Abstract
Background
Reducing clinically relevant post-operative pancreatic fistula (CR-POPF) incidence after pancreatic resections has been a topic of great academic interest. Optimizing post-operative drain management is a potential strategy in reducing this major complication.
Methods
Studies involving pancreatic resections, including both pancreaticoduodenectomy (PD) and distal pancreatic resections (DP), with intra-operative drain placement were screened. Early drain removal was defined as removal before or on the 3rd post-operative day (POD) while late drain removal was defined as after the 3rd POD. The primary outcome was CR-POPF, International Study Group of Pancreatic Surgery (ISGPS) Grade B and above. Secondary outcomes were all complications, severe complications, post-operative haemorrhage, intra-abdominal infections, delayed gastric emptying, reoperation, length of stay, readmission, and mortality.
Results
Nine studies met the inclusion criteria and were included for analysis. The studies had a total of 8574 patients, comprising 1946 in the early removal group and 6628 in the late removal group. Early drain removal was associated with a significantly lower risk of CR-POPF (OR: 0.24, p < 0.01). Significant reduction in risk of post-operative haemorrhage (OR: 0.55, p < 0.01), intra-abdominal infection (OR: 0.35, p < 0.01), re-admission (OR: 0.63, p < 0.01), re-operation (OR: 0.70, p = 0.03), presence of any complications (OR: 0.46, p < 0.01), and reduced length of stay (SMD: −0.75, p < 0.01) in the early removal group was also observed.
Conclusion
Early drain removal is associated with significant reductions in incidence of CR-POPF and other post-operative complications. Further prospective randomised trials in this area are recommended to validate these findings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Despite the development of numerous strategies to improve the safety and outcomes of pancreatic resections, clinically relevant post-operative pancreatic fistula (CR-POPF) remains a common major complication. A recent systematic review and meta-analysis reported that 21% of patients undergoing pancreatic resections experience varying severity of CR-POPF [1]. CR-POPF can cause serious complications such as haemorrhage and sepsis, leading to increased length of stay, re-operation, and morbidity [2], substantially increasing healthcare costs for the patient and the hospital system [3, 4]. CR-POPF also carries a 1% mortality risk, and this risk is increased to 25% in higher grades of CR-POPF [1].
CR-POPF, according to the ISGPS 2016 guidelines [5], requires both clinical and biochemical events, such as increased drain amylase level, requirement of interventions to manage bleeding and infection, organ failure, and mortality.
The underlying pathophysiology of CR-POPF is the leakage of pancreatic fluid into the abdomen from loss of mechanical barrier integrity, either through a faulty pancreatoenteric anastomosis or through the pancreatic parenchyma directly. Some identified risk factors for CR-POPF post-pancreatic resections include soft gland texture, non-malignant pathology, non-chronic pancreatitis pathology, small pancreatic duct diameter, and high intra-operative blood loss. These risk factors have been formulated into a fistula risk score (FRS) for easy stratification of high-risk patients [6]. Some other risk factors not included in the FRS but may affect CR-POPF incidence include positive drain fluid culture [7], octreotide therapy [8], and preoperative serum albumin level [9]. To reduce CR-POPF incidence, many strategies have been studied. For example, specific to pancreaticoduodenectomy, these include different ways of performing the pancreatoenteric anastomosis, usage of pancreatic stents, and minimally invasive approaches [10]. To this end, post-operative abdominal drain management has been identified as a potential strategy to reduce CR-POPF incidence.
