Abstract
Purpose
To detect pre- and postoperative retinal changes in fundus autofluorescence (AF) and spectral domain optical coherence tomography (SD-OCT) and to correlate these with functional outcome in patients with primary rhegmatogenous retinal detachment (RRD).
Methods
A prospective, 30-month study of patients operated with 25-gauge vitrectomy for primary RRD. Patients were examined preoperatively and after 6 and 30 months, using ultrawide-field AF images (UWFI) (Optos 200Tx) and SD-OCT (Topcon 3D OCT-2000) imaging.
Results
Of 84 patients (84 eyes) included at baseline, 100.0 and 86.9% were re-examined at month 6 and 30, respectively.
Preoperative findings such as macular attachment, detachment > 750 μm from foveola, lack of intraretinal separation, and subfoveal elevation ≤ 500 μm were all associated with better BCVA at months 6 and 30. Postoperative disruption of the photoreceptor layer was associated with poor BCVA at month 6 (p < 0.001) but not at month 30.
At baseline, AF-demarcation of RRD was demonstrated by a hyperfluorescent edge in 92.0% and was associated with visual impairment at months 6 (p = 0.003) and 30 (p = 0.003).
Visual outcome at month 30 was good (≤ 0.3 logMAR (≥ 20/40 Snellen)), regardless of the preoperative, macular status. However, with significantly better visual outcome in patients with macula attachments versus partly or totally macular detachments (p < 0.001).
Conclusion
Fundus AF and SD-OCT is able to identify retinal reestablishment up to 30 months after primary RRD, with good correlation to BCVA. These findings emphasize the importance of long-term studies for final visual recovery.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rhegmatogenous retinal detachment (RRD) is an acute ophthalmic condition, which may cause severe visual impairment or blindness. Anatomical success rates have improved remarkably during the last decades and final anatomical success rates are now close to 100% [1,2,3]. However, despite anatomical success, functional outcome is still variable [4,5,6]. Visual impairment after RRD have previously been associated with macular status [6, 7], area of detachment [6,7,8,9], duration of macular detachment [9, 10], height of macular detachment [10], preoperative proliferative vitreoretinopathy (PVR) [7, 8, 10], cystic macular edema [10], retinal folds [10, 11], subretinal fluid [10], epiretinal membranes [10], and disruption of the inner segment/outer segment (IS/OS) junction [12].
Spectral domain optical coherence tomography (SD-OCT) provides high-resolution images of the macular architecture. This can be used to visualize and evaluate the microstructural changes before and after successful reattachment. Likewise, fundus autofluorescence (AF) can be used to detect retinal changes due to hyperfluorescent, stress-induced lipofuscin-accumulation in the retinal pigment epithelium (RPE) [13]. Studies have used fundus AF to detect macular displacement following RRD [14, 15]. They found that macular displacement, following successful RRD surgery, was associated with visual disturbance, as distortion of lines or objects appearing smaller or narrower.
Short-term prospective studies have identified important risk factors for postsurgical failure [16], but long-term studies of retinal markers for functional outcome are needed.
Hence, we aimed to evaluate whether results from SD-OCT and fundus AF images could serve as non-invasive predictors for long-term visual acuity in RRD.
Methods
Study design and patient population
We performed a clinical, prospective long-term follow-up study of patients who underwent surgery for primary RRD at the Department of Ophthalmology, Odense University Hospital, Odense, Denmark. The inclusion period was between 1st of January 2013 and 12th of July 2013, and follow-up time was 30 months. At the time of the study, the department was the only vitreoretinal unit in the Region of Southern Denmark, an area with approximately 1.2 million inhabitants. Patients younger than 16 years were referred to other Danish hospitals according to Danish health care regulations.
This study was approved by the Research Ethics Committee of the Region of Southern Denmark and by the Danish Data Protection Agency. All parts of the study were conducted in accordance with the criteria of Helsinki II Declaration and in accordance with good epidemiological and clinical practice. Written informed consent was obtained from all patients.
