Abstract
Purpose
This literature review and meta-analysis aims to compare intraocular pressure (IOP) lowering efficacy, failure rates and loss of light perception (LP) rates 6 months after an IOP-lowering surgical procedure in neovascular glaucoma (NVG) eyes.
Methods
MEDLINE and EMBASE were used as data sources. Only studies including NVG patients who underwent two different surgical approaches were considered. The treatment effect measures were (i) weighted mean difference (WMD) for IOP reduction, (ii) risk ratio (RR) for failure rates and (iii) risk difference (RD) for loss of LP. Outcome measures were reported with a 95% confidence interval (CI) and P < 0.05 was considered statistically significant. Analysis was performed using RevMan v5.0.
Results
No RCT were retrieved. Seven comparative non-randomised studies were eligible. In glaucoma drainage devices (GDDs) vs cyclophotocoagulation arm, there was no statistical difference in IOP-lowering efficacy (WMD = − 3.63; CI [− 8.69, 1.43], P = 0.16), although failure rates and loss of LP were lower in the GDDs group (RR = 0.64, CI [0.41, 0.99], P = 0.05; and RD = − 0.15, CI [− 0.25, − 0.05], P = 0.004, respectively). In the Ahmed glaucoma valve (AGV) vs trabeculectomy arm, there was no statistical difference in IOP-lowering efficacy and loss of LP (WMD = 0.78, CI [− 2.29, 3.85], P = 0.62 and RD of 0.04, CI [− 0.05, 0.14], P = 0.34, respectively), but failure rates were lower in trabeculectomy group (RR of 2.25, CI [1.14, 3.71], P = 0.02).
Conclusions
There is lack of high-quality evidence on the subject as no RCT were retrieved comparing two different IOP-lowering procedures in NVG patients. Our findings are based, therefore, on non-RCT studies and should be interpreted with caution. There appears to be no difference in IOP-lowering efficacy between GDDs and cyclophotocoagulation, although GDDs appear to be safer. AGV and trabeculectomy also seem to provide similar IOP-lowering results with trabeculectomy showing lower failure rates.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Neovascular glaucoma (NVG) is an aggressive type of glaucoma generally associated with poor visual prognosis [1]. NVG is secondary to a number of diseases that affect the eye, the most common being diabetic retinopathy, ischemic central retinal vein occlusion and ocular ischemic syndrome [2]. The common feature to all is a hypoxic posterior segment, which leads to increased vascular endothelial growth factor (VEGF) formation. In turn, this cytokine-rich environment promotes the formation of fibrovascular tissue over the trabecular meshwork, resulting in aqueous humour (AH) outflow impairment and consequently increased intraocular pressure (IOP) [3]. While initially an open-angle condition, the myofibroblasts proliferation eventually creates a synechial angle-closure and further IOP elevation [4].
The management of NVG includes both controlling of the underlying ischemic process and decreasing IOP [5]. The first one is directed at reduction of ischemic drive that induces formation of new blood vessels, usually by panretinal photocoagulation or intravitreal anti-angiogenic administration [6]. The second key aspect is the successful IOP management [6]. This can be achieved with medical therapy and surgery, when ocular hypotensive drugs are insufficient.
Surgical options include cyclodestructive procedures, filtering surgery and glaucoma drainage devices (GDDs). Most cyclodestructive techniques are based on partial destruction of the ciliary body which decreases AH production, and therefore lowers the IOP [5]. Several procedures are available, such as cryotherapy, ultrasound cycloplasty and transcleral/endophotocoagulation [5]. Alternatively, both filtering surgery and GDDs create an alternative route for AH drainage, thus bypassing the blockade and restoring the AH outflow. The most commonly performed filtering surgery is trabeculectomy. However, NVG alone is considered a risk factor for bleb failure after trabeculectomy [7]. In consequence, GDDs have gained popularity as their success is less dependent on control of intraocular inflammation and the failure of filtering bleb [8]. Various GDDs (e.g. Molteno implant, Baerveldt implant, Ahmed glaucoma valve [AGV]), have been tried in the treatment of NVG. However, there is no evidence of superiority of one over another [6]. All in all, current evidence comparing surgical techniques in NVG is limited and the selection of the surgical procedure is based primarily on the individual surgeon’s judgement and consideration of patients’ variables [1].
The purpose of this review was to compare the best available evidence on the comparative efficacy and safety of the surgical techniques used in NVG.
Material and methods
Eligibility criteria for considering studies for this review
Studies including patients with NVG who underwent a surgical intervention for IOP control were included. Only studies comparing two groups with different surgical approaches were included. Both randomised clinical trials (RCTs) and non-randomised studies were included. No restrictions were made based on patients’ age, ocular comorbidities or NVG aetiology. The primary outcome was the mean difference in IOP reduction 6 months after surgery, with or without anti-glaucoma medication. Our secondary outcomes were the rates of surgical failure and loss of light perception (LP) at the last visit.
