Abstract
Purpose
To determine the impact of altering dietary sodium intake for 3 days preceding exercise on sweat sodium concentration [Na+], and cardiovascular and thermoregulatory variables.
Methods
Fifteen male endurance athletes (runners n = 8, cyclists n = 7) consumed a low (LNa, 15 mg kg−1 day−1) or high (HNa, 100 mg kg−1 day−1) sodium diet, or their usual free-living diet [UDiet, 46 (37–56) mg kg−1 day−1] for 3 days in a double-blind, randomized cross-over design, collecting excreted urine (UNa) and refraining from exercise. On day 4, they completed 2 h running at 55% \(\dot{V}\)O2max or cycling at 55% maximum aerobic power in Tamb 35 °C. Pre- and post-exercise blood samples were collected, and sweat from five sites using absorbent patches along the exercise protocol.
Results
UNa on days 2–3 pre-exercise [mean (95% CI) LNa 16 (12–19) mg kg−1 day−1, UDiet 46 (37–56) mg kg−1 day−1, HNa 79 (72–85) mg kg−1 day−1; p < 0.001] and pre-exercise aldosterone [LNa 240 (193–286) mg kg−1 day−1, UDiet 170 (116–224) mg kg−1 day−1, HNa 141 (111–171) mg kg−1 day−1; p = 0.001] reflected sodium intake as expected. Pre-exercise total body water was greater following HNa compared to LNa (p < 0.05), but not UDiet. Estimated whole-body sweat [Na+] following UDiet was 10–11% higher than LNa and 10–12% lower than HNa (p < 0.001), and correlated with pre-exercise aldosterone (1st h r = − 0.568, 2nd h r = − 0.675; p < 0.01). Rectal temperature rose more quickly in LNa vs HNa (40–70 min; p < 0.05), but was similar at the conclusion of exercise, and no significant differences in heart rate or perceived exertion were observed.
Conclusions
Three day altered sodium intake influenced urinary sodium excretion and sweat [Na+], and the rise in rectal temperature, but had no effect on perceived exertion during moderate-intensity exercise in hot ambient conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
During endurance exercise, metabolic heat production results in the production of sweat, to reduce body temperature through evaporation from the skin (Sawka et al. 2007). The composition of sweat includes significant quantities of sodium (Na+), which is the most abundant cation, present in typical concentrations of 12–105 mmolL−1 (Baker et al. 2016). As a result, endurance exercise, particularly in hot ambient conditions, can lead to substantial sodium losses, albeit proportionally less than water (Shirreffs and Sawka 2011). This has placed much emphasis on the dietary sodium needs of endurance athletes by researchers (Baker et al. 2016; Shirreffs and Sawka 2011), athletes, and their support networks (McCubbin et al. 2019). However, to date, there are no quantifiable guidelines for sodium intake before, during, or after endurance and ultra-endurance exercise (Hoffman et al. 2018; Thomas et al. 2016).
There are several factors that can influence the sodium concentration ([Na+]) in sweat during exercise. The initial sweat produced by the secretory coil of the sweat gland is generally similar [Na+] to plasma (Sato et al. 1989). Therefore, factors that affect fluid balance, and hence plasma [Na+] both before and during exercise, are likely to influence sweat [Na+] (Morgan et al. 2004). Sweat composition is also altered through ion reabsorption in the reabsorptive duct as it travels towards the skin surface (Sato et al. 1989). The flow rate of sweat through the duct impacts on the ability for ion reabsorption, such that high flow rates, as occurs from increased rates of sweat production, reduce the completeness of reabsorption and result in higher sweat [Na+] (Buono et al. 2008). The rate of sweat production is influenced by multiple factors during exercise, including exercise intensity, ambient conditions, and airflow over the skin (Holmes et al. 2016; Saunders et al. 2005; Sawka et al. 2007). The reabsorptive capacity of the sweat gland is also at least partially regulated, with heat acclimation shown to significantly increase Na+ reabsorption, resulting in lowered sweat [Na+] (Chinevere et al. 2008).
Dietary sodium intake is also thought to influence the reabsorptive capacity of sweat glands, with McCance (1938), showing that inducing sodium deficiency through the combination of dietary restriction and sweating results in a progressive reduction in sweat [Na+] to conserve total body sodium stores. However, this and other earlier studies primarily collected sweat samples during low-intensity exercise or over both exercise and rest periods, in sedentary or untrained populations, and with sodium intakes that do not reflect those typical of endurance athletes (Conn et al. 1946; Ramanathan et al. 1956; Robinson et al. 1950, 1955). A recent study that surveyed endurance athletes found that 58% intended to either deliberately, or unintentionally through increased overall food intake, increase sodium consumption compared to their usual diet in the days preceding competition, and for a typical period of 2–5 days, whereas only 3% intended to reduce sodium intake (McCubbin et al. 2019). For researchers studying the effect of sweat sodium replacement during exercise, the impact of altered dietary sodium intake in the days preceding exercise also represents a potential confounding variable, in that attempts to replace a specific proportion of sweat sodium losses relies on accurate estimations of expected losses in the first place. For this to occur, the impact of dietary sodium intake on sweat [Na+] must be understood and controlled for if necessary.
The influence of dietary sodium intake on sweat [Na+], in athletes and specifically during endurance exercise, remains unclear (McCubbin and Costa 2018). The varied results reported between studies may be due to several factors, including poor validation of sodium balance in the days preceding exercise, collection of sweat samples from limited regional sites, averaging sweat [Na+] data across multiple body sites or collection days, and insufficient statistical analysis or reporting (McCubbin and Costa 2018). Furthermore, to date, no studies have provided participants with dietary sodium proportional to body mass. Given that both total body sodium stores (Kennedy et al. 1983) and total body water (TBW) (Watson et al. 1980) are proportional to body mass, it would seem prudent for dietary sodium interventions to follow this approach, to achieve consistency in whole-body sodium balance between participants.
Changes in dietary sodium intake preceding exercise may also influence cardiovascular function and thermoregulation, through changes in plasma volume (Pv), plasma osmolality (POsm), and plasma [Na+] (Sims et al. 2007a, b; Hamouti et al. 2014; Armstrong et al. 1985; Koenders et al. 2017). Sodium loading 2–3-h pre-exercise (38 mg kg−1) has been shown to expand Pv 2–5% when consumed with 10 mL kg−1 water, with minimal or no change in POsm (Sims et al. 2007a, b; Hamouti et al. 2014). However, few studies have altered sodium intake for 1–5 days as frequently practiced by athletes (McCubbin et al. 2019), or have done so during a simultaneous period of heat acclimation (Armstrong et al. 1985; Konikoff et al. 1986), creating difficulties in their interpretation. An increased Pv prior to exercise in response to increasing sodium intake, if maintained throughout the exercise bout, has potential to increase stroke volume and cutaneous blood flow, resulting in reductions in heart rate and core body temperature, respectively (Trangmar and González-Alonso 2017). Altered POsm may also influence sweat rate, independently of Pv. (Takamata et al. 1995, 2001). However, to date, the effect of POsm has not specifically been studied in an exercise model of heat stress, using the 1–5-day time-frame in which athletes typically alter dietary sodium intake.
The purpose of this study was, therefore, to investigate the effect of 3 days of high and low dietary sodium intakes, proportional to body mass, on sodium balance and associated variables (urinary sodium excretion (UNa), plasma and sweat [Na+]) before and during endurance exercise in the heat, and also to investigate the subsequent effects on heart rate, rectal temperature, thermal comfort and perceived exertion, and to compare these to the participant’s usual free-living diet. We hypothesized that there would be a significant difference in UNa, plasma, and sweat [Na+] between dietary conditions, despite only a 3-day dietary intervention, but minimal difference in hydration status, cardiovascular, or thermoregulatory variables.