Baseline examinations
The attending ophthalmologist at the department performed standard eye examination, including best corrected visual acuity (BCVA) as measured by Snellen at 6 m with subjective refraction and custom clip-on trial frame with glasses, intraocular pressure (IOP) (Goldmann applanation tonometry), and slit-lamp biomicroscopy using the 90 diopter and three-mirror lens. The presumed duration of macular detachment (partial/total) was defined as self-reported time from significant loss of vision to primary RRD surgery. All examinations were performed as close to primary operation as possible. Images were captured after mydriatic eye drops (one drop of tropicamide 1%, and one drop of epinephrine 10%) in each eye.
Patient baseline examinations were performed by the study investigator (CP). Examination included auto refraction (Canon RK-F2, Full Auto Ref-Keratometer, Amstelveen, The Netherlands), SD-OCT (3D OCT-2000, Topcon, Tokyo, Japan) and ultra-widefield imaging (Optos 200Tx, Optos plc, Dunfermline, Scotland, UK). The macular status (attached/partly detached/detached) was determined by SD-OCT. In cases where macula could not be detected by SD-OCT, surgical descriptions were used. Intraretinal separation was graded in patients with macula detached or partly detached. It was defined/graded as separation of the intraretinal microstructures and layers (yes/no). Patients with no intraretinal separation had near-normal appearance of the macula.
SD–OCT images were graded by the same trained grader (MPP). Fifteen percent of SD-OCT images were re-graded by another trained grader (CP) to test for agreement. Intraclass correlation coefficient (ICC) was very high for subfoveal and central retinal thickness measurements (> 0.99 for both), and kappa values were fair (subfoveal bleb 0.33), moderate (intraretinal separation 0.46), good (cystic macular edema 0.75), and very good (macular status 1.00, detachment distance from fovea 1.00), respectively. Color and AF images were graded by the same trained grader (CP) by a standardized protocol. PVR was graded by the classification system defined by Machemer et al. [17] in grades A, B, and C. Retinal area (clock hours) and type of detachment (bullous, shallow or combined) were graded using color ultra-widefield images.
Surgical information
Patients were allocated for operation as soon as possible with respect to established guidelines. Patients with macula attached were admitted to bed rest, with allowance to restroom visits and to sit while eating. If patients were admitted Friday, Saturday, or during holidays, they were operated the next following weekday. Patients with macula detached were sent home with information of minimal physical exertion until the time of operation. This was performed within a week and preferably as soon as possible.
Patients were treated with pars plana vitrectomy (PPV), using a standard 25-gauge three-port system (Constellation, Alcon, Fort Worth, TX, USA). Peeling of the internal limiting membrane (ILM) was, at the time of study, a standard combination to PPV. Silicon oil (1000 cSt) or sulfur hexafluoride gas was used for tamponade. Gas was the preference but oil was used in eyes with inferior tears, severe or complicated cases, or at the discretion of the surgeon.
During the study, the vitreoretinal unit consisted of one experienced and four highly experienced vitreoretinal surgeons. Intraoperative data were collected using the surgical descriptions.
Follow-up examinations
Follow-ups were performed by the study investigator (CP) at months 6 and 30. Examinations included a complete ophthalmologic examination including auto refraction, BCVA measured by the Early Treatment Diabetic Retinopathy Study (ETDRS) scale, IOP, slit-lamp biomicroscopy, SD-OCT, and ultra-widefield imaging.
Data and statistical analyses
For statistical analysis, Snellen and ETDRS values were converted to logarithm of minimal angle of resolution (logMAR) using the base of the reciprocal of the Snellen fraction and a 0.02 logMAR-unit for each letter correctly read on the ETDRS chart. We allocated logMAR values for counting fingers (logMAR = 2.5), hand movements (logMAR = 2.7), and light perception (logMAR = 3.0). Good BCVA was defined as ≤ 0.3 logMAR (≥ 20/40 Snellen) and poor BCVA as > 0.3 logMAR (< 20/40 Snellen).