Placement of abdominal drains is a common standard practice in many centres post-pancreatic resection [11, 12]. Drains are purported to reduce post-operative complications by detecting leaks and sentinel bleeds, preventing fluid accumulation and subsequent infection. However, drain placement following pancreatic resections remains a matter of controversy. It has been found that drains do not provide these benefits consistently and may even increase the incidence of certain complications such as CR-POPF, re-admission, morbidity, and mortality, especially in patients with low risk of CR-POPF [11, 13, 14]. Prolonged placement of a drain has been suspected to contribute to CR-POPF due to the negative pressure exerted [15, 16]. Besides causing pain and discomfort to the patient, drains may also serve as portals for retrograde intra-abdominal infection [17,18,19]. In addition, there has been a move towards no drain placement or early drain removal in other major abdominal surgeries as part of Enhanced Recovery After Surgery (ERAS) protocols, such as in colorectal resections [20], closures of perforated duodenal ulcers [21], cholecystectomies [22], and deep pelvic procedures [23]. Many systematic reviews have inconclusive and conflicting conclusions on prophylactic abdominal drainage in pancreatic resection [13, 14, 24, 25].
Against this backdrop, this review aims to systematically review the literature and evaluate the effects of early versus late drain removal after pancreatic resection on post-operative outcomes. These findings may have a direct impact on post-operative clinical management and guideline recommendation.
Methods
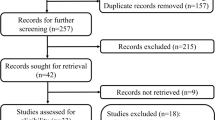
The protocol is registered with PROSPERO (CRD42022297746). This systematic review and meta-analysis was performed in line with the PRISMA (Preferred Reporting Items for Systematic Review and Meta-Analysis) 2020 checklist [26] (Fig. 1) and Cochrane Handbook for Systematic Reviews of Interventions v6.3 [27].
Literature search
The electronic databases of PubMed, Cochrane Library, and EMBASE were searched systematically until 17 May 2023 for relevant studies, using the concepts of pancreatic surgery and drain removal (Supplementary Table 1). No restrictions were placed on language, region, or publication date. References of identified studies were screened in order to identify further eligible studies.
Eligibility criteria
The inclusion criteria for this study were (1) all pancreatic resections ranging from pancreaticoduodenectomy (PD) to distal pancreatic resection (DP), regardless of whether surgeries were open or laparoscopic; (2) drains were placed intra-operatively; (3) early drain removal was defined as drain removal before or on the third post-operative day (POD); (4) late drain removal was defined as any drain removal after the third POD. Randomised, prospective and retrospective study designs were included, while case reports, conference abstracts, and reviews were excluded.
Titles and abstracts retrieved were independently screened by two reviewers (AW and KYF), with disagreements de-conflicted by discussion or via a third reviewer (RM). Full texts were assessed in detail for final inclusion in the review.
Outcomes
The primary end point was CR-POPF, that is International Study Group of Pancreatic Surgery (ISGPS) Grade B and above [5], or similar criteria [28, 29]. This is defined as drain amylase more than three times the upper institutional limit of normal of serum amylase, with clinically relevant conditions related directly to the CR-POPF, such as prolonged stay, use of therapeutic agents (for example, parenteral nutrition), or infection and organ failure. Biochemical leaks without clinical implications are not included.
Secondary outcomes were intra-abdominal infections, haemorrhage, length of stay, delayed gastric emptying (DGE), re-operation, re-admission, any complications, and Clavien-Dindo class III and above complications. Data pertaining to study characteristics, baseline characteristics, and quality characteristics were also extracted by two reviewers (AW and KYF) with the aid of a predefined electronic data extraction sheet.
Statistical analysis
For dichotomous outcomes, respective odds ratios (ORs) and their 95% confidence intervals (95% CI) were used for meta-analysis. For continuous outcomes, mean differences with 95% CIs were considered. Forest plots were generated based on random-effects models. Missing means and standard deviations were imputed from the reported median and range, or median and interquartile range, according to methods previously outlined by Hozo et al [30] and Wan et al [31], respectively.
Statistical heterogeneity was assessed via the I2 statistic and classified as low, moderate, or considerable for I2 < 40%, 40–75%, and >75%, respectively [32]. In addition, subgroup analyses were done for the main endpoint of CR-POPF, looking at PD only, low-risk patients only (defined as drain fluid amylase (DFA) <5000 on POD 1), and randomised controlled trials and propensity score matched (RCT/PSM) studies only.