Search methods for identifying studies
Search protocols were elaborated for MEDLINE and EMBASE databases (see Appendix 1 and 2 for detail). We electronically searched Journals@Ovid Full Text <March 18, 2018>, Ovid MEDLINE(R) Epub Ahead of Print, In-Process & Other Non-Indexed Citations, Ovid MEDLINE(R) Daily, Ovid MEDLINE and Versions, EMBASE Classic and EMBASE <1947 to 2018 March 18. The last electronic search was performed on the 18th of March of 2018. No data or language restrictions were used. Additionally, a manual search throughout the bibliography of relevant studies was performed to include other potential studies.
Study selection
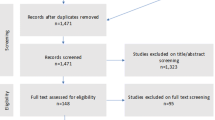
Two reviewers independently screened the titles and abstracts of studies identified by electronic and manual searches. Each reviewer classified the results as “Yes”, “Maybe” or “No” on whether to include or not to include studies for full text analysis. The differences in classification were resolved through mutual consensus, and whenever needed, a third reviewer was consulted. The same procedure was applied to full text analysis. All studies excluded from our review were excluded with reasons (see Fig. 1—PRISMA flow diagram). Whenever needed, additional information was requested from study investigators.
Data collection and risk of bias assessment
The risk of bias assessment was performed using ROBINS-I tool for non-randomised studies of interventions [9]. The first reviewer performed data extraction which included methods (study design, number of participants, randomisation, case matching), intervention details (definitions and time points) and outcome details (i.e. IOP at different time points, success and failures rates and proportion of patients with loss of LP). The second author reviewed the extracted data and existing conflicts were resolved by consensus.
Data synthesis and analysis
The measures of treatment effect were weighted mean difference (WMD) for IOP reduction, risk ratio (RR) for failure rates and risk difference (RD) for loss of LP. Outcome measures were reported with a 95% confidence interval (CI). P < 0.05 was considered statistically significant for the overall effect. Data analysis was performed with RevMan v5.0.
Results
Search results and baseline characteristics
A total of eight studies were analysed in this review (Fig. 1). No RCTs were retrieved (see Table 1 for baseline characteristics of the included studies). Of the eight eligible studies, six were retrospective and two were prospective in nature. The risk of bias assessment is summarised in Table 2. The studies were aggregated into two groups based on the type of intervention. The first consisted of three studies comparing GDDs with cyclophotocoagulation: (i) Chalam 2002 [10] compared pars plana modified Baerveldt valve with neodymium: YAG cyclophotocoagulation; (ii) Eid 1997 [11] compared tube-shunt surgery (which included eight eyes with double-plate Molteno implant, eight with AGV, six with Baerveldt valve and 2 with Schocket procedure) with neodymium: YAG cyclophotocoagulation; and (iii) Yildirim 2009 [12] compared AGV with diode laser cyclophotocoagulation. The second group consisted of four studies comparing AGV with trabeculectomy: (i) Engin 2011 [13] compared trabeculectomy with AGV; (ii) Liu 2011 [14] compared trabeculectomy combined with ranibizumab and mitomycin C (MMC) with AGV; (iii) Shen 2009 [15] compared trabeculectomy combined with MMC with AGV and (iv) Sun 2017 [16] compared intravitreal ranizumab with panretinal photocoagulation followed by trabeculectomy with AGV.
Yalvac 2007 [17] study, a retrospective study that compared AGV and Molteno valve in NVG, being the only identified study to compare two different GDDs was not considered for quantitative analysis, as data from this study could not be pooled with any of the two existing groups.
GDDs vs cyclophotocoagulation
IOP reduction
Mean IOP before and after surgical intervention at various time points was considered for quantitative analysis. In order to reduce bias, we selected only the common follow-up time point between all studies: mean IOP reduction 6 months after surgery. No statistically significant difference was observed between the two interventions (WMD = − 3.63; CI [− 8.69, 1.43]; P = 0.16) (Fig. 2).
Failure rates
In addition to IOP reduction, we also analysed failure rates in each study, which take into consideration not only IOP reduction, but also other factors such as loss of LP, necessity of an additional procedure and occurrence of complications (e.g. hypotony or phthisis bulbi). However, as seen in (Fig. 3), the criteria of failure applied in the reports were slightly heterogeneous. In cases in which no clear failure criteria were presented, we considered as a failure the cases that did not achieve the success criteria. We did not use the authors’ success rates in our analysis, due to an even wider heterogeneity.
Data concerning failure rates at several time points was extracted, but because there was no common time point, we used information available at the last visit for statistical analysis (Fig. 4) which showed RR favouring the GDDs group (RR = 0.64, CI [0.41, 0.99], P = 0.05).
