Abstract
Purpose
Retinal sensitivity (RS) can be a valuable indicator of retinal function in response to intravitreal steroid or anti-VEGF treatment in the eyes with diabetic macular edema (DME), macular edema post retinal vein occlusion (RVO), or uveitis.
Methods
This prospective longitudinal study included 68 patients (96 eyes) with macular edema (ME) secondary to diabetes mellitus (42 eyes), uveitis (36 eyes), or RVO (18 eyes). In addition to best corrected visual acuity (BCVA) and retinal thickness, Nidek MP1 microperimetry was used to quantify RS at baseline visit and to look at the mean difference (MD) at 3–6 months and 1–2 years post intravitreal therapy with corticosteroids or anti-VEGF.
Results
There was a significant negative correlation between the central RS and BCVA (r = − 0.47, p < 0.001), including DME (r = − 0.42, p = 0.006) and uveitis (r = − 0.60, p < 0.001), but not RVO (r = − 0.37, p = 0.12). At 2-year follow-up, the overall CST was reduced from baseline (MD − 147 μm, 95% C.I − 192 to − 102, p < 0.001) with improved BCVA (MD − 0.12 LogMAR, 95% C.I − 0.23 to − 0.01, p = 0.01), but no improvement in the RS in any of the disorders. Both anti-VEFG and steroid groups showed significant improvement in CST at 2 years from baseline (MD − 101 μm, p = 0.001 and − 167 μm, p < 0.001, respectively) with only improvement in BCVA among anti-VEGF group (MD − 0.16 LogMAR, 95% C.I − 0.26 to − 0.07, p = 0.008).
Conclusion
The long-term follow-up of ME cases did not show a significant improvement in RS following treatment even with reduced macular thickness at 2-year follow-up.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Macular edema (ME) is a leading cause of vision loss among many patients with diabetic retinopathy (DR) [1], retinal vein occlusion (RVO) [2], and uveitis [3]. Preserving vision is the primary objective of treating ME, and testing visual function is an important tool in assessing successful treatment. Best corrected visual acuity (BCVA) is the most commonly used functional outcome measure of treating ME, both in clinical practice and trials. While BCVA measurement is an easily applicable and reproducible test, it does not always correlate with what patients report as their daily visual experience [4,5,6]. Because BCVA mainly reflects foveal function, it can overestimate macular function and the visual experience of patient in response to treatment [7].
Microperimetry (MP) is a method for testing visual function that focuses on the macula, whereby contrast sensitivity is measured for specific macular regions by focal light stimulation. By using both static and dynamic stimuli, the test creates detailed maps of retinal sensitivity (RS) reflecting the visual function of the entire macula. In the eyes with diabetic macular edema (DME), a progressive reduction in RS can occur independent of visual acuity [8]. The RS in DME patients is influenced by retinal thickness, with a reported reduction of 0.83 dB for every 10% deviation of retinal thickness from normal levels [9]. RS was also one of the measured outcomes in the DA VINCI study, which assessed clinical outcome following treatment of DME by either anti-vascular endothelial growth factor (anti-VEGF) injections or macular laser photocoagulation [10]. Correlations between RS, BCVA, and retinal thickness have also been demonstrated in the eyes with ME secondary to RVO [11] and UV [12], suggesting it may be a valuable tool in assessing these patients.
There have been few studies that examined changes in RS with relation to macular thickness and BCVA following intravitreal therapy for ME secondary to DME [10, 13, 14] or RVO [15, 16]. Furthermore, any pathology-specific differences in RS change in response to treatment remain unknown, with no clear understanding of the long-term changes. There is also limited information regarding the influence of intravitreal steroid therapy on RS in ME secondary to uveitis.
The aim of this prospective study was to assess changes in RS with relation to macular thickness and BCVA in the eyes with ME secondary to RVO, uveitis, or DME. We also aimed to examine the long-term effect of intravitreal therapy with corticosteroids and anti-VEGF on RS compared with their effect on BCVA and macular thickness.
