Abstract
The role of the trigeminal system in facial and dural sensitivity has been recognized for a long time. More recently, the trigeminal system has also been considered a prominent actor in brain nociceptive innervation. It is the anatomical substrate of several frequent conditions, such as primary or secondary headaches, trigeminal neuralgia, and other orofacial pains. Appreciation of the delicate anatomical arrangement of the trigeminal pathway is one of the keys to understanding these conditions’ pathophysiology and to proposing innovative treatments. This review provides a structured presentation of existing knowledge about the trigeminal system, from classical anatomical data to the most recent literature. First, we present the organization of the trigeminal pathway from the trigeminal divisions, nerve, and nuclei to the thalamus and somatosensory cortex. We describe the neurons and fibers’ repartition at each level, depending on the location (somatotopic organization) and the type of receptors (modal organization). Such a dual somatotopic-modal arrangement of the trigeminal fibers is especially clear for the juxtapontine segment of the trigeminal nerve and the trigeminal nuclei of the brainstem. It has significant clinical consequences both for diagnosis and treatment. Second, we explore how the trigeminal system is modulated and involved in reflexes, for instance the trigemino-cardiac and the trigemino-autonomic reflexes, which could play an essential role in the autonomic symptoms observed in cluster headache. Finally, we present how to interact with this complex system to relieve pain mediated by the trigeminal system. This section covers both neuromodulatory and lesional approaches.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The trigeminal system is well known to support face, dura, and pia sensitivity [1,2,3,4,5,6,7], as well as motor control of chewing muscles [8]. The understanding of its involvement in brain innervation has more recently dramatically expanded, as it is now considered to be a major actor for brain nociceptive innervation [6, 9,10,11], as well as playing a role in neuroinflammation, blood flow regulation, and control of arterial wall permeability. [12,13,14,15,16].
Despite sharing a single anatomical support—the trigeminal system—primary or secondary headaches, trigeminal neuralgia, and other orofacial pains have distinct characteristics. One of the keystones in understanding the pathophysiology of these conditions is the somatotopic and modality-specific arrangement of fibers within the trigeminal system. The somatotopic arrangement refers to a point-for-point correspondence of an area of the face to a specific point of the trigeminal system. The modality-specific arrangement corresponds to the segregation of fibers depending on the modality (tact, proprioception, pain, temperature) they carry.
Methods
A PubMed search was performed, using the phrases “Trigeminovascular system”, “Trigeminal system”, “Dura mater innervation”, “Brain innervation”, “Trigeminal pathway” “Trigeminal nerve”, “Trigeminal ganglion”, “Trigeminal nuclei”, “Trigeminal tract” trigeminal nuclear complex”, trigemino-cervical complex”, “trigeminal thalamus”, “trigeminal somatosensory cortex”, “trigeminal convergence” from 1929 to 2021.
Systematic reviews, historical, experimental animal, clinical but also more recent functional imaging studies were used to clarify the arrangement of fibers at different levels of the trigeminal system. The references of these articles were examined for additional sources, and we ended up selecting 194 articles for this review. Here we will focus on specific aspects of this organization, which are important—or could become important—in managing pain of the craniofacial area.
Trigeminal nerve
This section focuses on the delicate anatomy of the trigeminal system, from its three divisions to its cortical endings. It will not cover its peripheral part, including brain vessels and meningeal innervation. Readers interested in this trigeminovascular system and its possible implication in migraine may refer to our previous dedicated review [7].
Trigeminal divisions
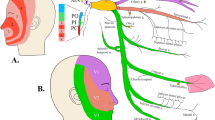
Taken together, the three divisions of the trigeminal nerve constitute the largest cranial nerve: the mandibular division (V3) counts 78,000 myelinated fibers, the maxillary division (V2) 50,000 fibers, and the ophthalmic division (V1) only 26,000. The first well-known level of somatotopy is the distribution of the dermatomes innervated by each of these divisions (Fig. 1) [17].
-
The V1 is responsible for the sensitive innervation of the upper-facial skin and cornea, and of most of the dura mater (anterior cranial fossa, falx, tentorium, venous sinuses, and occasionally parieto-occipital region) [18, 19], brain proximal arteries, pial arteries, and probably “pia arachnoid,” constituting the trigeminovascular system [1, 2, 4, 9,10,11, 20,21,22].
-
The V2 is in charge of the sensitive innervation of the mid-facial part (skin of the lower eyelid, nose, upper lip, sinuses, nasal and oral mucosa, upper gums, and teeth) and of a limited region of the dura mater (dura covering the middle cerebral fossa and the lesser wing of the sphenoid bone) [18, 23, 24].
-
The V3 innervates the lower-facial part (lower gums and teeth, skin in the temporal region, part of the auricle, lower lip) and a limited area of the temporal dura mater [18, 23, 24]. The mandibular nerve (V3) also contains efferent motor fibers for masticatory muscles.
Topographic somatotopy of the trigeminal divisions.The three trigeminal divisions (ophthalmic or V1, maxillary or V2, and mandibular or V3) of the trigeminal nerve get afferents from the mucosa and skin (a) and the dura matter (b), the dural territory of V1 being the largest one. In addition, afferent fibers from the large cerebral and pial arteries (c), and probably from the “pia arachnoid” (trigeminovascular system), reach the V1
Each of the trigeminal divisions carries fibers for touch, proprioception, pressure, vibration, and nociception from the face, and nociception from the meninges and brain blood vessels [8, 25] without any clear fibers arrangement by modality. Nevertheless, fibers of the trigeminovascular system innervating brain vessels and meninges are exclusively nociceptive and run within V1 branches [9]. This explains why the section of the V1 division has been reported to be frequently efficient in reducing headache, whereas disconnection of V2 or V3 would not affect its prevalence [4, 18]. Interestingly, some of these meningeal nociceptive fibers give collaterals reaching the extracranial space via calvarial sutures. They then join somatosensory trigeminal peripheral neurons innervating extracranial tissues such as the periosteum or the muscles [26]. This convergence of meningeal nociceptive inputs and external inputs at the level of the trigeminal peripheral fibers possibly contributes to the perception of pain originating from an intracranial stimulus (such as migraine) in the periorbital and occipital region (referred pain). [27].
