Abstract
Background
Photic retinopathy may lead to permanent foveal structural injury, leading to irreversible visual acuity loss.
Method
This prospective observational study evaluated 51 eyes of 30 patients with photic retinopathy. Optical coherence tomography (OCT) imaging was performed for all the eyes at baseline and final follow-up. All the eyes showed a focal outer retinal defect on spectral-domain OCT (SD-OCT) at the junction of the inner and outer photoreceptor segments. SD-OCT was used to measure central macular thickness, maximum horizontal dimension of the defect, maximum defect thickness, and the photic retinopathy index (PRI) through the foveal raster scan.
Results
Although PRI improved significantly at the final follow-up from baseline with a 17% improvement in PRI after a period of 6 months in photic retinopathy eyes, visual acuity declined for 14% of the patients and was stable for 84%. There was mild correlation of visual acuity with baseline PRI. Baseline PRI was significantly higher in eyes with poorer presenting visual acuity (VA). In total, 33.3% of the eyes showed partial ellipsoid zone recovery at 6 months. The area under curve of the receiver operator characteristic curve for partial ellipsoid zone recovery with the mean baseline PRI as the independent variable was poor at 0.612.
Conclusion
OCT-based ultrastructural features in photic retinopathy seem to have poor correlation with presenting or final visual acuity. To the best of our knowledge, this is the largest study on longitudinal OCT evaluation of photic retinopathy eyes in literature.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.

Introduction
The neurosensory retina is extremely sensitive to high intensities of light, with toxic levels leading to photic retinopathy. Although sunlight is the most common cause of photic retinopathy, newer causes like welding light and laser pointers are increasingly becoming common today. The first cases of photic maculopathy were described with sungazing or solar eclipse viewing, and hence the term “solar retinopathy” and “eclipse retinopathy” have become synonymous to each other. However, the clinical presentation of such patients is quite variable, which makes determination of incidence or prevalence of the disease difficult. Stokkermans and Dunbar have described an incidence of 0.14% in patients visiting a non-specialized eye care center [1]. Solar retinopathy may be associated with sungazing in mental disorders, and in subjects using psychotropic drugs, or sensitizing medications [2]. Eclipse-induced retinopathy is usually bilateral and asymmetric. Patients generally present with bilateral asymmetric blurring of vision, scotoma, metamorphopsia, or afterimages/photopsias [3,4,5]. After the advent of modern high-resolution optical coherence tomography, specific ultrastructural features of photic retinopathy have been described [6,7,8,9,10]. Spectral-domain optical coherence tomography (OCT) studies have observed that eyes undergo the following changes; in the acute phase, hyperreflective areas appear to be extending from the outer retinal layer to the inner layers, which in the chronic phase get substituted by a hyporeflective cyst-like space in the ellipsoid zone, indicating photoreceptor damage [11, 12]. Mostly photic retinopathy patients do not require treatment; while some clinicians prefer starting steroids, there is no consensus for the same. The visual prognosis of photic retinopathy is generally good, with majority of the eyes recovering the lost visual acuity or maintaining the same [13]. This study was planned to understand the ultrastructural features of the eyes presenting with clinically diagnosed photic retinopathy and confirmed on OCT imaging, and to try and describe prognostic features of anatomical and functional improvement on longitudinal follow-up.
Methods
This prospective observational study was conducted in the retina clinic of a tertiary care eye center in South India on patients of photic retinopathy who presented to the center between June 2017 and June 2019. The study was conducted after taking clearance from the Institutional Review Board and the study adhered to the tenets of the Declaration of Helsinki. All the patients gave informed consent before being included in this study.
A clinical diagnosis of photic retinopathy based on history of exposure to sunlight, eclipse, or welding light with the naked eye, followed by development of complaint of a scotoma in the visual field or frank diminution of visual acuity, was confirmed by the presence of an outer retinal defect in the ellipsoid zone on OCT in all the affected eyes. At diagnosis, all the patients underwent a complete ophthalmological examination including visual acuity using the Snellen chart, intraocular pressure, and slit-lamp biomicroscopy. SD-OCT (Spectralis HRA HD-OCT, Heidelberg Engineering) (16 horizontal raster scan) was performed in both eyes of each patient to evaluate the ultrastructural changes in each eye. Patients also underwent fundus autofluorescence imaging. All the patients received antioxidant medication and were followed up every 2 months until a period of 6 months. SD-OCT was repeated at 6 months to analyze the longitudinal ultramicroscopic changes.