Critical appraisal
Critical appraisal of the methodological quality of RCTs was conducted using the Cochrane Risk of Bias 2 tool [33]. For retrospective studies, Joanna Briggs Institute Critical Appraisal tools were used [34]. The overall quality of evidence was judged by means of the GRADE approach [35]. Funnel plots were used to visually inspect the small-study effects and potential publication bias for CR-POPF and its related subgroups. All analyses were conducted with Review Manager version 5.4 and R 4.1.2. All tests were two-sided with a 5% significance level.
Results
Study selection
The systematic literature search yielded 1534 studies. After the removal of 250 duplicates, the remaining 1284 articles were reviewed by title and abstract. Of these, 26 articles underwent full-text review. Finally, nine studies met the inclusion criteria and were included for analysis [36,37,38,39,40,41,42,43,44]. The studies had a total of 8574 patients, comprising 1946 in the early removal group and 6628 in the late removal group (Table 1). There are three RCTs [37, 39, 40], one retrospective study with propensity score matching (PSM) [38], and five retrospective studies [36, 41,42,43,44], from, Italy, Japan, USA, Netherlands, China, and Korea. Three studies [37, 39, 41] included populations from both pancreaticoduodenectomy and distal pancreatectomy, six studies included [38,39,40,41, 43, 44] results from only pancreaticoduodenectomy, and three studies [36, 40, 42] included results from only distal pancreatectomy. Only three studies [39, 40, 43] included data on fistula risk score.
Clinically relevant post-operative pancreatic fistula (CR-POPF)
Among nine studies (8574 patients) [36,37,38,39,40,41,42,43,44], early drain removal was associated with a significantly lower risk of CR-POPF with moderate heterogeneity observed (OR: 0.24, 95% CI: 0.14–0.40, p < 0.01, I2 = 59%) (Fig. 2A). This outcome is moderate quality evaluated by GRADE.
In the PD-only subgroup, among six studies (3460 patients) [38,39,40,41, 43, 44], there was similarly a lower risk of CR-POPF with early drain removal with moderate heterogeneity seen (OR: 0.27, 95% CI 0.14–0.53, p < 0.01, I2 = 51.0%) (Fig. 2B). This outcome is moderate quality evaluated by GRADE.
Amongst low-risk patients, across seven studies (3846 patients) [37,38,39,40, 42,43,44], there was also significant decreased risk of CR-POPF with early drain removal (OR: 0.29, 95% CI 0.14–0.57, p < 0.01, I2 = 61.0%) (Fig. 2C). For this subgroup, a moderate level of heterogeneity was found. This outcome is moderate quality evaluated by GRADE.
Amongst randomised controlled trials and propensity score-matched studies, across four studies (802 patients) [37,38,39,40], there was no statistically significant difference found between the two groups (OR: 0.30, 95% CI 0.06–1.55, p = 0.15, I2 = 65.0%) (Fig. 2D). For this subgroup, a moderate level of heterogeneity was found. This outcome is moderate quality evaluated by GRADE.
Complications
The presence of any complications was reported in seven studies (3800 patients) [37,38,39,40,41, 43, 44], with lower risk amongst those with early drain removal (OR: 0.46, 95% CI: 0.33–0.63, p < 0.01, I2 = 73%) (Fig. 3A). This outcome is moderate quality evaluated by GRADE.
Analysis of severe complications (Clavien-Dindo Grade ≥ III), reported in four studies (977 patients) [36, 39, 40, 44], showed no significant difference between the two groups (OR: 0.49, 95% CI: 0.21–1.12, p = 0.09, I2 = 60%) (Fig. 3B). This outcome is moderate quality evaluated by GRADE.
The presence of post-operative haemorrhage was reported in five studies (3113 patients) [37, 39,40,41, 43]. There was a significantly lower occurrence of haemorrhage with early drain removal (OR: 0.55, 95% CI: 0.41–0.73, p < 0.01, I2 = 0%) (Fig. 3C). This outcome is moderate quality evaluated by GRADE.