Loss of LP
We also analysed the rates of loss of LP in each group. We considered the data available at the last visit for statistical analysis (Fig. 5). The results showed RD favouring the GDDs group (RD = − 0.15, CI [− 0.25, − 0.05], P = 0.004). However, heterogeneity was significant in this case (I2 = 90%).
AGV vs trabeculectomy
IOP reduction
Data concerning mean IOP values before and after surgical intervention was extracted for various time points. Shen 2011 study was excluded from this analysis due to missing data at the intermediary time points. No statistically significant difference was observed between two interventions (WMD = 0.78; CI [− 2.29, 3.85], P = 0.62) (Fig. 6).
Failure rates
As in the first case, failure rates were analysed for each intervention. Similarly, in cases in which no clear failure criteria were presented, we considered as a failure the cases that did not achieve the success criteria. Once again, the criteria of failure were not homogeneous (Fig. 3) and we did not use the authors’ success rates in our analysis, due to an even wider heterogeneity.
We extracted data concerning failure rates at several time points for each group, but because there was no common time point, we used data available at last visit for statistical analysis which showed RR favouring the trabeculectomy group (RR = 2.25, CI [1.14, 3.71], P = 0.02) (Fig. 7).
Loss of LP
Rates of loss of LP were analysed for both interventions. We considered the number of eyes with loss of LP at last visit to run the statistical analysis. It showed no statistically significant difference between the two groups (RD = 0.04, CI [− 0.05, 0.14], P = 0.34) (Fig. 8).
Discussion
In the present review and meta-analysis, seven non-RCT studies in which two different IOP-lowering procedures compared head-to-head were analysed. In GDDs vs cyclophotocoagulation group, we found no statistically significant difference in IOP-lowering capacity between the two techniques. However, failure rates and proportion of patients with loss of LP were favourable to the GDDs group. The fact that all the three studies in this group were non-RCTs creates a potential selection bias, since cyclophotocoagulation procedures are traditionally reserved for patients with more advanced NVG and already limited visual acuity, whereas GDDs are normally implanted in patients thought of having better prognosis and in whom there is still some preserved visual acuity [8]. Thus, our results are consistent with the mainstream practice, as GDDs were superior concerning the failure rates and loss of LP, translating more serious complication profile of cyclophotocoagulation. However, GDDs is a non-specific term, covering at least three types of GDDs, each with its specificities, ranging from different plate sizes to valved and non-valved mechanisms. The fact that different types of GDDs were used make the GDDs vs cyclophotocoagulation comparison even more difficult. In regard to this, several authors conducted literature reviews with an objective to compare different GDDs [18] and a recent a Cochrane review found no evidence of superiority of one over another [19]. It is worth mentioning that all the above included mixed glaucoma patients (primary and secondary) and none was directed exclusively to NVG patients. In this specific segment of patients, Yalvac 2007 [12] found both AGV and single-plate Molteno implants to be successful in early and intermediate IOP control, although long-term follow-up showed that both implants had poor results in maintaining clinical success. A recent RCT by Christakis et al. [20] compared AGV with Baerveldt implants in inadequately controlled glaucoma and in patients with previously failed trabeculectomy that included 50 NVG patients and had a follow-up of 5 years. A univariate analysis reported NVG to be a risk factor for failure in this study. Notwithstanding, a multivariate analysis showed no difference in success rates between devices for neovascular cases. In our case, the results concerning the IOP-lowering capacity need a more attentive interpretation due to a possible selection bias, as previously stated. In addition, heterogeneity, as seen by I2 values, was high in the results. Variability in success and failure criteria applied among the studies may account partially for it. Another potential source is the fact that different types of cyclophotocoagulation were used in this arm, although literature supports the concept that diode laser and Nd-YAG to be equivalent in terms of efficacy [21].
In the AGV vs trabeculectomy arm, there was also no statistically significant difference in IOP-lowering capacity between the two procedures, and while we did not identify any significant difference in LP rates, there was a statistically significant difference in failure rates, which favoured the trabeculectomy group. These results are surprising, given that recently published data from a Survey of the American Glaucoma Society shows a substantial increase in the use of GDDs with proportional decrease in trabeculectomies [22]. Nevertheless, our results should be interpreted with caution. The non-RCT nature of the retrieved studies is a potential source of selection bias, since the severity of the disease may have influenced the choice of surgical procedure. Just as in the previous case, the criteria defining surgical success and failure were heterogenous among the studies. Another aspect that should be mentioned is the differences seen in adjuvant administration of anti-VEGF agent and MMC. Engin 2011 [13], for instance, did not use MMC in trabeculectomies, a practice that is uncommon nowadays. Anti-VEGF administration also varied among the studies. As such, prospective controlled studies are needed to clarify this question.