Descriptive statistics were used for distribution of demographic and clinical characteristics. Non-parametric data was expressed in median (with range). Differences in categorical data were analyzed using the chi-square test or Fisher’s exact test for small samples. Differences in continuous data were analyzed using Mann-Whitney U test, and Kruskal-Wallis was used for three-way comparisons. For OCT grading, inter-rater agreement was determined by kappa (categorical variables) and ICC (continuous variables).
Due to small subsample sizes, regression analysis was not performed. P values < 0.05 were considered as statistically significant. All statistics was performed by Stata 13.0 (StataCorp, College Station, TX, USA).
Results
A total of 110 patients with primary RRD were seen during inclusion. We excluded patients who were not operated for social or medical reasons (n = 3), patients with pre-existing retinal or macular pathology and reduced vision before RRD (≤ 20/40 Snellen, n = 9), patients with previous eye trauma (n = 2), and patients with no follow-up data (n = 5). Eighty-four patients (84 eyes) were included in the study (Fig. 1). Mean age of included patients was 63.3 years (SD 9.9). Table 1 presents baseline demographics and characteristics for included patients.
Gradable images
Baseline SD-OCT and fundus AF images were obtained in 82 (97.6%) and 80 (95.2%) patients. Of these, 11 (12.3%) and 2 (2.5%) were non-gradable due to poor image quality. Patients with gradable images at baseline and follow-up were included for long-term analysis of median BCVA according to structural finding in SD-OCT and AF images at baseline and follow-up. Three patients could only attend follow-up after 2 months. We included these patients in month 6 evaluations as the results did not significantly change when including them.
Inclusions versus exclusions
Baseline characteristics for included (n = 84) and excluded patients (n = 26), as well as for attendees at month 30 (n = 73) and patients who dropped out during follow-up (n = 11), were analyzed. Excluded patients and patients who dropped out were more likely to have macula detached at the time of operation (p = 0.005 and p = 0.02). In addition, non-attendees at month 30 were more likely to be older (p = 0.004) and to be pseudophakic (p = 0.06) as compared to attendees at month 30.
Surgery
Patients with PVR grade B or C were more likely to be treated with PPV and silicon oil (n = 25, 43.1%) as compared to those without PVR or with grade A PVR (n = 7, 26.9%, p = 0.16). Peeling of the internal limiting membrane was performed in most eyes (n = 78, 92.9%). Nearly all patients who were phakic at baseline had cataract surgery prior to month 30 (42 of 43 patients). Six patients (7.1%) were operated for retinal re-detachment within 3 months from the primary operation. All re-detachments were surgically re-attached. However, one patient still had silicon oil in the eye at month 30. The rate of anatomical success for attendees at month 30 was 98.6% (72 of 73 patients). Patients with re-detachment were included in the evaluation. The inclusion did not significantly change the results.
Pre- and postoperative changes detected by SD-OCT
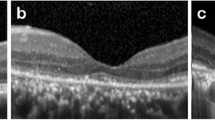
Median foveal thickness at month 6 in patients with macula attached and detached/partly detached was 261 μm (range 132–543 μm) and 226 μm (range 87–310 μm), respectively (p = 0.08). At month 30, median foveal thickness was increased to 269 μm (range 173–1112 μm) and 245 μm (range 158–571 μm), respectively (p = 0.13). These changes were not statistically significant, as compared to month 6 (p = 0.28 and p = 0.052 for patients with macula attached and detached/partly detached, respectively), but indicate regeneration. Figure 2 illustrates different structural changes in UWFI, AF, and SD-OCT images.
