Abstract
Purpose
To assess treatment effects following intravitreal injection of ocriplasmin for vitreomacular traction (VMT), with or without full-thickness macular hole (FTMH), in real-life setting.
Methods
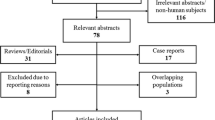
This is a monocentric, retrospective, consecutive series of 82 eyes from 82 patients who underwent ocriplasmin treatment between July 2013 and December 2016. We included 57 eyes with pure VMT, 17 eyes with small FTMHs, and eight eyes with medium FTMHs. Primary outcome measures were VMT release and MH closure rates. Secondary outcomes were visual acuity (VA), morphological changes, and subjective visual impairment after 1, 3, and 6 months and at last follow-up.
Results
After a median follow-up of 10 months, VMT release was achieved by pharmacologic vitreolysis in 57% of all eyes, whereas the macular hole closure rate was 32%. In those presenting with five or more positive prognostic factors (PPF), eyes with pure VMT showed nonsurgical traction release in 88%, and FTMHs were released in 93%, with a closure rate of 20%. Small FTMHs closed in 41% and medium FTMHs in 13%. The mean change in VA (LogMAR) was −0.07 ± 0.24 (median − 0.10) in all eyes. Subretinal fluid accumulation and ellipsoid zone changes were seen in 31% and 37% of all eyes, respectively. They were more frequent in eyes with traction release, but were self-limited.
Conclusions
In a real-life setting, release of VMT by ocriplasmin injection can be achieved in the majority of eyes, relying on a strict patient selection. Closure of FTMHs rather correlates with hole diameter than with presence of PPF, and remains a rare finding in medium FTMHs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pharmacological vitreolysis with ocriplasmin (Jetrea®, ThromboGenics USA, Alcon/Novartis EU, Basel, Switzerland) is a treatment option for eyes with vitreomacular traction (VMT) ≤ 1500 μm with or without associated full-thickness macular hole (FTMH) ≤ 400 μm. Until 2013, when ocriplasmin was not available, vitrectomy with or without internal limiting membrane (ILM) removal was the only therapeutic strategy to restore visual function in these eyes [1,2,3].
Conducted as multicentric, randomized, sham injection-controlled, double-blinded studies, MIVI-TRUST trials evaluated the efficacy and safety of ocriplasmin injection for the pharmacological treatment of VMT and FTMH [4,5,6,7]. Stalmans et al. reported that release of VMT was achieved in 26.5% at day 28 compared to 10.1% in the control group [5]. Notably, post-marketing clinical studies revealed a much higher rate of traction release presenting numbers ranging from 30 to 78% of eyes [8,9,10,11,12,13,14]. An improvement in visual function in ocriplasmin treated eyes was shown in most cases. Still, there is an ongoing discussion on the predictability of VMT release and closure of FTMHs in subgroups of patients.
Adverse events such as visual acuity loss, photopsia or dyschromatopsia were frequently reported [15,16,17,18,19,20,21]. Decline in visual function was found to be associated with structural outer retinal layer changes such as ellipsoid zone disturbances or subretinal fluid accumulation on spectral-domain optical coherence tomography (SD-OCT) analysis. Similar to electroretinogram abnormalities, these issues were reported to be correlated with VMT release [15]. However, the variability in duration of OCT changes and the occurrence of severe adverse events such as retinal detachment prompted a discussion on the risk/benefit profile of ocriplasmin treatment. To date, there is no proven information on the underlying pathogenesis of visual impairment and OCT changes in some of the treated eyes. A toxic effect of ocriplasmin on the interphotoreceptor layer has been suggested [22].
Data regarding real-life experiences are still limited, mostly due to a restricted number of cases or a limited period of follow-up. Therefore, the purpose of this study was to evaluate the real-life morphological and functional outcomes of a consecutive series of patients who were treated with a single intravitreal ocriplasmin injection, to determine subgroups of eyes that had best responded to treatment, and to describe the frequency and nature of adverse events in order to achieve better patient selection and patient information.
Patients and methods
This is a retrospective study of a consecutive series of 82 eyes of 82 patients who underwent a single ocriplasmin injection (125 μg) for focal VMT associated with or without FTMH between July 2013 and December 2016 at the Department of Ophthalmology, Ludwig-Maximilians-University, Munich, Germany. We included 57 eyes with pure VMT, 17 eyes with small FTMH, and eight eyes with medium FTMH in association with focal VMT. Primary outcome measures were VMT release and MH closure rates. Secondary outcomes were visual acuity (VA) changes, morphological OCT changes, and subjective visual disturbances.
