Abstract
Background and aims
Intraductal tubulopapillary neoplasms (ITPNs) of the pancreas are rare. The purpose of this study was to collate and analyze published data on ITPNs of the pancreas to determine the clinicopathological features of the tumors and the surgical outcomes of patients.
Patients and methods
We searched MEDLINE and Igakuchuo-Zasshi for the period of 1980 to 2015 for case reports on surgical resection for ITPN of the pancreas. We evaluated the clinicopathological data associated with pancreatic ITPNs, the prognosis for each patient, and surgical outcomes described in the case reports.
Results
We obtained clinicopathological data for 58 patients (33 men and 25 women) with a mean age of 61 years (range, 35–84 years) who had undergone surgical resection for ITPN of the pancreas, including one patient from our clinic. Although ITPNs of the pancreas have different clinicopathological features to intraductal papillary mucinous neoplasms, the treatment strategy for patients with ITPNs is the same as for patients with other cystic neoplasms of the pancreas. The immunohistochemical features of ITPNs included testing positive for cytokeratin 7 and/or cytokeratin 19 and negative for trypsin, MUC2, MUC5AC, and fascin. The overall 1-, 3-, and 5-year survival rates after surgery for the 37 cases with available data were 97.3, 80.7, and 80.7 %, respectively.
Conclusion
Surgical treatment is the only curative management option for patients with ITPN of the pancreas. To determine the best management strategy for this tumor and improve accuracy of prognosis for patients, we will continue to collect and analyze epidemiological and pathological data.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Intraductal tubulopapillary neoplasms (ITPNs) of the pancreas are very rare tumors that are characterized by intraductal tubulopapillary growth, ductal differentiation, scant intracellular mucin production, and cellular dysplasia. ITPNs are estimated to account for <1 % of all pancreatic exocrine tumors and 3 % of all pancreatic intraductal neoplasms [1, 2]. ITPN was first described in 2009 [2], and according to 2010 World Health Organization classification, there are two subtypes of intraductal neoplasms of the pancreas: intraductal papillary mucinous neoplasms (IPMNs) and ITPNs [1]. ITPNs include intraductal tubular carcinomas and are defined as grossly visible and tubule-forming epithelial neoplasms with high-grade dysplasia and ductal differentiation without overt production of mucin [1]. If a lesion has a component of invasive carcinoma, it is referred to as an “ITPN with an associated invasive carcinoma” [1].
We conducted a retrospective analysis of published cases of ITPN of the pancreas to determine the clinicopathological features of the tumors and the surgical outcomes, including survival rates, for patients with these tumors.
Method
Eligibility criteria
The reporting of this systematic review was performed according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement [3]. Observational studies written in English or written in Japanese with an English abstract were eligible for inclusion. There were no limitations regarding the date of publication of the included studies. Only articles for which full-text versions could be retrieved were included. For a study to be suitable for the qualitative synthesis, it had to contain information on patients being surgically treated for ITPN of the pancreas. In addition, the study had to describe a defined follow-up period exceeding the primary hospital stay, in which there had to be assessment of the patients’ prognosis after surgical management. Any study not meeting the above-mentioned requirements was not eligible for inclusion.
Literature search
A literature search was performed using the following terms: “intraductal tubulopapillary neoplasms,” “pancreas,” and “intraductal neoplasm without mucin.” The latter are now defined as ITPNs. The following databases were searched from 2009 to 2015 for case reports on surgical resection for ITPN of the pancreas: MEDLINE and Igakuchuo-Zasshi (a database of Japanese articles with English abstracts). Studies were included regardless of publication status. Studies in the reference lists of the retrieved articles, including relevant systematic reviews, were also searched.
After carefully reading articles in detail, cases of intraductal neoplasm without mucus that were published before 2009, when the term “intraductal tubulopapillary neoplasm” was introduced, were considered ITPNs. This enabled us to collect and analyze more clinical data, although retrospective re-diagnosis of patients is not ideal.
Data collection and assessment
To collect data, a standard data extraction form was developed. Information from included studies was systematically extracted and added to a customized Microsoft Excel spreadsheet. We extracted data on the following: patient demographics, including age and sex; tumor location and size; type of operation; and survival times. We also extracted data on the published prognosis for each patient.
Statistical analysis
Survival rates were generated using the Kaplan–Meier method and compared using the log-rank test. All analyses were performed using SPSS® (SPSS Inc, Chicago, IL). Values are expressed as mean ± standard deviation. P values <0.05 were considered statistically significant.
