Abstract
Aim
Reduced physical function and increased risk of falls in older adults are accompanied by age-related reductions in torque development of leg muscles, although the mechanisms and potential sex differences are not understood.
Purpose
To determine the mechanistic origins (neural vs. muscular) for the age-related reduction in torque development, we compared the peak rates of torque development (RTD) during electrically-evoked and fast voluntary contractions of the knee extensors between young and older men and women.
Methods
Sets of single- and double-pulse electrical stimulations evoked contractions of the knee extensor muscles in 20 young (23.0 ± 0.8 years; 10 women) and 20 older adults (78.2 ± 1.5 years; 10 women), followed by voluntary isometric knee extension contractions with torque development as fast as possible that matched the torque during electrically-evoked contraction (10–40% maximal torque).
Results
Peak RTD during fast-voluntary contractions was 41% less than electrically-evoked contractions (p < 0.001), but more so for older adults (44%) than young (38%, p = 0.04), with no sex differences. Peak RTD during fast-voluntary contractions was more variable between contractions for the older than young adults (77%MVC s−1 vs. 47%MVC s−1, p < 0.001). Additionally, older women exhibited greater variability than older men (81%MVC s−1 vs. 72%MVC s−1, p = 0.04) with no sex-related differences within the young adults.
Conclusion
Older adults had slower and more variable RTD during voluntary contractions than young adults, particularly older women. The limited age-related differences in electrically-evoked RTD suggest the primary mechanism for the slower torque development of the knee extensor muscles in older men and women involve reduced neural activation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human aging is associated with reductions in maximal muscle strength and power (Doherty 2003; Sundberg et al. 2018a; Hunter et al. 2016) and a slowed ability to generate forces rapidly, particularly in lower limb muscle groups (Thompson et al. 2013, 2014). The age-related reductions in physical performance are primarily attributed to an age-related loss of skeletal muscle mass [sarcopenia; (Doherty 2003)], and atrophy of fast fibers (Sundberg et al. 2018a). In addition, older adults exhibit greater variability between repeated motor tasks and within a single motor task, including increased variability of muscle contraction velocity and torque (Hunter et al. 2016). The large variability between repeated trials is attributed to greater variability in activation of the muscles (Hunter et al. 2016; Rozand et al. 2017) and impedes consistent and rapid power production by lower limb skeletal muscles.
In addition to age-related reductions in maximal muscle power, aging is accompanied by a reduced ability to produce force quickly, and this can be quantified as the rate of torque development (RTD) (Gerstner et al. 2017). The maximal RTD of lower limb muscles is a strong predictor of physical function in older age (Thompson et al. 2013; LaRoche et al. 2010). For example, the age-related reduction in RTD contributes to an impaired ability to recover from losses of balance to avoid a fall (Aagaard 2003; Bento et al. 2010) and diminished walking performance in particular in older women (Osawa et al. 2018). The mechanisms for the age-related reduction in RTD, however, are not well understood but likely involve contractile slowing with the preferential loss of skeletal muscle expressing myosin heavy chain (MHC II) in both older men and women (Hunter et al. 1999; Lexell et al. 1988; Purves-Smith et al. 2014; Sundberg et al. 2018a) and deficits in muscle activation (Wallace et al. 2016; Rozand et al. 2019). The level of voluntary activation (neural drive) can be quantified with the interpolated twitch technique of the involved muscles by either electrically-stimulating the nerve or the muscle (Taylor 2009), or stimulating the motor cortex using transcranial magnetic stimulation (TMS) (Todd et al. 2016). A recent meta-analysis reported that for maximal isometric voluntary contractions, older adults have deficits in voluntary activation of skeletal muscles compared with young adults for many muscle groups including the knee extensors (Rozand et al. 2020). Nonetheless, there is limited knowledge on the ability of older adults to activate skeletal muscle during rapid force development of a task. This is in part because of the technical limitations of directly assessing voluntary activation during dynamic contractions (Rozand et al. 2017) and during rapid force development during isometric tasks. While reductions in global surface electromyography (EMG) activity have been associated with lower RTD during dynamic fatiguing contractions in older men compared with young men (Wallace et al. 2016), EMG has some limitations in representing the net neural activation to the muscle (Farina et al. 2004, 2014).
Insight to age-related deficits in activation of skeletal muscle can be gained by comparing the RTD during voluntary isometric contractions with the RTD during involuntary contractions elicited by stimulation of the agonist muscle (Maffiuletti et al. 2016). Involuntary, evoked contractions stimulated via the lower motor neuron, represents the RTD of the skeletal muscle largely independent of neural contributions. Thus, an age-related reduction (young vs. older) in maximal RTD during voluntary contractions compared with involuntary contractions would indicate neural deficits in rapid activation of the muscle in the older adults compared with the younger adults. During voluntary contractions, older adults exhibit reductions in peak RTD compared with young adults (Klass et al. 2008; Barry et al. 2005; Izquierdo et al. 1999; Thompson et al. 2013). Collectively, however, the findings as to whether there are age differences in RTD during involuntary contractions evoked by electrical stimulation the RTD were inconclusive. Jenkins et al. (2014) for example reported similar RTD for young and older men in vastus lateralis muscle (Jenkins et al. 2014). In contrast, the RTD was reported to be less with age in plantar flexor muscles of women (Paasuke et al. 2000), and the dorsiflexor muscles of men and women (Klass et al. 2008). Thus, whether the slower RTD observed during voluntary contractions is due to contractile mechanisms alone or due to deficits in activation is not known. Furthermore, whether there are sex differences that are exacerbated with aging is also not known.