The institutional review board and the Ethics Committee of the Ludwig-Maximilians-University Munich approved the retrospective review of the patients’ data (No. 471–14). This study was conducted according to the tenets of the Declaration of Helsinki.
Patients’ records were reviewed for age, gender, history of surgical interventions, VA in ETDRS letters [converted to ETDRS letters when necessary (Gregory 2010)], follow-up period, status of the lens, and presence of other significant pathology. Particular attention was given to pre- and post-injection ocular findings such as subjective VA loss, metamorphopsia, photopsia, and dyschromatopsia, evaluated within 1, 3, and 6 months, and at the time of last follow-up. Similarly, SD-OCT scans during the same visits were re-evaluated with documentation of presence and length of VMT, presence and diameter of macular hole, presence of epiretinal membrane (ERM), and presence of intraretinal cysts and subretinal fluid accumulation, as well as ellipsoid zone changes.
The presence of positive prognostic factors (PPF) was documented as follows: (1) age < 65 years, (2) female gender, (3) phakic lens status, (4) absence of epiretinal membrane, (5) FTMH size <250 μm, (6) adhesion diameter < 1500 μm.
Inclusion and exclusion criteria
Patients were consecutively included in this series if they had undergone ocriplasmin injection for VMT, with or without FTMH, from July 2013 to December 2016. Patients were excluded if they were lost to follow-up for 6 months or longer.
High-resolution spectral-domain optical coherence tomography
SD-OCT images were reviewed using the Heidelberg Spectralis OCT (Heidelberg Engineering, Heidelberg, Germany). According to the classification system of the International Vitreomacular Traction Study Group, FTMH were graded as small (< 250 μm), medium (250–400 μm), or large (> 400 μm), with or without VMT. Vitreomacular traction with vitreous attachment was graded as focal (≤ 1500 μm) or broad (> 1500 μm). Macular hole diameter was measured at the narrowest point of the retinal layers. All measurements were performed using the calliper function at each image.
Ocriplasmin injection protocol
All patients received a single intravitreal injection of ocriplasmin (Jetrea, 125 μg, Alcon/Novartis, EU) following the standard recommendations for intravitreal injection. Injection was performed under sterile conditions through the pars plana (3.5 or 4.0 mm from the limbus for pseudophakic or phakic eyes, respectively). Patients were discharged if ocular discomfort was observed for at least 30 min after the injection and if intraocular pressure was measured below 25 mmHg.
Surgical procedure
If patients underwent vitrectomy during the follow-up period, the surgical technique comprised a standard pars plana 23-gauge vitrectomy. In a subset of patients, combined phaco-vitrectomy was performed depending on cataract formation. In these cases, lens surgery was followed by vitrectomy. The operative records were reviewed for the intraoperative use of brilliant blue (BB; Brilliant Peel, 0.025% brilliant blue G, Fluoron GmbH, Neu-Ulm, Germany; or Membrane Blue Dual, 0.15% trypan blue, 0.025% brilliant blue G, 4% polyethylene glycol, DORC, Düsseldorf, Germany) for internal limiting membrane (ILM) peeling, removal of an ERM, type of vitreous tamponade, postoperative positioning, and intra- or postoperative complications.
Data collection and statistical analysis
Raw clinical data and SD-OCT data were extracted from medical records and evaluated blinded. For statistical analysis, VA measurements were converted to the logarithm of minimum angle of resolution units (LogMAR). Statistical analysis was performed using IBM® SPSS® Statistics 23.0 Software (SPSS Inc., IBM Software Group. Chicago, IL, USA. 2015). Statistical significance was proven with Fisher’s exact test, and nonparametric tests including the Wilcoxon and Mann-Whitney tests. P-values <0.05 were considered to be statistically significant.
Results
Patient characteristics
Fifty-three women and 29 men were included in this study, corresponding to 31 right eyes and 51 left eyes. The mean patient age was 72 ± 10 years (median 73 years, range 50–90 years). The mean follow-up period was 14.1 ± 8.8 months (median 10 months, range 6–32 months). Patient baseline characteristics are presented in Table 1.
The mean adhesion length of VMT in eyes with pure VMT was 552.2 ± 567.9 μm (median 467 μm, range 51–3829 μm). The mean macular hole diameter in eyes with VMT and FTMH was 202.3 ± 91.1 μm (median 195.0 μm, range 50–397 μm).