Among five studies (2342 patients) [37,38,39,40, 43], early drain removal resulted in a lower risk of intra-abdominal infection compared to late drain removal (OR: 0.35, 95% CI: 0.22–0.56, p < 0.01, I2 = 31%) (Fig. 3D). This outcome is moderate quality evaluated by GRADE.
DGE occurrence was reported in seven studies (8057 patients) [37,38,39,40,41,42,43], and no statistically significant difference was found between the two groups (OR: 0.73, 95% CI: 0.47–1.13, p = 0.16, I2 = 66%) (Fig. 3E). This outcome is moderate quality evaluated by GRADE.
Meta-analysis of re-operation rates across seven studies (8040 patients) [37,38,39,40,41,42,43] showed a significantly lower rate of re-operation (OR: 0.70, 95% CI: 0.50–0.96, p = 0.03, I2 = 0%) (Fig. 3F). This outcome is moderate quality evaluated by GRADE.
Hospitalization outcomes
Length of stay was reported in seven studies (7329 patients) [36, 37, 39, 40, 42,43,44]. Early drain removal significantly decreased the length of stay compared to late drain removal (SMD: −0.75, 95% CI: −1.05 to −0.44, p < 0.01, I2 = 94%) (Fig. 3G]. This outcome is moderate quality evaluated by GRADE.
Re-admission rates were compared in nine studies (8554 patients) [36,37,38,39,40,41,42,43,44]. Meta-analysis of this data showed that patients with early drain removal had a lower rate of re-admission (OR: 0.63, 95% CI: 0.50–0.80, p < 0.01, I2 = 18%) (Fig. 3H). This outcome is moderate quality evaluated by GRADE.
Mortality
Mortality rate was compared in three studies (2773 patients) [38, 41, 43]. No significant difference was observed between the two groups across these studies (OR: 1.70, 95% CI: 0.90–3.24, p = 0.10, I2 = 0%) (Fig. 3I). This outcome is moderate quality evaluated by GRADE.
Risk of bias, publication bias, and quality of evidence
Risk of bias of RCTs was assessed using ROB2 [33], where overall risk is decided based on randomisation process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of reported results. Risk of bias of retrospective studies was assessed using JBI [34], where overall risk is decided based on selection and matching, identification of participants, exposure duration and measurement, identification of confounding factors, assessment of outcome, and statistical analysis methodology. Of the included studies, only two [36, 41] were deemed to have moderate risk of bias, due to poor and unclear study methodology, whilst others were rated to have low risk of bias.
Funnel plots for each outcome is plotted, and the plots of the primary outcome and the subgroups are attached in Fig. 4. Potential publication bias was observed in funnel plots A and C on visual inspection. Egger’s test and other tests for funnel plot asymmetry were not performed due to a small number of studies.
For overall quality of evidence, GRADE approach [35] was used, as an aggregate of factors such as study design, risk of bias, inconsistency, indirectness, imprecision, and publication bias. Overall, four studies [37,38,39,40] have a high quality of evidence, two [43, 44] with moderate, and three [36, 41, 42] low.
Discussion
In this review, early drain removal was associated with significant reductions in the incidence of CR-POPF. Further subgroup analyses revealed a similar significant observation amongst patients who underwent PD and patients considered low risk in the immediate post-operative period (defined as DFA <5000 on POD 1). Besides this, early drain removal also decreased the incidence of post-operative haemorrhage, intra-abdominal infection, re-admission, re-operation, presence of any complications, and reduced length of stay.
CR-POPF is a common and devastating adverse complication that plaque a significant portion of patients undergoing pancreatic resections of any extent. Other than reducing the number of CR-POPF occurred, the early removal of drains especially in lower risk population can further decrease any adverse sequelae following the operation, with and without the occurrence of CR-POPF, such as needing for drainage or re-operation, haemorrhage, sepsis, and infections.