The purpose of this study was to (1) compare age and sex-related differences in RTD between fast voluntary contractions and electrically-evoked contractions to provide insight into the contribution of contractile and neural mechanisms to the RTD, and (2) determine the variability of peak RTD during fast voluntary and electrically-evoked of the knee extensor muscles in young and older men and women. We hypothesized that both young and older adults would have reduced peak RTD for voluntary compared with electrically-evoked contractions and this reduced RTD during voluntary compared with electrically-evoked contractions would be exacerbated in older adults compared with young adults. Given the larger variability in maximality of activation during isometric and slow dynamic contractions with aging (Hunter et al. 2008, 2016; Rozand et al. 2017) we also hypothesized that variability of RTD would be larger for older compared to young adults, particularly for older women (Osawa et al. 2018). We tested both young and older men and women to determine whether there were sex-related differences in RTD with aging, and the possible contractile and neural origins. Global surface EMG was recorded during voluntary contractions to provide insight into muscle activation (Maffiuletti et al. 2016).
Materials and methods
Participants
Forty individuals participated in this study: 20 young adults (19–29 years: 23.0 ± 0.8 years, 10 men and 10 women) and 20 older adults (71–93 years: 78.2 ± 1.5 years, 10 men and 10 women). Prior to participation, each participant provided written informed consent and completed a general health screening survey, which included questions regarding demographic background, past medical history, and current prescription drug therapy. All Participants were healthy, moderately active, non-smoking, community-dwelling adults free of any known neurological, musculoskeletal or cardiovascular diseases and screened Body Mass Index (BMI) and %Bodyfat index using DEXA (General Electric Healthcare, Lunar Prodigy, full-body scanner, Madison, WI) recording. In addition to meeting these inclusion criteria, on the day of the experimental session participants were instructed to: (1) refrain from caffeine for 8 h and alcohol for 24-h prior to testing, (2) consume a normal diet based on their daily routine, and (3) stay hydrated prior to the experimental session. In addition, water was provided ad libitum during the experimental session. All participants self-reported biological sex and self-identified gender, with no participant reporting a difference between their biological sex and self-identified gender. In addition, all participants reported right limb dominance (Oldfield 1971). All the experiments were performed in the afternoon of the day. All The experimental session was completed with the dominant (right) leg knee extensor muscles. All experimental procedures were approved by the Marquette University Institutional Review Board (HR-2945) in accordance with the Declaration of Helsinki for human experimentation.
Experimental protocol
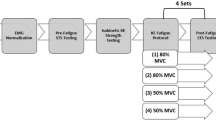
Each participant visited the laboratory for one experimental session that lasted ~ 2 h. The experimental session began with informed consent, followed by familiarization of the procedures, including a demonstration and verbal explanation of each procedure. As an overview, the following procedures were then performed with the knee extensors of the dominant leg: (1) electrical stimulation (ESTIM) of the femoral nerve to evoke twitch contractions; (2) maximum voluntary isometric contractions (MVC); (3) ESTIM coupled with an MVC (MVC-ESTIM); and (4) fast-voluntary isometric contractions at three submaximal torque levels (fast-voluntary contraction). Below is a description of the experimental setup and equipment, followed by more detailed descriptions of the procedures outlined above to obtain the experimental data on the voluntary and involuntary (electrically evoked) RTD.
Experimental set up
Each participant was seated in an upright position (90˚ of hip flexion) in the Biodex system 4 dynamometer (Biodex Medical, Shirley, NY) and secured with two shoulder straps and a strap across the hips to minimize movement and isolate knee extension contractions throughout the experiment (Fig. 1a). The dominant leg was positioned at 90˚ of knee flexion and this axis of rotation of the knee was aligned with the axis of rotation of the dynamometer. The shank of the leg was secured to the lever arm of the dynamometer with a Velcro strap ~ 3.8 cm proximal to the lateral malleolus. Each participant received visual feedback of knee extensor torque via a 24-inch monitor (S240HL, Acer America Corporation, San Jose, CA) placed 1.5 m away at eye level of each participant. The analog signal corresponding to isometric torque of the knee extensor muscles was digitized (500 Hz) via auxiliary output of the dynamometer and recorded through a Micro 1401 analog-to-digital converter (Cambridge Electronic Design, Cambridge, UK) with Spike2 software (Version 6.18, Cambridge Electronic Design, Cambridge, UK).
Experimental protocol. a Schematic of the experimental setup. b Representative data analysis. Shown is the record of a fast voluntary contraction of knee extensors (top). The peak torque was quantified when the first derivative of the torque (middle) crossed zero. The peak RTD quantified as the peak of the first derivative of the torque signal. Interference EMG signal (bottom) of vastus lateralis muscle (light grey) was rectified (dark grey) and 6 Hz low-pass cut-off (black line). c Schematic of the experimental protocol to measure rate of torque development (RTD) during electrically-evoked and fast voluntary contractions. S: single pulse electrical nerve stimulation, D: double pulse electrical nerve stimulation. Numbers indicate the number of trials in each task
Electromyography (EMG)
The EMG signal was recorded during the fast-voluntary contractions and during electrically-evoked twitches contractions to determine the compound muscle action potential (Mwave) of the VL. The EMG signal of the vastus lateralis (VL) was recorded with bipolar Ag/AgCl EMG electrodes (Grass Products, Natus Neurology, Warwich, RI). After cleaning the skin over the muscle belly with alcohol swabs, bipolar electrodes were placed in line with the orientation of the VL muscle fibers with a 20 mm center-to-center distance between the electrodes according to international standards (Hermens et al. 2000) and secured with medical tape to prevent displacement. The EMG signals were amplified (× 100), online-filtered (13–1000 Hz; Coulbourn Instruments, Allentown, PA) and sampled at 2000 Hz with Spike2 software.