One of the most importing findings in our study is probably the fact that very little high-quality evidence exists concerning surgical procedures in NVG patients. We conducted an extensive electronic search for articles comparing different IOP-lowering surgical procedures in NVG population; however, not a single RCT was retrieved, meaning that current practice is largely based on case series and expert opinions, rather than well-designed clinical trials. Our study also highlights the heterogeneity of the success and failure criteria in the retrieved studies. More standardised definitions should be used in the upcoming studies to allow future systematic reviews and meta-analyses. Also, limited duration of the follow-up is another issue that should be addressed in the future studies, as it seems that most of the surgical procedures are effective in early post-operative time; however, little information exists regarding their long-term efficacy. Over the last years, there has been a significant increase in use of anti-VEGF agents for NVG, both as stand-alone or adjunctive agents to photocoagulation, trabeculectomy or GDDs [23]. Despite its widespread use, there is still lack of high-quality evidence regarding its role in the treatment of NVG, as seen by recently conducted Cochrane systematic review by Simha et al. [24]. The authors studied the role of anti-VEGF agent in NVG and not a single study was retrieved meeting their inclusion criteria.
Conclusion
There is lack of high-quality evidence on the subject as no RCT were retrieved comparing two different IOP-lowering procedures in NVG patients. Our findings are based, therefore, on non-RCT studies and should be interpreted with caution. There appears to be no difference in IOP-lowering efficacy between GDDs and cyclophotocoagulation; however, GDDs seem to be associated with a smaller number of complications. Trabeculectomy and AGV also showed a similar IOP-lowering capacity, with failure rates favouring the trabeculectomy group. Given the non-RCT nature and heterogeneity of included studies, these results need to be interpreted with caution. There is a great need of prospective controlled trials, using well-established reporting protocols from the International Ophthalmological Societies to clarify which is the best surgical option in NVG.
Abbreviations
- IOP:
-
intraocular pressure
- LP:
-
light perception
- NVG:
-
neovascular glaucoma
- WMD:
-
weighted mean difference
- RR:
-
risk ratio
- RD:
-
risk difference
- CI:
-
confidence interval
- GDDs:
-
glaucoma drainage devices
- AGV:
-
Ahmed glaucoma valve
- VEGF:
-
vascular endothelial growth factor
- AH:
-
aqueous humour
- RCTs:
-
randomised clinical trials
- MMC:
-
mitomycin C
References
Havens SJ, Gulati V (2016) Neovascular glaucoma. Dev Ophthalmol 196–204
Shazly TA, Latina MA (2009) Neovascular glaucoma: etiology, diagnosis and prognosis. Semin Ophthalmol 24:113–121. https://doi.org/10.1080/08820530902800801
Brown GC, Magargal LE, Schachat A, Shah H (1984) Neovascular glaucoma. Ophthalmology 91:315–320. https://doi.org/10.1016/S0161-6420(84)34293-2
John T, Sassani JW, Eagle RC (1983) The myofibroblastic component of rubeosis iridis. Ophthalmology 90:721–728
Sivak-Callcott JA, O’Day DM, Gass JD, Tsai JC (2001) Evidence-based recommendations for the diagnosis and treatment of neovascular glaucoma. Ophthalmology 108:1767–1776; quiz1777, 1800. https://doi.org/10.1016/S0161-6420(01)00775-8
Hayreh SS (2007) Neovascular glaucoma. Prog Retin Eye Res 26:470–485. https://doi.org/10.1016/j.preteyeres.2007.06.001
Mietz H, Raschka B, Krieglstein GK (1999) Risk factors for failures of trabeculectomies performed without antimetabolites. Br J Ophthalmol 83:814–821
Olmos LC, Lee RK (2011) Medical and surgical treatment of neovascular glaucoma. Int Ophthalmol Clin 51:27–36. https://doi.org/10.1097/IIO.0b013e31821e5960
Sterne JA, Hernán MA, Reeves BC et al (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4919. https://doi.org/10.1136/BMJI4919
Chalam KV, Gandham S, Gupta S et al (2002) Pars plana modified Baerveldt implant versus neodymium:YAG cyclophotocoagulation in the management of neovascular glaucoma. Ophthalmic Surg, Lasers Imaging Retin 33:383–393. https://doi.org/10.3928/1542-8877-20020901-08
Eid TE, Katz LJ, Spaeth GL, Augsburger JJ (1997) Tube-shunt surgery versus neodymium: YAG cyclophotocoagulation in the management of neovascular glaucoma. Ophthalmology 104:1692–1700. https://doi.org/10.1016/S0161-6420(97)30078-5