Trigeminal ganglion
The three trigeminal divisions (V1, V2, and V3) reach the trigeminal ganglion, containing 20,000–150,000 cell bodies for most of the trigeminal sensory axons [17, 25]. These trigeminal neurons are pseudo-unipolar cells with two axonal branches, one extending distally towards one of the trigeminal divisions and the other running proximally up to brainstem nuclei (Figs. 2 and 3) [8]. Due to these anatomical features, trigeminal neurons are supposed to be capable of conducting action potentials in both directions: orthodromically, towards the brain stem, for touch and pain perception, but also antidromically, towards branches of the trigeminal divisions. This antidromic conduction mediates the peripheral release of inflammatory neuropeptides by the trigeminal system occurring in migraine. [6, 28].
Modal somatotopy within the juxtapontine portion. The three trigeminal divisions (ophthalmic or V1, maxillary or V2, and mandibular or V3) of the trigeminal nerve contain proprioceptive (dark blue), epicritic (light blue), and nociceptive (green) fibers. In addition, V3 includes motor (red) fibers for masticatory muscles. Within the juxtapontine portion of the trigeminal nerve, fibers are somatotopically organized depending on their modality: motor fibers originate from the motor trigeminal nucleus (MotN), run in the pars minor, at the dorsomedial aspect of the nerve, and finally join the V3 to distribute to the masticatory muscles. The pars intermediaris is located at the dorsomedial aspect of the V juxtapontine portion. It contains epicritic and proprioceptive fibers. Epicritic fibers join the principal (PN) and rostral part of the spinal (SN, not represented) trigeminal nuclei, whereas proprioceptive fibers reach the mesencephalic (MN) trigeminal nucleus. Cell bodies of epicritic and proprioceptive neurons connected to Golgi tendon organs and extraocular muscle spindles are located within the trigeminal ganglion (not represented). Cell bodies of the proprioceptive neurons connected to jaw muscles spindles are found within the mesencephalic proprioceptive nucleus of the brainstem. They send projections to the MotN for bite control (jaw-jerk closing reflex) and the thalamus (not represented). The pars major is located at the ventrolateral aspect of the nerve root. It contains nociceptive fibers, which follow the trigeminal tract (Tr Tract) to reach the spinal trigeminal nucleus (SN). Nociceptive fibers from V1, including those from the cornea, are located at the dorsomedial third of the pars major, whereas nociceptive fibers from V2 and V3 are more ventral and lateral. This explains the preservation of the corneal sensitivity after a neurotomy limited to the ventrolateral part of the V juxtapontine part
Trigeminal tract and spinal trigeminal nucleus afferences and somatotopy. A. Afferents fibers run through the three trigeminal divisions (ophthalmic or V1, maxillary or V2, and mandibular or V3) of the trigeminal nerve. V1, V2, and V3 run posteriorly towards the trigeminal ganglion (Tr gangl., primary neurons) and then the trigeminal root (Tr root). The trigeminal root converges to the pons and fibers bifurcate: fibers for touch reach the principal nucleus (PN), proprioceptive fibers enter the mesencephalic nucleus (MN, not represented), whereas nociceptive fibers follow a descending course up to C2 or C3 within the trigeminal tract (Tr tract) to finally reach the spinal trigeminal nucleus (SN). The trigeminal tract fibers are somatotopically organized, V1 being ventral, V2 intermediate, and V3 dorsal. The same ventrodorsal somatotopic organization exists in the pars oralis (SNo), interpolaris (SNi), and caudalis (SNc) of the spinal trigeminal nucleus. No clear somatotopic organization was described for PN. For figure clarity, the trigeminal tract is represented dorsally to the spinal nucleus, whereas it is lateral. C1 vent: ventral root of the first cervical spinal nerve; C2 vent: ventral root of the second cervical spinal nerve; C2 dors: dorsal root of the second cervical spinal nerve. B. Onion skin pattern of Dejerine. The face is divided into concentric rings from the perioral to the preauricular regions (a–f). Nociceptors of the central facial zone (a, b) project rostrally within the SN, whereas the most peripheral facial area is represented in the caudal SN (e, f). The rostrocaudal extent of these projections varies across publications and is drawn here on the whole SN. Nociceptive fibers from the upper cervical roots (circles) and the V1 (frontal areas e and f, and anterior skull base dura mater, superior sagittal and transverse sinuses) converge onto the same cervical central neurons. Their nociceptive stimulation induces a referred pain, which is perceived in the frontal area regardless of the intracranial or cervical site of stimulation. The precise cutaneous projections of areas a-f onto the SN are largely unknown, and the limits displayed here are only indicative
The trigeminal ganglion also contains the somata of proprioceptive neurons connected to Golgi tendon organs and extraocular muscle spindles, but not to jaw muscles spindles [29], located within the mesencephalic proprioceptive nucleus of the brainstem. These proprioceptive fibers for jaw muscles are involved in a reflex arc modulating the degree of bite [8]. They run through the motor trigeminal root, which is ventral to the trigeminal ganglion, and then follows the V3 division. [30].
Functional MRI has confirmed a somatotopic arrangement of ganglionic cells, ophthalmic cells being located superomedially, mandibular cells posterolaterally, and maxillary cells in between [25, 31].
Trigeminal root
The trigeminal sensory root extends from the trigeminal ganglion to the ventrolateral aspect of the pons. At about 12 mm from the pons, central myelin replaces peripheral one, and this transitional area (Trigeminal Root Entry Zone or TREZ) is considered to be more vulnerable, and the site of most neurovascular compressions. [32].
The percentage of myelinated fibers involved in touch and proprioception is higher (50%) within the trigeminal root than in the dorsal root of spinal nerves (20–30%), possibly because of the high tactile discrimination in the face [33]. Fibers tend to group according to their modality (Fig. 2) [34,35,36,37,38]. As a result, at the juxtapontine level, three groups of fibers can be distinguished based on their modality, from lateral to medial: pars major (nociceptive fibers), pars intermediaris (epicritic and proprioceptive fibers), and pars minor (motor fibers). Corneal fibers have classically been considered to run within the pars intermediaris dedicated to epicritic sensitivity [34] and reach pontine and spinal trigeminal nuclei. Nevertheless, electrophysiological and microscopical studies have demonstrated a purely nociceptive corneal innervation (free-endings, non-myelinated C, and small myelinated Aδ fibers), which pleads for an exclusive localization of corneal fibers within the pars major. [38].