ESTIM task
The femoral nerve was electrically stimulated to evoke twitch contractions of the dominant knee extensor muscles and record Mwave from the VL. The femoral artery was palpated in the area of the femoral triangle of the upper thigh and the cathode electrode (Ambu Neuroline electrodes, Denmark; 1.5 cm diameter) was placed lateral to the femoral artery over the femoral nerve. The anode electrode was placed over the greater trochanter of the femur. Single- (200 μs duration, 400 V) and double-pulse (100 Hz, 200 μs duration, 400 V) ESTIM evoked twitch contractions via constant-current stimulator (DS7AH, Digitimer, Ltd., Welwyn Garden City, UK). The intensity of the single-pulse ESTIM was determined by increasing the current until both the twitch contraction torque and the peak-to-peak amplitude of the Mwave of the VL plateaued (Mmax). The current was increased further by 20% to ensure maximal excitation of the femoral nerve (120–660 mA). This supramaximal current determined with single pulse ESTIM was used for single- and double-pulse ESTIM, as has been performed in the knee extensors previously (Cometti et al. 2016).
After the supramaximal intensity was established, three single-pulse ESTIMs and three double-pulse ESTIMs were performed with ~ 10 s between each evoked contraction. For single and double pulse ESTIM, the peak electrically-evoked twitch contraction torque was measured. The peak torque value of the electrically-evoked twitch from each of the three single-pulse ESTIM trials was averaged, and the average peak torque value was used for the fast-voluntary contraction task (see below). Mmax of the VL was elicited with the single-pulse ESTIM and recorded with surface EMG, see Fig. 1b.
MVC task
Participants performed a minimum of three brief (2–3 s) MVC trials. Additional MVC trials (3–6 total trials) were performed until the two highest MVC torque values were within 5%. Each MVC trial was separated by at least 60 s rest. Participants were provided strong verbal encouragement and visual feedback on their performance. The greatest torque achieved by each participant was recorded as the MVC torque and used as the reference to calculate the submaximal target torques. Each MVC performed during the MVC task was performed without electrical stimulation, see Fig. 1c.
MVC-ESTIM task
Participants performed MVC trials with superimposed ESTIM. Single- and double-pulse ESTIM were performed ~ 3 s before and immediately after (~ 3 s) each MVC (four ESTIMs), to obtain a range of involuntary electrically-evoked twitch torque values. The double-pulse ESTIM was expected to elicit a greater twitch torque than single-pulse ESTIM because of the summation of torque, and both single- and double-pulse evoked contractions were expected to be potentiated immediately after the brief MVC (Baudry and Duchateau 2007; Baudry et al. 2008). Participants performed three trials of the MVC-ESTIM task with 2 min rest between each trial, see Fig. 1c.
Fast-voluntary isometric contraction task
Participants performed 10 trials of fast-voluntary contractions at three submaximal torque levels (30 contractions total) increasing the torque as fast as possible. The submaximal torque levels included 25% MVC, 50% MVC and a torque level that was matched to the torque evoked by a non-potentiated single-pulse ESTIM which on average was 17.6 ± 1.0%MVC (100% ESTIMsingle). The submaximal target torque level for each block of 10 voluntary contractions was block-randomized.
During each submaximal fast-voluntary contraction, participants were instructed to perform the submaximal knee extension contractions “as fast as possible”, because the speed of force generation was the primary aim of this task. A horizontal target torque line was displayed on a monitor with visual feedback of knee extension torque during practice trials only, because presenting the target line during the task may reduce the speed of force generation (Fitts 1954). Participants performed practice trials until participants were able to relatively approximate the target torque (± 5% MVC) as fast as possible prior to performing the block of 10 fast-voluntary contractions. After 5 min rest following the practice trials, the target line was removed from the visual feedback and participants performed the block of 10 fast-voluntary contractions. Participants rested for 5 s between each submaximal contraction and 3 min between each block of 10 submaximal contractions, see Fig. 1c.
Data analysis
Data were analyzed using custom algorithms (Matlab® R2017a, Math Works™ Inc., Natick, Massachusetts, USA). MVC torque was quantified as the average value over a 0.5 s before and after the peak torque achieved during the MVC task. For submaximal contractions (both fast-voluntary and ESTIM-evoked contractions), the peak torque was calculated as the torque value at which the first derivative of the torque signal crossed zero (Klass et al. 2008). The peak RTD was calculated using a 10-ms window as the peak value of the first derivative of the torque signal between the start and end of the contraction (Fig. 1b).
The EMG burst during the fast-voluntary contraction was quantified as the rectified and smoothed interference EMG using a fourth-order Butterworth digital filter with 6 Hz cutoff frequency (Kwon et al. 2014). The rate of EMG rise was calculated using 10-ms window as the peak value of the first derivative of the EMG burst signal between the start and the end of the contraction. The onset of EMG burst signal was determined as the first point exceeded 3 standard deviations (SD) above the quiescent EMG value during a period of inactivity of the VL. The EMG of the VL during fast-voluntary contractions was normalized to the peak-to-peak amplitude of Mmax elicited by ESTIM, to normalize for differences between groups in EMG amplitude due to physiological differences in impedance (e.g., subcutaneous fat) (Lanza et al. 2018).