Yildirim N, Yalvac IS, Sahin A et al (2009) A comparative study between diode laser cyclophotocoagulation and the Ahmed glaucoma valve implant in neovascular glaucoma: a long-term follow-up. J Glaucoma 18:192–196. https://doi.org/10.1097/IJG.0b013e31817d235c
Engin KN, Yılmazlı C, Engin G, Bilgiç L (2011) Results of combined cyclectomy/trabeculectomy procedure compared with ahmed glaucoma valve implant in neovascular glaucoma cases. ISRN Ophthalmol 2011:680827. https://doi.org/10.5402/2011/680827
Liu L, Xu Y, Huang Z, Wang X (2016) Intravitreal ranibizumab injection combined trabeculectomy versus Ahmed valve surgery in the treatment of neovascular glaucoma: assessment of efficacy and complications. BMC Ophthalmol 16:1–7. https://doi.org/10.1186/s12886-016-0248-7
Shen CC, Salim S, Du H, Netland PA (2011) Trabeculectomy versus Ahmed Glaucoma valve implantation in neovascular glaucoma. Clin Ophthalmol 5:281–286
Sun J-T, Liang H-J, An M, Wang D-B (2017) Efficacy and safety of intravitreal ranibizumab with panretinal photocoagulation followed by trabeculectomy compared with Ahmed glaucoma valve implantation in neovascular glaucoma. Int J Ophthalmol 10:400–405. https://doi.org/10.18240/IJO.2017.03.12
Yalvac IS, Eksioglu U, Satana B, Duman S (2007) Long-term results of Ahmed glaucoma valve and Molteno implant in neovascular glaucoma. Eye (Lond) 21:65–70
Gedde S, Panarelli J, Banitt M, Lee R (2013) Evidenced-based comparison of aqueous shunts. Curr Opin Ophthalmol 24:87–95
Tseng VL, Coleman AL, Chang MY, Caprioli J (2017) Aqueous shunts for glaucoma. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD004918.pub3
Christakis PG, Kalenak JW, Tsai JC et al (2016) The Ahmed versus Baerveldt study: five-year treatment outcomes. Ophthalmology 123:2093–2102. https://doi.org/10.1016/j.ophtha.2016.06.035
Oguri A, Takahashi E, Tomita G et al (1998) Transscleral cyclophotocoagulation with the diode laser for neovascular glaucoma. Ophthalmic Surg Lasers 29:722–727
Vinod K, Gedde SJ, Feuer WJ et al (2017) Practice preferences for glaucoma surgery. J Glaucoma 26:687–693. https://doi.org/10.1097/IJG.0000000000000720
SooHoo JR, Seibold LK, Kahook MY (2013) Recent advances in the management of neovascular glaucoma. Semin Ophthalmol 28:165–172. https://doi.org/10.3109/08820538.2012.730103
Simha A, Braganza A, Abraham L, et al (2013) Anti-vascular endothelial growth factor for neovascular glaucoma. Cochrane database Syst Rev CD007920
Acknowledgments
We thank Joana Alarcão MD for her contribution in elaborating the search strategy for this review. We also thank Filipe Rodrigues MD for assistance in the electronic search.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1. MEDLINE search strategy
-
1.
exp. neovascular glaucoma/
-
2.
((glaucoma* or angle* or iris or anterior) adj4 neovascular*).tw.
-
3.
(NVG or NVI or NVA).tw.
-
4.
1 or 2 or 3
-
5.
exp. filtering surgery/
-
6.
filtering surgery.tw.
-
7.
glaucoma surgery.tw.
-
8.
trabeculectomy.tw.
-
9.
exp. glaucoma drainage implants/
-
10.
exp. molteno implants/
-
11.
(drainage adj2 (valve or implant or device or shunt)).tw.
-
12.
(ahmed adj2 (valve or implant or device or shunt)).tw.
-
13.
(molteno adj2 (valve or implant or device or shunt)).tw.
-
14.
(krupin adj2 (valve or implant or device or shunt)).tw.
-
15.
exp. angiogenesis inhibitors/
-
16.
(macugen* or pegaptanib* or lucentis* or rhufab* or ranibizumab* or bevacizumab* or avastin* or aflibercept* or eylea*).tw.
-
17.
(anti adj2 VEGF*).tw.
-
18.
(baerveldt adj2 (valve or implant or device or shunt)).tw.
-
19.
5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17 or 18
-
20.
4 and 19
-
21.
(animal$ not human$).sh,hw.
-
22.
20 not 21
Appendix 2. EMBASE search strategy
-
1.
exp. neovascular glaucoma/
-
2.
((glaucoma* or angle* or iris or anterior) adj4 neovascular*).tw.
-
3.
(NVG or NVI or NVA).tw.
-
4.
or/1–3
-
5.
exp. filtering surgery/
-
6.
filtering surgery.tw.
-
7.
glaucoma surgery.tw.
-
8.
trabeculectomy.tw.
-
9.
exp. glaucoma drainage implants/
-
10.
exp. molteno implants/
-
11.
(drainage adj2 (valve or implant or device or shunt)).tw.
-
12.
(ahmed adj2 (valve or implant or device or shunt)).tw.
-
13.
(molteno adj2 (valve or implant or device or shunt)).tw.
-
14.
(baerveldt adj2 (valve or implant or device or shunt)).tw.
-
15.
(krupin adj2 (valve or implant or device or shunt)).tw.