Additionally, nociceptive fibers of the juxtapontine pars major are somatotopically organized, with an arrangement of fibers from ophthalmic (V1), maxillary (V2), and mandibular (V3) from dorsomedial to ventrolateral. Nociceptive fibers from the V1 division—including corneal ones—usually occupy the dorsomedial third of the pars major, whereas the remaining ventrolateral two-thirds contain fibers from the V2 and V3 divisions [38]. This anatomical arrangement explains why trigeminal neuralgia without vascular conflict can be successfully treated by a rhizotomy limited to the ventrolateral two-thirds of the pars major without impairment of corneal sensitivity nor of the corneal reflex (see below) [38]. By contrast, the corneal reflex usually weakens or disappears after the whole section of the pars major and preservation of pars intermediaris [36, 39, 40]. Similarly, headache improvement after total section of the pars major, but not a rhizotomy limited to its ventrolateral two-thirds, pleads for a location of trigeminovascular fibers within the dorsomedial third of the pars major.
Trigeminal tract
Shortly after entering the brainstem, some trigeminal fibers bifurcate to give a short ascending branch reaching the principal trigeminal nucleus and a long one, descending up to C2 or C3 within the trigeminal tract (Fig. 3) [25]. This tract is adjacent to the spinal trigeminal nucleus to which it distributes fibers [30]. It is caudally continued by a similar distributing system for the spinal cord, the tract of Lissauer.
In the trigeminal tract, fibers from V1 are ventral, V2 intermediate, and V3 dorsal. A somatotopic organization exists for the entire rostrocaudal extend of the nucleus, fibers conveying inputs from V1, V2, and V3 being present throughout the trigeminal tract. [30].
Clinical evidence also suggests a distribution by fibers’ modality, nociceptive fibers being lateral to heat-sensitive ones [30]. In addition, fewer myelinated fibers—dedicated to touch—have been observed in the caudal region of the trigeminal tract, as compared to its rostral region. [17].
Trigeminal nuclear complex
The trigeminal nuclear complex has been anatomically studied in animal [41,42,43,44,45,46] and by imaging studies in humans [25, 47,48,49]. It is distinct from the motor trigeminal nucleus, not included in this review. It is a long column of cells, located in the dorsolateral brainstem, subdivided into (Fig. 3):
-
The mesencephalic nucleus (MN) extending along the lateral border of the central gray, from the superior colliculus level in the upper midbrain to the middle pons. It is anterolateral to the fourth ventricle and medial to the sensory trigeminal nucleus [8].
-
The sensory trigeminal complex gathering, from rostral to caudal:
-
The principal trigeminal nucleus (PN), which is a small nucleus located in the middle pons, at the level of the entrance of the trigeminal nerve, and dorsolaterally to the motor nucleus of the trigeminal nerve [48].
-
The spinal trigeminal nucleus (SN), which is a long nucleus located in the dorsolateral region of the brainstem. It extends from the midpons (caudal end of the PN) to the cervical spinal cord levels C2 to C4, where it is continued by the dorsal horn of the cervical spinal cord [30]. SN is anterolateral to the fourth ventricle and medial to the spinal trigeminal tract [8, 25, 30, 48]. This nucleus is further subdivided into three cytoarchitecturally and functionally defined parts, from rostral to caudal [8, 25, 30, 48]: the pars oralis (SNo, from the mid-pons to the rostral third of the inferior olive), the pars interpolaris (SNi, from the rostral third of the inferior olive to the obex of the fourth ventricle), and the pars caudalis (SNc, from the obex to C2–C4 level) [50, 51]. Transitions between the various part of the sensory trigeminal complex and between the trigeminal nucleus and upper cervical spinal cord are gradual without clear limits. [50, 51].
All these nuclei have more or less pronounced—or known—modal and somatotopic arrangements.
Arrangement of the trigeminal nuclear complex by modality
The MN receives proprioceptive fibers from the teeth, periodontium, hard palate, temporomandibular joint capsule, and spindles and Golgi tendinous organs from extraocular and masticatory muscles [8]. As stated above, cell bodies of fibers connected to masticatory muscles spindles are located within the MN, whereas the others are in the trigeminal ganglion. The MN mainly projects to the motor trigeminal nucleus and is involved in bite control, but it also projects to thalami with an ipsilateral predominance. These thalamic projections can be direct or occur via the PN and are participate in proprioceptive sensation.
The PN receives fibers connected to low-threshold mechanoreceptors having small receptive fields involved in facial touch perception. It mainly projects to the VPM nucleus of the thalamus [17] and is regarded as the analogous of dorsal column nuclei, specialized in epicritic sensitivity.
Finally, the SN is mainly dedicated to nociceptive and thermoceptive fibers for the face. A finer arrangement by modality exists within the three SN subnuclei:
-SNo plays an essential role in intraoral and dental sensation, including intraoral pain, but it also receives epicritic signals from the face [50, 52, 53]. SNo cells are associated with widespread receptive fields and can show modality convergence, for instance, from both cutaneous and tooth receptors [17]. They are more specialized in processing short-duration than tonic nociceptive information after stimulation by noxious chemicals such as formalin. [17]
-SNi receives cutaneous nociceptive or low-threshold mechanoreceptive neurons, but its function remains unclear [54]. Animal studies show that PNi enlarges when the vibrissae in rodents are well developed and may be involved in deep oral pain perception, similarly to SNo [55]. It also projects to PN and SNc and may be necessary for internuclear integration.
-SNc is similar to the dorsal horn of the spinal cord for pain and thermal perception of the facial and cranial tissues [8, 25, 30, 48], and shares a similar laminar organization. It contains [30, 50, 52] a marginal zone (resembling Rexed’s lamina (1), a substantia gelatinosa (resembling Rexed’s lamina (2), and a magnocellular layer (similar to Rexed’s lamina 3 and 4 of the dorsal horn).
A deeper zone corresponding to laminae 5 and 6 has also been described [17]. Similar to what occurs in the dorsal horn, most of the small-diameter myelinated (Aδ) and unmyelinated (C) fibers terminate in laminae 1, 2, 5, and 6 of pars caudalis. SNc is more involved in processing tonic signals than short-duration nociceptive stimuli (e.g., by noxious chemicals [17]). Aδ fibers are possibly involved in short-lasting non-inflammatory pain, whereas the C-fibers are probably engaged in long-lasting inflammatory pain [17].
Somatotopic arrangement of the trigeminal nuclear complex
Three different somatotopic arrangements have been described for the trigeminal nuclei:
First, fibers from the V1, V2, and V3 trigeminal divisions end, respectively in ventral, intermediate, and dorsal positions in the SN (Fig. 3A) [8, 25, 30, 48]. For instance, the dorsolateral region of the SNi receives input from the V3 branches, whereas its ventrolateral area receives input from V1 and V2 branches. [46, 48].