Results
Patient treated in our clinic
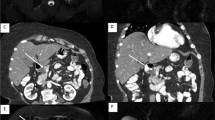
A 54-year-old woman with upper abdominal pain and anorexia was admitted to our hospital for examination and treatment of a developing abdominal cyst. Levels of tumor markers, including carcinoembryonic antigen and carbohydrate 19-9, were within normal ranges. Abdominal plain computed tomography (CT) revealed a cystic lesion in the head of the pancreas (Fig. 1a). Contrast-enhanced abdominal CT showed a cystic mass with a maximum diameter of 43 mm in the head of the pancreas, which seemed to contain mural nodule components (Fig. 1b). On magnetic resonance imaging (MRI), the cyst content was of low intensity on T1-weighted images (Fig. 2a) and high intensity on T2-weighted images (Fig. 2b). The mural nodule showed a high signal on diffusion-weighted imaging (DWI) (Fig. 2c). A magnetic resonance cholangiopancreatographic image showed interruption of the main pancreatic duct and mild dilation of the main pancreatic duct at the body and tail of the pancreas, indicating possible communication between the cystic lesion of the head of the pancreas and the main pancreatic duct (Fig. 2d). Considering the malignant potential of the cystic lesion based on preoperative radiological findings, especially the positive signal on DWI, a pancreaticoduodenectomy with regional lymphadenectomy was planned. Gross observation of the resected specimen revealed a cyst with a maximum diameter of 55 mm and a clear margin (Fig. 3a). Histopathological examination of the specimen revealed that the tumor was thickly composed of solid proliferating tumor cells with tubular, cribriform, or papillary formation and that the positive signal areas on DWI generally corresponded to the histological sites of malignancy (Fig. 3b, c). There was no lymph node metastasis, and mucin was not detectable. Immunohistochemical investigation showed that the tumor cells expressed cytokeratin (CK) 7, CK19, cancer antigen (CA) 19-9, MUC1, and MUC6. They tested negative for CK20, CA125, carcinoembryonic antigen, chromogranin A, synaptophysin, CD56, trypsin, p53, MUC2, MUC5AC, and CDX2 (Fig. 3d–f). More than 80 % of the neoplastic cells displayed Ki-67 immunoreactivity. The final diagnosis was ITPN with an associated invasive carcinoma in pathological stage T2N0M0 (p-stage 1B) according to the classification of the Union for International Cancer Control [4]. The patient’s postoperative course was uneventful, and she was discharged 13 days after the surgery. During the 24-month follow-up period, there was no evidence of recurrence or metastasis.
a Magnetic resonance (MR) image showing cyst content to be of low intensity on T1-weighted images. b MR image showing cyst content to be of high intensity on T2-weighted images. c MR image showing that the mural nodule had a high signal on diffusion-weighted imaging. d MR cholangiopancreatographic image showing the interruption of the main pancreatic duct and a mild dilation of the main pancreatic duct at the body and tail of the pancreas
a The resected specimen, revealing a cyst with a maximum diameter of 55 mm and a clear margin. b, c Histopathological images revealing that the tumor was thickly composed of solid proliferating tumor cells with tubular, cribriform, or papillary formation; there was no lymph node metastasis, and mucin was not detectable (b hematoxylin and eosin stain, low-magnification image; c hematoxylin and eosin stain, high-magnification image). d–f Histopathological images showing that neoplastic cells tested positive for MUC1 and MUC6 and negative for MUC5AC (d MUC1; e MUC5AC; f MUC6)
Literature review
We identified 23 articles available in electronic databases by searching PubMed and 16 articles from other sources (Fig. 4). We included 33 of these in our narrative synthesis and systematic review [2, 5–36]. We extracted comprehensive data on 58 cases of curative surgical resection for ITPN of the pancreas, which included a patient who we had treated in our clinic (Table 1). The published diagnoses for all of the patients were true indications for surgical resection.
Clinicopathological features of ITPNs
Of the 58 cases of ITPN of the pancreas for which we found comprehensive data, 33 occurred in men and 25 occurred in women. The mean patient age was 62 years (range, 35–84 years). The IPTNs were not associated with any specific clinical syndrome. The clinical characteristics of these cases of ITPN of the pancreas are shown in Table 1.