Treatment success, defined as VMT release following ocriplasmin treatment in eyes with pure VMT or macular hole closure in eyes with VMT and FTMH, was found in 31/82 (37.8%) eyes. During the follow-up period, 36/82 (43.9%) eyes underwent pars plana vitrectomy (PPV). In another four eyes, vitrectomy was recommended but patients refused surgery. Before ocriplasmin injection, 55/82 (67.1%) patients were phakic. At the time of last follow-up, 52/82 (63.4%) eyes were pseudophakic.
Vitreomacular traction release
In this study population, VMT was released in 45/82 (54.9%) eyes following ocriplasmin treatment during a median follow-up of 10 months. After 6 months of follow-up, 52% (43/82) of eyes showed nonsurgical release rat of VMT. In 70.5% of these 45 eyes, traction release was observed within 30 days after ocriplasmin injection. In the subgroup of eyes with four or more PPF, traction release was seen in 30/41 (73.2%) eyes. If five or more PPF were found, traction release occurred in 21/23 (91.3%) eyes.
In the subgroup of eyes with pure VMT, traction release was found in 23/57 (40.4%) eyes. The traction release rate was higher in smaller adhesion sites and correlated with the width of vitreomacular adhesion (Mann-Whitney test; P = 0.04). Of these, eyes with at least four PPF showed traction release in 60.0% (12/20 eyes), and eyes with five or more PPF in 87.5% (7/8 eyes). The rate of traction release significantly correlated with higher numbers of PPF (Mann-Whitney test; P = 0.001). In the subgroup of eyes with pure VMT and no ERM, on SD-OCT, traction release was achieved in 19/35 (54.3%) eyes. In the subgroup of eyes with pure VMT and no additional retinal pathology, traction release was seen in 11/31 (35.5%) eyes.
Macular hole closure
In eyes with FTMH, VMT was released in 88.0% (22/25 eyes). In contrast, macular holes closed in 32.0% (8/25 eyes). If four or more PPF were found, VMT was released in 85.7% (18/21 eyes) and macular hole closure was seen in 23.8% (5/21 eyes). If five or more PPF were present, VMT was released in 14/15 (93.3%) eyes and macular holes closed in 3/15 (20.0%) eyes. Small macular holes closed in 41.2% (7/17) and medium FTMH in 12.5% (1/8). Macular hole closure was related to macular hole diameter prior to ocriplasmin injection and tended to be found more frequently in smaller holes (Mann-Whitney test; P = 0.06). In contrast to the traction release rate that has been associated with higher numbers of PPF, as mentioned above, lower numbers of PPF were associated with macular hole closure (Mann-Whitney test; P = 0.03).
In cases where macular holes closed after ocriplasmin injection, 87.5% of eyes showed closure within 4 weeks after treatment. In the subgroup of patients without any other retinal pathology, such as age-related macular degeneration (AMD) or retinal vein occlusion, macular holes closured in 31.8% (7/22). In the absence of ERM on SD-OCT, the macular hole closure rate was 31.6% (6/19).
Analysis of visual function
Prior to ocriplasmin injection, the mean VA in eyes with pure VMT was LogMAR 0.40 ± 0.23 (median 0.30), and in eyes with FTMH, LogMAR 0.56 ± 0.19 (median 0.50). At last follow-up, the mean VA in eyes with pure VMT was LogMAR 0.38 ± 0.32 (median 0.30), and in eyes with FTMH, LogMAR 0.39 ± 0.28 (median 0.40). The mean change in VA from baseline to last FU in eyes with pure VMT and traction release was LogMAR −0.07 ± 0.17 (median 0.00), and in eyes with FTMH and nonsurgical macular hole closure it was LogMAR −0.25 ± 0.25 (median − 0.30) (Mann-Whitney test; P = 0.07).
Comparing VA change in eyes that presented with nonsurgical traction release after ocriplasmin injection (see above) and eyes with pure VMT that underwent vitrectomy for persistent VMT (LogMAR 0.00 ± 0.27; median − 0.05), there was no significant difference (Mann-Whitney test; P = 0.38). Comparing VA change in eyes that presented with nonsurgical macular hole closure after ocriplasmin injection (see above) and eyes that underwent vitrectomy for persistent FTMH (LogMAR −0.12 ± 0.25; median − 0.15), there was no significant difference (Mann-Whitney test; P = 0.29). Functional outcomes at specific time points (1, 3, and 6 months following ocriplasmin injection, and last follow-up), differentiating between VMT resolution and persistence, are illustrated in Table 2.