Second, different facial regions are represented by long rostrocaudal columns, each representing a distinct part of the face, with midline regions represented medially and lateral skin represented laterally. For instance, the SNo can be subdivided into a lateral portion, which receives afferents from more lateral facial structure (including vibrissae in animals), and a medial one, which receives afferents from intraoral and nasal structures. [45, 48, 57].
Finally, the SN presents a specific arrangement of trigeminal projections, called the “onion skin” pattern of Déjerine (Fig. 3B), [58] in which the face is divided into concentric rings from the perioral to the preauricular regions. The central facial zone (nose and mouth) projects rostrally within the SN, whereas the most peripheral facial area is represented in the caudal SN (Fig. 3) [30, 50]. This rostrocaudal somatotopic arrangement in the SN is supported by historical surgical studies (tractotomy at different levels) [51] and clinical observations (intramedullary lesions or acute demyelination). [30, 59, 60] The rostrocaudal extent of the projections of the onion skin pattern of Dejerine onto the SN varies across publications: it was initially limited to SNc [30], other papers showing an extension throughout the whole spinal nucleus [50, 61]. This discrepancy may be related to the fact that studies on trigeminal nociception have focused for a long time on nociceptive inputs from the facial skin but not from deep oral structures. This probably excluded SNo and SNi from somatotopic maps, even though they are now recognized to play an important role in deep oral structures and dental nociception [55]. Innervation of the middle cerebral fossa and the middle meningeal arteries is in agreement with this model: these territories are innervated by the V2 and V3 divisions and project onto the rostral two-thirds of the SNc and the caudal part of the SNi, but also to the infra-trigeminal region ventral to SNi, to the nucleus of the solitary tract, and the medullary reticular formation [25, 62,63,64,65]. Similarly, in the rat, nerve fibers innervating the middle cerebral artery are mainly deserved by the V2/V3 and project onto SNo and dorsocaudal two-fifths SNi [66]. More surprisingly, the same group of fibers also projects to the PN, suggesting a non-purely nociceptive intracranial innervation or a wider distribution of nociceptive inputs to the trigeminal sensory nuclear complex.
Following this model, the frontal area projects onto the lower SNc located in the upper cervical spinal cord. Consequently, nociceptive inputs from territories innervated by the V1 (frontal area, anterior skull base dura mater, superior sagittal, and transverse sinuses) and by the upper cervical roots (neck, posterior fossa dura mater) both converge onto the same cervical central neurons (Fig. 3B) [67, 68]. Their nociceptive stimulation induces a referred pain, which is perceived in the frontal area (V1 cutaneous territory) regardless of the intracranial or cervical site of stimulation [56, 67]. This reinforces the peripheral convergence, which occurs at the level of the trigeminal nerve (see Trigeminal nerve section). The morphological and functional continuum between the SN and dorsal horn of the cervical spine (Trigeminocervical Complex) [69] may account for a large part of the phenotype of primary headaches.
Contrary to the extensive literature focusing on the somatotopic arrangement of SN, no clear somatotopic organization has been described for PN, possibly due to its small size.
Despite this general organization, trigeminal projections do not follow a strict somatotopic repartition. For instance, in the rat and the monkey, the cornea projects heavily onto the SNi/SNc junction (the “peri obex” region), but also lightly to the SNc/C1 transition zone and sparsely to the dorsal horn of C1-C3 spinal cord segments [63,64,65]. Moreover, “local circuit” neurons connecting PN to SNc have been shown in the rat and support integration between subnuclei. [17].
Finally, projections of trigeminal fibers are not restricted to the trigeminal sensory nucleus. For instance, in the rat, trigeminal fibers innervating the middle cerebral artery also project onto ipsilateral dorsal motor nucleus of the vagus nerve, bilateral lateral nuclei of the solitary tract at the level of the obex, ventral periaqueductal gray including the dorsal raphe, and C2 dorsal horn. [66].
Thalamus and deep brain nuclei
Secondary neurons, located in PN and SN, follow the ventral (VTT) and dorsal (DTT) trigemino-thalamic tracts to join the ventro-posteromedial (VPM), and to a lesser degree, the ventro-posterolateral (VPL), dorsal and intralaminar nuclei of the thalamus (Fig. 4) [27, 30, 48, 70,71,72,73,74,75,76,77,78]:
-
The VTT, or trigeminal lemniscus, is a large tract arising from the ventral part of PN and SNo and SNi [47]. It crosses the midline along the median lemniscus (ML) dorsal border and ascends just posterior to ML [25, 47]. It mainly joins the contralateral VPM [47, 78] but also VPL. In agreement with its nuclei of origin (PN and SN), lesions of the VTT lead to a contralateral sensory loss for touch and pain. [79]
-
The DTT arises from the dorsal part of the PN and the caudal two-thirds of the SN and joins the ipsilateral thalamus. [48, 80]
Trigemino-thalamic neurons. The principal (PN) and spinal (SN) trigeminal nuclei, in charge of tact and pain respectively, project to the thalamus via three main pathways: the ventral trigemino-thalamic tract or trigeminal lemniscus (VTT) arises from PN and rostral SN. It crosses the midline and joins the dorsal aspect of the median lemniscus (ML). The dorsal trigemino-thalamic tract (DTT) arises from the dorsal part of PN and caudal two-thirds of SN (not represented) and reaches the ipsilateral thalamus. Finally, neurons from the caudal SN, involved in nociceptive and extreme thermal perception, have a similar organization as the dorsal horn of the cervical spinal cord: they cross the midline and join the spinothalamic tract to reach the contralateral thalamus. SN and PN mainly project to the ventro-posteromedial (VPM) and, to a lesser degree, the ventro-posterolateral (VPL), dorsal and intralaminar nuclei of the thalamus. cun fasc: cuneate fasciculus; cun nucl: cuneate nucleus; grac fasc: gracile fasciculus, grac nucl: gracile nucleus
Neurons of the caudal SN involved in nociceptive and extreme thermal perception have a similar organization as in the dorsal horn of the cervical spinal cord. They cross the midline and join the spinothalamic tract to reach the thalamus [25, 30]. This organization agrees with the morpho-functional continuum between the rostral cervical cord and the caudal SN (trigemino-cervical complex), presented in §2.2.