Methods
Ethical approval
This study conformed to the standards set by the Declaration of Helsinki, and was approved by the Monash University Human Research Ethics Committee. All participants gave written informed consent prior to participating in the study.
Participants
Fifteen non heat-acclimatized, endurance-trained male runners, cyclists, and triathletes volunteered to participate in this study (mean ± SD: age 40 ± 5 yr, height 179 ± 5 cm, body mass 77.1 ± 5.0 kg, body fat mass 17.1 ± 4.8%, training volume 8.6 ± 5.5 h wk−1, and V̇O2max 55.6 ± 4.7 mL kg−1 min−1). Participants were excluded if they had known Cystic Fibrosis, renal failure or other chronic conditions that impair kidney or sweat gland function, or musculoskeletal injury that would impair their ability to complete the required exercise task. Participants opted to either complete the experimental procedure cycling (n = 7) or running (n = 8), depending on their usual sporting participation and personal preference.
Preliminary measures and familiarization
Seven-to-fourteen days prior to the first experimental trial, participants attended the laboratory where height, nude body mass, fat, and fat-free mass were measured (Seca 515 MBCA; Seca Group, Hamburg, Germany). Maximum oxygen uptake (V̇O2max) was estimated by continuous incremental exercise test to volitional exhaustion (Vmax Encore Metabolic Cart; Carefusion, San Diego, Calif., USA), for runners on a motorized treadmill (Forma Run 500; Technogym, Seattle, WA, USA) as previously reported (Costa et al. 2009), and for cyclists on their own bicycle attached to a Wahoo KICKR cycle ergometer (Wahoo Fitness, Atlanta, GA, USA) previously validated in the power output range of all participants’ maximum aerobic power (MAP) (Zadow et al. 2016), using an incremental protocol previously reported (Currell and Jeukendrup 2008). Participants rode the Wahoo KICKR for approximately 10 min and then performed a spin-down calibration prior to testing. Running speed for experimental trials was determined as the speed at 1% gradient that produced approximately 55% of V̇O2max, verified from the oxygen uptake-work-rate relationship (8.9 ± 1.0 km h−1). Cycling power output for experimental trials was determined as 55% of MAP (163 ± 17 W), with MAP calculated as previously described (Hawley and Noakes 1992).
Participants then completed a 1-h exertional-heat stress familiarization trial, at the running speed or power output used in experimental trials, in an environmental chamber at 35.2 ± 0.5 °C ambient temperature (Tamb) and 22 ± 3% relative humidity (RH). Throughout the familiarization, participants drank water ad libitum, and completed psychophysical measures including Rating of Perceived Exertion (RPE) on a 6–20 Borg Scale (Borg 1982), thermal comfort rating [TCR; 13-point Likert-type thermal rating, with 7 indicative of comfortable, 10 indicative of hot, and 13 indicative of unbearably hot; adapted from Hollies and Goldman (1977)], and ratings of thirst and gastrointestinal symptoms (GIS) using a modified visual analogue scale (Gaskell et al. 2019). Nude body mass and water bottle mass were recorded before and after the familiarization to determine whole-body sweat rate, which was subsequently used to estimate fluid requirements for participants during the experimental trials.
Experimental procedure
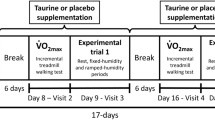
Participants were provided with 2-L urine collection containers and asked to collect all urine produced in the 3 days preceding each experimental trial, excluding the first void on the first collection day. The first void on the morning of the experimental trial was collected in a separate bottle to allow separate analysis of urine-specific gravity (USG). Participants were instructed to refrain from activities that caused significant perspiration during the urine collection period, to prevent sodium losses through thermoregulatory sweating. Prior to the low sodium diet (LNa) and high sodium diet (HNa) experimental visits, participants were provided with 3-day pre-prepared food (175 kJ kg−1 day−1, protein: 1.5 g kg−1 day−1, CHO: 6 g kg−1 day−1, and Na: 15 mg kg−1 day−1), as well as methylcellulose capsules providing either 85-mg kg−1 pharmaceutical grade NaCl (350-mg sodium per capsule) or placebo (caster sugar) in a randomized order. Capsules were consumed with main meals and snacks, and distributed evenly across the day, resulting in total dietary sodium intake of approximately 15 mg kg−1 day−1 (LNa) or 100 mg kg−1 day−1 (HNa). The LNa condition was chosen to reflect a sodium-restricted diet, and the HNa condition to reflect a sodium intake that was: (a) greater than the typical population sodium intake (Land et al. 2018) by the same magnitude that LNa was reduced, and (b) realistically achievable through conscious sodium loading by endurance athletes in the days preceding exercise (McCubbin et al. 2019). Participants and researchers interacting with participants were blinded to the content of capsules consumed before each experimental trial. Participants also completed an initial experimental trial, consuming their usual free-living diet (UDiet) whilst collecting all urine produced as described above. All other aspects of the experimental procedure were identical to HNa and LNa trials. The UDiet data have been included to allow comparison between UDiet and both increased and reduced sodium intakes.
Experimental visits were separated by 1–2 weeks, as previous work suggests that sodium balance stabilizes following abrupt changes in dietary intake within this timeframe (Conn and Arbor 1963). Participants arrived at the laboratory fasted between 0830 and 0930, but the time consistent for the same participant. They were instructed to consume 8 mL kg−1 water 2 h prior to arrival to ensure euhydration before exercise commencement. Upon arrival, they were immediately provided breakfast (CHO: 1.5 g kg−1, protein: 0.25 g kg−1, fluid: 250 mL, Na: 420 mg). Thirty minutes later, after bladder voiding, total body water (TBW) was measured using multi-frequency bioelectric impedance analysis (Seca 515 MBCA; Seca Group, Hamburg, Germany) and corrected by regression equation, as previously validated against deuterium dilution in endurance athletes with a constant error of 0.02 L. Forty-five minutes after consuming breakfast, and after sitting for 5 min, blood was collected by venepuncture from the antecubital vein in a vacutainer (6-mL, 1.5-IUmL−1 heparin), in a seated position. To monitor rectal temperature (Tre) during exercise, participants inserted a thermocouple 12 cm beyond the external anal sphincter (Grant REC soft insertion probe thermocouple; Grant 2010 Squirrel data logger, Shepreth, UK).
The experimental protocol consisted of 2 h running or cycling at 55% of V̇O2max (running) or 55% MAP (cycling), within an environmental chamber at 35.1 ± 0.6 °C Tamb, 21.8 ± 1.5% RH and fan airspeed ~ 10.6 km h−1 (running) or ~ 19.5 km h−1 (cycling). Participants consumed water (approximately 23 °C) of the same quantity in both trials, intended to limit body mass loss to 1.5%, based on sweat rate calculated during the familiarization. The required water volume was provided as four boluses, one given at the beginning of each 30-min period, and participants instructed to consume the water evenly throughout this time. Tre was recorded every 5 min throughout exercise, and RPE, thermal comfort, perceived thirst and GIS every 10 min. Following completion of the first hour of exercise, participants ceased exercising, and left the environmental chamber for 5 min to apply a second set of sweat patches (description below). A second blood sample was collected immediately post-exercise, as previously described within.