Structural pre- and postoperative changes in OCT are presented in Table 2. Patients with preoperative macula attachment had better visual outcome at months 6 and 30 as compared to patients with macula partly or totally detached. For those groups, median BCVA was 0.02 (20/21), 0.20 (20/32), and 0.37 logMAR (20/47 Snellen) at month 6 (p < 0.001) and 0.02 (20/21), 0.10 (20/25) and 0.26 logMAR (20/36 Snellen) at month 30 (p < 0.001), respectively. Likewise, preoperative RRD > 750 μm from foveola (p = 0.009 and p = 0.01), lack of intraretinal layer separation (p = 0.004 and p = 0.006), and subfoveal elevation ≤ 500 μm (p = 0.02 and p = 0.04) were all associated with a better outcome at month 6 and 30. At month 6, disruption of IS/OS junction was demonstrated in 50.0% and associated with a decreased BCVA (0.34 vs 0.06 (20/44 vs 20/23 Snellen), p < 0.001). However, at month 30, IS/OS junction disruption was only present in 25.0% and there was no association with decreased BCVA (p = 0.19). Postoperative changes like subfoveal bleb (p = 0.83 and p = 0.76), cystic macular edema (p = 0.47 and p = 0.17), or retinal folds (p = 0.40 and p = 0.58) were not associated with BCVA at month 6 and 30, respectively.
Pre- and postoperative changes detected by AF
Structural pre- and postoperative changes in fundus AF are presented in Table 3 and illustrated in Fig. 2. Notably, 69 patients (92.0%) had a hyperfluorescent edge (HFE, Fig. 2, V) of RRD prior to surgery. The HFE correlated with the area, judged by OCT, with impression of extent of fluid. As compared to patients without HFE, the HFE group had a decreased median BCVA at month 6 (0.22 vs. − 0.10 logMAR (20/33 vs. 20/16 Snellen), p = 0.003) and month 30 (0.14 vs. − 0.05 logMAR (20/28 vs. 20/18 Snellen), p = 0.003), respectively.
Other preoperative AF-findings like hypo- or hyperfluorescence of the area of RRD or PVR were not associated with postoperative BCVA. However, the presentation of bullous RRD’s and severe PVR (grade B and C) was highly hypofluorescent with well-defined borders.
Ghost vessels have previously been described [11, 14, 15] as hyperfluorescent lines in RPE parallel to retinal blood vessels localized in previous areas of detachment. Neither postoperatively detected ghost vessels nor HFE correlated with BCVA. Figure 3 illustrates ghost vessels in a patient with macula off at time of surgery.
Discussion
To the best of our knowledge, this is the first prospective study of substantial duration to evaluate the pre-and postoperative retinal changes by SD-OCT and AF UWFI in relation to long-term visual outcome, in patients operated with 25-gauge vitrectomy for primary RRD.
Macular detachment has been identified as the primary structural reason for visual impairment in RRD [18, 19]. However, retinal imaging technology has gone through a rapid development during the last 50 years, especially the developments of scanning laser ophthalmoscopy, AF, and SD-OCT. These techniques have made it possible to study microstructural changes which may be associated to visual impairment in patients with RRD.
In our study, SD-OCT showed to be a valuable imaging tool for the assessment of postoperative visual loss. We found a worse BCVA at months 6 and 30 in patients with preoperative macular detachment/partly detachment, retinal detachment ≤ 750 μm from foveola, a high subfoveal amplitude of retinal detachment (> 500 μm), and in patients with the presence of intraretinal layer separation in the detached macula. These findings are consistent with previous reports [6,7,8,9,10, 12, 20].
The preoperative presence of intraretinal separation in the detached macula has previously been found to correlate with higher incidence of postoperative disruption in the photoreceptor IS/OS junction and the ELM [20]. This study was not designed to investigate this correlation. However, we found that disruption of the IS/OS junction was present in half of the patients at month 6, compared to only one in four patients at month 30. Disruption of the IS/OS junction was only associated with visual impairment at month 6, indicating retinal repair and reestablishment of the photoreceptors and cone function between months 6 and 30. Disruption of the IS/OS junction has been associated with cone dysfunction in other retinal diseases [21].
Like previous studies, we found a high anatomical success rate of RRD [1,2,3]. Surprisingly, final functional outcome was good (≤ 0.3 logMAR (≥ 20/40 Snellen)) regardless of the preoperative, macular status, but patients with macula attached had significantly better final visual outcome, as compared to patients with partly or total macular detachment (p < 0.001). These findings were in accordance with findings in previous studies [6, 7, 10].