In 20 cases, the patients did not have symptoms; in the remaining 30 cases, the patients were diagnosed while they had clinical symptoms. The symptoms were abdominal pain or sense of fullness with either jaundice or diarrhea in 24 patients, jaundice in three, exacerbation of diabetes mellitus in one, excessive thirst in one, appetite loss in one, fever in one, and weight loss in one.
The tumor locations and operative methods are shown in Table 1. In 34 patients (59 %), the tumor was located in either the head of the pancreas alone (32 patients) or the head and body (two patients); in 19 patients (33 %), the tumor was in the body and/or tail of the pancreas; and in three patients, the tumor affected the whole pancreas. All patients underwent attempted curative resection. The surgical methods used included the following: pancreaticoduodenectomy (PD), including pylorus-preserving PD (PpPD) (20 patients [34 %]), distal pancreatectomy (10 [17 %]), and total pancreatectomy (five [9 %]).
Accurate preoperative diagnosis of ITPN of the pancreas is very difficult because no characteristics on imaging studies can be used to differentiate it from cystic neoplasm of the pancreas. In terms of size, ITPNs of the pancreas generally seem to be larger than ordinary pancreatic adenocarcinomas. The mean size of the ITPNs of the pancreas for 49 patients with available data was 4.5 ± 3.3 cm (range, 1–15 cm) (Table 1). To date, the most difficult discrimination lies between ITPN and IPMN pancreatobiliary types [2, 14, 37]. Tumor cells form tubulopapillae and contain little cytoplasmic mucin. Furthermore, ITPNs have some particular immunohistochemical features; they test positive for CK 7 and/or CK 19 and negative for trypsin, MUC2, MUC5AC, and fascin [2, 14, 37]. However, in two cases, the surgically resected specimen from patients with ITPN tested positive for MUC5AC [13, 18]. Of the 47 cases that we analyzed, 31 IPTNs had an invasive component, although involvement of an invasive component was not significantly associated with age, sex, or tumor size.
Survival outcomes of patients with ITPNs
Survival outcome data was only available for 37 cases of ITPNs of the pancreas, including the patient from our clinic (Table 1). The overall 1-, 3-, and 5-year survival rates after surgery were 97.3, 80.7, and 80.7 %, respectively (Fig. 5a). The overall 5-year survival rate was 81.5 % in patients with an invasive component (after pathologically complete resection of the ITPN of the pancreas) and 77.8 % in patients with a noninvasive tumor (p = 0.900) (Fig. 5b).
Discussion
We conducted a retrospective analysis of 58 cases with ITPN of the pancreas, with a focus on the clinicopathological features and surgical outcomes [2, 5–36]. Clinically, it is difficult to accurately diagnose ITPNs, although modern imaging modalities provide informative findings. IPMN, pancreatic intraepithelial neoplasia, MCN, acinar cell carcinoma, solid and pseudopapillary neoplasm (SPN), and tumor metastasis of renal cell carcinoma were part of the differential diagnosis for the treating physicians. The most difficult diagnoses to distinguish are ITPN and IPMN pancreatobiliary type [2, 14, 37]. Contrast-enhanced CT and MRI show ITPN as a cystic lesion with a dilated and irregular main pancreatic duct, but without an abundance of low attenuation mucin [18]. These findings are diagnostically significant for ITPNs. However, since IPMN, pancreatic intraepithelial neoplasia, MCN, acinar cell carcinoma, SPN, and tumor metastasis of renal cell carcinoma have similar radiological features, ITPNs of the pancreas are indistinguishable from other cystic tumors of the pancreas based on these CT findings. Moreover, because the cyst wall is sometimes thin, as was the case in our patient, enhancement of the inner cyst wall is not always seen. The preoperative CT scan for our patient did not provide information on the type of cystic lesion. MRI generally provides more information about cyst content than CT. However, in the cases that we analyzed, the nature of the fluid in the ITPN varied according to factors such as bleeding, chronic inflammation, and infection. Therefore, MRI seems to be of less use than expected for assessing lesions of the pancreas, especially mural nodules of cystic tumors, but is still useful for eliminating the possibility of malignant potential [38].