Sweat sample collection
Sweat samples were collected using the regional patch technique (Baker et al. 2009). Participants completed an 8–10-min warm-up at the same intensity and ambient conditions as the experimental trial, which allowed the onset of sweat production, and reduced the risk of sample contamination from minerals in the sweat pore (Baker 2017). Five sterile patches (Tegaderm + Pad, 3 M Health Care, Minnesota, USA) were pre-weighed (Quintix 313-1S, Sartorius, Goettingen, Germany), and then applied to the forehead (FH), right superior scapula (SS), upper chest (5 cm below the mid-point of the clavicle, UC), posterior mid-forearm (FA), and mid-thigh (MT) sites, as previously reported (Baker et al. 2009). Prior to application, each site was cleaned with an alcohol wipe, rinsed with deionized water, and dried with a clean laboratory wipe (Kimberly-Clark, Irving, TX, USA). Patches were removed with steel forceps that were pre-rinsed with deionized water and dried with clean laboratory wipes, when approximately 25% of the patch was visibly soaked with sweat, to prevent altered sample composition due to hidromeiosis (Baker 2017). Exercise time was stopped during patch removal (approximately 30 s) to ensure that the full 2 h of exercise was completed. Removed sweat patches were placed in pre-weighed glass petri dishes that had been rinsed in deionized water and air-dried. Local sweat rate (LSR) at each site was calculated from the change in patch mass before to after application, as previously reported (Smith and Havenith 2011). Following removal and weighing, patches were immediately transferred to airtight plastic tubes (Salivette, Sarstedt, Nümbrecht, Germany) and centrifuged at 4,000 RPM and 4 °C for 10 min to extract sweat.
Sweat and urine analysis
Sweat and urine [Na+] was determined by ion selective electrode (ISE) (LAQUATwin, Horiba, Kyoto, Japan), previously validated against ion chromatography for both sweat (Baker et al. 2014) and urine (Goulet and Asselin 2015) samples. Two-point calibration was undertaken as per manufacturer’s instructions. For calibration and measurement of sweat samples, the ISE surface was covered in a dry, pre-cut piece of laboratory wipe, and 45 μL samples pipetted onto the wipe. This technique compared to manufacturer-supplied sampling sheets with a coefficient of variation (CV) of 1.3%. For urine samples, 400 μL was pipetted directly onto the ISE surface following calibration with the same volume. The ISE surface was thoroughly washed with deionized water and dried with a clean laboratory wipe between each measurement. Urinary Na excretion (UNa) was calculated as the product of the urine [Na+] and volume in each container. For LNa and HNa trials, only UNa data from the final 2 days of collection were analysed, due to a period of renal adjustment to the altered sodium intake on the first day of collection. Sweat [Na+] was reported as individual patch site values and estimates of whole-body sweat [Na+], calculated using the regression equation developed by Baker et al. (2009) that incorporates data from all five sites (r = 0.97, ICC = 0.70).
Blood analysis
Whole-blood hemoglobin (Hb) (Hb201+, Hemocue AB, Ängelholm, Sweden) and hematocrit (HCT) (centrifuged capillary tubes, Propper, Long Island City, USA) were used to calculate changes in plasma volume (Pv) relative to baseline, and to correct plasma variables (Dill and Costill 1974). Remaining blood samples were centrifuged at 4000 RPM and 4 °C for 10 min, within 15 min of collection. Plasma was aliquoted into 1.7 mL micro-storage tubes and frozen at – 80 °C until analysis, except for 100 μL (2 × 50 μL) that was used to determine plasma osmolality (POsm), in duplicate (CV 0.8%), by freeze-point osmometry (Osmomat 030; Gonotec, Berlin, Germany). Plasma [Na+] was determined using ion selective electrodes (Cobas c 501, Roche Diagnostics, Risch-Rotkreuz, Switzerland) and analysed by local pathology services (Cabrini Pathology, Malvern, Victoria, Australia). Plasma aldosterone (DE5298; Demeditec Diagnostics GmbH, Kiel, Germany) and cortisol (RE52061; IBL International, Hamburg, Germany) were determined by ELISA. All variables were analysed as per the manufacturer’s instructions on the same day, with standards and controls on each plate, and each participant’s samples on the same plate. Aldosterone and cortisol CVs were 3.9% and 5.8%, respectively.
Calculation of sweat sodium secretion and reabsorption rate
To examine the contribution of indirect factors (plasma [Na+], sweat production rate) and direct factors (regulated Na+ reabsorption in the sweat gland) that contribute to regulation of sweat [Na+] as a result of altered dietary sodium intake, and across exercise time-period, calculations of sweat Na+ secretion and reabsorption rates were performed using the method developed by Sato (1977), and utilised by Buono et al. (2008):
Statistical analysis
Using published standard deviations for sweat [Na+] at regional patch sites (Dziedzic et al. 2014) and standard alpha of 0.05 and beta 0.85, it was calculated (G*Power v3.1.9.2, Universität Düsseldorf, Germany) that n = 9 would be required to provide adequate statistical power to detect a change of 25% in sweat [Na+], a magnitude of change consistent with existing literature comparing 14-day usual and low sodium intakes (Hargreaves et al. 1989; Yamazaki et al. 1994). Data are presented as mean and 95% confidence interval (CI), or mean ± SD, as indicated. The means of single time-point data were analyzed using repeated-measures ANOVA across the three dietary conditions. Data with multiple time-points were analyzed using two-way repeated-measures ANOVA to determine main effects of trial and time, and trial x time interactions, followed by Tukey’s HSD post hoc analysis for pairwise comparisons, where applicable. To determine the contributing role of plasma variables (plasma [Na+], POsm or Pv) and well as hormonal regulation of sweat gland function (plasma aldosterone and cortisol concentrations), Pearson correlation coefficients were calculated between individual variables and estimated whole-body sweat [Na+]. Analysis was performed using SPSS 25.0 (IBM Corp., Armonk, New York, USA) with significance accepted at p ≤ 0.05. There were no significant differences for control or outcome variables between cyclists and runners, and therefore, data were combined for the purpose of analysis and reporting. Blood samples could either not be collected or adequately analyzed for one participant in any trial and for three participants post-exercise in one trial, due to difficulties with venepuncture or insufficient sample volume, and therefore, these data were excluded from comparative analysis. Sweat samples at FH could not be obtained in one participant and MT in another due to very low LSR, and therefore, these data were also excluded from comparative analysis.
Results
Pre-exercise sodium and hydration status
All participants reported that they consumed 100% of the food and NaCl capsules provided during both LNa and HNa trials. One participant reported vomiting after ingestion of NaCl capsules at one meal, when NaCl capsules were not taken as instructed. There were no other reports of vomiting or severe nausea. UNa from the 3 days of UDiet was 46 (37–56) mg kg−1 day−1, similar to the mid-point between LNa and HNa trials (47.5 mg kg−1 day−1). UNa was significantly greater (p < 0.001) following HNa and lower following LNa (p < 0.001) compared to UDiet, and reflected the intended dietary Na intakes (Table 1). Pre-exercise nude body mass was lower following LNa compared to both UDiet (p < 0.05) and HNa (p < 0.01). TBW was lower following LNa compared to UDiet and HNa (p < 0.05). Compared to UDiet, pre-exercise Pv was 1.8% lower following LNa, and 2.7% higher following HNa. However, this effect was highly variable and differences did not reach statistical significance (Table 1). First-void USG was lower (p < 0.05) on the morning of exercise for LNa compared to both UDiet and HNa, likely as a function of the reduced UNa (Fig. 1).