Figure 1 presents functional outcomes of eyes at specific time points including 1, 3, and 6 months of follow-up, illustrating treatment success with ocriplasmin in eyes not requiring additional surgery and eyes that underwent vitrectomy for persistent VMT or FTMH.
Subjective visual impairment after ocriplasmin injection including sudden VA loss, dyschromatopsia, metamorphopsia, or photopsia was recorded in 43/82 (52.4%) eyes. Subjective complaints were documented in 25/57 (43.9%) eyes with pure VMT and 18/25 (72.0%) eyes with FTMH (Fisher’s exact test; P = 0.03). They were more frequent in eyes with VMT release than in eyes with persistent VMT (Fisher’s exact test; P < 0.001). Regarding subjective complaints in detail, in eyes with traction release (71.1%, 32/45 eyes), sudden vision loss was reported in 27.0% of patients(12/45 eyes), blurred vision in 37.7% (17/45 eyes), photopsia in 40.0% (18/45 eyes), dyschromatopsia in 17.8% (8/45 eyes), other optic complaints in 27.0% (12/45 eyes), metamorphopsia in 11.1% (5/45 eyes), and eye pain after injection in 6.7% (3/45 eyes). Acute vision loss and blurred vision resolved spontaneously after a maximum of 7 days in 91.1% and 86.7% of eyes, respectively. None of the patients complained of acute vision loss or blurred vision for more than 30 days. Among subjective complaints, photopsia was the most long-standing, and resolved spontaneously in 94% of eyes after 20 days.
Analysis of morphological changes
Following ocriplasmin injection, morphological changes in retinal layers as assessed by SD-OCT included subretinal fluid accumulation in 25/82 (30.5%) eyes, ellipsoid zone changes in 30/82 (36.6%), development of ERM in 3/82 (3.7%), and cystoid macular edema in 5/82 (6.1%). Subretinal fluid accumulation was more frequently seen in eyes with vitreomacular traction release (Fisher’s exact test; P = 0.003). Both subretinal fluid and ellipsoid zone changes resolved spontaneously, as shown in Fig. 2. Subretinal fluid and ellipsoid zone changes persisted in 4/82 (4.9%) eyes and 2/82 (2.4%) eyes, respectively, after a follow-up period of 6 months.
Follow-up examinations with spectral-domain optical coherence tomography (OCT) horizontal volume scans of same positions of a 59-year-old woman with pure VMT. a She presented with visual acuity of LogMAR 0.40 before ocriplasmin injection. b Six hours following ocriplasmin injection the patient complained of sudden vision loss and nyctalopia. On OCT scans obtained 15 h after the injection, subretinal fluid accumulation was demonstrated with VA of LogMAR 0.80. c One month following ocriplasmin treatment, subretinal fluid accumulation was reduced, and visual acuity had spontaneously improved to LogMAR 0.40. d Two months after ocriplasmin treatment, ellipsoid zone irregularity was still detected, but no subretinal fluid. Visual acuity was LogMAR 0.20. Six months following ocriplasmin injection, OCT showed neither irregularity of retinal layers nor subretinal fluid. Visual function had spontaneously recovered to LogMAR 0.10
In eyes with FTMHs, hole diameter increased significantly, from 202 ± 91 μm (median 195 μm, range 50–397 μm) to 363 ± 178 μm (median 382 μm, range 190–660 μm), following ocriplasmin injection (Wilcoxon test; P < 0.001). Retinal detachment was observed in 4/82 (4.9%) eyes, and a lamellar macular defect developed in one eye (1.2%) with prior pure VMT.
Vitrectomy following ocriplasmin injection
During follow-up, macular surgery was recommended in 40/82 (48.8%) eyes. Of these, 36 eyes (43.9%) underwent vitreoretinal surgery during that follow-up period. In eyes with persistent VMT, vitrectomy was performed after a mean period of 136 ± 110 days (median 90 days, range 50–300 days) after ocriplasmin injection. In eyes with persistent FTMH, vitrectomy was performed after a mean period of 56 ± 42 days (median 45 days, range 6–166 days) after ocriplasmin injection. Four eyes were operated for retinal detachment within 8 days after ocriplasmin injection. Eighteen of 55 eyes (32.7%) underwent PPV with BB-assisted ILM peeling for persistent VMT, 9/16 (56.3%) eyes for persistent small FTMH, and 5/7 (71.4%) eyes for persistent medium FTMH. Combined vitrectomy was performed in 24/35 (68.6%) eyes.