Patients and method
This prospective study recruited patients with ME secondary to either DR, uveitis, or RVO who attended Moorfields Eye Hospital in London, UK, during the period between May 2014 and December 2016. The study protocol was approved by the institutional review board (LIGS1026, Visual Outcome of Retinal Oedema Treatment Study) and was conducted in compliance with regulations of the Health Insurance Portability and Accountability Act and the Declaration of Helsinki. All patients signed a written consent form before initiation of study-specific procedures. Patients were included in the study if ≥ 18 years old at time of recruitment and diagnosed with either DR, RVO, or uveitis complicated with ME defined as central retinal thickness ≥ 320 μm in the central subfield measured using optical coherence tomography (OCT). BCVA was measured using a Snellen chart which was later converted to its equivalent logarithm of minimum angle of resolution (LogMAR) for statistical analysis convenience.
Retinal sensitivity was measured by the fundus-monitored MicroPerimeter 1 (MP-1; Nidek Technologies, Albignasego, Italy). A 4-2-1-strategy with Goldmann III size stimulus with 200 ms duration and distributed over a total of 28 loci radiating from the fovea. These were divided into four loci within the central 0.6° and 12 loci used for each of the 1.2° and 2.5° radius circles. A red cross was used as a fixation target. Pupils were dilated and patients spent 5 min in a dim room before being tested to allow for visual adaptation.
Using OCT scans (Topcon 3D OCT-2000 Ver 8.11), the average retinal thickness was taken from the central subfield thickness (CST) 500 μm from the foveal center, as well as four inner subfields (within 500 to 1500 μm of the center) and four outer subfields (within 1500 and 3000 μm of the center). The mean RS and retinal thickness were measured from the central OCT subfield alone (1-mm-diameter circle centered on the fovea), the central subfield together with the inner four OCT subfields (3-mm diameter circle), and the central subfield together with the 4 inner and outer OCT subfield (6-mm diameter circle) of the macula (Fig. 1).
Following the baseline visit, patients who received intravitreal injections of steroids or anti-VEGF for ME had repeat measurements of their BCVA, RS, and macular thickness at 3 months, 6 months, 1 year, and 2 years. Changes in BCVA, CST, and RS were calculated per eye to determine response to treatment and to avoid large between-group variations. Treatment was given as per clinical guideline, with anti-VEGF injections given as a loading dose of 3 monthly injections, followed by series of three injections given as needed. For steroid implant injections a single implant was inserted and repeated as needed.
Statistical analysis
Analyses were performed with the SPSS statistical software (version 22, IBM, USA). Normal distribution was tested using the Shapiro-Wilk and Kurtosis tests. Continuous variables were presented as mean ± standard deviation for normally distributed data otherwise presented as median and Interquartile range (IQR). Kruskal Wallis Test was used to compare continuous variables between groups at baseline visit. Pearson’s correlation coefficient was used in the analysis of correlation among normally distributed baseline parameters. For not-normally distributed parameters the Spearman rank correlation coefficient was used. To assess change in BCVA, RS, and macular thickness post intravitreal therapy from baseline, a repeated measurement analysis was performed using generalized estimating equations (GEE), and a p value less than 0.05 was considered to be statistically significant.
Results
Among the 68 patients (96 eyes) included in the study, 42 (61.8%) were male. Average age at time of first assessment was 56.3 ± 14.5 years, ranging from 21.8 to 82.8 years. The three subgroups included 24 DME patients (42 eyes), 26 uveitis patients (36 eyes), and 18 RVO patients (18 eyes). The RVO cases were mainly branch RVO (11 eyes), and the remaining were central RVO (7 eyes). Data among RVO cases was normally distributed for CST and RS, but not for BCVA. Idiopathic intermediate uveitis was the most common form of uveitis, involving 24 eyes (66.8%), while the remaining uveitis cases ranged between HLA B-27 associated uveitis (4 eyes), birdshot chorioretinopathy (2 eyes), Vogt Koyanagi Harada syndrome (2 eyes), multiple sclerosis (2 eyes), sarcoidosis (1 eye), and tuberculosis hypersensitivity uveitis (1 eye). No cataract was observed at baseline visit in 40 eyes (41.6%) of cases, while 31 eyes (32.2%) were pseudophakic and the remaining eyes (26.2%) had some degree of cataract not enough to obscure the view for macular scans.
The overall median BCVA at baseline was 0.30 (IQR 0.18–0.30) LogMAR, with a median of 0.30 (IQR 0.18–0.77) LogMAR in the DME group, 0.30 (IQR 0.18–0.48) LogMAR in the RVO group, and 0.30 (IQR 0.18–0.60) LogMAR in the uveitis group. There was no significant difference in the average BCVA at baseline between the three groups (p = 0.81).