Statistical analysis
Two-way mixed model analyses of variances (ANOVAs) with two between subject factors [sex (men vs. women) and age (young vs. older)] and two within subject factors [contraction type (voluntary vs. electrically-evoked) and torque level (25%MVC, 50%MVC, 100%ESTIMsingle)] were used to compare contractile function (peak twitch force and RTD), muscle activation (EMG burst and peak rate of EMG rise) and variability of RTD. For each ANOVA the sphericity of data was verified, and technical corrections were performed if necessary. Post hoc analyses [Tukey’s Honest Significant Difference (HSD)] was used to test for differences between torque levels (25%MVC, 50%MVC, 100%ESTIMsingle) when significant main effects or interactions of torque level were identified and post hoc Students t test were used to test for interactions of age and sex. All alpha level for all statistical tests was 0.05 and Bonferroni corrected P values for multiple comparisons (P < 0.025) were used for all post hoc analyses. The effect sizes were yield as partial eta squared (\(\eta_{{\text{p}}}^{2}\)).
The electrically-evoked twitch amplitudes from the twelve stimulations [4 ESTIMs (2 single- and 2 double pulse) × 3 trials] elicited during the MVC-ESTIM task were torque-matched with twelve voluntary twitch amplitudes from the fast-voluntary contraction task. Thus, only twelve fast contraction trials were selected out of 30 fast contraction trials (10 trials × 3 torque levels) and these data were pooled across participants to compare the RTD across a range of torque values. Linear regression models were used to determine associations between the RTD and EMG burst compared with torque for voluntary and electrically-evoked contractions for these torque-matched twitches. The slopes of the linear regression models between RTD from the voluntary and electrically-evoked contractions were compared separately for each group (young and older men and women) using an analysis of covariance (ANCOVA).
Variability of the RTD was quantified using the residual of each data point from the regression model of each twitch contraction as function of torque level. The residual indicated the difference between the actual data point to the predicted data point from the regression model and the sum and mean of the residuals are theoretically zero. Thus, the variability of RTD with increasing level of torque was quantified as Root Mean Square Error (RMSE) of residual. The following formula was used to calculate RMSE:
where n = number of data points. Significance was determined at P < 0.05, and all the analyses were performed with IBM Statistical Package for Social Sciences version 24 statistical package (IBM, Armonk, NY, USA). Data are presented as mean ± SD in the text and tables and mean ± standard error (SEM) in the figures.
Results
Muscle strength and peak twitch torque
The peak knee extension MVC torque was greater for men compared with women (F1,36 = 7.33, p = 0.01, \(\eta_{{\text{p}}}^{2}\) = 0.13) and greater for the young than older adults (F1,36 = 36.22, p < 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.68), see Table 1. The age-related deficit in MVC torque was greater for men than women (age × sex: − 51% vs. − 35%, F1,36 = 25.3, p < 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.5).
The torque during electrically-evoked twitch contractions was greater for men compared with women for single-pulse (F1,36 = 17.04, p = 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.21) and double-pulse (F1,36 = 26.89, p < 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.32) stimulations and for young compared with older adults for single- (F1,36 = 18.6, p < 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.26) and double-pulse stimulations (F1,36 = 28.9, p < 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.34), see Table 1. However, the relative peak torque of electrically-evoked twitch contractions (%MVC) was not different between men and women for single- (F1,36 = 0.5, p = 0.83, \(\eta_{{\text{p}}}^{2}\) < 0.01) or double-pulse (F1,36 = 0.001, p = 0.97, \(\eta_{{\text{p}}}^{2}\) < 0.01) stimulations nor for young compared with older adults for single- (F1,36 = 1.84, p = 0.19, \(\eta_{{\text{p}}}^{2}\) < 0.01) or double-pulse (F1,36 = 1.59, p = 0.28, \(\eta_{{\text{p}}}^{2}\) < 0.01), see Table 1.
As expected, independent of age and sex, the torque produced during the fast-voluntary contractions was greater with increased level of target torque (F2,34 = 153.6, p < 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.7). Post hoc analysis indicated that 50% MVC twitch was greater than 25% MVC twitch (t = − 8.88, p < 0.001), and both were lesser than the 100% ESTIMsingle (vs. 25% MVC, t = -5.58, p < 0.001; vs. 50% MVC, t = − 8.91 p < 0.001).
Similar to the fast-voluntary contractions, a range of submaximal torques was elicited with the ESTIM. The double-pulse ESTIM evoked a larger twitch torque response than the single-pulse ESTIM (36.9 ± 2.2%MVC vs. 20.5 ± 1.7%MVC, p < 0.001). Additionally, the amplitude of torque of the potentiated electrically-evoked twitches was greater than non-potentiated twitch torque for single- (24.4 ± 1.4% MVC vs. 16.7 ± 1.0%MVC, p < 0.001 and double-pulse ESTIM (39.3 ± 2.0% MVC vs. 34.6 ± 2.0%MVC, p = 0.05).
Peak rate of torque development
The peak RTD during the fast-voluntary contractions was greater for the young compared with older adults (F1,36 = 12.62, p = 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.13), but was not different between men and women (F1,36 = 0.39, p = 0.54, \(\eta_{{\text{p}}}^{2}\) < 0.01), see Table 1. During the electrically-evoked contractions, however, the peak RTD was not different for young compared with older adults (F1,36 = 1.31, p = 0.26, \(\eta_{{\text{p}}}^{2}\) < 0.01) nor between men and women (562.1 ± 20.1 vs. 540.1 ± 12.1%MVC·s−1; F1,36 = 0.22, p = 0.65, \(\eta_{{\text{p}}}^{2}\) < 0.01).