The results of this review show that early drain removal is likely to be a superior strategy compared to late drain removal. These benefits apply across the whole cohort of patients undergoing pancreatic resection, especially in low-risk patients. Practicing clinicians should seriously consider early drain removal in their clinical practice, amongst patients they deem appropriate. The practice of early drain removal would adhere to evidence-based ERAS principles of minimising drain placement, and while addressing the hesitancy, many surgeons may feel in not placing any drains at all in pancreatic resections [47]. Patients undergoing pancreatic resections are not uniform and homogenous; hence, the treatment and management that start from preoperative investigations should stratify patients into different bins that receive different treatments.
Prior studies have focused mainly on whether to use drains at all in pancreatic surgeries. Evidence from multiple meta-analyses on the use of prophylactic intraperitoneal drains in pancreatic surgery is conflicting. Some studies reported that prophylactic drain placement in pancreatic resections may benefit patients by reducing the incidence of complications such as CR-POPF, infections, and re-admissions [13, 15, 25, 46], while others reported no significant differences in complications such as CR-POPF [14, 24]. These studies omit the potential of an intermediate management strategy—placement of a drain with early removal. Such a strategy could potentially reap the benefits of drain placement, such as monitoring for any intra-abdominal events, while minimising risks of associated complications. This review addresses the knowledge gap, and the results hold practical clinical implications. This is also the first metanalysis on this topic that includes the subgroup analysis with low-risk population that explore this option in this specific group of patient and perform the analysis in a more controlled fashion by removing a significant confounder.
Although in the RCT/PSM subgroup, the improvement in outcomes is not statistically significant, but there is still a trend showing possible benefits, and this should be verified and explored in further studies. Future studies should also further investigate other risk factors for POPF that can influence decision-making regarding the duration of the drain, such as BMI, pancreas texture, duct size, operative methods, intra-operative blood loss, and intra-operative blood transfusion [47], to help guide drain management in patients at different risks of fistula formation, as our study provides evidence that early drain removal is beneficial for low-risk populations. Further studies can also be conducted to quantify the exact optimal time of drain removal for patients of each risk group of POPF. Results of this meta-analysis may provide an impetus for research into early drain removal not just in pancreatic surgery, but also in other major abdominal surgeries.
Limitations
This study has several limitations. Firstly, there was a paucity of RCTs available for inclusion. Although most studies favoured early drain removal, the subgroup analysis of PSM/RCT-only studies showed no statistically significant difference between the two groups; however, there is still a trend that favours early drain removal (Fig. 2D). Non-randomised studies may have inherent biases. For example, patients in retrospective cohorts may have longer drain durations if they were thought to have higher risks of pancreatic fistula or other complications, whilst those with shorter drain duration might have had a more straightforward clinical course. This selection bias may have a significant impact on the incidence of POPF in the study. Differences in inclusion criteria across different studies (Table 1) likely explain the moderate to high level of heterogeneity as evidenced by the I2 values across most meta-analyses. However, there is still a statistical reduction in CR-POPF rate in early drain removal in patients with low risk of CR-POPF (Fig. 2C). This subgroup analysis attempts to eliminate the bias incurred due to retrospective nature of studies and is able to standardise the baseline of the included population so that a fair comparison can be made at least within the low-risk population. Future studies can be conducted to further investigate the optimal candidate for early and late drain removal.
Another unaddressed issue was the type of drains used and their relations to the risk of CR-POPF formation and other complications. Commonly used drains include closed drains with active suction, such as Jackson-Pratt, Blake or Shirley drains, and open drains such as Penrose drains. We attempted to factor in the type of drain used in each study, whether it was passive drainage or active suction; however, this information is not available. However, it is noteworthy that a recent meta-analysis showed that the types of drain generally do not correlate with CR-POPF rates [48]. The effect of the duration of drainage for passive versus active drains remains a potential area to look into as the negative pressure applied may alter the clinical course and formation of CR-POPF.