-
16.
exp. angiogenesis inhibitors/
-
17.
(macugen* or pegaptanib* or lucentis* or rhufab* or ranibizumab* or bevacizumab* or avastin* or aflibercept* or eylea*).tw.
-
18.
(anti adj2 VEGF*).tw.
-
19.
or/5–18
-
20.
and/4,19
-
21.
(animal$ not human$).sh,hw.
-
22.
20 not 21
Appendix 3. References to studies excluded from this review
-
1.
Ayyala R, MD F, Zurakowski D, et al. (2002) Comparison of Double-Plate Molteno and Ahmed Glaucoma Valve in Patients With Advanced Uncontrolled Glaucoma. Ophthalmic Surg Lasers 33:94–101.
-
2.
Wang Q (2014) Study on the efficacy between cyclocryotherapy combined trabeculectomy and single cyclocryotherapy for neovascular glaucoma. [Chinese]. Int Eye Sci 14:1638–1640. https://doi.org/10.3980/j.issn.1672-5123.2014.09.23
-
3.
Christakis PG, Tsai JC, Kalenak JW, et al. (2013) The Ahmed Versus Baerveldt Study. Ophthalmology 120:2232–2240. https://doi.org/10.1016/j.ophtha.2013.04.018
-
4.
Ma KT, Yang JY, Kim JH, et al. (2012) Surgical results of ahmed valve implantation with intraoperative bevacizumab injection in patients with neovascular glaucoma. J Glaucoma 21:331–336. https://doi.org/10.1097/IJG.0b013e31820e2fd0
-
5.
Takihara Y, Inatani M, Kawaji T, et al. (2011) Combined intravitreal bevacizumab and trabeculectomy with mitomycin C versus trabeculectomy with mitomycin C alone for neovascular glaucoma. J. Glaucoma 20:196–201.
-
6.
Yang Z, Wang XH, Li RX (2012) Compared analysis of the efficacy of different treatment methods for neovascular glaucoma. Int J Ophthalmol 12:104–106. https://doi.org/10.1016/j.optcom.2009.12.042
-
7.
Li XJ, Hu SQ (2014) Therapeutic effect of modified trabeculectomy in treatment of neovascular glaucoma. [Chinese]. Int Eye Sci 14:745–746. https://doi.org/10.3980/j.issn.1672-5123.2014.04.48
-
8.
Chen C-H, Lai I-C, Wu P-C, et al. (2010) Adjunctive Intravitreal Bevacizumab-Combined Trabeculectomy Versus Trabeculectomy Alone in the Treatment of Neovascular Glaucoma. J Ocul Pharmacol Ther 26:111–118. https://doi.org/10.1089/jop.2009.0055
-
9.
Marey HM, Ellakwa AF (2011) Intravitreal bevacizumab with or without mitomycin C trabeculectomy in the treatment of neovascular glaucoma. Clin Ophthalmol 5:841–845. https://doi.org/10.2147/OPTH.S21453
-
10.
Sugimoto Y, Mochizuki H, Okumichi H, et al. (2010) Effect of intravitreal bevacizumab on iris vessels in neovascular glaucoma patients. Graefe’s Arch. Clin. Exp. Ophthalmol. 248:1601–1609.
-
11.
Teixeira SH, Doi LM, Freitas Silva AL De, et al. (2012) Silicone Ahmed glaucoma valve with and without intravitreal triamcinolone acetonide for neovascular glaucoma: Randomised clinical trial. J Glaucoma 21:342–348. https://doi.org/10.1097/IJG.0b013e31820d7e4e
-
12.
Du Q-W, Yang L-S (2015) Effects of intravitreal injection of Bevacizumab combined with complex trabeculectomy for advanced neovascular glaucoma. Int Eye Sci 15:1766–1768. https://doi.org/10.3980/j.issn.1672-5123.2015.10.24
-
13.
Mackenzie PJ, Schertzer RM, Isbister CM (2007) Comparison of silicone and polypropylene Ahmed glaucoma valves: Two-year follow-up. Can. J. Ophthalmol. 42:227–232.
-
14.
Eid TM, Radwan A, El-Manawy W, El-Hawary I (2009) Intravitreal bevacizumab and aqueous shunting surgery for neovascular glaucoma: Safety and efficacy. Can J Ophthalmol 44:451–456. https://doi.org/10.3129/i09-108
-
15.
Hinkle DM, Zurakowski D, Ayyala RS (2007) A comparison of the polypropylene plate Ahmed™ glaucoma valve to the silicone plate Ahmed™ glaucoma flexible valve. Eur. J. Ophthalmol. 17:696–701.
-
16.
Yazdani S, Mahboobipour H, Pakravan M, et al. (2016) Adjunctive Mitomycin C or amniotic membrane transplantation for Ahmed glaucoma valve implantation: A randomised clinical trial. J Glaucoma 25:415–421. https://doi.org/10.1097/IJG.0000000000000256
-
17.