Although recommended, four patients had not undergone vitrectomy at the time of last follow-up examination. Two aged patients with persistent pure VMT and one 84-year-old man refused macular surgery due to poor state of health, and one other patient with pure VMT died due to advanced gastrointestinal cancer disease.
Discussion
In this monocentric consecutive series, all patients who underwent ocriplasmin treatment between July 2013 and December 2016 were retrospectively included, except for those who were lost to follow-up for less than 6 months. Based on these criteria, our study results emphasize issues of daily clinical practice with implications for a better patient selection and patient information.
In our real-life analysis, release of VMT by ocriplasmin injection was achieved in the majority of eyes when relying on a strict patient selection. If five or more PPF were found, eyes with pure VMT showed traction release in 88%, whereas the overall VMT release rate following ocriplasmin treatment was 57% in all eyes after a median follow-up period of 10 months. In accordance with previous studies, our data emphasize the prognostic value of PPF in predicting traction release in pure VMT.
However, nonsurgical macular hole closure was achieved in 32% of this series. If five or more PPF were present, FTMHs showed traction release in 93%, with a lower closure rate of 20%. Our results indicate that PPF have limited value in the prognosis of macular hole closure. In contrast, significant correlation has been found between macular hole diameter and nonsurgical closure rate. In this series, small FTMHs closed in 42% and medium FTMHs in 13%. We emphasize that closure of FTMH correlates with hole diameter rather than with the presence of PPF. Since macular hole closure remains a rare finding in medium FTMH, there is discussion among vitreoretinal surgeons on recommending ocriplasmin treatment in these cases. Some of them prefer to proceed with macular surgery in eyes with VMT and medium FTMH. Although research has shown that surgical outcomes in patients with a prior history of ocriplasmin injection are comparable to those in patients who proceed directly to surgery without ocriplasmin treatment [23, 24], recommendation of ocriplasmin injection in eyes with VMT and FTMH remains an important issue in terms of patient information and clinical setting.
When discussing pharmacologic vitreolysis with ocriplasmin as a treatment option for VMT with our patients, their decisions are typically informed by the probability of avoiding surgery, along with their need to improve visual function. In patients with pure VMT, the traction release rate represents treatment success, and no additional treatment is necessary in most cases. In contrast, traction release in patients with VMT and FTMH does not guarantee treatment success, as demonstrated herein and in other studies [19, 20, 22,23,24]. The rate of macular hole closure has been reported between 17% and 78% [13,14,15, 18]. Despite VMT release, a large number of FTMHs have been shown to persist or even increase in diameter, with a significant drop of visual function [3, 13, 22,23,24,25,26].
For statistical analysis in this study, treatment success was defined as (1) release of VMT in eyes with pure VMT, and as (2) closure of macular hole in eyes with VMT and FTMH. To date, defining treatment success following ocriplasmin injection remains controversial. Traction release is a feasible and reliable measure, and indicates the efficacy of ocriplasmin as a reagent for pharmacologic vitreolysis. Nonetheless, in eyes with FTMHs, proof of traction release is not synonymous with treatment success or avoidance of surgery. Thus, to us it appears reasonable that eyes with pure VMT and eyes with VMT and FTMH will be discussed separately due to their different behaviour following ocriplasmin treatment.
Improvement in visual function is the overall goal in treating patients. However, in this study, as well as in other reports [25, 26], VA was shown to improve not only in cases with traction release, but also in eyes with persistent vitreomacular traction. Additionally, VA is influenced by multiple variables, such as refraction correction, state of the lens, and individual dependence on shape; thus VA does not appear to be an accurate parameter for assessing treatment success with ocriplasmin. Pre-marketing and post-marketing clinical studies have revealed an improvement in visual function in ocriplasmin-treated eyes, showing a release of traction in up to 78% of cases [13,14,15]. In this study, the mean change in VA (LogMAR) among all eyes was −0.08 ± 0.24 (median − 0.1). Our results are similar to those of other studies reporting an overall improvement in VA from baseline to last follow-up in eyes with VMT resolution [25, 26]. In this study, 55% of patients gained five letters or more, compared to 25% who lost five letters or more. However, in 36/85 (42%) of all eyes, vitrectomy was performed during follow-up, and macular surgery was recommended in an additional six eyes. Thus, VA change in eyes of this series resulted partly from surgical intervention during follow-up.