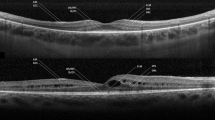
The parameters measured on SD-OCT were central macular thickness (CMT), maximum defect thickness (vertical depth), and maximum horizontal dimension of the outer retinal defect at the scan through the fovea, and these were measured from the images taken both at baseline and at 6-month follow-up (Fig. 1). “Photic retinopathy index” or PRI was obtained by dividing the maximum defect thickness in the foveal scan with the CMT (Fig. 1). All measurements were recorded by two independent graders and the readings were averaged to obtain the final value. The overall status of the ellipsoid zone (EZ) at fovea and whether it showed partial recovery at the final follow-up was also documented.
Data were entered into a Microsoft Excel Spreadsheet and analyzed using SPSS version 20.0 (IBM Inc.). Qualitative variables were expressed as mean ± standard deviation and categorical variables as percentages. Comparison of variables was performed using the t test for parametric data and Mann–Whitney U test for non-parametric data. A p value of less than 0.05 was considered as the significance level.
Results
Demography and clinical presentation
The current study evaluated 51 eyes of 30 subjects with photic retinopathy (Table 1). The mean age of the study population was 42.4 ± 13.22 years (range, 21–64 years; median = 40 years). A total of 27 eyes had solar retinopathy, 4 eyes had eclipse retinopathy, and 20 eyes had welder’s arc retinopathy. The subjects presented to the retina clinic within an average time period of 8.9 ± 4.7 days (median, 7.5 days; range, 3–20 days). The most common presenting symptoms were absolute scotoma (42 eyes, 51%), followed by diminution of vision (21 eyes, 41.2%). Twenty-one patients had bilateral retinopathy and 9 had unilateral. A total of 20 eyes had 6/6 Snellen visual acuity at presentation. On examination, all the eyes had normal anterior segment findings. In total, 47/51 eyes showed an altered foveal reflex on fundus examination. All the eyes showed a focal outer retinal defect on SD-OCT at the junction of inner and outer photoreceptor segments, involving the fovea. The vitreoretinal interface showed no abnormality in the eyes, and the retinal pigment epithelium and the choriocapillaris were structurally normal. In the region of the outer retinal defect, all the eyes showed a disruption of EZ and interdigitation zone.
All measurements of CMT, maximum horizontal dimension of defect, and maximum defect thickness through the foveal scan were performed by 2 independent graders (K.A., S.S.). The agreement analysis between the two graders has been summarized in Table 2. The central macular thickness at fovea at baseline was 238.05 ± 37.6 µm. The average horizontal dimension of the defect reduced from 262.55 ± 132.38 µm at baseline to 240.2 ± 129.46 µm at final follow-up (p = 0.39). The average maximum defect thickness reduced from 46.88 ± 16.4 µm at baseline to 40.79 ± 16.57 µm at final follow-up (p = 0.06).
We compared the recovery of the maximum defect thickness and horizontal dimension of the defect at final follow-up with baseline. In 17 eyes (33.3%) showing partial EZ recovery, the horizontal dimension improved from 260.44 ± 158.93 µm at baseline to 240.76 ± 163.34 µm at final follow-up (p = 0.72), and maximum defect thickness improved from 44.35 ± 16.54 µm at baseline to 37.09 ± 14.67 µm at final follow-up (p = 0.18). In 34 eyes without any EZ recovery, the horizontal dimension improved from 263.6 ± 119.59 µm at baseline to 239.9 ± 111.6. µm at final follow-up (p = 0.4) and the maximum defect thickness improved significantly from 48.15 ± 16.45 at baseline to 42.65 ± 17.34 at final follow-up (p = 0.18).
The baseline photic retinopathy index was measured as 0.199 ± 0.068 and the final PRI at 6 months follow-up was 0.172 ± 0.064 (p = 0.04). At baseline, a total of 33 eyes had VA of ≥ 6/9 with an average PRI of 0.17 ± 0.06 (median, 0.167), while 18 eyes (35.3%) had VA < 6/9 with an average PRI of 0.248 ± 0.05 (median, 0.257) (p < 0.001). Baseline PRI was significantly higher in eyes with poorer presenting VA. None of the eyes had worse than 6/36 Snellen VA at baseline.