Liu XN, Tong FF (2014) Study of surgical treatment on neovascular glaucoma. [Chinese]. Int Eye Sci 14:942–944. https://doi.org/10.3980/j.issn.1672-5123.2014.05.48
-
18.
Syed HM, Law SK, Nam SH, et al. (2004) Baerveldt-350 Implant versus Ahmed Valve for Refractory Glaucoma: A Case-Controlled Comparison. J. Glaucoma 13:38–45.
-
19.
Connor MA, Knape RM, Oltmanns MH, Smith MF (2010) Trainee Glaucoma Surgery: Experience with Trabeculectomy and Glaucoma Drainage Devices. Ophthalmic Surgery, Lasers, and Imaging 41:523–531. https://doi.org/10.3928/15428877-20100625-01
-
20.
Sevim MS, Buttanri IB, Kugu S, et al. (2013) Effect of intravitreal bevacizumab injection before Ahmed glaucoma valve implantation in neovascular glaucoma. Ophthalmologica 229:94–100. https://doi.org/10.1159/000345490
-
21.
Chen JP, Wang R, Xu HY, et al. (2013) Clinical observation of two operation methods for neovascular glaucoma. [Chinese]. Int Eye Sci 13:2259–2261. https://doi.org/10.3980/j.issn.1672-5123.2013.11.28
-
22.
Wu XQ, Guo Y, Gao JL (2016) Curative effect of Ranibizumab with Ex-PRESS drainage device for neovascular glaucoma. [Chinese]. Int Eye Sci 16:300–302. https://doi.org/10.3980/j.issn.1672-5123.2016.2.27
-
23.
Maris PJG, Ishida K, Netland PA (2007) Comparison of trabeculectomy with Ex-PRESS miniature glaucoma device implanted under scleral flap. J. Glaucoma 16:14–19.
-
24.
Zhou MW, Wang W, Huang W Bin, et al. (2013) Adjunctive with versus without intravitreal bevacizumab injection before ahmed glaucoma valve implantation in the treatment of neovascular glaucoma. Chin Med J (Engl) 126:1412–1417. https://doi.org/10.3760/cma.j.issn.0366-6999.20130011
-
25.
Bai Y, Wang M, Li Y, et al. (2011) [Clinical efficacy and safety of FP-7 Ahmed glaucoma valve implantation in neurovascular glaucoma patients]. Zhonghua Yan Ke Za Zhi 47:893–897. https://doi.org/10.3760/cma.j.issn.0412-4081.2011.10.007
-
26.
de França FO, Goya DH, de Camargo Penteado CL (2018) User profiling of the Twitter Social Network during the impeachment of Brazilian President. Soc Netw Anal Min 8:179–185. https://doi.org/10.1007/BF01206252
-
27.
Mahdy RA, Nada WM, Fawzy KM, et al. (2013) Efficacy of intravitreal bevacizumab with panretinal photocoagulation followed by ahmed valve implantation in neovascular glaucoma. J. Glaucoma 22:768–772.
-
28.
Marra K V., Wagley S, Omar A, et al. (2015) Case-matched comparison of vitrectomy, peripheral retinal endolaser, and endocyclophotocoagulation versus standard care in neovascular glaucoma. Retina 35:1072–1083.
-
29.
Kiddee W, Tantisarasart T, Wangsupadilok B (2012) Neovascular glaucoma: a retrospective review of 5-year experience in Songklanagarind Hospital. J Med Assoc Thai 95 Suppl 4:
-
30.
Yang Z, Wang XH, Li RX (2012) Compared analysis of the efficacy of different treatment methods for neovascular glaucoma. Int J Ophthalmol 12:104–106. https://doi.org/10.1016/j.optcom.2009.12.042
-
31.
Tao SW, Li H, Li J, et al. (2013) Treatment choice of different stages for neovascular glaucoma. Int Eye Sci 13:519–521. https://doi.org/10.3980/j.issn.1672-5123.2013.03.29
-
32.
Malik R, Ellingham RB, Suleman H, Morgan WH (2006) Refractory glaucoma - Tube or diode? Clin Exp Ophthalmol 34:771–777. https://doi.org/10.1111/j.1442-9071.2006.01339.x
-
33.
Goulet RJ, Phan ADT, Cantor LB, WuDunn D (2008) Efficacy of the Ahmed S2 Glaucoma Valve Compared with the Baerveldt 250-mm2 Glaucoma Implant. Ophthalmology 115:1141–1147. https://doi.org/10.1016/j.ophtha.2007.10.034
-
34.
Zhu Y, Li J, Xu SK (2015) Therapeutic effects of Ex-PRESS glaucoma filtration device implantation in neovascular glaucoma. [Chinese]. Int Eye Sci 15:534–536. https://doi.org/10.3980/j.issn.1672-5123.2015.3.42
-
35.