Both subretinal fluid accumulation and ellipsoid zone changes were more frequently found in eyes with traction release; they were self-limited and spontaneously resolved in the majority of eyes during follow-up. Structural outer retinal layer changes such as ellipsoid zone disturbances or subretinal fluid accumulation on SD-OCT analysis have been reported to be transient changes in other studies as well. They correlated with VMT release and VA loss. The underlying pathomechanism related to these symptoms and morphological findings is not fully understood. Ocriplasmin is suspected to cause disruption of the photoreceptor layer by interacting with laminin within the interphotoreceptor matrix [27, 28]. However, it remains to be elucidated whether ocriplasmin, as an unspecific serine-protease or one of its cleavage products, accounts for this possibly toxic effect on outer retinal layers [29, 30]. It is now well accepted that these changes are most likely responsible for subjective complaints such as vision loss, blurred vision, dyschromatopsia, and other visual impairment that all resolve spontaneously after a short period of time in the majority of eyes.
Limitations of this study were largely related to its retrospective nature and the documentation of corrected VA as well as a variable period of follow-up. A center-related bias is also possible because of the single-center design. However, our study represents a consecutive series from daily clinical practice without large dropout numbers, emphasizing the clinical setting and demonstrating real-life situations for instances when patients refuse surgery.
In conclusion, pharmacologic vitreolysis with ocriplasmin is an effective treatment option for eyes with vitreomacular traction. Treatment success is strongly related to patient selection based on positive predictive factors that are most sensitive in eyes with pure VMT. In eyes with VMT and FTMH, the adherence to positive predictive factors does not seem to be essential for predictability of treatment success, since traction release is not a prerequisite for macular hole closure. According to our results, it appears advisable to instead discuss the indication for ocriplasmin treatment in eyes with VMT and macular holes in the context of hole diameter.
References
Duker JS, Kaiser PK, Binder S et al (2013) The international Vitreomacular traction study group classification of vitreomacular adhesion, traction, and macular hole. Ophthalmology 120:2611–2619
Gandorfer A, Rohleder M, Sethi C et al (2004) Posterior vitreous detachment induced by microplasmin. Invest Ophthalmol Vis Sci 45:641–647
de Smet MD, Valmaggia C, Zarranz-Ventura J, Willekens B (2009) Microplasmin: ex vivo characterization of its activity in porcine vitreous. Invest Ophthalmol Vis Sci 50:814–819
de Smet MD, Gandorfer A, Stalmans P et al (2009) Microplasmin intravitreal administration in patients with vitreomacular traction scheduled for vitrectomy: the MIVI I trial. Ophthalmology 116:1349–1355
Stalmans P, Benz MS, Gandorfer A et al (2012) MIVI-TRUST study group. Enzymatic vitreolysis with ocriplasmin for vitreomacular traction and macular holes. N Engl J Med 367:606–615
Haller JA, Stalmans P, Benz MS et al (2015) Efficacy of intravitreal ocriplasmin for treatment of vitreomacular adhesion. Subgroup analyses from two randomized trials. Ophthalmology 122:117–122
Gandorfer A, Benz MS, Haller JA et al (2015) MIVI-TRUST study group. Association between anatomical resolution and functional outcomes in the MIVI-Trust studies using ocriplasmin to treat symptomatic vitreomacular adhesion/vitreomacular traction, including when associated with macular hole. Retina 35:1151–1157
Kim BT, Schwartz SG, Smiddy WE et al (2013) Initial outcomes following intravitreal ocriplasmin for treatment of symptomatic vitreomacular adhesion. Ophthalmic Surg Lasers Imaging Retina 44:334–343
Singh RP, Li A, Bedi R et al (2014) Anatomical and visual outcomes following ocriplasmin treatment for symptomatic vitreomacular traction syndrome. Br J Ophthalmol 98:356–360
Sharma P, Juhn A, Houston SK et al (2015) Efficacy of intravitreal ocriplasmin on vitreomacular traction and full-thickness macular holes. Am J Ophthalmol 159:861.e2–867.e2