Illustrative description of experimental procedures. UDiet Usual free-living diet (46 mg.kg−1 day−1), LNa low sodium diet (15 mg kg−1 day−1), HNa high sodium diet (100 mg kg−1 day−1), MAP maximal aerobic power, Tamb ambient temperature, RPE rating of perceived exertion, HR heart rate, GIS gastrointestinal symptoms, Tre rectal temperature, TBW total body water, MF-BIA multi-frequency bioelectric impedance analysis
Physiological markers, thirst, and gastrointestinal symptoms during exercise
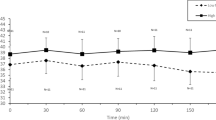
Body mass change, whole-body sweat rate, water intake, and Pv change during exercise are presented in Table 2. During all trials, Pv reduced as expected from the prescribed water intake. However, the Pv reduction was less during HNa compared to both LNa and UDiet (p < 0.05). There was no main effect of trial (p = 0.273), time (p = 0.569), or time × trial interaction (p = 0.424) for plasma [Na+]. A main effect of trial was present for POsm, which was significantly lower following LNa compared to UDiet (p < 0.05), but no effect of time, or trial × time interaction, was observed. No main effects were observed for perceived thirst. A main effect of trial (p = 0.044) and time (p < 0.001) was observed for Tre, as well as a time × trial interaction (p < 0.001). As a simple main effect, Tre was greater in LNa compared to HNa (p = 0.012) but not UDiet (p = 0.236), or between UDiet and HNa (p = 0.214). The increase in Tre from the onset of exercise occurred more rapidly in LNa than UDiet and HNa, such that pairwise differences appeared from 40–70 min (p < 0.05). A noticeable reduction in Tre occurred from 60–70 min, due to exercise cessation required for sweat patch application. Thereafter, Tre reached somewhat of a steady state during the 2nd h of exercise in all trials, with no pairwise differences from 80 min onwards (Fig. 2a). There was a main effect of time observed for HR (p < 0.001), which was elevated compared to 10 min at all subsequent time-points (p < 0.05) except for 70 min. Although visual inspection of the data suggested reduced HR with increasing sodium intake (Fig. 2b), the main effect of trial was not statistically significant (p = 0.124), nor was a time × trial interaction observed (p = 0.388). A main effect of time (p = 0.001), but not trial (p = 0.938) or interaction (p = 0.311), was also observed for RPE, which increased with exercise duration (Fig. 2c). A main effect of both trial (p = 0.011) and time (p = 0.031) was observed for TCR, with TCR generally increasing with exercise duration, and lower (more comfortable) during HNa at some but not all time-points (Fig. 2d).
Rectal temperature (a), heart rate (b), rating of perceived exertion (c), and thermal comfort rating (d) in response to 2 h of running and cycling at 55% \(\dot{V}\)O2max or MAP in Tamb 35 °C following HNa (black squares), UDiet (grey squares), or LNa (white squares). Mean ± SD: main effect of time #p < 0.05 and ##p < 0.01, main effect of trial §p < 0.05, time × trial interaction ^p < 0.01, ap < 0.05 and aap < 0.01 vs 10 min, bp < 0.05 and bbp < 0.01 LNa vs HNa, cp < 0.05 LNa vs UDiet, ddp < 0.01 UDiet vs HNa
A main effect of trial (p = 0.037) was observed for plasma cortisol concentration, with cortisol significantly greater in UDiet compared to HNa (p = 0.006) but not LNa (p = 0.105). No effect of time (p = 0.115) or interaction (p = 0.111) was observed. A main effect of trial (p = 0.001) and time (p < 0.001) was observed for plasma aldosterone (Table 3), but no interaction (p = 0.188). Simple main effects demonstrated aldosterone to be lower in UDiet (p = 0.017) and HNa (p = 0.002) compared to LNa, but not different between UDiet and HNa (p = 0.190). Incidence of any reported GIS was 27% following LNa, 40% following UNa and 20% following HNa. No effect of trial was observed for total GIS (p = 0.166), upper GIS (p = 0.125), lower GIS (p = 0.482), nausea (p = 0.412), or total gut discomfort (p = 0.114).
Sweat sodium concentration and local sweat rate
A main effect of time was observed for all patch sites (p ≤ 0.01), whereby sweat [Na+] was greater in the second hour of exercise compared to the first hour (Fig. 3a). A main effect of trial was also observed (p < 0.05 at all patch sites and estimated whole-body sweat [Na+]), with sweat [Na+] increasing in proportion to sodium intake across the three trials. For whole-body sweat [Na+], the difference between UDiet and LNa, and UDiet and HNa, was similar [first hour (mean ± SD): LNa 36 ± 13 mmol L−1, UDiet 41 ± 12 mmol L−1, HNa 47 ± 15 mmol L−1; 2nd h: LNa 44 ± 15 mmol L−1, UDiet 49 ± 13 mmol L−1, HNa 55 ± 17 mmol L−1; p < 0.001].
Sweat [Na+] (a), Local Sweat Rate (b), sweat sodium secretion (c), and reabsorption (d) rate during the 1st and 2nd h of exertional-heat stress (running and cycling at 55% maximal oxygen uptake/maximal aerobic power in 35 °C Tamb) following HNa (black bars), UDiet (grey bars), or LNa (white bars). Mean ± SD: ap < 0.05 and aap < 0.01 vs 1st h, bp < 0.05 and bbp < 0.01 vs LNa, cp < 0.05 vs UDiet
A main effect of time was observed for LSR at FH (p = 0.045), UC (p = 0.020), and FA (p = 0.025) patch sites. Simple main effects demonstrated LSR to be greater in the second hour of exercise at these sites (p < 0.05), although pairwise comparisons did not consistently demonstrate statistically significant differences (Fig. 3b). A main effect of trial was observed for LSR at the FH patch site only (p = 0.026), with LSR greater in HNa compared to LNa during the second hour (p = 0.014).
Sweat sodium secretion and reabsorption rates
No main effect of trial, time or time × trial interaction was observed for sweat Na+ secretion rate at any sweat patch site (Fig. 3c). A main effect of trial was observed for sweat Na+ reabsorption at the UC (p = 0.039), FA (p = 0.027) and MT (p = 0.040) sites, with Na+ reabsorption increased following LNa compared to HNa (p < 0.05 at all three sites) but not UDiet. There was no main effect of time or time × trial interaction for sweat Na+ reabsorption at any site. Pairwise differences are shown in Fig. 3d.
Correlation of variables with sweat [Na+] and sodium losses
The change in UNa between UDiet and LNa, and between UDiet and HNa, was not correlated with the subsequent change in WB sweat [Na+] (UDiet vs LNa: r = 0.21, p = 0.470; UDiet vs HNa: r = 0.293, p = 0.310). The correlation between variables implicated in sweat gland function or output (pre-exercise plasma [Na+], aldosterone, cortisol, POsm, and change in pre-exercise Pv between trials), and both the estimated whole-body sweat [Na+] and sweat sodium losses across all three trials, are presented in Table 4. Only pre-exercise plasma aldosterone (1st h r = − 0.568, p = 0.027; 2nd h r = − 0.675, p = 0.006) was correlated with sweat [Na+], and no variable was correlated with sweat sodium losses.
