, Watadani T, Nakagomi R, Kondo M, Nakatsuka T, Minami T, Sato M, Uchino K, Enooku K, Kondo Y, Asaoka Y, Tanaka Y, Ohtomo K, Shiina S, Koike K (2015) Sarcopenia, intramuscular fat deposition, and visceral adiposity independently predict the outcomes of hepatocellular carcinoma. J Hepatol 63(1):131–140. https://doi.org/10.1016/j.jhep.2015.02.031
Fujita M, Takahashi A, Hayashi M, Okai K, Abe K, Ohira H (2019) Skeletal muscle volume loss during transarterial chemoembolization predicts poor prognosis in patients with hepatocellular carcinoma. Hepatol Res 49(7):778–786. https://doi.org/10.1111/hepr.13331
Vogel A, Meyer T, Sapisochin G, Salem R, Saborowski A (2022) Hepatocellular carcinoma. Lancet 400(10360):1345–1362. https://doi.org/10.1016/s0140-6736(22)01200-4
(2018) EASL Clinical practice guidelines: Management of hepatocellular carcinoma. J Hepatol 69(1):182–236. https://doi.org/10.1016/j.jhep.2018.03.019
Marrero JA, Kulik LM, Sirlin CB, Zhu AX, Finn RS, Abecassis MM, Roberts LR, Heimbach JK (2018) Diagnosis, staging, and management of hepatocellular carcinoma: 2018 practice guidance by the American Association for the Study of Liver Diseases. Hepatology 68(2):723–750. https://doi.org/10.1002/hep.29913
Yoh T, Seo S, Taura K, Iguchi K, Ogiso S, Fukumitsu K, Ishii T, Kaido T, Uemoto S (2021) Surgery for recurrent hepatocellular carcinoma: achieving long-term survival. Ann Surg 273(4):792–799. https://doi.org/10.1097/sla.0000000000003358
Tsilimigras DI, Bagante F, Moris D, Hyer JM, Sahara K, Paredes AZ, Mehta R, Ratti F, Marques HP, Soubrane O, Lam V, Poultsides GA, Popescu I, Alexandrescu S, Martel G, Workneh A, Guglielmi A, Hugh T, Aldrighetti L, Endo I, Pawlik TM (2020) Recurrence patterns and outcomes after resection of hepatocellular carcinoma within and beyond the Barcelona Clinic Liver Cancer Criteria. Ann Surg Oncol 27(7):2321–2331. https://doi.org/10.1245/s10434-020-08452-3
Xu XF, Xing H, Han J, Li ZL, Lau WY, Zhou YH, Gu WM, Wang H, Chen TH, Zeng YY, Li C, Wu MC, Shen F, Yang T (2019) Risk factors, patterns, and outcomes of late recurrence after liver resection for hepatocellular carcinoma: a multicenter study from China. JAMA Surg 154(3):209–217. https://doi.org/10.1001/jamasurg.2018.4334
Tabrizian P, Jibara G, Shrager B, Schwartz M, Roayaie S (2015) Recurrence of hepatocellular cancer after resection: patterns, treatments, and prognosis. Ann Surg 261(5):947–955. https://doi.org/10.1097/sla.0000000000000710
Zheng J, Cai J, Tao L, Kirih MA, Shen Z, Xu J, Liang X (2020) Comparison on the efficacy and prognosis of different strategies for intrahepatic recurrent hepatocellular carcinoma: a systematic review and Bayesian network meta-analysis. Int J Surg 83:196–204. https://doi.org/10.1016/j.ijsu.2020.09.031
Erridge S, Pucher PH, Markar SR, Malietzis G, Athanasiou T, Darzi A, Sodergren MH, Jiao LR (2017) Meta-analysis of determinants of survival following treatment of recurrent hepatocellular carcinoma. Br J Surg 104(11):1433–1442. https://doi.org/10.1002/bjs.10597
Zou Q, Li J, Wu D, Yan Z, Wan X, Wang K, Shi L, Lau WY, Wu M, Shen F (2016) Nomograms for pre-operative and post-operative prediction of long-term survival of patients who underwent repeat hepatectomy for recurrent hepatocellular carcinoma. Ann Surg Oncol 23(8):2618–2626. https://doi.org/10.1245/s10434-016-5136-0
Wang K, Liu G, Li J, Yan Z, Xia Y, Wan X, Ji Y, Lau WY, Wu M, Shen F (2015) Early intrahepatic recurrence of hepatocellular carcinoma after hepatectomy treated with re-hepatectomy, ablation or chemoembolization: a prospective cohort study. Eur J Surg Oncol 41(2):236–242. https://doi.org/10.1016/j.ejso.2014.11.002
Zheng X, Jin W, Wang S, Ding H (2021) Progression on the roles and mechanisms of tumor-infiltrating T lymphocytes in patients with hepatocellular carcinoma. Front Immunol 12:729705. https://doi.org/10.3389/fimmu.2021.729705