At baseline, the overall average CST was 401 ± 156 μm, for the DME group, it was 373 ± 121 μm; for the RVO group, 448 ± 190 μm; and for the uveitis group, it was 408 ± 171 μm (p = 0.22, one-way ANOVA). For the entire cohort, there was a weak but significant correlation between CST and BCVA at baseline (r = 0.21, p = 0.033, Spearman corr, Fig. 2a). This correlation was more obvious for the DME cases (r = 0.42, p = 0.006) unlike the RVO (r = 0.20, p = 0.42) and uveitis (r = 0.05, p = 0.75) groups.
Macular retinal sensitivity (RS) measured at baseline was 8.8 ± 5.8 dB, including 8.4 ± 5.9 dB in DME group, 7.8 ± 5.3 dB in RVO group, and 9.8 ± 6.0 dB in uveitis group (p = 0.43, one-way ANOVA). There was a significant negative correlation between the central RS and BCVA (r = − 0.47, p < 0.001; Spearman corr, Fig. 2b). A similar correlation was seen when looking at the subgroups of DME (r = − 0.42, p = 0.006) and uveitis (r = − 0.60, p < 0.001), but did not reach a significant level for the smaller sample of 18 RVO cases (r = − 0.37, p = 0.12). There was no significant correlation at baseline between average RS and retinal thickness within the central 1 mm area (r = − 0.029, p = 0.84 Pearson corr), which was also the case within each of the three patient subgroups.
Out of the 68 patients seen at baseline, 50 eyes had at least one further follow-up visit after receiving intravitreal therapy (Table 1). For all cases, the average change in BCVA during the first 3–6 months was not statistically significant, with a mean difference (MD) of only 0.01 LogMAR (95% C.I − 0.09 to 0.11, p = 0.91) but reached a significant level at 1–2 years from baseline (MD − 0.12 LogMAR, 95% C.I − 0.23 to − 0.01, p = 0.01), which was also the case for the RVO group (MD − 0.17 LogMAR, 95% C.I − 0.31 to − 0.02, p = 0.02).
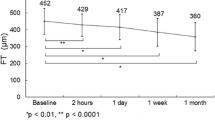
There was significant reduction in the average central retinal thickness at 1–2 years from baseline (MD − 147 μm, 95% C.I − 192 to − 102, p < 0.001). This was also noted within the subgroups of DME and uveitis, while the RVO group had a reduction in CST of the central 1 mm that only reached a significant level during the first 3–6 months. The average RS did not change significantly over the follow-up period within the total group nor did it change within the three subgroups (Table 1).
The eyes with ME were treated with either intravitreal anti-VEGF (27 eyes) or intravitreal steroids (23 eyes). At 3–6 months post commencing anti-VEGF therapy, there was no significant change in the median BCVA, CST, or RS compared with baseline. This changed at 1- to 2-year follow-up when there was an improvement in BCVA (MD − 0.16 LogMAR, 95% C.I − 0.26 to − 0.07, p = 0.008). During the same period there was a reduction in the median CST (MD − 101 μm, 95% C.I − 159 to − 44, p = 0.001) and 3 mm area of the macula. This however was not associated with a concomitant improvement in the median RS within similar measured areas of the macula (Table 2). Similarly, among those eyes treated with intravitreal corticosteroids, there was a significant improvement in the average CST at 3–6 months (MD − 122 μm, 95% C.I − 209 to − 35, p = 0.006) and at 1–2 years (MD − 167 μm, 95% C.I − 231 to − 103, p < 0.001). However, the change in BCVA and RS did not reach a statistically significant level (Table 2). Within the subgroup of the eyes receiving either intravitreal triamcinolone acetonide (9 eyes) or intravitreal dexamethasone implant (14 eyes), there was improvement in the CST by 1- to 2-year follow-up but no similar improvement in BCVA and RS (Table 3).
Discussion
This study aimed to examine the change in RS if any in response to intravitreal therapy for ME. Our results showed that (1) there is a significant moderate correlation between retinal sensitivity and visual acuity in the eyes with ME, including those with DME and uveitis. (2) There was a weak correlation between BCVA and central retinal thickness at baseline, which was moderate in the DME group while not significant in the uveitis and RVO groups. (3) No significant correlation was observed at baseline between RS and central retinal thickness. (4) Improvements in CST and/or BCVA at 2 years post intravitreal treatment was not associated with concomitant improvement in RS.