Neurons located in Ventral Posterior nucleus of the thalamus (VP) are somatotopically arranged: VPM and VPL nuclei, respectively, receive input from the contralateral half of the head and body [30, 79]. A more precise somatotopic organization (thalamic homunculus) has been described in animals, including primates [17, 70, 81, 82]. For instance, in VPM, intraoral tactile receptors (from the tongue, teeth, and oral cavity) project medially to the lips and upper face [17]. The proximity of regions of VPL (responsive to finger and hand surface) and VPM (responsive to the oral surface) explains the sensory disturbance of the contralateral face and hand (cheiro-oral syndrome) observed after limited vascular lesions in the area [17]. This somatotopic organization of the VPN/VPM system is consistent with localizing nociceptive stimuli.
Interestingly, only 6% of VPM neurons respond to noxious stimuli, suggesting that pain processing may occur in additional sites [83]. One candidate for this is the posterior part of the ventromedial nucleus (VMpo), located infero-medially to VPM, which is involved in perceiving pain and cold in the contralateral body and face. A somatotopic arrangement exists in VMpo, with facial input being anteromedial and limbs posterolateral.
The organization is entirely different for trigeminal projections to the intralaminar and dorsal thalamic nuclei, that lack any organization by somatotopy or by modality, reflecting the involvement of these nuclei in affective-motivational aspects of nociception, without any important role for pain localization. [80].
The trigeminal nuclei also project onto the hypothalamus; the trigemino-hypothalamic tract originates mainly from nociceptive multimodal neurons and more rarely from non-nociceptive neurons of the trigeminal nuclear complex [84, 85]. The trigemino hypothalamic tract ascends homolaterally in the brainstem to reach the hypothalamus. There, half of the fibers decussates and reaches the lateral and medial hypothalamus. The areas receiving trigeminal inputs are those regulating homeostasis and integrating pain with visceral afferent input [84,85,86].
Furthermore, the trigeminal system is connected to the limbic system (amygdala, lenticular nucleus, nucleus accumbens) and the periaqueductal gray via a polysynaptic pathway. These projections are probably involved in the transmission of visceral pain and emotions and may play a role in affective aspects of facial nociception: [85, 87].
Cortical projections
Tertiary trigeminal neurons located in various thalamic nuclei project onto different areas of the cerebral cortex. For instance, VPM projects to primary and secondary somatosensory (S1/S2) cortices and the insula. By contrast, neurons in posterior, lateral, or dorsal nuclei of the thalamus project to multiple cortical areas, such as motor, parietal association, retrosplenial, somatosensory, auditory, visual, and olfactory cortices [27, 88,89,90].
The sensation of pain and thermal extremes becomes conscious at the thalamic level, but localization and perception of intensity and quality of noxious stimuli require primary and secondary somatosensory cortices [30]. In line with this role in pain localization, these cortices have a clearly defined somatotopic organization.
The primary somatosensory cortex (SI) dedicated to the face, located in the ventral part of the postcentral gyrus, has been extensively investigated for a specific spatial organization [91,92,93]. The exact somatotopy within the face area (sensory homunculus) is still discussed despite the various techniques used such as direct cortical stimulation, sensory-evoked potentials, functional MRI, magnetoencephalography (MEG), or positron emission tomography (PET). First described by Penfield and Boldrey, the face representation was depicted as non-inverted, “with forehead superior and lower lip inferior.” [17] Nevertheless, evoked potentials recorded from the cortical surface, analysis of Jacksonian sensory seizures in patients, and fMRI studies have suggested an inverted face representation within the somatosensory cortex with the upper face inferior to the lips and oral cavity [30, 94]. Moulton et al. suggested a concentric ring or “onion skin” pattern for the facial primary somatosensory cortical representation: anteromedial parts of the face (nose) project ventrally within the post-central gyrus, whereas postero-lateral facial regions (ear) project more dorsally. [92] Such an arrangement is consistent with results by Nguyen et al., which found the nose and lips represented ventrally to other facial areas” [95, 96].
Deafferentation following amputation or nerve section induces a somatotopic reorganization of body representation. During this phenomenon, the representation of a body part adjacent to a denervated area expands into the deafferented representation [97, 98]. For instance, after hand amputation, the facial area may shift towards the region normally associated with responses from the hand, explaining why stimulation of the face evokes sensations within the “phantom” hand [79]. Similarly, referred facial sensation on the hand has been recently observed after full-face transplantation [99]: the patient felt sensations in the area from around the lips to the forehead and ear after finger stimulation by brush. Two months after the examination of referred sensation, fMRI was performed and indicated that cortical areas for face and hand overlapped. Uysal [99] proposed that sensory input from the hand “invades” the cortical territory corresponding to the denervated face. Consequently, touch stimuli applied to the hand activate the area of the brain initially dedicated to the face and are therefore interpreted as coming from the face.
The secondary somatosensory cortex (SII), located within the parietal operculum, is probably involved in tactile object recognition and memory, and pain perception [100, 101]. Its somatotopy is controversial: some fMRI studies have confirmed a gross face-hand-foot somatotopic organization, whereas others could not discriminate clusters activated by hand and foot. [102].
Modulation of trigeminal signals and trigeminal reflexes
The complexity of the trigeminal system is also due to the multiple connections and modulations which occur along the trigeminal pathway, and that are still only partially understood.
The brainstem is a significant processing site for nociceptive input and can play a crucial role in the pathophysiology of various headache disorders. For instance, in an fMRI study after noxious stimulation, Schulte et al. observed activations of multiple areas involved in trigeminal nociceptive processing: spinal trigeminal nuclei, thalamus, secondary somatosensory cortex, insular cortex, and cerebellum [103]. They also observed activation of a pain-modulating network, including the periaqueductal gray, hypothalamus, locus coeruleus, and cuneiform nucleus. Interestingly, the left cuneiform nucleus activations positively correlated with pain intensity rating, and trigeminal noxious stimulation enhanced functional connectivity between bilateral spinal trigeminal nuclei and between the cuneiform nucleus and rostral ventromedial medulla [103].
The cerebellum’s role in pain processing remains poorly understood [104,105,106,107]: in an fMRI study, Mehnert et al. provided evidence of specific cerebellar areas activation during nociceptive trigeminal input. They also described robust functional connectivity between the cerebellum and rostral pons and between the periaqueductal grey and thalamus. These functional relationships with areas involved in the descending antinociceptive network plead for an important role of the cerebellum in pain transmission, perception, and control [104].