Gil-Carrasco F, Jiménez-Román J, Turati-Acosta M, et al. (2016) Comparative study of the safety and efficacy of the Ahmed glaucoma valve model M4 (high density porous polyethylene) and the model S2 (polypropylene) in patients with neovascular glaucoma. Arch la Soc Española Oftalmol (English Ed 91:409–414. https://doi.org/10.1016/j.oftale.2016.05.012
-
36.
P. H, W. W, Y. S, E. L (2015) Comparison of clinical efficacy between intravitreal bevacizumab combined with trabeculectomy and intravitreal bevacizumab combined with cyclophotocoagulation for late stage neovascular glaucoma. Zhonghua Shiyan Yanke Zazhi/Chinese J Exp Ophthalmol 33:362–366. https://doi.org/10.3760/cma.j.issn.2095-0160.2015.04.015
-
37.
Gedde SJ, Heuer DK, Parrish RK (2010) Review of results from the tube versus trabeculectomy study. Curr. Opin. Ophthalmol. 21:123–128.
-
38.
Kato N, Takahashi G, Kumegawa K, et al. (2015) Indications and postoperative treatment for Ex-PRESS® insertion in Japanese patients with glaucoma: Comparison with standard trabeculectomy. Clin Ophthalmol 9:1491–1498. https://doi.org/10.2147/OPTH.S86504
-
39.
Wang RF, Li X, Gao XW, et al. (2009) Ahmed glaucoma valve implantation combined with anterior vitrectomy for treatment of neovascular glaucoma. Int J Ophthalmol 9:2332–2334. https://doi.org/10.3969/j.issn.1672-5123.2009.12.023
-
40.
Wan DH, Zhao Q (2015) Clinical observation of trabeculectomy with mitomycin and interferon therapy on neovascular glaucoma. [Chinese]. Int Eye Sci 15:146–148. https://doi.org/10.3980/j.issn.1672-5123.2015.1.44
-
41.
Sahyoun M, Azar G, Khoueir Z, et al. (2015) Long-Term Results of Ahmed Glaucoma Valve in Association With Intravitreal Bevacizumab in Neovascular Glaucoma. J. Glaucoma 24:383–388.
-
42.
Sheha H, Kheirkhah A, Taha H (2008) Amniotic membrane transplantation in trabeculectomy with mitomycin C for refractory glaucoma. J. Glaucoma 17:303–307.
-
43.
Wan C, Liu NN, Zhao N, et al. (2011) Comparison of surgical effectiveness between two kinds of triple operation for neovascular glaucoma. [Chinese]. Int J Ophthalmol 11:614–616. https://doi.org/10.3969/j.issn.1672-5123.2011.04.016
-
44.
Bikbov MM, Khusnitdinov II (2016) The results of glaucoma drainage tube surgery in patients with diabetes. Diabetes Mellit 19:237–241. https://doi.org/10.14341/DM2003414-16
-
45.
Yazdani S, Hendi K, Pakravan M, et al. (2009) Intravitreal bevacizumab for neovascular glaucoma: A randomised controlled trial. J Glaucoma 18:632–637. https://doi.org/10.1097/IJG.0b013e3181997211
-
46.
Christakis PG, Tsai JC, Kalenak JW, et al. (2013) The Ahmed Versus Baerveldt Study. Ophthalmology 120:2232–2240. https://doi.org/10.1016/j.ophtha.2013.04.018
-
47.
Park UC, Park KH, Kim DM, Yu HG (2011) Ahmed glaucoma valve implantation for neovascular glaucoma after vitrectomy for proliferative diabetic retinopathy. J. Glaucoma 20:433–438.
-
48.
Sisto D, Vetrugno M, Trabucco T, et al. (2007) The role of antimetabolites in filtration surgery for neovascular glaucoma: Intermediate-term follow-up. Acta Ophthalmol Scand 85:267–271. https://doi.org/10.1111/j.1600-0420.2006.00810.x
-
49.
Yue ZX, Li Y, Liu ZC (2011) Clinical oberservation of ciliary body cryotherapy, trabeculectomy and 5-FU in treatment of neovascular glaucoma. [Chinese]. Int J Ophthalmol 11:162–163. https://doi.org/10.1053/comp.2003.50033
-
50.
Tang M, Fu Y, Wang Y, et al. (2016) Efficacy of intravitreal ranibizumab combined with Ahmed glaucoma valve implantation for the treatment of neovascular glaucoma Glaucoma. BMC Ophthalmol 16:7. https://doi.org/10.1186/s12886-016-0183-7
Rights and permissions
About this article
Cite this article
Shchomak, Z., Cordeiro Sousa, D., Leal, I. et al. Surgical treatment of neovascular glaucoma: a systematic review and meta-analysis. Graefes Arch Clin Exp Ophthalmol 257, 1079–1089 (2019). https://doi.org/10.1007/s00417-019-04256-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-019-04256-8