The methodology associated with studies comparing early vs late drain removal meant that patients with late removal of drains may have higher rates of diagnosis of CR-POPF as the presence of drains allow for DFA to be sent in the event of an unexpected clinical course. CR-POPF may therefore be under-diagnosed in the early drain removal group, as symptoms from milder CR-POPF may be misattributed to other diagnosis and not fully investigated, leading to underdiagnosis of CR-POPF in the delayed removal group. Regardless, secondary outcomes studied such as re-admission and re-operation rates were still lower in the early drain removal group. This may point towards an overall decrease in post-operative complications in pancreatic surgeries, regardless if CR-POPF is diagnosed or not, if drain is removed early.
Lastly, all but one study [36] favours early drain removal regardless of subgroup analysis with regards to POPF. In this study [36], there are only two reported cases of POPF in the early removal group versus none in the delayed group, and this causes the 95% confidence interval to be widespread due to the 0 used in the calculation. The same author later repeated the RCT study with a larger group of patients, and the new multicenter RCT study significantly favours early drain removal. This inevitably leads to a skewing of results especially in subgroup analyses with less number of total patients and studies. Although we cannot discount this study, more studies should be performed and included in subsequent analysis to reduce the effect of a single study on the results in a metanalysis.
Conclusions
This review suggests that early drain removal after pancreatic surgery is advantageous compared to late drain removal, especially in low-risk patients. Future randomised trials in this area should be conducted to further validate the findings of this meta-analysis.
References
Pedrazzoli S (2017) Pancreatoduodenectomy (PD) and postoperative pancreatic fistula (POPF): a systematic review and analysis of the POPF-related mortality rate in 60,739 patients retrieved from the English literature published between 1990 and 2015. Medicine (Baltimore) 96(19):e6858
Malleo G, Pulvirenti A, Marchegiani G, Butturini G, Salvia R, Bassi C (2014) Diagnosis and management of postoperative pancreatic fistula. Langenbeck's Arch Surg 399(7):801–810
McMillan MT, Christein JD, Callery MP et al (2016) Comparing the burden of pancreatic fistulas after pancreatoduodenectomy and distal pancreatectomy. Surgery. 159(4):1013–1022
Ma LW, Dominguez-Rosado I, Gennarelli RL et al (2017) The cost of postoperative pancreatic fistula versus the cost of pasireotide: results from a prospective randomized trial. Ann Surg 265(1):11–16
Bassi C, Marchegiani G, Dervenis C et al (2017) The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 years after. Surgery. 161(3):584–591
Callery MP, Pratt WB, Kent TS, Chaikof EL, Vollmer CM Jr (2013) A prospectively validated clinical risk score accurately predicts pancreatic fistula after pancreatoduodenectomy. J Am Coll Surg 216(1):1–14
Kawai M, Okada KI, Miyazawa M et al (2023) Positive drain fluid culture on postoperative day 1 predicts clinically relevant pancreatic fistula in early drain removal with higher drain fluid amylase after pancreaticoduodenectomy. Surgery 173(2):511–520
McMillan MT, Christein JD, Callery MP et al (2014) Prophylactic octreotide for pancreatoduodenectomy: more harm than good? HPB (Oxford) 16(10):954–962
Shen J, Zhang Y, Hu J, Wei R, Wu H (2020) Albumin difference as a new predictor of pancreatic fistula following distal pancreatectomy: a retrospective study of 211 consecutive patients. Langenbeck's Arch Surg 405(1):55–62
Nahm CB, Connor SJ, Samra JS, Mittal A (2018) Postoperative pancreatic fistula: a review of traditional and emerging concepts. Clin Exp Gastroenterol 11:105–118