Additionally, the trigeminal sensory nuclei are directly connected to various posterior fossa structures (superior colliculus, cerebellar cortex, and deep nuclei [25]) and via tegmental interneurons. The latter connect the trigeminal sensory nuclei to the ocular motor nuclei, the motor nuclei of the trigeminal nerve, the facial, glossopharyngeal, vagal, hypoglossal nuclei, and the vestibular nuclei [25, 108,109,110,111,112]. For instance, Henssen et al. reviewed the central connections of the vagus nerve to explain the effectiveness of non-invasive vagal nerve stimulation (VNS) in primary headache disorders (see below) [113]. The vagus nerve contains visceromotor (thoracoabdominal parasympathetic) fibers from the dorsal motor nucleus and somatomotor fibers for the 6th branchial arch (larynx muscles, but crico-thyroïdian). It also contains sensory fibers which enter the brainstem and form the solitary tract. These fibers establish synapses in the solitary nucleus, which sends output to the reticular formation and several cranial nerve nuclei, including SN [80]. The four nuclei sending or receiving fibers to/from the trigeminal nerve (dorsal motor and ambiguus nuclei, nucleus of the solitary tract, and SN) are extensively interconnected. This is especially true for the interpolaris and caudal parts of SN, PN, and the solitary tract nucleus, as demonstrated by animal studies [111, 113,114,115].
As a consequence of these rich interconnections, the trigeminal system is involved in several reflexes. The jaw-jerk closing monosynaptic reflex (Fig. 2) is the closure of jaws after a sudden stretching of the masseter muscle. Input neurons (located in the trigeminal mesencephalic nucleus) receive impulses from peripheral masticatory muscle spindles and project onto the trigeminal motor nucleus. Output neurons are motor neurons situated within the motor trigeminal nucleus. They follow the pars minor to reach the V3 and the masticatory muscles. Closure of the mouth implies a bilateral contraction of the muscles, which depends on commissural internuclear connections between trigeminal motor nuclei [30, 116]. The corneal polysynaptic reflex is the bilateral closure of eyelids after a light touch of the cornea. Primary nociceptive corneal neurons located within the trigeminal ganglion project to the whole extent of the spinal nucleus. They indirectly connect via lateral reticular formation interneurons to both facial nuclei, which finally project to orbicularis oculi muscles ipsilateral (direct corneal response) and contralateral to corneal stimulation (consensual corneal response) [30, 38]. The primary neurons (located in the trigeminal ganglion) transmit the impulse to the secondary neurons located at all levels of the spinal nucleus. The secondary neurons then transmit their impulse to bilateral facial nuclei via internuclear fibers of the lateral reticular formation. The efferent fibers from each facial nucleus reach their corresponding orbicularis oculi muscle [30, 38].
The trigemino-cardiac reflex (TCR) is the sudden onset of bradycardia, hypotension, apnea, and gastric hypermotility after physical or chemical stimulation of any branches of the trigeminal nerve [117,118,119,120], or during stimulation of the brainstem trigeminal pathway [119, 121]. Afferent signals reach the trigeminal sensory nucleus, which connects to reticular interneurons. The latter join premotor neurons of the nucleus ambiguus and the dorsal motor nucleus of the vagus nerve. This efferent pathway sends cardio-inhibitory fibers to the myocardium [122]. Based on experimental evidence, TCR could be mediated initially in the trigeminal nucleus caudalis. [117, 119, 123].
The trigemino-autonomic reflex activates the parasympathetic system after stimulation of the trigeminal complex, leading to conjunctival injection, lacrimation, rhinorrhea, and cranial vasodilatation. The caudal trigeminal nucleus and upper cervical spinal cord are connected to the muco-lacrimo-nasal/superior salivatory nucleus, which sends preganglionic fibers to the facial nerve. These fibers exit the facial nerve at the level of the geniculate ganglion to enter the greater petrosal nerve and end within the pterygopalatine ganglion. There, they connect to postganglionic fibers, which project to lacrimal and nasal glands. This reflex is supposed to play an essential role in parasympathetic symptoms observed in cluster headaches and may be triggered by the hypothalamus, which projects onto the superior salivatory nucleus [124,125,126].
Trigeminal nerve: which target for which pain?
Modern pharmacological approaches successfully manage some of headaches or facial pain. Still, some are resistant to drugs and require alternative handling. Several procedures have been developed in the last 50 years, including, for instance, trigeminal surgical decompression in the cerebellopontine angle [127]. Apart from these classical treatments, invasive lesional and, more recently, less-invasive neuromodulatory approaches have been proposed to deal with resistant headaches and facial pain. This section reviews lesional, invasive neuromodulatory (implanted device), and non-invasive neuromodulatory approaches in light of the anatomical organization of the trigeminal system.
Lesional procedures
Lesional procedures for trigeminal neuralgia
According to ICHD-3, trigeminal neuralgia (TN) is defined as “recurrent unilateral brief electric shock-like pains, abrupt in onset and termination, limited to the distribution of one or more divisions of the trigeminal nerve and triggered by innocuous stimuli” [85, 128] Three categories can be recognized: classic TN, consecutive to vascular compression of the trigeminal nerve at its root entry zone; secondary TN, straight to various diseases such as multiple sclerosis or tumor compressing the trigeminal nerve; and idiopathic TN, which cause remains unknown [129]. Most patients are relieved by pharmacological treatments such as carbamazepine or other antiepileptic drugs. Several surgical approaches can be proposed when TN becomes pharmacoresistant or when drug side effects are too debilitating. Shortly it is a non-ablative surgical procedure [130], which can be offered to patients with classic TN (vascular compression) with a cure rate of 80–98% without hypoesthesia but 10–30% recurrence [131]. By contrast, patients with secondary or idiopathic TN are eligible for lesional procedures, such as radiofrequency thermal rhizotomy, retrogasserian glycerol rhizotomy, trigeminal balloon compression, stereotactic radiation therapy, and radiosurgery. These methods use various mechanisms but have a common target: limiting pain transmission by directly lesioning small nociceptive fibers. The nociceptive nature of the corneal innervation, conveyed by the V1, restricts these methods to the V2 and V3 trigeminal divisions. They offer pain relief but with a high rate of facial sensory loss with numbness and frequent—possibly neuropathic—pain recurrence, which often requires medication continuation or reoperation [38, 132,133,134,135,136,137]. Results are variable [130, 138,139,140,141,142] and depend on multiple factors [141] (type of trigeminal neuralgia, type of compression, disease duration). Comorbidity and surgical team’s habits are also involved in the choice of the technique. For more details, refer to Table 1.