Discussion
The aims of the present study were to investigate the impact of 3-day high (HNa) and low (LNa) dietary sodium intake, in comparison to each other and to usual habitual diet (UDiet), on aspects of sodium balance before and during exercise; as well as the effect on cardiovascular, thermoregulatory, and gastrointestinal variables. As hypothesized, UNa increased progressively from LNa to HNa, and was reflected in pre-exercise plasma aldosterone concentration differences. Increasing sodium intake tended to result in greater pre-exercise nude body mass, Pv and POsm, but not plasma [Na+], although these differences were smaller and less consistent when comparing HNa to UDiet. During exercise, a smaller reduction in Pv was observed following HNa compared to UDiet and LNa, and the rate of rise in Tre was attenuated, although final Tre was not different between trials, and differences in HR between trials failed to reach statistical significance. Changes in plasma aldosterone, cortisol, POsm, and plasma [Na+] from pre- to post-exercise were not affected by trial, nor was GIS. In accordance with our hypothesis, the main finding was a clear effect of dietary sodium intake on sweat [Na+], with LNa resulting in a reduction in estimated whole-body sweat [Na+] of 10–11%, and HNa an increase of 10–12%, compared to UDiet. To the best of our knowledge, the present study is the first to provide competitive recreational endurance athletes with a controlled dietary sodium intake, blinded and proportional to body mass, over a timeframe (3 days) that reflects the period of altered sodium ingestion before competition (McCubbin et al. 2019). Collecting sweat samples using previously reported patch sites allowed estimation of whole-body sweat [Na+] from established regression equations (Baker et al. 2009), showing that in response to dietary sodium intake, altered sweat Na+ reabsorption, rather than secretion, was the most likely reason for the observed difference between trials.
Considering the multifactorial nature of sweat [Na+] regulation, several potential mechanisms could potentially contribute to the differences observed between dietary sodium intakes, including differences in plasma [Na+], POsm, or Pv, as well as changes in hormonal regulation of sweat gland reabsorptive function. The present study observed minimal differences in pre-exercise plasma [Na+] between interventions, although POsm and Pv were both greater following UDiet and HNa compared to LNa. Previous research has shown that increased POsm can increase the Tre threshold for the onset of sweating, and lower the sweat rate itself, independent of Pv in both passive (Takamata et al. 1995, 2001) and exercise-based (Sawka et al. 1985; Fortney et al. 1984) models of heat stress. Altered sweat rate could then influence sweat Na+ secretion and, therefore, sweat [Na+] (Buono et al. 2008). However, in the current study, as in others specifically investigating the effect of sodium intake on sweat sodium losses (Armstrong et al. 1985; Hargreaves et al. 1989; Koenders et al. 2017; Konikoff et al. 1986), differences in whole-body and local sweat rates were not observed between trials despite differences in Pv and POsm, nor was plasma [Na+] affected by dietary sodium intake.
In contrast, differences in pre-exercise aldosterone and cortisol concentration have been suggested to play a role in regulating sweat Na+ reabsorption, through expression of ion channels on the luminal surface of the gland (Sato et al. 1989; Castro-Sepulveda et al. 2019). In the present study, the concentration of plasma aldosterone was lower, and plasma cortisol higher, following 3-day LNa compared to UDiet and HNa. However, only plasma aldosterone concentration was correlated with sweat [Na+]. The correlation between pre-exercise aldosterone and sweat [Na+] is in accordance with previous research (Yoshida et al. 2006). Supporting a causative role of aldosterone on sweat Na+ reabsorption, intervention studies that have administered local or systemic exogenous aldosterone have reported significant reductions in sweat [Na+]; albeit at least 6 h after administration (Sato and Dobson 1970; Collins, 1966).
The results from the present study also raise the question as to the mechanism underlying differences in pre-exercise POsm between trials. POsm can be predicted from plasma concentrations of sodium, potassium, glucose, and urea (Hooper et al, 2015). No differences between trials were observed for pre-exercise plasma [Na+] (Table 3), potassium or glucose (data not shown), leaving urea the likely contributor to POsm differences. Although urea was not measured in the current study, previous work suggests that urea plays a key role in the renal regulation of sodium balance, with increased urea production a response to increasing sodium intake, as it facilitates increased water reabsorption in the renal medulla, preventing a significant diuresis from accompanying the upregulated natriuresis (Rakova et al. 2017).
The effect of dietary sodium intake in the days preceding exercise on thermoregulatory and cardiovascular variables during the exercise bout is also of relevance to athletes, particularly when exercising at high intensities and in hot ambient conditions. The observed pattern of change in Tre in particular is of interest. Whilst all trials reached a similar Tre at the conclusion of exercise, the initial rise in Tre was more rapid in the LNa trial. There are two potential explanations for this finding. First, the increased pre-exercise TBW in UDiet and HNa trials would require a greater degree of energy expenditure to raise core body temperature to the same extent (i.e., increasing exercise duration, as seen in HNa and UDiet compared to LNa). Second, the difference in measured Tre may also be at least in part due to differences in blood flow to the rectum, which can be altered by pre-exercise Pv (Taylor et al. 2014). Regardless of the mechanism, exercise-induced changes in Tre were small in the present study, mean Tre did not exceed 38.5 °C in any trial or time-point, and appeared to reach a steady state in the 2nd h of exercise, a pattern consistent with similar exercise protocols (Costa et al 2014; Gill et al. 2016; Alcock et al 2018; Snipe et al 2017; Snipe et al 2018a; 2018b). A lack of clear effect of pre-exercise sodium intake on Tre is also consistent with previously published data from similar intensity steady-state exercise (Hamouti et al. 2014), and for sodium ingestion during exercise when water intake is fixed (Earhart et al. 2015). In contrast, the effect of sodium-influenced differences in pre-exercise TBW and Pv is more likely to be evident during higher intensity exercise, where the rate of heat production is greater, and thermoregulation is likely to become a limiting factor to performance (Racinais et al. 2019). Previous studies employing an acute sodium loading strategy (20–40 mgkg−1, 1–2 h prior to exercise) increased pre-exercise Pv, reduced the rate of rise in HR and Tre during a time to exhaustion test at 70% V̇O2peak, the performance of which was improved as a result (Sims et al. 2007a, b). Even following 2 h of moderate-intensity steady-state exercise, acute pre-exercise sodium loading resulted in improved time-trial performance of approximately 10-min duration (Hamouti et al. 2014). Whilst differences in HR and Tre were minimal even at the completion of the time trial, differences were apparent in stroke volume and, therefore, cardiac output (Hamouti et al. 2014). It would appear that the relevance of pre-exercise sodium intake for athletes may depend on the specific demands of their sport, with shorter, higher intensity endurance events (e.g., marathon or shorter distance running events and Olympic distance triathlon), or longer events with interspersed high intensity efforts (e.g., road cycling) most likely to benefit from a higher sodium intake, especially when gastrointestinal tolerance or opportunities to drink during exercise are limited.
The current finding of a 23–28% difference in estimated whole-body sweat [Na+] between LNa and HNa, and 10–12% between UDiet and both LNa and HNa, is in contrast to some of the previous studies on the topic, recently summarized in a systematic review (McCubbin and Costa 2018). Possible explanations for this outcome include: first, the current study compared sodium intakes that were both substantially lower and higher than typical intakes (i.e., 15 mgkg−1day−1 and 100 mgkg−1day−1), and a greater difference in sodium intake compared with many other studies. Second, the current study utilized the regional patch technique for sweat collection rather than the whole-body washdown (WBW) method, which is considered the reference method for obtaining samples during exercise (Baker et al. 2018). The reasons for using the regional patch technique were that the WBW method is unsuitable for treadmill running, because shoes cannot be worn during this technique, and both the participant and equipment must be thoroughly rinsed with several liters of solution following the exercise period, a technique clearly unsuitable for a motorized treadmill (Shirreffs and Maughan 1997). The regional patch method with five sites allowed us to observe if the change in sweat [Na+] occurred universally across body sites, which was found to be the case. In addition, the use of the regional patch method allowed the investigation of the effects of the intervention on sweat gland function, providing further insight into the mechanisms underlying the altered sweat [Na+] that has not been reported in dietary intervention studies to date.