Dugel PU, Tolentino M, Feiner L et al (2016) Results of the 2-year ocriplasmin for treatment for symptomatic vitreomacular adhesion including macular hole (OASIS) randomized trial. Ophthalmology 123:2232–2247
Warrow DJ, Lai MM, Patel A et al (2015) Treatment outcomes and spectral-domain optical coherence tomography findings of eyes with symptomatic vitreomacular adhesion treated with intravitreal ocriplasmin. Am J Ophthalmol 159:20–30
Maier M, Abraham S, Frank C et al (2016) Pharmakologische Vitreolyse mit Ocriplasmin als Behandlungsoption bei symptomatischer fokaler vitreomakulärer Traktion mit oder ohne Makulaforamen (≤400 μm) im Vergleich zur transkonjunktivalen Vitrektomie. Ophthalmologe 114:148–154 German
Stalmans P, Duker JS, Kaiser PK et al (2013) OCT-based interpretation of the vitreomacular interface and indications for pharmacologic vitreolysis. Retina 33:2003–2011
Hahn P, Chung MM, Flynn HW et al (2015) Safety profile of ocriplasmin for symptomatic vitreomacular adhesion: a comprehensive analysis of premarketing and postmarketing experiences. Retina 35:1128–1134
Shah SP, Jeng-Miller KW, Fine HF et al (2016) Post-marketing survey of adverse events following ocriplasmin. Ophthalmic Sug Lasers Imaging Retina 47:156–160
Beebe DC (2015) Understanding the adverse effects of ocriplasmin. JAMA Ophthalmol 133:229
Nudleman E, Franklin MS, Wolfe JD et al (2016) Resolution of subretinal fluid and outer retinal changes in patients treated with ocriplasmin. Retina 36:738–743
Quezada-Ruiz C, Pieramici DJ, Nasir M et al (2015) Outer retina reflectivity changes on SD-OCT after intravitreal ocriplasmin for vitreomacular traction and macular hole. Retina 35:1144–1150
Itoh Y, Ehlers JP (2016) Ellipsoid zone mapping and outer retinal characterization after intravitreal ocriplasmin. Retina 36:2290–2296
Itoh Y, Kaiser PK, Singh RP et al (2014) Assessment of retinal alterations after intravitreal ocriplasmin with spectral-domain optical coherence tomography. Ophthalmology 121:2506.e2-2507.e2
Figueira J, Martins D, Pessoa B et al (2016) The Portuguese experience with ocriplasmin in clinical practice. Opthalmic Res 56:186–192
Greven MA, Garg S, Chiu B et al (2016) Vitrectomy after ocriplasmin for VitreOmacular adhesion or macular hole (VAVOOM) study. Br J Ophthalmol 100:1211–1215
Schumann RG, Wolf A, Hoerauf H et al (2017) Vitrectomy for persistent macular holes following ocriplasmin injection: a comparative multicenter study. Retina. doi:10.1097/IAE.0000000000001473
Khan MA, Haller JA (2016) Ocriplasmin for treatment of vitreomacular traction: an update. Ophthalmol Ther 5:147–159
Chatziralli I, Theodossiadis G, Xanthopoulou P, Miligkos M, Siviprasad S, Theodossiadis P (2016) Ocriplasmin use for vitreomacular traction and macular hole: a meta-analysis and comprehensive review on predictive factors for vitreous release and potential complications. Graefes Arch Clin Exp Ophthalmol 254:1247–1256
Chen W, Mo W, Sun K et al (2009) Microplasmin degrades fibronectin and laminin at the vitreoretinal interface and outer retina during enzymatic vitrectomy. Curr Eye Res 34:1057–1064
Libby RT, Lavallee CR, Balkema GW et al (1999) Disruption of laminin beta2 chain production causes alterations in morphology and function in the CNS. J Neurosci 19:9399–9411
Schumann RG, Wolf A, Mayer WJ et al (2015) Pathology of internal limiting membrane specimens following invtravitreal injection of ocriplasmin. Am J Ophthalmol 160:767–778
Vielmuth F, Schumann RG, Spindler V et al (2016) Biomechanical properties of the internal limiting membrane after intravitreal Ocriplasmin treatment. Ophthalmologica 235:233–240
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this research.
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licencing arrangements) or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Schumann, R.G., Langer, J., Compera, D. et al. Assessment of intravitreal ocriplasmin treatment for vitreomacular traction in clinical practice. Graefes Arch Clin Exp Ophthalmol 255, 2081–2089 (2017). https://doi.org/10.1007/s00417-017-3747-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-017-3747-1