Shirabe K, Motomura T, Muto J, Toshima T, Matono R, Mano Y, Takeishi K, Ijichi H, Harada N, Uchiyama H, Yoshizumi T, Taketomi A, Maehara Y (2010) Tumor-infiltrating lymphocytes and hepatocellular carcinoma: pathology and clinical management. Int J Clin Oncol 15(6):552–558. https://doi.org/10.1007/s10147-010-0131-0
Hernandez-Gea V, Toffanin S, Friedman SL, Llovet JM (2013) Role of the microenvironment in the pathogenesis and treatment of hepatocellular carcinoma. Gastroenterology 144(3):512–527. https://doi.org/10.1053/j.gastro.2013.01.002
Brunner SM, Rubner C, Kesselring R, Martin M, Griesshammer E, Ruemmele P, Stempfl T, Teufel A, Schlitt HJ, Fichtner-Feigl S (2015) Tumor-infiltrating, interleukin-33-producing effector-memory CD8(+) T cells in resected hepatocellular carcinoma prolong patient survival. Hepatology 61(6):1957–1967. https://doi.org/10.1002/hep.27728
Fu J, Zhang Z, Zhou L, Qi Z, Xing S, Lv J, Shi J, Fu B, Liu Z, Zhang JY, Jin L, Zhao Y, Lau GK, Zhao J, Wang FS (2013) Impairment of CD4+ cytotoxic T cells predicts poor survival and high recurrence rates in patients with hepatocellular carcinoma. Hepatology 58(1):139–149. https://doi.org/10.1002/hep.26054
Sideras K, Biermann K, Verheij J, Takkenberg BR, Mancham S, Hansen BE, Schutz HM, de Man RA, Sprengers D, Buschow SI, Verseput MC, Boor PP, Pan Q, van Gulik TM, Terkivatan T, Ijzermans JN, Beuers UH, Sleijfer S, Bruno MJ, Kwekkeboom J (2017) PD-L1, Galectin-9 and CD8(+) tumor-infiltrating lymphocytes are associated with survival in hepatocellular carcinoma. Oncoimmunology 6(2):e1273309. https://doi.org/10.1080/2162402x.2016.1273309
Gao Q, Zhou J, Wang XY, Qiu SJ, Song K, Huang XW, Sun J, Shi YH, Li BZ, Xiao YS, Fan J (2012) Infiltrating memory/senescent T cell ratio predicts extrahepatic metastasis of hepatocellular carcinoma. Ann Surg Oncol 19(2):455–466. https://doi.org/10.1245/s10434-011-1864-3
Daitoku N, Miyamoto Y, Hiyoshi Y, Tokunaga R, Sakamoto Y, Sawayama H, Ishimoto T, Baba Y, Yoshida N, Baba H (2022) Preoperative skeletal muscle status is associated with tumor-infiltrating lymphocytes and prognosis in patients with colorectal cancer. Ann Gastroenterol Surg 6(5):658–666. https://doi.org/10.1002/ags3.12570
Kitano Y, Yamashita YI, Saito Y, Nakagawa S, Okabe H, Imai K, Komohara Y, Miyamoto Y, Chikamoto A, Ishiko T, Baba H (2019) Sarcopenia affects systemic and local immune system and impacts postoperative outcome in patients with extrahepatic cholangiocarcinoma. World J Surg 43(9):2271–2280. https://doi.org/10.1007/s00268-019-05013-y
Nishikawa H, Shiraki M, Hiramatsu A, Moriya K, Hino K, Nishiguchi S (2016) Japan Society of Hepatology guidelines for sarcopenia in liver disease (1st edition): recommendation from the working group for creation of sarcopenia assessment criteria. Hepatol Res 46(10):951–963. https://doi.org/10.1111/hepr.12774
Youden WJ (1950) Index for rating diagnostic tests. Cancer 3(1):32–35. https://doi.org/10.1002/1097-0142(1950)3:1%3c32::aid-cncr2820030106%3e3.0.co;2-3
Zhang X, Li H, He M, Wang J, Wu Y, Li Y (2022) Immune system and sarcopenia: presented relationship and future perspective. Exp Gerontol 164:111823. https://doi.org/10.1016/j.exger.2022.111823
Nelke C, Dziewas R, Minnerup J, Meuth SG, Ruck T (2019) Skeletal muscle as potential central link between sarcopenia and immune senescence. EBioMedicine 49:381–388. https://doi.org/10.1016/j.ebiom.2019.10.034
Tuttle CSL, Thang LAN, Maier AB (2020) Markers of inflammation and their association with muscle strength and mass: a systematic review and meta-analysis. Ageing Res Rev 64:101185. https://doi.org/10.1016/j.arr.2020.101185
Huang SW, Xu T, Zhang CT, Zhou HL (2020) Relationship of peripheral lymphocyte subsets and skeletal muscle mass index in sarcopenia: a cross-sectional study. J Nutr Health Aging 24(3):325–329. https://doi.org/10.1007/s12603-020-1329-0