A summary of the final VA status is summarized in Table 3. Final VA of ≥ 6/9 was seen in 28 eyes (54.9%), whose average baseline PRI was 0.167 ± 0.06 (median, 0.155) and final VA < 6/9 was seen in 23 eyes (45.1%) with average baseline PRI of 0.209 ± 0.06 (median, 0.211) (p = 0.021). Hence, the eyes with higher baseline PRI seemed to have poorer final VA. Figure 2a and b demonstrate the linear regression between baseline logMAR VA and final logMAR VA with PRI respectively. The R2 in Fig. 2a was 0.3 and that of Fig. 2b was 0.33, indicating that the correlation of PRI with visual acuity was not strong. Figure 2c demonstrates the correlation between baseline and final PRI and we found a strong correlation (R2 = 0.76), indicating that the baseline defect morphology affected the final anatomical status. Moreover, this linear regression also suggested a 17% improvement in PRI after a period of 6 months in photic retinopathy eyes. We also performed a linear regression between baseline and final VA and observed that the R2 was 0.74, indicating a similar strong correlation (Fig. 2d).
a Correlation between baseline PRI and baseline visual acuity of patients. b Correlation between baseline PRI and final follow-up visual acuity of patients. c Correlation between baseline PRI and final PRI. d Correlation between baseline visual acuity and final visual acuity. e Correlation between baseline PRI and baseline maximum horizontal dimension of outer retinal defect
A total of 17 eyes (33.3%) showed a partial EZ recovery at 6 months. Baseline VA of better than or equal to 6/9 Snellen was seen in 13/17 eyes (76.4%) of the eyes showing partial EZ recovery, and in 15/34 (55.9%) eyes in the eyes without EZ recovery. Eyes showing partial EZ recovery had a baseline average PRI of 0.18 ± 0.06 (median, 0.169) which was not significantly different from eyes without EZ recovery (final PRI average = 0.207 ± 0.07, median = 0.205) (p = 0.18). We also compared the baseline horizontal dimension between the groups and found no significant difference (p = 0.8). We observed that baseline PRI showed a weak positive correlation with baseline horizontal dimension of the outer retinal defect (R2 = 0.155) (Fig. 2e). Further correlations were performed between baseline horizontal dimension and baseline VA (R2 = 0.05) and final VA (R2 = 0.02), which were quite weak.
We performed a ROC analysis with partial EZ recovery as the dependent variable and baseline PRI as the determinant variable, and found that the area under curve for baseline PRI was 0.612 signifying a poor discriminant function (95% CI = 0.453 – 0.772) (Fig. 3). At a cutoff value for baseline PRI of 0.173, the sensitivity and specificity for the discriminant function of baseline PRI towards partial EZ recovery were 67.6% and 58.8%. Eyes which had PRI of less than or equal to 0.173 (n = 22) had a final VA of 0.084 ± 0.175 logMAR and eyes which had PRI of more than 0.173 (n = 29) had a final VA of 0.247 ± 0.188 logMAR (p = 0.003). The comparison of final VA in these two groups was statistically significant.
Receiver operator characteristic curve for partial ellipsoid zone recovery with independent variables—mean horizontal dimension of defect at baseline (AUC = 0.549), mean defect thickness (vertical) at baseline (AUC = 0.574), and mean baseline PRI (AUC = 0.612). The AUC was maximum for mean baseline PRI
Discussion
Outer retinal defect in photic retinopathy is a pathognomonic feature for clinching the diagnosis, and is said to be quite specific [14, 15]. The authors have shown that all photic retinopathy–affected eyes may show this defect, associated with foveal atrophy and abnormalities in photoreceptor cells and retinal pigment epithelium at the fovea [7, 16]. To date, there is no consensus regarding treatment of photic retinopathy eyes, and probably the long-term overall prognosis without any therapy is also optimal. Some cases may have persistent vision loss along with central scotoma over prolonged periods of time, although studies have shown that most cases may recover completely within a few weeks or months [17, 18]. Most of the eyes tend to maintain the visual acuity after initial diminution to around 6/12. We observed that 64.7% of the current study eyes presented with equal to or better than 6/9 Snellen VA at baseline and none of the rest of the eyes had worse than 6/36 Snellen VA. A total of 84.3% of total eyes showed no change in VA at the final follow-up, while 13.7% eyes showed worsening.