The peak RTD was greater for electrically-evoked compared with fast-voluntary contractions (F1,36 = 384, p < 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.8) for each contraction intensity (25% MVC, 50% MVC, 100% ESTIMsingle, torque × intensity interaction, F2,72 = 242, p < 0.001, \(\eta_{{\text{p}}}^{2}\) < 0.78) when trials were pooled across the groups (young and older men and women). The greater RTD for electrically-evoked compared with fast-voluntary contractions was not different for men (37 ± 4%) and women (sex effect, 36 ± 2%, F1,36 = 0.19, p = 0.89, \(\eta_{{\text{p}}}^{2}\) < 0.01). However, the difference between RTD for electrically-evoked and fast-voluntary contractions was greater for the older adults (43.7 ± 2.3%) compared with the young adults (38.4 ± 2.7%; age effect, F1,36 = 4.58, p = 0.04 \(\eta_{{\text{p}}}^{2}\) = 0.06), see Table 1.
Association between torque and peak RTD
Peak RTD was positively associated with relative submaximal torque for fast-voluntary (R2 = 0.73, p < 0.001) and electrically-evoked twitch contractions (R2 = 0.88, p < 0.001) for all groups. The linear slope of the regression models between peak RTD and submaximal torque was greater for electrically-evoked twitches than the fast voluntary twitch contraction (y = 18.46x + 10.31 vs. y1 = 11.33x1 + 27.19; F1,36 = 8.61, p = 0.004) for both men and women (sex effect, F1,36 = 0.45, p = 0.84). However, the magnitude of the correlation coefficient for electrically-evoked compared with fast voluntary twitch contractions was larger for older adults (F1,36 = 60.58, p < 0.001) compared with younger adults (F1,36 = 8.39, p = 0.03), see Fig. 2.
Association of RTD and Torque. For fast voluntary (unfilled circles) and electrically-evoked (filled circles) contractions, the rate of torque development (RTD) was associated peak, relative torque produced (% maximal voluntary contraction, MVC) by the contraction. For both older (a) and young (b) men (blue circles) and women (red circles), the correlation coefficient was greater (steeper slope) and the variance was less (larger R2) for electrically-evoked than fast voluntary contractions
EMG rise
During the fast-voluntary contractions, the rate of EMG rise was greater with increasing level of torque (F2,72 = 26.14, p < 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.41). The young adults exhibited a greater rate of EMG rise compared with the older adults (41.0 ± 3.9%Mmax s−1 vs. 19.3 ± 2.3%Mmax s−1 age effect, F1,36 = 12.43, p < 0.001, \(\eta_{{\text{p}}}^{2}\) = 0.27). Similarly, men exhibited a greater rate of EMG rise compared with women (38.4 ± 4.3%Mmax s−1 vs. 28.0 ± 3.7%Mmax s−1: sex effect, F1,36 = 4.23, p = 0.04, \(\eta_{{\text{p}}}^{2}\) = 0.04).
Associations between RTD and EMG rise
The rate of EMG rise was positively associated with the RTD during the fast-voluntary contractions (R2 = 0.15, p < 0.001). However, when the young and older groups were examined separately, the rate of EMG rise was associated with the RTD for the young (R2 = 0.32, p < 0.001) but not the older adults (R2 = 0.01, p = 0.3), see Fig. 3.
Variability of RTD
During the fast-voluntary contractions, the RMSE of the RTD was greater for the older adults compared with the young (F1,3 = 14.23, p = 0.03, \(\eta_{{\text{p}}}^{2}\) = 0.03). The variability of the RTD was greater for women compared with men (F1,3 = 10.51, p = 0.04, \(\eta_{{\text{p}}}^{2}\) = 0.03). The post hoc analysis indicated that the variability of RTD was greater for older women compared with the older men (t = 4.35, p < 0.001) with no differences between the young men and women (t = − 1.88, p = 0.06).
During the electrically-evoked contractions, the variability of the RTD was similar between the young and older adults (F1,3 = 0.59, p = 0.5, \(\eta_{{\text{p}}}^{2}\) = 0.02). The variability of the RTD was similar for women and men (F1,3 = 1.56, p = 0.3, \(\eta_{{\text{p}}}^{2}\) = 0.03) in both age groups, see Fig. 4.
Variability of RTD. During the electrically-evoked contractions, there were no differences in the variability of the rate of torque development (RTD) for young (filled circles) and older (unfilled circles) men (blue circles) (a), and young and older women (red circles) (c). However, during the fast-voluntary contractions, overall older exhibited greater RTD variability than young but specifically in older adults, older women (d: red unfilled circles) was greater RTD variability than older men (b). Dashed line represents 95% confidential line for young (dark) and older (grey) and # represents between group significance at the level of 0.05
Discussion
The novel findings of this study are that there were no age-related or sex-related differences in peak RTD for the electrically-evoked twitch contractions of the knee extensor muscles, but during the fast-voluntary contractions, the peak RTD was less for older compared with young adults. Furthermore, the peak RTD during the fast-voluntary contractions, was more variable between trials among the older than the young adults, particularly the older women, with no age differences in variability for the electrically-evoked contractions. These data suggest that neural mechanisms primarily contributed to the age-related reductions in RTD of the knee extensor muscles. Because the twitch torque and RTD were normalized to maximal strength, the observed lower peak RTD of older adults during the fast-voluntary contractions was not due to strength-related differences between young and older adults. Furthermore, the greater RTD during the voluntary isometric contractions was positively associated with a larger rate of rise of EMG burst for the young adults, but not the older adults. Together, these observations, provide new evidence that the slower torque development in older men and women is due to limitations in the ability to provide rapid neural drive to the knee extensor muscles, and this is independent of age-related strength reductions.