Chatraw JH, Wherry EJ, Ahmed R, Kapasi ZF (2008) Diminished primary CD8 T cell response to viral infection during protein energy malnutrition in mice is due to changes in microenvironment and low numbers of viral-specific CD8 T cell precursors. J Nutr 138(4):806–812. https://doi.org/10.1093/jn/138.4.806
Rundqvist H, Veliça P, Barbieri L, Gameiro PA, Bargiela D, Gojkovic M, Mijwel S, Reitzner SM, Wulliman D, Ahlstedt E, Ule J, Östman A, Johnson RS (2020) Cytotoxic T-cells mediate exercise-induced reductions in tumor growth. Elife 9. https://doi.org/10.7554/eLife.59996
Wang D, Hu X, Xiao L, Long G, Yao L, Wang Z, Zhou L (2021) Prognostic nutritional index and systemic immune-inflammation index predict the prognosis of patients with HCC. J Gastrointest Surg 25(2):421–427. https://doi.org/10.1007/s11605-019-04492-7
Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, Kudo M, Breder V, Merle P, Kaseb AO, Li D, Verret W, Xu DZ, Hernandez S, Liu J, Huang C, Mulla S, Wang Y, Lim HY, Zhu AX, Cheng AL (2020) Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med 382(20):1894–1905. https://doi.org/10.1056/NEJMoa1915745
Schumacher TN, Schreiber RD (2015) Neoantigens in cancer immunotherapy. Science 348(6230):69–74. https://doi.org/10.1126/science.aaa4971
Schumacher TN, Scheper W, Kvistborg P (2019) Cancer neoantigens. Annu Rev Immunol 37:173–200. https://doi.org/10.1146/annurev-immunol-042617-053402
Coelho MPP, de Castro P, de Vries TP, Colosimo EA, Bezerra JMT, Rocha GA, Silva LD (2023) Sarcopenia in chronic viral hepatitis: from concept to clinical relevance. World J Hepatol 15(5):649–665. https://doi.org/10.4254/wjh.v15.i5.649
Ruiz-Margáin A, Román-Calleja BM, Moreno-Guillén P, González-Regueiro JA, Kúsulas-Delint D, Campos-Murguía A, Flores-García NC, Macías-Rodríguez RU (2021) Nutritional therapy for hepatocellular carcinoma. World J Gastrointest Oncol 13(10):1440–1452. https://doi.org/10.4251/wjgo.v13.i10.1440
Takami T, Yamasaki T, Saeki I, Matsumoto T, Suehiro Y, Sakaida I (2016) Supportive therapies for prevention of hepatocellular carcinoma recurrence and preservation of liver function. World J Gastroenterol 22(32):7252–7263. https://doi.org/10.3748/wjg.v22.i32.7252
Hashida R, Kawaguchi T, Koya S, Hirota K, Goshima N, Yoshiyama T, Otsuka T, Bekki M, Iwanaga S, Nakano D, Niizeki T, Matsuse H, Kawaguchi A, Shiba N, Torimura T (2020) Impact of cancer rehabilitation on the prognosis of patients with hepatocellular carcinoma. Oncol Lett 19(3):2355–2367. https://doi.org/10.3892/ol.2020.11345
Author information
Authors and Affiliations
Contributions
Study conception and design: SD, SY. Acquisition of data: YM, TS, SN, MN, KN, TT, YK, SN. Analysis and interpretation of data: SD, SY. Drafting of manuscript: SD. Critical revision of manuscript: SY, MS.
Corresponding author
Ethics declarations
Ethical approval
This retrospective study was approved by the Institutional Review Board of Nara Medical University (No. 3474).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
423_2023_3170_MOESM1_ESM.tiff
Supplementary file1. Figure S1 Overall survival of the patients based on the treatment type. (A) Surgery group (p = 0.003); (B) RFA group (p = 0.002); (C) TACE group (p = 0.455). Abbreviations: RFA, radiofrequency ablation; TACE, transcatheter arterial chemoembolization (TIFF 348 KB)
423_2023_3170_MOESM2_ESM.tiff
Supplementary file2. Figure S2 Recurrence-free survival in patients categorized based on the median values of CD4+, CD8+, and CD45RO+. (A) CD4+ high and low groups (p< 0.001); (B) CD8 the high and low groups (p = 0.018); (C) CD45RO+ high and low groups (p = 0.015) (TIFF 358 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Doi, S., Yasuda, S., Matsuo, Y. et al. Clinical impact of sarcopenia in early-stage intrahepatic recurrent hepatocellular carcinoma: an association with impaired host immunity. Langenbecks Arch Surg 408, 433 (2023). https://doi.org/10.1007/s00423-023-03170-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-023-03170-2