To deal with extreme cases of TN in therapeutic failure, Dandy proposed in 1929 to partially section the trigeminal nerve (partial sensory rhizotomy) [143] in order to disconnect nociceptive fibers before they enter the brainstem. This method suffered from a lack of precise anatomical knowledge on trigeminal somatotopy and was consequently not fully codified; from procedure to procedure, rhizotomy of the pars major of the trigeminal nerve was very partial or complete and performed either at the juxtapontine portion or not. Non-surprisingly, these various procedures had highly variable results for pain relief and hypoesthesia, including loss of the corneal reflex [38], and were gradually abandoned.
The detailed description of the somatotopy and modal fiber arrangement in the juxta-pontine portion of the trigeminal nerve has led to targeted partial sensory rhizotomy [38]. In the latter, only the ventrolateral two-thirds of the pars major of the trigeminal nerve is sectioned at its juxtapontine portion, preserving both the corneal nociceptive fibers (dorsomedial third of the pars major) and epicritic fibers (pars intermediaris) (Fig. 2). Compared to other lesional methods for secondary or idiopathic TN, this procedure provides an excellent outcome in terms of pain relief (86.4%), postoperative partial and focalized hypoesthesia (22.7%), preservation of corneal reflex (100%), and recurrence rate (31.5% at five years). [38].
Lesional procedures for cluster headaches
Cluster headache (CH) is a primary headache belonging to the trigeminal autonomic cephalalgias group. According to ICHD-3, CH is a “severe or very severe unilateral orbital, supraorbital and/or temporal pain lasting 15–180 min”. It is associated with ipsilateral prominent parasympathetic features (conjunctival injection, lacrimation, rhinorrhea or nasal congestion), myosis and/or ptosis, and agitation [128, 144]. The most frequent one (80–90%), episodic CH, is characterized by seven days to one year periods in which recurring attacks occur. These “active” periods are separated by pain-free periods lasting at least 3 months. Chronic CH is defined as attacks occurring for one year or longer without remission or with remission periods lasting less than three months. The pathophysiology of CH is only partially understood, and the current hypothesis is a dual activation of the trigeminal (ophthalmic branch of the trigeminal nerve) and autonomic systems (sphenopalatine ganglion). This dual activation is probably induced by a generator of attacks, potentially located in the posterior hypothalamic gray matter. It is then propagated from the trigeminal to the autonomic systems via the trigemino-autonomic reflex (see above, modulation of trigeminal signals and trigeminal reflexes) [144,145,146]. The pharmacological CH management includes prophylactic (verapamil or lithium) and abortive treatments (subcutaneous injection of sumatriptan, oxygen inhalation). Episodic CH is the most frequent form (85–90%) but can become chronic in 10–15%. Refractory chronic CH represents 15–20% of chronic CH. This condition, in which pharmacological therapies do not provide pain remission, or become inoperant, is often referred to as “suicidal headache”. [125, 144, 146, 147].
Various lesional procedures for CH, such as surgical rhizotomy/section of the trigeminal nerve or thermolesion of the sphenopalatine ganglion, induced only short-term pain relief and high risks of complications and were therefore abandoned [144]. Nevertheless, lesions of the sphenopalatine ganglion were considered more efficient on CH attacks than those of the trigeminal nerve [144], pleading to focus non-lesional procedures on this target (see next section).
Invasive neuromodulation
Invasive neuromodulation is mainly used in CH. It uses implanted electrodes to stimulate various anatomical targets in cranial pain circuits (occipital nerve, sphenopalatine ganglion, hypothalamus) and is usually reserved for the most severe patients [143, 148].
Initially proposed for occipital nerve neuralgia, Occipital nerve stimulation (ONS) is now the preferred technique in refractory chronic CH patients and has also been used for chronic migraine [144, 145]. ONS, which delivers a continuous electrical stimulation to the greater and/or the lesser occipital nerves via a subcutaneous electrode, induces paresthesia in the occipital region [144, 149, 150]. Its exact mechanism of action remains unknown, but several arguments suggest that ONS could involve a “gate control theory-like” mechanism through modulation of convergent nociceptive inputs into the trigemino-cervical complex [145, 146]. ONS is similar to prophylactic treatment, in that it decreases CH attacks frequency and intensity, but does not stop them after had they begun. [144, 151].
Sphenopalatine ganglion (SPG) neuromodulation is an interesting option in refractory chronic CH. A miniaturized device is implanted within the pterygopalatine fossa in contact with the sphenopalatine ganglion, which contains sensory, parasympathetic, and sympathetic neurons [144, 152, 153]. It possibly modulates the trigeminal autonomic reflex, in which the parasympathetic efferent component is mediated through the sphenopalatine ganglion. [144] In a double-blinded trial, 68% of patients were significantly clinically improved after stimulation, with pain relief in more than 50% and/or reduction in attack frequency in more than 50% [154]. SPG stimulation remains efficient over 24 months in the majority of patients [155]. The most frequent side effect (81%) is a transient mild/moderate loss of sensation in the V2 territory, which usually resolves within three months [144, 154].
Deep brain stimulation (DBS) targets the posteroinferior hypothalamus, a region activated during CH attacks. It aims to inhibit the presumed pain-attacks generator and modulate non-specific antinociceptive systems, including mesencephalic gray substance or orexinergic system [144, 156,157,158,159] (for more information, please report to the review by Fontaine et al. [144]). The overall response rate (pain-free patients or decrease in more than 50% of attack frequency) is 60% [144]. Few stimulation-related side effects have been reported. Still, in the limited published literature to date, the risk of clinically significant cerebral hemorrhage is slightly superior in hypothalamic DBS than movement disorder DBS (1–5%) [144, 160]. DBS is therefore restricted to refractory chronic CH patients after ONS failure.
Motor cortex stimulation (MCS) has emerged in the last decades as an alternative technique to treat patients with facial chronic neuropathic pain (FCNP) [161]. FCNP is a devastating condition characterized by resistant pain located in any of the territories of the trigeminal nerve branches (V1, V2, or V3) or a non-dermatomeric area of the face. It is referred to as resistant trigeminal neuralgia, trigeminal neuropathic pain, trigeminal deafferentation pain, or atypical facial pain [162,163,164,165,166,167]. It can be the consequence of any injury or pathological conditions all along the trigeminal system. The surgical procedure consists in implanting an electrode over the covering of the targeted area (motor cortex). The mode of action of MCS in FCNP remains largely unknown but could involve a central analgesic mechanism [161, 168,169,170].