The peak RTD during the electrically-evoked contractions was similar for young and older adults, demonstrating that the maximal RTD was maintained in older age. Despite the maintained RTD, older adults have a weaker, less powerful and slower muscle compared to young adults (Hunter et al. 2016) primarily due to an age-related reduction in the proportional area of ‘fast fibers’ (myosin-heavy chain II skeletal muscle fibers) (Larsson et al. 1979, 1997; Sundberg et al. 2018b; Hunter et al. 1999). Additionally, the age-related reduction in the number and function of lower motor neurons (Hepple and Rice 2016) can contribute to lower maximal contractile capacity of older than young adults. Accordingly, we found that because of the age-related attenuation in muscle strength and power, the absolute RTD during the electrically-evoked contractions was ~ 60% lower for older (584.1 ± 18.4 Nm s−1) adults compared with the young (953.8 ± 35.0 Nm s−1) although there was no difference in the relative RTD between young and older adults. The attenuated RTD during the fast-voluntary contractions compared with electrically-evoked contractions was greater in older compared to young adults and this age-related difference in RTD was exacerbated during higher force contractions. These observations of an age difference in the RTD between the evoked and fast-voluntary contraction are consistent with previous studies in elbow flexor (Barry et al. 2005), dorsiflexor (Klass et al. 2008) and leg extensor muscles (Izquierdo et al. 1999) and suggest that older adults have a slowing in the activation of the muscle.
Older adults also exhibited a greater variability of RTD only during the fast-voluntary contractions, particularly the older women. This greater variability for older adults was exacerbated at higher force levels. Thus, older adults have a reduced and more variable RTD (particularly older women) of the knee extensor muscles during the fast-voluntary contractions which are exacerbated at higher force levels and may pose a significant impairment in the ability to recover from tripping and increased risk of falling. Because our data suggest this age-related variability is of neural original, voluntary RTD may be improved with exercise training targeting the central nervous system (Barry et al. 2005).
The age-related reductions in maximal RTD are likely due to the aggregate effects of multiple mechanisms along the motor pathway from the motor cortex through the corticospinal tract down to the skeletal muscle fibers (Hunter et al. 2016). Recent meta-analysis found that older adults have reduced voluntary activation during maximal contractions with the knee extensors among other muscles (Rozand et al. 2020); however, the data on fast contractions was limited due to technical difficulties (Rozand et al. 2017). Although we recently found reduced corticospinal excitability in older compared with young adults (Rozand et al. 2019), the age-related differences were observed for the first dorsal interosseous and not the knee extensor muscles (Hassanlouei et al. 2017). However, Klass et al. reported a blunted motor unit discharge frequency and number of doublet discharges that likely contributed to reduced maximal RTD in the ankle dorsiflexor muscles of older adults (Klass et al. 2008). More recently, Del Vecchio et al. described the strategies used by the central nervous system attain maximal RTD using high-density surface EMG in young men (Del Vecchio et al. 2019b). Del Vecchio et al. found that maximal RTD is limited by synaptic input received by the lower, alpha motor neurons before force generations is initiated (Del Vecchio et al. 2019b). Additionally, Del Vecchio et al. also found that it is possible to increase motor neuron output from the spinal cord to the muscle with strength training in young men (Del Vecchio et al. 2019a). Thus, although our data and others show that neural drive is reduced during voluntary contractions, it may be possible to ‘train’ the motor cortical pathway to augment motor unit recruitment/discharge rates to increase RTD in older adults.
In conclusion, we found the peak RTD was similar for young and older men and women during the electrically-evoked twitch contractions; however, the RTD for the fast- voluntary contractions was slower for older adults than young adults indicating neural deficits with aging in the ability of older adults to activate the knee extensors muscles. Additionally, older women were more variable in the ability to rapidly develop voluntarily torque with the knee extensors muscles compared with young women and young and older men. Taken together with the associations between muscle activation (EMG) and peak RTD in the young but not older adults, our findings provide robust evidence that the age-related reduction in peak RTD during voluntary contractions of the lower limb muscle was due to deficits in neural activation in older men and women. Thus, healthy and community dwelling older men, and, particularly older women, have a reduced and more variable ability to activate their muscle rapidly, possibly predisposing them to a greater risk of falling.