Two additional practical implications for athletes have emerged from these findings. First, increasing sodium intake substantially in the days preceding exercise had minimal impact on both absolute plasma [Na+], and the change in plasma [Na+] during exercise, when only water was consumed. However, whilst a deliberate increase in sodium intake in the days preceding exercise increased Pv and may offer a potential thermoregulatory benefit, the increased sweat [Na+] in the HNa condition would theoretically increase the sodium intake requirement during exercise, to maintain plasma [Na+]. Future research is warranted to better understand these potential trade-offs. Second, for athletes undergoing sweat composition testing in training, to inform expected sodium losses during competition, any difference in dietary sodium intake between these time-points is likely to result in only a modestly inaccurate estimation of sweat sodium losses. In fact, such differences (i.e., less than 12% between UDiet and HNa in the present study) fall within the typical day-to-day variability observed in previous reliability studies of sweat [Na+] testing (Baker 2017); albeit dietary sodium intake was not controlled in such studies. The observed differences in sweat [Na+] in the current study may still be of relevance to researchers during laboratory studies of sodium replacement during exercise, in that careful control of dietary sodium intake in the days preceding exercise would ensure more predictable sweat sodium excretion (and therefore replacement) occurs.
There are some limitations with the current study. First, the method of sweat sample collection, using the regional patch method rather than WBW as the reference method, has already been discussed. Second, the application of both running and cycling in the current study was a deliberate choice, to identify any differences between exercise modes on sweat losses, and cardiovascular and thermoregulatory variables. Whilst the authors acknowledge this as a potential limitation, these variables were found not to be different between exercise modalities, and therefore, data were combined for presentation purposes. Finally, the use of free-living diet (UDiet) as a comparator to both LNa and HNa means that nutrient intake during UDiet was not standardized between participants, or between UDiet and the other dietary conditions in the study. This difference could potentially influence pre-exercise TBW and during-exercise RPE with UDiet compared to LNa and HNa, due to differences in muscle glycogen content and associated intracellular water (Olsson and Saltin 1970). Caution should be made, therefore, in interpreting differences in TBW between UDiet and the other conditions. It was noted that RPE was not different between any of the conditions in the study, including the highly controlled LNa and HNa conditions, suggesting that any effect of muscle glycogen content is unlikely to influence interpretation of RPE. UNa resulting from UDiet was also more variable between participants than LNa and HNa due to the lack of control of dietary sodium intake. However, the magnitude of increase or decrease in UNa between UDiet and both LNa and HNa did not correlate with the magnitude of change in sweat [Na+], suggesting that the homeostatic response to changes in sodium intake also varies between individuals.
Conclusion
Three days of a high sodium diet (100 mg kg−1 day−1) prior to steady-state endurance exercise in the heat resulted in a reduced rate of rise in Tre compared to 3 days of low sodium diet (15 mg kg−1 day−1). However, differences between usual and high sodium intakes were minimal despite increased total body water and plasma volume from the high sodium diet. In addition, sweat [Na+] was reduced when restricting dietary sodium intake, and increased when sodium intake was increased from usual levels, and these changes correlated with pre-exercise plasma aldosterone. Future research should aim to assess the practical significance of these physiological changes on exercise performance in hot ambient conditions, across a range of exercise modalities, intensities, and durations relevant to competitive athletes.
Abbreviations
- CHO:
-
Carbohydrate
- CI:
-
Confidence interval
- CV:
-
Coefficient of variation
- FA:
-
Forearm
- FH:
-
Forehead
- GIS:
-
Gastrointestinal symptoms
- Hb:
-
Haemoglobin
- HCT:
-
Haematocrit
- HNa:
-
High sodium diet (100 mg kg−1 day−1)
- HR:
-
Heart rate
- ISE:
-
Ion selective electrode
- LNa:
-
Low sodium diet (15 mg kg−1 day−1)
- LSR:
-
Local sweat rate
- MAP:
-
Maximum aerobic power
- MT:
-
Mid-thigh
- Na+ :
-
Sodium
- [Na+]:
-
Sodium concentration
- NaCl:
-
Sodium chloride
- P Osm :
-
Plasma osmolality
- P v :
-
Plasma volume
- RPE:
-
Rating of perceived exertion
- SD:
-
Standard deviation
- SS:
-
Superior scapula
- T amb :
-
Ambient temperature
- TBW:
-
Total body water
- TCR:
-
Thermal comfort rating
- T re :
-
Rectal temperature
- UC:
-
Upper chest
- UDiet:
-
Usual free-living diet (mean: 46 mg kg−1 day−1)
- UNa:
-
Urinary sodium excretion
- USG:
-
Urine specific gravity
- \(\dot{V}\)O2max :
-
Maximal oxygen uptake
- \(\dot{V}\)O2peak :
-
Peak oxygen uptake
- WB:
-
Whole body
- WBW:
-
Whole body washdown
References
Alcock R, McCubbin A, Camões-Costa V, Costa RJS (2018) Case study: providing nutritional support to an ultraendurance runner in preparation for a self-sufficient multistage ultramarathon: rationed versus full energy provisions. Wilderness Environ Med 29(4):508–520. https://doi.org/10.1016/j.wem.2018.06.004
Armstrong LE, Costill DL, Fink WJ (1985) Effects of dietary sodium on body and muscle potassium content during heat acclimation. Eur J Appl Physiol Occup Physiol 54(4):391–397
Baker LB (2017) Sweating rate and sweat sodium concentration in athletes: a review of methodology and intra/interindividual variability. Sports Med 47(Suppl 1):111–128. https://doi.org/10.1007/s40279-017-0691-5
Baker LB, Barnes KA, Anderson ML, Passe DH, Stofan JR (2016) Normative data for regional sweat sodium concentration and whole-body sweating rate in athletes. J Sports Sci 34(4):358–368. https://doi.org/10.1080/02640414.2015.1055291
Baker LB, De Chavez PJD, Ungaro CT, Sopeña BC, Nuccio RP, Reimel AJ, Barnes KA (2018) Exercise intensity effects on total sweat electrolyte losses and regional vs. whole-body sweat [Na+], [Cl−], and [K+]. Eur J Appl Physiol. https://doi.org/10.1007/s00421-018-4048-z (in press)
Baker LB, Stofan JR, Hamilton AA, Horswill CA (2009) Comparison of regional patch collection vs. whole body washdown for measuring sweat sodium and potassium loss during exercise. J Appl Physiol 107(3):887–895. https://doi.org/10.1152/japplphysiol.00197.2009
Baker LB, Ungaro CT, Barnes KA, Nuccio RP, Reimel AJ, Stofan JR (2014) Validity and reliability of a field technique for sweat Na+ and K+ analysis during exercise in a hot-humid environment. Physiol Rep 2(5):e12007. https://doi.org/10.14814/phy2.12007
Borg GA (1982) Psychophysical bases of perceived exertion. Med Sci Sports Exerc 14(5):377–381