Guidelines on the management of pancreatic cystic neoplasms [39] will be updated this year. However, our systematic review shows that the biological behavior of ITPNs might be different from that of MCNs and/or IPMNs. Although it is difficult to obtain a radiological preoperative diagnosis of tumor type, it is important to detect “worrisome features” and “high-risk stigmata” that suggest surgical resection for ITPNs rather than obtain a diagnosis of tumor type in the preoperative setting. Diffusion-weighted MRI exploits the random motion of water molecules in biological tissues; the signal intensity reflects the impeded diffusion of water molecules. Pancreatic adenocarcinoma is usually associated with low apparent diffusion coefficient (ADC) in comparison to healthy pancreatic parenchyma because of the presence of fibrosis and increased cell density in these malignant lesions, which are associated with impeded free water diffusion. In contrast, lesions with increased fluid content may show increased ADCs due to increased random motion of water molecules. Diffusion-weighted MRI is recommended for detecting pancreatic cancer and characterizing pancreatic masses [40]. Our results suggest that, compared with conventional MRI alone, adding DWI to conventional MRI improves diagnostic accuracy by improving detection of malignant lesions and differentiation of malignant lesions from benign neoplasms of the pancreas.
The current overall 5-year survival rate of 81.5 % in patients with an invasive component is comparable to or better than the survival rates for invasive malignant IPMNs in other reports, although some of the cases that we examined in this study included patients who met criteria for surgical treatment with intent to cure IPMNs [37, 41, 42]. Furthermore, even if ITPNs had an invasive component, excellent curative treatment was achieved for patients compared to patients with IPMNs.
In terms of immunohistochemical features, ITPNs tested positive for CK 7 and/or CK 19 and negative for trypsin, MUC2, MUC5AC, and fascin [2, 14, 37]. Neither acinar differentiation nor aberrant expression of beta-catenin was observed. After attempted curative resection, a good prognosis in patients with ITPNs might be more likely when the lesion tests negative for MUC5AC in immunohistochemical studies. It has been reported that MUC5AC is aberrantly expressed in premalignant and malignant lesions, and several pancreatic cancer cell lines, and that MUC5AC is frequently expressed in malignant IPMNs, whereas no intense MUC5AC expression is detected in normal or hyperplastic lesions [43]. It has been suggested that expression of MUC5AC plays a role in the invasiveness of cancers by direct involvement in immune suppression that enables tumor cells to avoid the host immune system [44]. Several authors have suggested that overexpression of MAC5AC may reduce survival time in patients with pancreatic cancer [43–46].
This study had some limitations associated with errors and biases inherent in a small retrospective study. A major limitation of this study is that the results cannot simply be extrapolated because it included case reports published before diagnostic guidelines for ITPN were defined. ITPN of the pancreas is a rare disease, and because we included cases published before 2009 and cases of “intraductal tubular neoplasm without mucus,” our selection criteria might have contributed to heterogeneous outcomes. Another limitation is the lack of re-evaluation of the specimens by a dedicated pathologist. A definite diagnosis of ITPN is a pathological diagnosis that requires high specialization in pancreatic disease. Owing to the relatively recent description of ITPNs and the fact that they are extremely rare, accurate diagnosis is challenging, even for high-volume pancreatic pathologists. Therefore, determining the best management strategy for this tumor and improving the accuracy of prognosis for patients will require collection and analysis of further epidemiological and pathological data.
In conclusion, we treated a patient with ITPN of the pancreas with an associated invasive carcinoma. In this and other cases, it would have been difficult to diagnose ITPN preoperatively, even with the various imaging modalities that are available. Currently, surgical treatment is the only curative management option for ITPNs of the pancreas. In patients with a cystic lesion of the pancreas, ITPN should be considered part of the differential diagnosis.