Abbreviations
- ANCOVA:
-
Analysis of covariance
- ANOVA:
-
Analysis of variance
- EMG:
-
Electromyography
- ESTIM:
-
Electrical stimulation
- MVC:
-
Maximum voluntary isometric contraction
- RMSE:
-
Root mean square error
- RTD:
-
Rate of Torque Development
- SD:
-
Standard deviation
- SEM:
-
Standard error of measurement
References
Aagaard P (2003) Training-induced changes in neural function. Exerc Sport Sci Rev 31(2):61–67
Barry BK, Warman GE, Carson RG (2005) Age-related differences in rapid muscle activation after rate of force development training of the elbow flexors. Exp Brain Res 162(1):122–132. https://doi.org/10.1007/s00221-004-2127-3
Baudry S, Duchateau J (2007) Postactivation potentiation in a human muscle: effect on the rate of torque development of tetanic and voluntary isometric contractions. J Appl Physiol (Bethesda, Md: 1985) 102(4):1394–1401. https://doi.org/10.1152/japplphysiol.01254.2006
Baudry S, Klass M, Duchateau J (2008) Postactivation potentiation of short tetanic contractions is differently influenced by stimulation frequency in young and elderly adults. Eur J Appl Physiol 103(4):449–459. https://doi.org/10.1007/s00421-008-0739-1
Bento PC, Pereira G, Ugrinowitsch C, Rodacki AL (2010) Peak torque and rate of torque development in elderly with and without fall history. Clin Biomech (Bristol, Avon) 25(5):450–454. https://doi.org/10.1016/j.clinbiomech.2010.02.002
Cometti C, Babault N, Deley G (2016) Effects of constant and doublet frequency electrical stimulation patterns on force production of knee extensor muscles. PLoS ONE 11(5):e0155429. https://doi.org/10.1371/journal.pone.0155429
Del Vecchio A, Casolo A, Negro F, Scorcelletti M, Bazzucchi I, Enoka R, Felici F, Farina D (2019a) The increase in muscle force after 4 weeks of strength training is mediated by adaptations in motor unit recruitment and rate coding. J Physiol 597(7):1873–1887. https://doi.org/10.1113/jp277250
Del Vecchio A, Negro F, Holobar A, Casolo A, Folland JP, Felici F, Farina D (2019b) You are as fast as your motor neurons: speed of recruitment and maximal discharge of motor neurons determine the maximal rate of force development in humans. J Appl Physiol 597(9):2445–2456. https://doi.org/10.1113/JP277396
Doherty TJ (2003) Invited review: aging and sarcopenia. J Appl Physiol 95(4):1717–1727. https://doi.org/10.1152/japplphysiol.00347.2003
Farina D, Merletti R, Enoka RM (2004) The extraction of neural strategies from the surface EMG. J Appl Physiol (Bethesda, Md: 1985) 96(4):1486–1495. https://doi.org/10.1152/japplphysiol.01070.2003
Farina D, Merletti R, Enoka RM (2014) The extraction of neural strategies from the surface EMG: an update. J Appl Physiol (Bethesda, Md: 1985) 117(11):1215–1230. https://doi.org/10.1152/japplphysiol.00162.2014
Fitts PM (1954) The information capacity of the human motor system in controlling the amplitude of movement. J Exp Psychol 47(6):381–391
Gerstner GR, Thompson BJ, Rosenberg JG, Sobolewski EJ, Scharville MJ, Ryan ED (2017) Neural and Muscular contributions to the age-related reductions in rapid strength. Med Sci Sports Exerc 49(7):1331–1339. https://doi.org/10.1249/MSS.0000000000001231
Hassanlouei H, Sundberg CW, Smith AE, Kuplic A, Hunter SK (2017) Physical activity modulates corticospinal excitability of the lower limb in young and old adults. J Appl Physiol (Bethesda, Md: 1985) 123(2):364–374. https://doi.org/10.1152/japplphysiol.01078.2016
Hepple RT, Rice CL (2016) Innervation and neuromuscular control in ageing skeletal muscle. J Appl Physiol 594(8):1965–1978. https://doi.org/10.1113/JP270561
Hermens FL, Merletti R, Hägg G, Stegeman D, Blok JRCD-K (2000) SENIAM 8: European recommendations for surface ElectroMyoGraphy. Roessingh Research and Development, Enschede
Hunter SK, Thompson MW, Ruell PA, Harmer AR, Thom JM, Gwinn TH, Adams RD (1999) Human skeletal sarcoplasmic reticulum Ca2+ uptake and muscle function with aging and strength training. J Appl Physiol (Bethesda, Md: 1985) 86(6):1858–1865
Hunter SK, Todd G, Butler JE, Gandevia SC, Taylor JL (2008) Recovery from supraspinal fatigue is slowed in old adults after fatiguing maximal isometric contractions. J Appl Physiol 105(4):1199–1209. https://doi.org/10.1152/japplphysiol.01246.2007
Hunter SK, Pereira HM, Keenan KG (2016) The aging neuromuscular system and motor performance. J Appl Physiol (Bethesda, Md: 1985) 121(4):982–995. https://doi.org/10.1152/japplphysiol.00475.2016
Izquierdo M, Aguado X, Gonzalez R, Lopez JL, Hakkinen K (1999) Maximal and explosive force production capacity and balance performance in men of different ages. Eur J Appl Physiol 79(3):260–267. https://doi.org/10.1007/s004210050504
Jenkins ND, Buckner SL, Cochrane KC, Bergstrom HC, Palmer TB, Johnson GO, Schmidt RJ, Housh TJ, Cramer JT (2014) Age-related differences in rates of torque development and rise in EMG are eliminated by normalization. Exp Gerontol 57:18–28. https://doi.org/10.1016/j.exger.2014.04.015
Klass M, Baudry S, Duchateau J (2008) Age-related decline in rate of torque development is accompanied by lower maximal motor unit discharge frequency during fast contractions. J Appl Physiol (Bethesda, Md: 1985) 104(3):739–746. https://doi.org/10.1152/japplphysiol.00550.2007
Kwon M, Chen YT, Fox EJ, Christou EA (2014) Aging and limb alter the neuromuscular control of goal-directed movements. Exp Brain Res 232(6):1759–1771. https://doi.org/10.1007/s00221-014-3868-2