Buono MJ, Claros R, Deboer T, Wong J (2008) Na+ secretion rate increases proportionally more than the Na+ reabsorption rate with increases in sweat rate. J Appl Physiol 105(4):1044–1048. https://doi.org/10.1152/japplphysiol.90503.2008
Castro-Sepulveda M, Cancino J, Fernández-Verdejo R, Pérez-Luco C, Jannas-Vela S, Ramirez-Campillo R, del Coso J, Zbinden-Foncea H (2019) Basal serum cortisol and testosterone/cortisol ratio are related to rate of Na+ lost during exercise in elite soccer players. Int J Sports Nutr Exerc Metab. https://doi.org/10.1123/ijsnem.2019-0129 (in press)
Chinevere TD, Kenefick RW, Cheuvront SN, Lukaski HC, Sawka MN (2008) Effect of heat acclimation on sweat minerals. Med Sci Sports Exerc 40(5):886–891. https://doi.org/10.1249/MSS.0b013e3181641c04
Collins KJ (1966) The action of exogenous aldosterone on the secretion and composition of drug-induced sweat. Clin Sci 30(2):207–221
Conn JW, Arbor A (1963) Aldosteronism in man—some clinical and climatological aspects—Part I. JAMA 183:775–781
Conn JW, Johnston MW, Louis LH (1946) Relationship between salt intake and sweat salt concentration under conditions of hard work in humid heat. Fed Proc 5(1):230
Costa RJ, Oliver SJ, Laing SJ, Waiters R, Bilzon JL, Walsh NP (2009) Influence of timing of postexercise carbohydrate-protein ingestion on selected immune indices. Int J Sport Nutr Exerc Metab 19(4):366–384
Costa RJ, Gill SK, Hankey J, Wright A, Marczak S (2014) Perturbed energy balance and hydration status in ultra-endurance runners during a 24 h ultra-marathon. Br J Nutr 112(3):428–437. https://doi.org/10.1017/S0007114514000907
Currell K, Jeukendrup AE (2008) Superior endurance performance with ingestion of multiple transportable carbohydrates. Med Sci Sports Exerc 40(2):275–281. https://doi.org/10.1249/mss.0b013e31815adf19
Dill DB, Costill DL (1974) Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J Appl Physiol 37(2):247–248
Dziedzic CE, Ross ML, Slater GJ, Burke LM (2014) Variability of measurements of sweat sodium using the regional absorbent-patch method. Int J Sports Physiol Perform 9(5):832–838. https://doi.org/10.1123/ijspp.2013-0480
Earhart EL, Weiss EP, Rahman R, Kelly PV (2015) Effects of oral sodium supplementation on indices of thermoregulation in trained, endurance athletes. J Sports Sci Med 14(1):172–178
Fortney SM, Wenger CB, Bove JR, Nadel ER (1984) Effect of hyperosmolality on control of blood flow and sweating. J Appl Physiol Respir Environ Exerc Physiol 57(6):1688–1695. https://doi.org/10.1152/jappl.1984.57.6.1688
Gaskell SK, Snipe RMJ, Costa RJS (2019) Test re-test reliability of a modified visual analogue scale assessment tool for determining incidence and severity of gastrointestinal symptom in response to exercise stress. Int J Sport Nutr Exerc Metab. https://doi.org/10.1123/ijsnem.2018-0215 (in press)
Gill SK, Allerton DM, Ansley-Robson P, Hemmings K, Cox M, Costa RJ (2016) Does short-term high dose probiotic supplementation containing lactobacillus casei attenuate exertional-heat stress induced endotoxaemia and cytokinaemia? Int J Sport Nutr Exerc Metab 26(3):268–275. https://doi.org/10.1123/ijsnem.2015-0186
Goulet ED, Asselin A (2015) Reliability and validity of a low cost, pocket-sized and battery operated sodium analyzer in measuring urinary sodium concentration. Technol Health Care 23(6):881–891. https://doi.org/10.3233/THC-151028
Hamouti N, Fernandez-Elias VE, Ortega JF, Mora-Rodriguez R (2014) Ingestion of sodium plus water improves cardiovascular function and performance during dehydrating cycling in the heat. Scand J Med Sci Sports 24(3):507–518. https://doi.org/10.1111/sms.12028
Hargreaves M, Morgan T, Snow R, Guerin M (1989) Exercise tolerance in the heat on normal and low sodium diets. Clin Sci 76:553–557
Hawley JA, Noakes T (1992) Peak power output predicts maximal oxygen uptake and performance time in trained cyclists. Eur J Appl Physiol 65(1):79–83
Hoffman MD, Stellingwerff T, Costa RJS (2018) Considerations for ultra-endurance activities: part 2—hydration. Res Sports Med. https://doi.org/10.1080/15438627.2018.1502189
Hollies NRS, Goldman RF (eds) (1977) Psychological scaling in comfort assessment. In: Clothing comfort: interaction of thermal, ventilation, construction, and assessment factors. Science Publishers Inc., Ann Arbor
Holmes N, Bates G, Zhao Y, Sherriff J, Miller V (2016) The effect of exercise intensity on sweat rate and sweat sodium and potassium losses in trained endurance athletes. Ann Sports Med Res 3(2):1063–1066
Hooper L, Abdelhamid A, Ali A, Bunn DK, Jennings A, John WG, Kerry S, Lindner G, Pfortmueller CA, Sjöstrand F, Walsh NP, Fairweather-Tait SJ, Potter JF, Hunter PR, Sheepstone L (2015) Diagnostic accuracy of calculated serum osmolarity to predict dehydration in older people: adding value to pathology laboratory reports. BMJ Open 5:e008846. https://doi.org/10.1136/bmjopen-2015-008846
Kennedy NSJ, Eastell R, Smith MA, Tothill P (1983) Normal levels of total body sodium and chlorine by neutron activation analysis. Phys Med Biol 28(3):215–221
Koenders EE, Franken CPG, Cotter JD, Thornton SN, Rehrer NJ (2017) Restricting dietary sodium reduces plasma sodium response to exercise in the heat. Scand J Med Sci Sports 27(11):1213–1220. https://doi.org/10.1111/sms.12748
Konikoff F, Shoenfeld Y, Magazanik A (1986) Effects of salt loading during exercise in a hot dry climate. Biomed Pharmacother 40(8):296–300
Land MA, Neal BC, Johnson C, Nowson CA, Margerison C, Petersen KS (2018) Salt consumption by Australian adults: a systematic review and meta-analysis. Med J Aust 208(2):75–81. https://doi.org/10.5694/mja17.00394
McCance RA (1938) The effect of salt deficiency in man on the volume of the extracellular fluids, and on the composition of sweat, saliva, gastric juice and cerebrospinal fluid. J Physiol 92:208–218
McCubbin AJ, Costa RJS (2018) The impact of dietary sodium intake on sweat sodium concentration in response to endurance exercise: a systematic review. Int J Sports Sci 8(1):25–37. https://doi.org/10.5923/j.sports.20180803.05
McCubbin AJ, Cox GR, Costa RJS (2019) Sodium intake beliefs, information sources and intended practices of endurance athletes before and during exercise. Int J Sport Nutr Exerc Metab. https://doi.org/10.1123/ijsnem.2018-0270 (in press)
Morgan R, Patterson M, Nimmo M (2004) Acute effects of dehydration on sweat composition in men during prolonged exercise in the heat. Acta Physiol Scand 182(1):37–43. https://doi.org/10.1111/j.1365-201X.2004.01305.x
Olsson K, Saltin B (1970) Variation in total body water with muscle glycogen changes in man. Acta Physiol Scand 80(1):11–18. https://doi.org/10.1111/j.1748-1716.1970.tb04764.x
Rakova N, Kitada K, Lerchl K, Dahlmann A, Birukov A, Daub S, Kopp C, Pedchenko T, Zhang Y, Beck L, Johannes B, Marton A, Müller DN, Rauh M, Luft FC, Titze J (2017) Increased salt consumption induces body water conservation and decreases fluid intake. J Clin Invest 127(5):1932–1943. https://doi.org/10.1172/JCI88530