Good vision is influenced by the presence of normal foveal photoreceptor function and an uninterrupted neural network. While BCVA has been the main tool to test visual function, it mainly focuses on central foveal function. Microperimetry assessment of RS and fixation is able to asses a wider parafoveal area and correlate with other complex binocular functions such as reading [17]. In this study, we found RS sensitivity to correlate with BCVA in the presence of centrally involved ME within the overall group as well as in DME and uveitis which agrees with other studies that RS can provide additional information about macular function in ME cases, especially DME [18]. In addition, there was a correlation between CST in the eyes with ME > 320 μm and RS within the same central area. This corresponds with other studies that found a decrease in macular sensitivity by an average of 0.01 to 0.05 dB for every 1 μm increase in thickness above 280–300 μm, [19, 20] particularly in the presence of CME [20]. Once the central ME resolves BCVA may also improve, as foveal function is restored. However, macular function of peri-foveal regions may still be affected, resulting in visual dysfunction that would not be reflected by BCVA. In such cases, RS would remain reduced and reflect more closely to patients own experience of disparity between an improved BCVA and a remaining sense of visual impairment, which is reflected in poor visual quality of life scores [5, 6, 21].
In this study, the morphologic retinal features demonstrated a positive response to the intreavitreal therapies with a significant reduction in the retinal thickness at 1–2 years. However, this was not consistent with the retinal function parameter. It has been reported that the correlation between retinal thickness and RS can be weak when there is an associated photoreceptor disruption in a thinned atrophic retina < 300 μm. This is unlike edematous retina in which the thickness remains a strong predictor of poor RS independent of photoreceptor defect [20]. The LUCIDATE study looked at the effect of ranibizumab on the structural and functional outcome of 33 patients with DME over 34 weeks of follow-up and reported an improvement in RS among the ranibizumab group when compared with those managed with focal laser photocoagulation [22]. However, it was not clear if the change in RS post ranibizumab did reach a statistically significant level when compared with baseline measurement. In cases of DME managed with intravitreal dexamethasone implants, Mastropasqua et al. reported improvement in RS within the central 10° together with a reduction in the central macular thickness but only up to 4 months post injection, following which the effect was lost. Interestingly, there was no significant improvement in the electrophysiological results obtained from the pattern and multifocal electroretinograms, which even worsened after 4 months. These studies support the approach that visual function is related to more than only foveal thickness and that photoreceptor integrity and extra-foveal macular function are also responsible for patient’s visual experience.
This study did not include patients self-reporting of vision-related quality of life assessment, as well as other functional tests, such as reading speed. Exploring the effect of treating ME on these and correlations to RS may support its role in the functional assessment of patients with ME. Furthermore, the small study cohort limits the significance of any individual sub-population analysis, particularly that of the RVO group.
In conclusion, our study shows that treatment of ME may not result in improvement in RS, despite an effect on foveal thickness and BCVA. This suggests that there may be a role for expanding the functional assessment of patients to have a more encompassing representation of their visual function. Microperimetry can add to the information obtained from other vision assessment tools due to its ability to assess multiple macular areas including parafoveal areas, which are difficult to be tested by BCVA alone, thus improving patient assessment.