Fundus AF UWFI was a reliable method to demark RRD and to identify areas of advanced PVR (grade B and C). These findings may be explained by the fact that lipofucin accumulates due to stress and that RPE cell damage leads to loss of lipofuscin, respectively [13]. In addition, the preoperative presence of a hyperfluorescent edge was demonstrated in more than 90% of patients. A previous study found that the presence of HLE corresponded with the presence of shallow detachment with subretinal fluid in OCT [22], However, they did not correlate preoperative HLE to visual outcome. In our study, preoperative HLE was associated with visual impairment at month 6 and at month 30. As far as we know, this has not been examined in other studies of RRD. However, in non-exudative age-related macular degeneration, focal hyper-autofluorescence has been identified in the junctional zone surrounding patches of atrophy [23] and has been given as an indicator of upcoming atrophy and visual dysfunction [24]. The association between HLE and worse visual outcome is an interesting finding that warrants upcoming studies with larger subsamples.
The key strength of this study is the long follow-up period and the high attendance at included patients throughout the study (86.9% at month 30). Other strengths are the same grader/examiner for all images, and the re-grading of SD-OCT images, followed by a test for agreement between graders (ICC and kappa). This was done in order to obtain a high reliability, and to validate the grading. The inter-rater agreement showed reasonable results but also implied that some grading was biased by the grader.
The study was limited by the non-randomized design and the relatively small sample size. These limitations induce the risk of bias and the possibility of not detecting small differences, respectively.
For the baseline values, the most important limitation lies in the fact that some patients had to wait more than 24 h from baseline measurements to primary operation. This delay induces the risk of incorrect baseline data, as the RRD may have progressed with a risk of macula involvement and thereby the risk of worse baseline BCVA. It was not practically possible to reduce the time delay. However, in order to address the risk of macular progression, we cross-matched the macular status in baseline SD-OCT images, with the surgical description. This cross-match did not detect any changes in the macular status from baseline to the time of surgery.
The baseline BCVA was measured by the attending ophthalmologist in Snellen. At follow-ups, the BCVA was measured by the same examiner (CP) using the ETDRS scale. To ensure comparable values, we converted Snellen and ETDRS values to logMAR. This change in measurements induces the risk of inaccurate and incorrect values.
Spectral domain-OCT and AF UWFI showed to be reliable methods that demonstrate retinal repair and reestablishment of functional outcome up until 30 months after primary surgery for RRD. Furthermore, findings strengthen the idea that a detached macula with near-normal appearance has more visual potential as compared to the detached macula with a disrupted intraretinal appearance. Taken together, these findings suggest a role for preoperative SD-OCT and UWFI, especially in the selection of patients who might benefit from early surgery, despite macula off. Furthermore, retinal images may also help the surgeon in preoperative preparations and in counseling patients when informed about their RRD.
Upcoming randomized studies should test SD-OCT and AF methods as non-invasive markers of long-term functional outcome in RRD.