Racinais S, Moussay S, Nichols D, Travers G, Belfekih T, Schumacher YO, Periard JD (2019) Core temperature up to 41.5 °C during the UCI road cycling world championships in the heat. Br J Sports Med 53(7):426–429. https://doi.org/10.1136/bjsports-2018-099881
Ramanathan NL, Sen Gupta A, Lundgren NP (1956) Changes in the salt concentration of sweat during exercise in monsoon weather. Indian J Med Res 44(2):377–384
Robinson S, Kincaid RK, Rhamy RK (1950) Effect of salt deficiency on the salt concentration in sweat. J Appl Physiol 3(2):55–62
Robinson SN, Nicholas JR, Smith JH, Daly WJ, Pearcy M (1955) Time relation of renal and sweat gland adjustments to salt deficiency in men. J Appl Physiol 8(2):159–165
Sato K (1977) The physiology, pharmacology, and biochemistry of the eccrine sweat gland. Rev Physiol Biochem Pharmacol 79:51–131
Sato K, Dobson RL (1970) Regional and individual variations in the function of the human eccrine sweat gland. J Invest Dermatol 54(6):443–449. https://doi.org/10.1111/1523-1747.ep12259272
Sato K, Kang WH, Saga K, Sato KT (1989) Biology of sweat glands and their disorders. I. Normal sweat gland function. J Am Acad Dermatol 20(4):537–563
Saunders AG, Dugas JP, Tucker R, Lambert MI, Noakes TD (2005) The effects of different air velocities on heat storage and body temperature in humans cycling in a hot, humid environment. Acta Physiol Scand 183:241–255
Sawka MN, Young AJ, Francesconi RP, Muza SR, Pandolf KB (1985) Thermoregulatory and blood responses during exercise at graded hypohydration levels. J Appl Physiol 59(5):1394–1401. https://doi.org/10.1152/jappl.1985.59.5.1394
Sawka MN, Burke LM, Eichner ER, Maughan RJ, Montain SJ, Stachenfeld NS (2007) American College of Sports Medicine position stand. Exercise and fluid replacement. Med Sci Sports Exerc 39(2):377–390. https://doi.org/10.1249/mss.0b013e31802ca597
Shirreffs SM, Maughan RJ (1997) Whole body sweat collection in humans: an improved method with preliminary data on electrolyte content. J Appl Physiol 82(1):336–341
Shirreffs SM, Sawka MN (2011) Fluid and electrolyte needs for training, competition, and recovery. J Sports Sci 29(Suppl 1):S39–S46. https://doi.org/10.1080/02640414.2011.614269
Sims ST, Rehrer NJ, Bell ML, Cotter JD (2007a) Preexercise sodium loading aids fluid balance and endurance for women exercising in the heat. J Appl Physiol (1985) 103(2):534–541. https://doi.org/10.1152/japplphysiol.01203.2006
Sims ST, van Vliet L, Cotter JD, Rehrer NJ (2007b) Sodium loading aids fluid balance and reduces physiological strain of trained men exercising in the heat. Med Sci Sports Exerc 39(1):123–130. https://doi.org/10.1249/01.mss.0000241639.97972.4a
Smith CJ, Havenith G (2011) Body mapping of sweating patterns in male athletes in mild exercise-induced hyperthermia. Eur J Appl Physiol 111(7):1391–1404. https://doi.org/10.1007/s00421-010-1744-8
Snipe RMJ, Khoo A, Kitic CM, Gibson PR, Costa RJS (2017) Carbohydrate and protein intake during exertional heat stress ameliorates intestinal epithelial injury and small intestine permeability. Appl Physiol Nutr Metab 42(12):1283–1292. https://doi.org/10.1139/apnm-2017-0361
Snipe RMJ, Khoo A, Kitic CM, Gibson PR, Costa RJS (2018a) The impact of mild heat stress during prolonged running on gastrointestinal integrity, gastrointestinal symptoms, systemic endotoxin and cytokine profiles. Int J Sports Med. https://doi.org/10.1055/s-0043-122742 (in press)
Snipe RMJ, Khoo A, Kitic CM, Gibson PR, Costa RJS (2018b) The impact of exertional-heat stress on gastrointestinal integrity, gastrointestinal symptoms, systemic endotoxin and cytokine profile. Eur J Appl Physiol 118(2):389–400. https://doi.org/10.1007/s00421-017-3781-z
Takamata A, Mack GW, Gillen CM, Jozsi AC, Nadel ER (1995) Osmoregulatory modulation of thermal sweating in humans: reflex effects of drinking. Am J Physiol Regul Integr Comp Physiol 268(2):R414–R422. https://doi.org/10.1152/ajpregu.1995.268.2.R414
Takamata A, Yoshida T, Nishida N, Morimoto T (2001) Relationship of osmotic inhibition in thermoregulatory responses and sweat sodium concentration in humans. Am J Physiol Regul Integr Comp Physiol 280(3):R623–R629. https://doi.org/10.1152/ajpregu.2001.280.3.R623
Taylor NAS, Tipton MJ, Kenny GP (2014) Considerations for the measurement of core, skin and mean body temperatures. J Therm Biol 46:72–101. https://doi.org/10.1016/j.jtherbio.2014.10.006
Thomas DT, Erdman KA, Burke LM (2016) American College of Sports Medicine joint position statement. Nutrition and athletic performance. Med Sci Sports Exerc 48(3):543–568. https://doi.org/10.1249/mss.0000000000000852
Trangmar SJ, González-Alonso J (2017) New insights into the impact of dehydration on blood flow and metabolism during exercise. Exerc Sport Sci Rev 45(3):146–153. https://doi.org/10.1249/JES.0000000000000109
Watson PE, Watson ID, Batt RD (1980) Total body water volumes for adult males and females estimated from simple anthropometric measurements. Am J Clin Nutr 33(1):27–39
Yamazaki F, Fujii N, Sone R, Ikegami H (1994) Mechanisms of potentiation in sweating induced by long-term physical training. Eur J Appl Physiol 69:228–232
Yoshida T, Shin-ya H, Nakai S, Yorimoto A, Morimoto T, Suyama T, Sakurai M (2006) Genomic and non-genomic effects of aldosterone on the individual variation of the sweat Na+ concentration during exercise in trained athletes. Eur J Appl Physiol 98(5):466–471. https://doi.org/10.1007/s00421-006-0295-5
Zadow EK, Kitic CM, Wu SS, Smith ST, Fell JW (2016) Validity of power settings of the Wahoo KICKR power trainer. Int J Sports Physiol Perform 11(8):1115–1117. https://doi.org/10.1123/ijspp.2015-0733
Acknowledgements
The authors would like to thank the participants for their significant time commitment in completing this study, as well as Alexandra Stone, Alice Mika, Ashleigh Gale, Camilo Cayazaya, Rebecca Sue, and Holly Bolton for their assistance in the laboratory during data collection.
Funding
Funding for this work was provided by the Monash University, Be Active, Sleep Eat (BASE) Strategic Grants Scheme, and an Australian Government Research Training Program (RTP) Scholarship.
Author information
Authors and Affiliations
Contributions
AM, GC, JC, and RC were involved in the study design. AM recruited participants. AM and ML collected the data. AM, ML, and RC analyzed all samples. AM and ML analyzed all data. Data interpretation and manuscript preparation were undertaken by AM, JC, GC, and RC. All authors approved the final version of the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest, financial or otherwise, to declare.
Additional information
Communicated by George Havenith.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
McCubbin, A.J., Lopez, M.B., Cox, G.R. et al. Impact of 3-day high and low dietary sodium intake on sodium status in response to exertional-heat stress: a double-blind randomized control trial. Eur J Appl Physiol 119, 2105–2118 (2019). https://doi.org/10.1007/s00421-019-04199-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-019-04199-2