References
Klein R, Klein BE, Moss SE, Cruickshanks KJ (1995) The Wisconsin epidemiologic study of diabetic retinopathy. XV The long-term incidence of macular edema. Ophthalmology 102:7–16
Rehak J, Rehak M (2008) Branch retinal vein occlusion: pathogenesis, visual prognosis, and treatment modalities. Curr Eye Res 33:111–131
Tomkins-Netzer O, Talat L, Bar A et al (2014) Long-term clinical outcome and causes of vision loss in patients with uveitis. Ophthalmology 121:2387–2392
Lamoureux EL, Tai ES, Thumboo J et al (2010) Impact of diabetic retinopathy on vision-specific function. Ophthalmology 117:757–765
Okamoto Y, Okamoto F, Hiraoka T, Oshika T (2014) Vision-related quality of life and visual function following intravitreal bevacizumab injection for persistent diabetic macular edema after vitrectomy. Jpn J Ophthalmol 58:369–374
Suñer IJ, Bressler NM, Varma R et al (2017) Responsiveness of the National Eye Institute Visual Function Questionnaire-25 to visual acuity gains in patients with diabetic macular edema: evidence from the ride and rise trials. Retina 37:1126–1133
Squirrell DM, Mawer NP, Mody CH, Brand CS (2010) Visual outcome after intravitreal ranibizumab for wet age-related macular degeneration: a comparison between best-corrected visual acuity and microperimetry. Retina 30:436–442
Kube T, Schmidt S, Toonen F et al (2005) Fixation stability and macular light sensitivity in patients with diabetic maculopathy: a microperimetric study with a scanning laser ophthalmoscope. Ophthalmologica 219:16–20
Vujosevic S, Midena E, Pilotto E et al (2006) Diabetic macular edema: correlation between microperimetry and optical coherence tomography findings. Invest Ophthalmol Vis Sci 47:3044–3051
Gonzalez VH, Boyer DS, Schmidt-Erfurth U et al (2015) Microperimetric assessment of retinal sensitivity in eyes with diabetic macular edema from a phase 2 study of intravitreal aflibercept. Retina 35:687–694
Noma H, Mimura T, Shimada K (2013) Retinal function and morphology in central retinal vein occlusion with macular edema. Curr Eye Res 38:143–149
Roesel M, Heimes B, Heinz C et al (2011) Comparison of retinal thickness and fundus-related microperimetry with visual acuity in uveitic macular oedema. Acta Ophthalmol 89:533–537
Senturk F, Ozdemir H, Karacorlu M et al (2010) Microperimetric changes after intravitreal triamcinolone acetonide injection for macular edema due to central retinal vein occlusion. Retina 30:1254–1261
Grenga P, Lupo S, Domanico D, Vingolo EM (2008) Efficacy of intravitreal triamcinolone acetonide in long standing diabetic macular edema: a microperimetry and optical coherence tomography study. Retina 28:1270–1275
Noma H, Funatsu H, Mimura T, Shimada K (2012) Macular sensitivity and morphology after intravitreal injection of triamcinolone acetonide for macular edema with branch retinal vein occlusion. Retina 32:1844–1852
Yamaike N, Tsujikawa A, Sakamoto A et al (2009) Retinal sensitivity after intravitreal injection of bevacizumab for the treatment of macular edema secondary to retinal vein occlusion. Retina 29:757–767. https://doi.org/10.1097/IAE.0b013e31819d4fbf
Finger RP, Charbel Issa P, Fimmers R et al (2009) Reading performance is reduced by parafoveal scotomas in patients with macular telangiectasia type 2. Invest Ophthalmol Vis Sci 50:1366–1370
Deák GG, Bolz M, Ritter M et al (2010) A systematic correlation between morphology and functional alterations in diabetic macular edema. Invest Ophthalmol Vis Sci 51:6710–6714
Hatef E, Colantuoni E, Wang J et al (2011) The relationship between macular sensitivity and retinal thickness in eyes with diabetic macular edema. Am J Ophthalmol 152:400–405.e2
Yohannan J, Bittencourt M, Sepah YJ et al (2013) Association of retinal sensitivity to integrity of photoreceptor inner/outer segment junction in patients with diabetic macular edema. Ophthalmology 120:1254–1261
Ramu J, Chatziralli I, Yang Y et al (2017) Health-related quality of life, visual function and treatment satisfaction following intravitreal dexamethasone implant for diabetic macular edema. Patient Prefer Adherence 11:579–586
Comyn O, Sivaprasad S, Peto T et al (2014) A randomized trial to assess functional and structural effects of ranibizumab versus laser in diabetic macular edema (the LUCIDATE study). Am J Ophthalmol 157:960–970
Funding
This work was supported in part by an IMPACT award from Allergan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Moorfields Eye Hospital, London, UK, and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
This type of study does not require informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sharief, L., Chen, YH., Lightman, S. et al. Prospective study of morphologic and functional parameter changes post intravitreal therapy for macular edema. Graefes Arch Clin Exp Ophthalmol 258, 1941–1947 (2020). https://doi.org/10.1007/s00417-020-04715-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-020-04715-7