Authors have seen that VA improvement may begin from the first week to 10 days after the insult, and may continue to improve until 6 months, after which the VA may become stationary [2, 13, 18]. Similar to observations regarding persistent VA loss, the outer retinal defect may also persist up to 1 year in almost 80% of the eyes, with only a few patients showing complete healing after 1 year [13, 19,20,21]. SD-OCT helps in diagnosing and following up these eyes longitudinally and may also help in prognosticating the eyes. VA has been previously correlated with foveal thickness, and tissue disruption [7, 22, 23]. Interestingly, subjective VA improvement may be reported despite persistence of ellipsoid zone defect on OCT, even after 50 years of exposure, and visual acuity may not always correlate with structural findings [7, 24, 25]. It is also believed that different outcomes in the eyes in terms of rapid VA recovery and persistent dysfunction may be dependent on the degree of initial exposure, geographic location, retinal pigmentation, etc. [26].
In our study, we found that final visual acuity depended on baseline visual acuity. There was somewhat of a weak correlation between PRI and visual acuity (Figs. 1 and 2); however, VA correlated even more weakly with the horizontal dimension of the defect (R2 ~ 0.02–0.05). Moreover, the PRI also correlated very weakly with the horizontal dimension of the defect, indicating that PRI and maximum defect thickness may be better predictors of visual acuity. Around 33% eyes (1/3) may show recovery of EZ disruption after 6 months, with a statistically significant recovery of PRI by 17%. The PRI may be used to understand the magnitude of the initial insult of photic retinopathy and how much visual morbidity may occur in the long run. However, in our study subjects, visual acuity declined for 14% of the patients and was stable for 84% (1 patient had 2-line vision gain). PRI value of more than 0.173 may have prognostic value for visual outcome; however at a AUC value of 0.612 in our dataset, currently, we cannot conclude the same with strong confidence. Unless stronger data is shown in the future, as of now we might infer that OCT-based ultrastructural features in photic retinopathy have poor correlation with presenting or final visual acuity.
PRI may be an indirect marker of tissue loss, since it is a proportion of the patient’s central macular thickness. Although over a prolonged period of follow-up partial recovery of ellipsoid zone may be noted, and the horizontal dimension of the defect may reduce, we did not observe any significant reduction in the PRI in these eyes, indicating again the poor correlation between horizontal dimension and maximum defect thickness in terms of recovery.
The limitations of our study are that most of the patients presented days to weeks later; hence, the OCT manifestation may mostly reflect late changes. Moreover, the period of follow-up is relatively short and longer periods of time may be required to effectively understand the final anatomical and functional outcome of photic retinopathy eyes. However, the strength of the study is that the patients were followed up regularly and a uniform follow-up is available for all patients.
Based on our results, it will be precautious to assume that one may estimate the extent of horizontal tissue defect and EZ disruption by measuring the PRI (since the correlation was weak). However, our study is novel in that we have developed an index for characterization of the disease based on OCT ultrastructural findings, apart from the other commonly used parameter, that is EZ disruption, and this needs to be further characterized in larger sample size studies with longer periods of follow-up. Also, to the best of our knowledge, this is the largest series of OCT-based documentation of longitudinal follow-up of photic retinopathy eye reported to date.
Data availability
Will be made available on request.
References
Stokkermans TJ, Dunbar MT (1998) Solar retinopathy in a hospital-based primary care clinic. J Am Optom Assoc 69:625–636
Chen JC, Lee LR (2004) Solar retinopathy and associated optical coherence tomography findings. Clin Exp Optom 87:390–393
Penner R, McNair JN (1966) Eclipse blindness. Report of an epidemic in the military population of Hawaii. Am J Ophthalmol 61:1452–1457
Rai N, Thuladar L, Brandt F, Arden GB, Berninger TA (1998) Solar retinopathy. A study from Nepal and from Germany. Doc Ophthalmol 95:99–108
Kamali K, el Hoshy M, Rifai ME (1975) Eclipse blindness. Bull Ophthalmol Soc Egypt 68:373–379