References
Bosman FT, Carneiro F, Hruba RH et al (2010) WHO classification of tumours of the digestive system, 4th edn. IARC Press, Lyon, pp 304–313
Yamaguchi H, Shimizu M, Ban S et al (2009) Intraductal tubulopapillary neoplasms of the pancreas distinct from pancreatic intraepithelial neoplasia and intraductal papillary mucinous neoplasms. Am J Surg Pathol 33:1164–1172
Moher D, Liberati A, Tetzlaff J, PRISMA Group et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6, e1000097
Sobin LH, Gospodarowicz MK, Wittekind C (eds) (2009) TNM classification of malignant tumors, 7th edn. Wiley-Backwell, New York
Suda K, Hirai S, Matsumoto Y et al (1996) Variant of intraductal carcinoma (with scant mucin production) is of main pancreatic duct origin: a clinicopathological study of four patients. Am J Gastroenterol 91:798–800
Tajiri T, Tate G, Kunimura T et al (2004) Histologic and immunohistochemical comparison of intraductal tubular carcinoma, intraductal papillary-mucinous carcinoma, and ductal adenocarcinoma of the pancreas. Pancreas 29:116–122
Tajiri T, Tate G, Inagaki T et al (2005) Intraductal tubular neoplasms of the pancreas: histogenesis and differentiation. Pancreas 30:115–121
Ito K, Fujita N, Noda Y et al (2005) Intraductal tubular adenocarcinoma of the pancreas diagnosed before surgery by transpapillary biopsy: case report and review. Gastrointest Endosc 61:325–329
Itatsu K, Sano T, Hiraoka N et al (2006) Intraductal tubular carcinoma in an adenoma of the main pancreatic duct of the pancreas head. J Gastroenterol 41:702–705
Thirot-Bidault A, Lazure T, Ples R et al (2006) Pancreatic intraductal tubular carcinoma: a sub-group of intraductal papillary-mucinous tumors or a distinct entity? A case report and review of the literature. Gastroenterol Clin Biol 30:1301–1304
Hisa T, Nobukawa B, Suda K et al (2007) Intraductal carcinoma with complex fusion of tubular glands without macroscopic mucus in main pancreatic duct: dilemma in classification. Pathol Int 57:741–745
Oh DK, Kim SH, Choi SH et al (2008) Intraductal tubular carcinoma of the pancreas: a case report with the imaging findings. Korean J Radiol 9:473–476
Terada T (2009) Intraductal tubular carcinoma, intestinal type, of the pancreas. Pathol Int 59:53–58
Yamaguchi H, Kuboki Y, Hatori T et al (2011) Somatic mutations in PIK3CA and activation of AKT in intraductal tubulopapillary neoplasms of the pancreas. Am J Surg Pathol 35:1812–1817
Shimizu S, Tsukamoto T, Kanazawa A et al (2011) A case of intraductal tubular carcinoma (ITC). Jpn J Gastroenterol Surg 44:1158–1164
Bhuva N, Wasan H, Spalding D et al (2011) Intraductal tubulopapillary neoplasm of the pancreas as a radiation induced malignancy. BMJ Case Rep
Jokoji R, Tsuji H, Tsujimoto M et al (2012) Intraductal tubulopapillary neoplasm of pancreas with stromal osseous and cartilaginous metaplasia; a case report. Pathol Int 62:339–343
Urata T, Naito Y, Nagamine M et al (2012) Intraductal tubulopapillary neoplasm of the pancreas with somatic BRAF mutation. Clin J Gastroenterol 5:413–420
Tajiri T, Tate G, Matsumoto K et al (2012) Diagnostic challenge: intraductal neoplasms of the pancreatobiliary system. Pathol Res Pract 208:691–696
Shibasaki Y, Sakaguchi T, Inaba K et al (2012) Intraductal tubulopapillary neoplasm of the pancreas successfully treated with total pancreatectomy. J Jpn Surg Assoc 73:2061–2067
Motosugi U, Yamaguchi H, Furukawa T et al (2012) Imaging studies of intraductal tubulopapillary neoplasms of the pancreas: 2-tone duct sign and cork-of-wine-bottle sign as indicators of intraductal tumor growth. J Comput Assist Tomogr 36:710–717
Kasugai H, Tajiri T, Takehara Y et al (2013) Intraductal tubulopapillary neoplasms of the pancreas: case report and review of the literature. J Nippon Med Sch 80:224–229
Furuhata A, Minamiguchi S, Mikami Y et al (2014) Intraductal tubulopapillary neoplasm with expansile invasive carcinoma of the pancreas diagnosed by endoscopic ultrasonography-guided fine needle aspiration: a case report. Diagn Cytopathol 42:314–320
Matsushita K, Morimoto O, Motoki Y et al (2013) A case of intraductal tubulopapillary neoplasm of the pancreas (ITPN) which with difficult to diagnose preoperatively. Jpn J Gastroenterol Surg 46:196–202
Matsuda M, Watanabe G, Hashimoto M et al (2014) A case of intraductal tubulopapillary neoplasm of the pancreas with portal vein tumor thrombus. J Jpn Pancreas Soc 29:729–735