Lanza MB, Balshaw TG, Massey GJ, Folland JP (2018) Does normalization of voluntary EMG amplitude to MMAX account for the influence of electrode location and adiposity? Scand J Med Sci Sports 28(12):2558–2566. https://doi.org/10.1111/sms.13270
LaRoche DP, Cremin KA, Greenleaf B, Croce RV (2010) Rapid torque development in older female fallers and nonfallers: a comparison across lower-extremity muscles. J Electromyogr Kinesiol 20(3):482–488. https://doi.org/10.1016/j.jelekin.2009.08.004
Larsson L, Grimby G, Karlsson J (1979) Muscle strength and speed of movement in relation to age and muscle morphology. J Appl Physiol Respir Environ Exerc Physiol 46(3):451–456. https://doi.org/10.1152/jappl.1979.46.3.451
Larsson L, Li X, Frontera WR (1997) Effects of aging on shortening velocity and myosin isoform composition in single human skeletal muscle cells. The American journal of physiology 272(2 Pt 1):C638–649
Lexell J, Taylor CC, Sjostrom M (1988) What is the cause of the ageing atrophy? Total number, size and proportion of different fiber types studied in whole vastus lateralis muscle from 15- to 83-year-old men. J Neurol Sci 84(2–3):275–294. https://doi.org/10.1016/0022-510x(88)90132-3
Maffiuletti NA, Aagaard P, Blazevich AJ, Folland J, Tillin N, Duchateau J (2016) Rate of force development: physiological and methodological considerations. Eur J Appl Physiol 116(6):1091–1116. https://doi.org/10.1007/s00421-016-3346-6
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9(1):97–113
Osawa Y, Studenski SA, Ferrucci L (2018) Knee extension rate of velocity development affects walking performance differently in men and women. Exp Gerontol 112:63–67. https://doi.org/10.1016/j.exger.2018.09.005
Paasuke M, Ereline J, Gapeyeva H, Sirkel S, Sander P (2000) Age-related differences in twitch contractile properties of plantarflexor muscles in women. Acta Physiol Scand 170(1):51–57. https://doi.org/10.1046/j.1365-201x.2000.00753.x
Purves-Smith FM, Sgarioto N, Hepple RT (2014) Fiber typing in aging muscle. Exerc Sport Sci Rev 42(2):45–52. https://doi.org/10.1249/JES.0000000000000012
Rozand V, Senefeld JW, Hassanlouei H, Hunter SK (2017) Voluntary activation and variability during maximal dynamic contractions with aging. Eur J Appl Physiol 117(12):2493–2507. https://doi.org/10.1007/s00421-017-3737-3
Rozand V, Senefeld JW, Sundberg CW, Smith AE, Hunter SK (2019) Differential effects of aging and physical activity on corticospinal excitability of upper and lower limb muscles. J Neurophysiol 122(1):241–250. https://doi.org/10.1152/jn.00077.2019
Rozand V, Sundberg CW, Hunter SK, Smith AE (2020) Age-related deficits in voluntary activation: a systematic review and meta-analysis. Med Sci Sports Exerc 52(3):549–560. https://doi.org/10.1249/MSS.0000000000002179
Sundberg CW, Hunter SK, Trappe SW, Smith CS, Fitts RH (2018a) Effects of elevated H(+) and Pi on the contractile mechanics of skeletal muscle fibres from young and old men: implications for muscle fatigue in humans. J Physiol 596(17):3993–4015. https://doi.org/10.1113/JP276018
Sundberg CW, Kuplic A, Hassanlouei H, Hunter SK (2018b) Mechanisms for the age-related increase in fatigability of the knee extensors in old and very old adults. J Appl Physiol (Bethesda, Md: 1985) 125(1):146–158. https://doi.org/10.1152/japplphysiol.01141.2017
Taylor JL (2009) Point: the interpolated twitch does/does not provide a valid measure of the voluntary activation of muscle. J Appl Physiol (Bethesda, Md: 1985) 107(1):354–355. https://doi.org/10.1152/japplphysiol.91220.2008
Thompson BJ, Ryan ED, Sobolewski EJ, Conchola EC, Cramer JT (2013) Age related differences in maximal and rapid torque characteristics of the leg extensors and flexors in young, middle-aged and old men. Exp Gerontol 48(2):277–282. https://doi.org/10.1016/j.exger.2012.10.009
Thompson BJ, Ryan ED, Herda TJ, Costa PB, Herda AA, Cramer JT (2014) Age-related changes in the rate of muscle activation and rapid force characteristics. Age (Dordrecht, Netherlands) 36(2):839–849. https://doi.org/10.1007/s11357-013-9605-0
Todd G, Taylor JL, Gandevia SC (2016) Measurement of voluntary activation based on transcranial magnetic stimulation over the motor cortex. J Appl Physiol (Bethesda, Md: 1985) 121(3):678–686. https://doi.org/10.1152/japplphysiol.00293.2016
Wallace JW, Power GA, Rice CL, Dalton BH (2016) Time-dependent neuromuscular parameters in the plantar flexors support greater fatigability of old compared with younger males. Exp Gerontol 74:13–20. https://doi.org/10.1016/j.exger.2015.12.001
Acknowledgements
We thank Jose D. Delgadillo for the assistance with data acquisition.
Funding
This work was support by National Institute of Aging Grant (R01- AG048262) to SKH.
Author information
Authors and Affiliations
Contributions
MK, JWS and SKH conceived and designed research; MK performed experiments; MK analyzed data; MK, JWS and SKH interpreted results of experiments; MK prepared figures; MK, JWS and SKH drafted manuscript; MK, JWS and SKH edited and revised manuscript; MK, JWS and SKH approved final version of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest, financial or otherwise, are declared by the authors.
Additional information
Communicated by William J. Kraemer.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kwon, M., Senefeld, J.W. & Hunter, S.K. Attenuated activation of knee extensor muscles during fast contractions in older men and women. Eur J Appl Physiol 120, 2289–2299 (2020). https://doi.org/10.1007/s00421-020-04451-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-020-04451-0