Bechmann M, Ehrt O, Thiel MJ, Kristin N, Ulbig MW, Kampik A (2000) Optical coherence tomography findings in early solar retinopathy. Br J Ophthalmol 84:547–8
Hossein M, Bonyadi J, Soheilian R, Soheilian M, Peyman GA (2011) Spectral-domain optical coherence tomography features of mild and severe acute solar retinopathy. Ophthal Surg Lasers Imaging 42:e84–e86
Jain A, Desai RU, Charalel RA, Quiram P, Yannuzzi L, Sarraf D (2009) Solar retinopathy: comparison of optical coherence tomography (OCT) and fluorescein angiography (FA). Retina 29:1340–1345
Comander J, Gardiner M, Loewenstein J (2011) High-resolution optical coherence tomography findings in solar maculopathy and the differential diagnosis of outer retinal holes. Am J Ophthalmol 152:413–9.e6
Cho HJ, Yoo ES, Kim CG, Kim JW (2011) Comparison of spectral-domain and time-domain optical coherence tomography in solar retinopathy. Korean J Ophthalmol 25:278–281
Sherman J, Slotnick S (2012) Physiological causes of solar maculopathy. Optometry 83:6 ([letter, author reply])
Abdellah MM, Mostafa EM, Anber MA, Saman ISE, Eldawla ME (2019) Solar maculopathy: prognosis over one year follow up. BMC Ophthalmol 19:201
Klemencic S, McMahon J, Upadhyay S, Messner L (2011) Spectral domain optical coherence tomography as a predictor of visual function in chronic solar maculopathy. Optom Vis Sci 88:1014–1019
Suhr CL, Buffano RM, Sellers A (2011) The use of optical coherence tomography to aid in diagnosing solar maculopathy. Optometry 82:481–484
Comander J, Gardiner M, Loewenstein J (2011) High-resolution optical coherence tomography findings in solar maculopathy and the differential diagnosis of outer retinal holes. Am J Ophthalmol 152:413–9.e6
Eke T, Wong SCK (2001) Resolution of visual symptoms in eclipse retinopathy. Lancet 358(9282):674
Atmaca LS, Idil A, Can D (1995) Early and late visual prognosis in solar retinopathy. Graefes Arch Clin Exp Ophthalmol 233(12):801–804
Drake-Casanova P, Bolivar-de-Miguel G, Castro-Rebollo M, Clement-Corral M, Dapena-Sevilla I, Pareja-Esteban J (2007) Eclipse retinopathy: three case reports. Arch Soc Esp Oftalmol 82:575–577
Wong SC, Eke T, Ziakas NG (2001) Eclipse burns a prospective study of solar retinopathy following the 1999 solar eclipse. Lancet. 357:199e200
Doyle E, Sahu D, Ong G (2002) Solar retinopathy after the 1999 solar eclipse in East Sussex. Eye (Lond) 16:203e6
Kallmark FP, Ygge J (2005) Photo-induced foveal injury after viewing a solar eclipse. Acta Ophthalmol Scand 83:586e9
Gulkilik G, Taskapili M, Kocabora S, Demirci G, Muftuoglu GI (2009) Association between visual acuity loss and optical coherence tomography findings in patients with late solar retinopathy. Retina 29:257–261
Jorge R, Costa RA, Quirino LS, Paques MW, Calucci D, Cardillo JA et al (2004) Optical coherence tomography findings in patients with late solar retinopathy. Am J Ophthalmol 137:1139–1143
Bachmeier I, Helbig H, Greslechner R (2017) Eclipse retinopathy: a case series after the partial solar eclipse on 20 March 2015. Ophthalmology 114:44–48
Gregory-Roberts E, Chen Y, Harper CA, Ong T, Maclean MA, Fagan XJ et al (2015) Solar retinopathy in children. J Am Assoc Pediatr Ophthalmol Strabismus 19(4):349–351
Yannuzzi LA, Fisher YL, Krueger A, Slakter J (1987) Solar retinopathy: a photobiological and geophysical analysis. Trans Am Ophthalmol Soc 85:120–158
Author information
Authors and Affiliations
Contributions
KK: concept design, data collection and analysis, final approval. SS: concept design, data collection and analysis, final approval. KA: data collection and analysis, drafting manuscript, final approval. RPR: concept design, data collection and analysis, final approval. NBK: data collection and analysis, final approval. KR: concept design, final approval.
Corresponding author
Ethics declarations
Ethics approval
This study was reviewed and approved by the Ethics Committee of the Aravind Eye Hospital.
Informed consent
Patients were not required to give informed consent to the study because the analysis used anonymous clinical data that were obtained after each patient agreed to treatment by written consent.
Consent for publication
All the authors have given their consent for publication and authorship.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(XLSX 34 kb)
Rights and permissions
About this article
Cite this article
Kumar, K., Sen, S., Anudeep, K. et al. Anatomical and functional features of photic retinopathy: a spectral domain optical coherence tomography–based longitudinal study. Graefes Arch Clin Exp Ophthalmol 260, 415–423 (2022). https://doi.org/10.1007/s00417-021-05228-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-021-05228-7