Cheng Y, Xia J, Lai M, Cheng N, He S (2016) Prophylactic abdominal drainage for pancreatic surgery. Cochrane Database Syst Rev 10(10):CD010583-CD010583
He S, Xia J, Zhang W, Lai M, Cheng N, Liu Z, Cheng Y ( 2021) Prophylactic abdominal drainage for pancreatic surgery. Cochrane Database Syst Rev 12(12):CD010583
Liu X, Chen K, Chu X, Liu G, Yang Y, Tian X (2021) Prophylactic intra-peritoneal drainage after pancreatic resection: an updated meta-analysis. Front Oncol 11:658829
Wang YC, Szatmary P, Zhu JQ et al (2015) Prophylactic intra-peritoneal drain placement following pancreaticoduodenectomy: a systematic review and meta-analysis. World J Gastroenterol 21(8):2510–2521
Conlon KC, Labow D, Leung D et al (2001) Prospective randomized clinical trial of the value of intraperitoneal drainage after pancreatic resection. Ann Surg 234(4):487–493
Crippa S, Salvia R, Falconi M, Butturini G, Landoni L, Bassi C (2007) Anastomotic leakage in pancreatic surgery. HPB (Oxford) 9(1):8–15
Kawai M, Tani M, Terasawa H et al (2006) Early removal of prophylactic drains reduces the risk of intra-abdominal infections in patients with pancreatic head resection: prospective study for 104 consecutive patients. Ann Surg 244(1):1–7
Inoue M, Uchida K, Otake K et al (2011) Placement of prophylactic drains after laparotomy may increase infectious complications in neonates. Pediatr Surg Int 27(9):975–979
Jeekel J (1992) No abdominal drainage after Whipple's procedure. Br J Surg 79(2):182
Tsujinaka S, Konishi F (2011) Drain vs no drain after colorectal surgery. Indian J Surg Oncol 2(1):3–8
Pai D, Sharma A, Kanungo R, Jagdish S, Gupta A (1999) Role of abdominal drains in perforated duodenal ulcer patients: a prospective controlled study. Aust N Z J Surg 69(3):210–213
Gurusamy KS, Koti R, Davidson BR (2013) Routine abdominal drainage versus no abdominal drainage for uncomplicated laparoscopic cholecystectomy. Cochrane Database Syst Rev 3(9):CD006004
Charoenkwan K, Kietpeerakool C (2017) Retroperitoneal drainage versus no drainage after pelvic lymphadenectomy for the prevention of lymphocyst formation in women with gynaecological malignancies. Cochrane Database Syst Rev 6(6):Cd007387
Hüttner FJ, Probst P, Knebel P et al (2017) Meta-analysis of prophylactic abdominal drainage in pancreatic surgery. Br J Surg 104(6):660–668
Lyu Y, Cheng Y, Wang B, Zhao S, Chen L (2020) Peritoneal drainage or no drainage after pancreaticoduodenectomy and/or distal pancreatectomy: a meta-analysis and systematic review. Surg Endosc 34(11):4991–5005
Page MJ, Moher D, Bossuyt PM et al (2021) PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ. 372:n160
Higgins JPT TJ, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors) Cochrane handbook for systematic reviews of interventions version 6.3 (updated February 2022). Cochrane. Published 2022. http://www.training.cochrane.org/handbook
McMillan MT, Vollmer CM Jr, Asbun HJ et al (2016) The characterization and prediction of ISGPF grade C fistulas following pancreatoduodenectomy. J Gastrointest Surg 20(2):262–276
Bassi C, Dervenis C, Butturini G et al (2005) Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery. 138(1):8–13
Hozo SP, Djulbegovic B, Hozo I (2005) Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol 5(1):13
Wan X, Wang W, Liu J, Tong T (2014) Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 14(1):135
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560
Sterne JAC, Savović J, Page MJ et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898
Moola SMZ, Tufanaru C, Aromataris E, Sears K, Sfetcu R, Currie M, Qureshi R, Mattis P, Lisy K, Mu P-F. Chapter 7: systematic reviews of etiology and risk. In: Aromataris E, Munn Z (Editors). JBI Manual for Evidence Synthesis. Published 2020. https://synthesismanual.jbi.global
Guyatt GH, Oxman AD, Vist GE et al (2008) GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 336(7650):924–926