References
Heimann H, Bartz-Schmidt KU, Bornfeld N, Weiss C, Hilgers RD, Foerster MH (2008) Primary pars plana vitrectomy. Techniques, indications, and results. Ophthalmologe 105(1):19–26. https://doi.org/10.1007/s00347-007-1672-0
Wong CW, Wong WL, Yeo IY, Loh BK, Wong EY, Wong DW, Ong SG, Ang CL, Lee SY (2014) Trends and factors related to outcomes for primary rhegmatogenous retinal detachment surgery in a large asian tertiary eye center. Retina 34(4):684–692. https://doi.org/10.1097/IAE.0b013e3182a48900
Eibenberger K, Georgopoulos M, Rezar-Dreindl S, Schmidt-Erfurth U, Sacu S (2018) Development of surgical management in primary rhegmatogenous retinal detachment treatment from 2009 to 2015. Curr Eye Res 43(4):517–525. https://doi.org/10.1080/02713683.2018.1428996
Falkner-Radler CI, Myung JS, Moussa S, Chan RV, Smretschnig E, Kiss S, Graf A, D'Amico DJ, Binder S (2011) Trends in primary retinal detachment surgery: results of a bicenter study. Retina 31(5):928–936. https://doi.org/10.1097/IAE.0b013e3181f2a2ad
Heimann H, Bartz-Schmidt KU, Bornfeld N, Weiss C, Hilgers RD, Foerster MH (2007) Scleral buckling versus primary vitrectomy in rhegmatogenous retinal detachment: a prospective randomized multicenter clinical study. Ophthalmology 114(12):2142–2154. https://doi.org/10.1016/j.ophtha.2007.09.013
Pastor JC, Fernandez I, Rodriguez de la Rua E, Coco R, Sanabria-Ruiz Colmenares MR, Sanchez-Chicharro D, Martinho R, Ruiz Moreno JM, Garcia Arumi J, Suarez de Figueroa M, Giraldo A, Manzanas L (2008) Surgical outcomes for primary rhegmatogenous retinal detachments in phakic and pseudophakic patients: the retina 1 project—report 2. Br J Ophthalmol 92(3):378–382. https://doi.org/10.1136/bjo.2007.129437
Heimann H, Zou X, Jandeck C, Kellner U, Bechrakis NE, Kreusel KM, Helbig H, Krause L, Schuler A, Bornfeld N, Foerster MH (2006) Primary vitrectomy for rhegmatogenous retinal detachment: an analysis of 512 cases. Graefes Arch Clin Exp Ophthalmol 244(1):69–78. https://doi.org/10.1007/s00417-005-0026-3
Mitry D, Awan MA, Borooah S, Siddiqui MA, Brogan K, Fleck BW, Wright A, Campbell H, Singh J, Charteris DG, Yorston D (2012) Surgical outcome and risk stratification for primary retinal detachment repair: results from the Scottish retinal detachment study. Br J Ophthalmol 96(5):730–734. https://doi.org/10.1136/bjophthalmol-2011-300581
Park DH, Choi KS, Sun HJ, Lee SJ (2018) Factors associated with visual outcome after macula-off rhegmatogenous retinal detachment surgery. Retina 38(1):137–147. https://doi.org/10.1097/iae.0000000000001512
Abouzeid H, Wolfensberger TJ (2006) Macular recovery after retinal detachment. Acta Ophthalmol Scand 84(5):597–605. https://doi.org/10.1111/j.1600-0420.2006.00676.x
Dell'Omo R, Mura M, Lesnik Oberstein SY, Bijl H, Tan HS (2012) Early simultaneous fundus autofluorescence and optical coherence tomography features after pars plana vitrectomy for primary rhegmatogenous retinal detachment. Retina 32(4):719–728. https://doi.org/10.1097/IAE.0b013e31822c293e
Shimoda Y, Sano M, Hashimoto H, Yokota Y, Kishi S (2010) Restoration of photoreceptor outer segment after vitrectomy for retinal detachment. Am J Ophthalmol 149(2):284–290. https://doi.org/10.1016/j.ajo.2009.08.025
Sparrow JR, Boulton M (2005) RPE lipofuscin and its role in retinal pathobiology. Exp Eye Res 80(5):595–606. https://doi.org/10.1016/j.exer.2005.01.007
Lee E, Williamson TH, Hysi P, Shunmugam M, Dogramaci M, Wong R, Laidlaw DA (2013) Macular displacement following rhegmatogenous retinal detachment repair. Br J Ophthalmol 97(10):1297–1302. https://doi.org/10.1136/bjophthalmol-2013-303637
Shiragami C, Shiraga F, Yamaji H, Fukuda K, Takagishi M, Morita M, Kishikami T (2010) Unintentional displacement of the retina after standard vitrectomy for rhegmatogenous retinal detachment. Ophthalmology 117(1):86–92.e81. https://doi.org/10.1016/j.ophtha.2009.06.025