Guan H, Gurda G, Lennon AM et al (2014) Intraductal tubulopapillary neoplasm of the pancreas on fine needle aspiration: case report with differential diagnosis. Diagn Cytopathol 42:156–160
Del Chiaro M, Mucelli RP, Blomberg J et al (2014) Is intraductal tubulopapillary neoplasia a new entity in the spectrum of familial pancreatic cancer syndrome? Fam Cancer 13:227–229
Ahls MG, Niedergethmann M, Dinter D et al (2014) Case report: intraductal tubulopapillary neoplasm of the pancreas with unique clear cell phenotype. Diagn Pathol 9:11
Ito H, Imai J, Yazaki T et al (2014) A case of pancreatic intraductal tubulopapillary neoplasm (ITPN) mimicking a small ductal carcinoma. Prog Dig Endosc 84:200–201,221
Chang X, Jiang Y, Li J, Chen J (2014) Intraductal tubular adenomas (pyloric gland-type) of the pancreas: clinicopathologic features are similar to gastric-type intraductal papillary mucinous neoplasms and different from intraductal tubulopapillary neoplasms. Diagn Pathol 9:172
Someya Y, Nakamoto Y, Nakatani K et al (2014) 18F-FDG uptake in intraductal tubulopapillary neoplasm of the pancreas. Clin Nucl Med 39:e277–e280
Takayama S, Maeda T, Nishihara M et al (2015) A case of intraductal tubulopapillary neoplasm of pancreas with severe calcification, a potential pitfall in diagnostic imaging. Pathol Int 65:501–506
Yoshida Y, Matsubayashi H, Sasaki K et al (2015) Intraductal tubulopapillary neoplasm of the pancreatic branch duct showing atypical images. J Dig Dis 16:357–361
Kitaguchi K, Kato Y, Kojima M et al (2015) A resected case of intraductal tubulopapillary neoplasm of the pancreas: report of a case. Int Surg 100:281–286
Matthews Y, McKenzie C, Byrne C et al (2015) Intraductal tubulopapillary neoplasm of pancreas with associated invasive carcinoma, lymph node, rectal and hepatic metastases. Pathology 47:169–171
Kölby D, Thilén J, Andersson R et al (2015) Multifocal intraductal tubulopapillary neoplasm of the pancreas with total pancreatectomy: report of a case and review of literature. Int J Clin Exp Pathol 8:9672–9680
Kloppel G, Basturk O, Schlitter AM et al (2014) Intraductal neoplasms of the pancreas. Semin Diagn Pathol 31:452–466
Lee NK, Kim S, Kim GH et al (2012) Diffusion-weighted imaging of biliopancreatic disorders: correlation with conventional magnetic resonance imaging. World J Gastroenterol 18:4102–4117
Tanaka M, Fernández-del Castillo C, Adsay V et al (2012) International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology 12:183–197
Barral M, Taouli B, Guiu B et al (2015) Diffusion-weighted MR imaging of the pancreas: current status and recommendations. Radiology 274:45–63
Ceppa EP, Roch AM, Cioffi JL et al (2015) Invasive, mixed-type intraductal papillary mucinous neoplasm: superior prognosis compared to invasive main-duct intraductal papillary mucinous neoplasm. Surgery 158(937–944):944–945
Koh YX, Zheng HL, Chok AY et al (2015) Systematic review and meta-analysis of the spectrum and outcomes of different histologic subtypes of noninvasive and invasive intraductal papillary mucinous neoplasms. Surgery 157:496–509
Kanno A, Satoh K, Kimura K et al (2006) The expression of MUC4 and MUC5AC is related to the biologic malignancy of intraductal papillary mucinous neoplasms of the pancreas. Pancreas 33:391–396
Hoshi H, Sawada T, Uchida M et al (2013) MUC5AC protects pancreatic cancer cells from TRAIL-induced death pathways. Int J Oncol 42:887–893
Takikita M, Altekruse S, Lynch CF et al (2009) Associations between selected biomarkers and prognosis in a population-based pancreatic cancer tissue microarray. Cancer Res 69:2950–2955
Aloysius MM, Zaitoun AM, Awad S et al (2010) Mucins and CD56 as markers of tumour invasion and prognosis in periampullary cancer. Br J Surg 97:1269–1278
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by the Kochi Organization for Medical Reformation and Renewal Grants.
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethics committee of the Kochi Health Sciences Center and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. A patient provided written informed consent.
Rights and permissions
About this article
Cite this article
Date, K., Okabayashi, T., Shima, Y. et al. Clinicopathological features and surgical outcomes of intraductal tubulopapillary neoplasm of the pancreas: a systematic review. Langenbecks Arch Surg 401, 439–447 (2016). https://doi.org/10.1007/s00423-016-1391-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-016-1391-6