Adachi T, Kuroki T, Kitasato A et al (2015) Safety and efficacy of early drain removal and triple-drug therapy to prevent pancreatic fistula after distal pancreatectomy. Pancreatology. 15(4):411–416
Bassi C, Molinari E, Malleo G et al (2010) Early versus late drain removal after standard pancreatic resections: results of a prospective randomized trial. Ann Surg 252(2):207–214
Beane JD, House MG, Ceppa EP, Dolejs SC, Pitt HA (2019) Variation in drain management after pancreatoduodenectomy: early versus delayed removal. Ann Surg 269(4):718–724
Dai M, Liu Q, Xing C et al (2020) Early drain removal after major pancreatectomy reduces postoperative complications: a single-center, randomized, controlled trial. J Pancreatol 3(2)
Dai M, Liu Q, Xing C et al (2022) Early drain removal is safe in patients with low or intermediate risk of pancreatic fistula after pancreaticoduodenectomy: a multicenter, randomized controlled trial. Ann Surg 275(2):e307–e314
Linnemann RJA, Patijn GA, van Rijssen LB et al (2019) The role of abdominal drainage in pancreatic resection - a multicenter validation study for early drain removal. Pancreatology. 19(6):888–896
Seykora TF, Liu JB, Maggino L, Pitt HA, Vollmer CM Jr (2020) Drain management following distal pancreatectomy: characterization of contemporary practice and impact of early removal. Ann Surg 272(6):1110–1117
Xourafas D, Ejaz A, Tsung A, Dillhoff M, Pawlik TM, Cloyd JM (2019) Validation of early drain removal after pancreatoduodenectomy based on modified fistula risk score stratification: a population-based assessment. HPB (Oxford) 21(10):1303–1311
Yoon SJ, Yoon SK, Jung JH et al (2021) Realistic advantages of early surgical drain removal after pancreatoduodenectomy: a single-institution retrospective study. J Clin Med 10(12):2716
Pędziwiatr M, Mavrikis J, Witowski J et al (2018) Current status of enhanced recovery after surgery (ERAS) protocol in gastrointestinal surgery. Med Oncol 35(6):95
Pedrazzoli S, Brazzale AR (2020) Systematic review and meta-analysis of surgical drain management after the diagnosis of postoperative pancreatic fistula after pancreaticoduodenectomy: draining-tract-targeted works better than standard management. Langenbeck's Arch Surg 405(8):1219–1231
Peng Y-P, Zhu X-L, Yin L-D et al (2017) Risk factors of postoperative pancreatic fistula in patients after distal pancreatectomy: a systematic review and meta-analysis. Sci Rep 7(1):185
Gachabayov M, Gogna S, Latifi R, Dong XD (2019) Passive drainage to gravity and closed-suction drainage following pancreatoduodenectomy lead to similar grade B and C postoperative pancreatic fistula rates. A meta-analysis. Int J Surg 67:24–31
Author information
Authors and Affiliations
Contributions
AGRW participated in the conception of the study, data collection, data analysis and manuscript writing. RM participated in data collection, data analysis and manuscript writing. KYF participated in data collection, data analysis and manuscript writing. ZC participated in data collection, data analysis and manuscript writing. GKB participated in conception of the study, manuscript writing and editing. AWCK participated in conception of the study, manuscript writing and editing. ISG participated in conception of the study, manuscript writing and editing. NQP participated in conception of the study, data analysis, manuscript writing and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary file 1
Supplementary Table 1. Full search strategies for the respective databases. (DOCX 14 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, A.G., Mohan, R., Fong, K.Y. et al. Early vs late drain removal after pancreatic resection—a systematic review and meta-analysis. Langenbecks Arch Surg 408, 317 (2023). https://doi.org/10.1007/s00423-023-03053-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-023-03053-6