Wickham L, Ho-Yen GO, Bunce C, Wong D, Charteris DG (2011) Surgical failure following primary retinal detachment surgery by vitrectomy: risk factors and functional outcomes. Br J Ophthalmol 95(9):1234–1238. https://doi.org/10.1136/bjo.2010.190306
Machemer R, Aaberg TM, Freeman HM, Irvine AR, Lean JS, Michels RM (1991) An updated classification of retinal detachment with proliferative vitreoretinopathy. Am J Ophthalmol 112(2):159–165
Wilkinson CP (1981) Visual results following scleral buckling for retinal detachments sparing the macula. Retina 1(2):113–116
Burton TC (1982) Recovery of visual acuity after retinal detachment involving the macula. Trans Am Ophthalmol Soc 80:475–497
Joe SG, Kim YJ, Chae JB, Yang SJ, Lee JY, Kim JG, Yoon YH (2013) Structural recovery of the detached macula after retinal detachment repair as assessed by optical coherence tomography. Korean J Ophthalmol 27(3):178–185. https://doi.org/10.3341/kjo.2013.27.3.178
Hood DC, Zhang X, Ramachandran R, Talamini CL, Raza A, Greenberg JP, Sherman J, Tsang SH, Birch DG (2011) The inner segment/outer segment border seen on optical coherence tomography is less intense in patients with diminished cone function. Invest Ophthalmol Vis Sci 52(13):9703–9709. https://doi.org/10.1167/iovs.11-8650
Witmer MT, Cho M, Favarone G, Chan RV, D'Amico DJ, Kiss S (2012) Ultra-wide-field autofluorescence imaging in non-traumatic rhegmatogenous retinal detachment. Eye (Lond) 26(9):1209–1216. https://doi.org/10.1038/eye.2012.122
Holz FG, Bellman C, Staudt S, Schutt F, Volcker HE (2001) Fundus autofluorescence and development of geographic atrophy in age-related macular degeneration. Invest Ophthalmol Vis Sci 42(5):1051–1056
Schmitz-Valckenberg S, Bindewald-Wittich A, Dolar-Szczasny J, Dreyhaupt J, Wolf S, Scholl HP, Holz FG, Fundus Autofluorescence in Age-Related Macular Degeneration Study G (2006) Correlation between the area of increased autofluorescence surrounding geographic atrophy and disease progression in patients with AMD. Invest Ophthalmol Vis Sci 47(6):2648–2654. https://doi.org/10.1167/iovs.05-0892
Funding
This study was funded by grants from Alice Rasmussen Memorial Fund, the Foundation of A. P. Møller and Chastine Mc-Kinney Møller, the Research Grant at the University of Southern Denmark, Director Jakob Madsen and Wife Olga Madsen Foundation, King Christian the X Foundation, Henry and Astrid Møller’s Foundation, A.J. Andersen and Wife Foundation, Einar Willumsen’s Memorial Fund, Department of Clinical Research Foundation at Odense University Hospital, the Region of Southern Denmark, and the Institute of Clinical Research at the University of Southern Denmark.
The included data has not been presented before.
Author information
Authors and Affiliations
Contributions
CDP, JG, TP, and AG contributed to the concept and design of the study. CDP contributed to the acquisition of data. MPP contributed to the grading of SD-OCT images.
Data analyses were performed by CDP, who also wrote the initial draft of the paper. AG, TP, and JG revised the paper critically for intellectual content. All the authors approved the final version of the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This study was approved by the Danish Data Protection Agency and the Research Ethics Committee of the Region of Southern Denmark. All parts of the study were conducted in accordance with the Helsinki declaration II and in accordance with good clinical practice.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Poulsen, C.D., Petersen, M.P., Green, A. et al. Fundus autofluorescence and spectral domain optical coherence tomography as predictors for long-term functional outcome in rhegmatogenous retinal detachment. Graefes Arch Clin Exp Ophthalmol 257, 715–723 (2019). https://doi.org/10.1007/s00417-018-04222-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-018-04222-w