A review on MCS for FCNP [161] showed a positive response in 44–100% of MCS-treated patients, but the response to stimulation decreased over time in some patients. The authors concluded that this technique is safe and efficient when other options failed. Nevertheless, the published studies are highly heterogenous for the origin of the pain syndromes, surgical procedures, and methods for assessing clinical outcomes. More extensive randomized controlled multicentric studies are needed, but MCS could be considered in FCNP after the failure of less invasive options and performed by an experienced surgical team. [161, 162, 171].
Non-invasive neuromodulation
Non-invasive stimulation uses transcutaneous electrical or magnetic stimulation to target anatomical structures involved in pain transmission/modulation, for instance, the vagal and supraorbital nerves or cortex [148] (for more information, please report to the review of Hoffman et al.) [145]. They may be a safe and valuable complementary support to the pharmaceutical strategy. Still, their action mechanisms remain partially unknown, and randomized and sham-controlled studies have to confirm positive results observed in open-label trials [145].
Non-invasive vagus nerve stimulation (VNS) has been proposed to manage migraine and cluster headache, in addition to some of the other trigeminal autonomic cephalalgias (TACs) [172]. A small handheld device is placed on the neck (GammaCore® device) or within the upper part of the ear concha (cymba concha (NEMOS ® device), ipsilaterally to the pain. It and delivers an electrical current inducing a light cutaneous tingling sensation. Transauricular VNS stimulates the auricular branch of the vagal nerve, which innervates the concha, and transcervical VNS stimulates the cervical segment of the vagus nerve through the neck [173,174,175,176]. According to Silberstein et al., VNS possibly uses the autonomic trigeminal reflex circuits to inhibit the cortical spread depression underlying migraine aura, regulate neurotransmitter release, and modulate nociception by suppressing the acute nociceptive activation of the trigemino-cervical system [172]. VNS efficiency is quite challenging to assess because evaluation criteria highly vary across trials [177,178,179,180,181,182]. However, it has recently been shown to significantly decrease CH mean attack frequency (64%), duration (43%), and severity (23%) [172, 183]. It has recently been recommended by the National Institute for Health and Care Excellence (United Kingdom) as an effective treatment for CH and is FDA approved in the USA for both attack and preventive therapy in CH [172]. In migraine, non-invasive VNS was associated with a significant reduction in monthly migraine days (p = 0.043), monthly headache days (p = 0.045) and acute medication days (p = 0.039). The pain-free rate at 120 min increased by 10% in non-invasive VNS vs. sham [172]. In a recent review, Silberstein et al. concludes that non-invasive VNS should be considered «a first-line treatment for both acute and preventive treatment of cluster headache, an effective option for acute treatment of migraine, and a highly relevant, practical option for migraine prevention» [172].
The supraorbital nerve stimulation (Cefaly® device) was proposed for acute and preventive treatment of migraine. The handheld device is placed on the forehead and delivers current pulses. The mode of action is unknown, but FDG-PET and fMRI plead for functional changes in the orbitofrontal cortex and the rostral anterior cingulate cortex. [145, 184,185,186].
Transcranial magnetic stimulation was developed for acute and preventive treatment of migraine with aura and neuropathic pain. A small device applied to the scalp delivers a single magnetic pulse to the underlying cortex to disrupt the cortical spread depression (CSD), responsible for migraine with aura. A single-pulse transcranial magnetic stimulation can be applied after the beginning of the aura to abort a migraine attack [149, 187,188,189,190]. In contrast, repetitive-pulse transcranial magnetic stimulation is expected to have a prophylactic effect by modulating cortical excitability [145, 190]. A recent meta-analysis of randomized controlled trials [187] suggests that single-pulse transcranial magnetic stimulation is efficient for the acute treatment of migraine with aura (more patients were pain-free at two hours post-treatment), but not for chronic migraine. Nevertheless, more randomized controlled trials are needed to evaluate TMS efficiency on migraine. [187].
Conclusion
The trigeminal system is the principal actor for facial tactile and nociceptive innervation, and for meninges and brain vessels nociceptive innervation. It has a complex somatotopic and modality-specific arrangement along its entire path, from the trigeminal nerve to the somatosensory cortex. This fine anatomical organization could be one of the keys to understanding the pathophysiology of several diseases with distinct clinical features but sharing a common anatomical trigeminal substrate. It can also be the starting point to propose innovative therapeutic approaches targeting various levels of the trigeminal system when pharmacological strategy fails to relieve pain or induces debilitating side effects. Non-invasive and invasive modulatory methods have been mainly proposed for CH and migraine and must be thoroughly evaluated before a wider diffusion. By contrast, lesional approaches are mainly limited to intractable TN, targeted partial sensory rhizotomy being, for instance, a direct application of a more precise anatomical description of trigeminal fiber arrangement.
References
Ray BS, Wolff HG (1940) Experimental studies on headache: pain-sensitive structures of the head and their significance in headache. Arch Surg 41(4):813–856. https://doi.org/10.1001/archsurg.1940.01210040002001
Feindel W, Penfield W, McNAUGHTON F (1960) The tentorial nerves and localization of intracranial pain in man. Neurology 10:555–563
Steiger HJ, Tew JM, Keller JT (1982) The sensory representation of the dura mater in the trigeminal ganglion of the cat. Neurosci Lett 31(3):231–236
Mayberg MR, Zervas NT, Moskowitz MA (1984) Trigeminal projections to supratentorial pial and dural blood vessels in cats demonstrated by horseradish peroxidase histochemistry. J Comp Neurol 223(1):46–56. https://doi.org/10.1002/cne.902230105
Edvinsson L (2011) Tracing neural connections to pain pathways with relevance to primary headaches. Cephalalgia Int J Headache 31(6):737–747. https://doi.org/10.1177/0333102411398152
Olesen J, Burstein R, Ashina M, Tfelt-Hansen P (2009) Origin of pain in migraine: evidence for peripheral sensitisation. Lancet Neurol 8(7):679–690. https://doi.org/10.1016/S1474-4422(09)70090-0
Terrier L-M, Hadjikhani N, Velut S et al (2021) The trigeminal system: the meningovascular complex—a review. J Anat. https://doi.org/10.1111/joa.13413
Joo W, Yoshioka F, Funaki T, Mizokami K, Rhoton AL (2014) Microsurgical anatomy of the trigeminal nerve. Clin Anat 27(1):61–88. https://doi.org/10.1002/ca.22330
Fontaine D, Almairac F, Santucci S et al (2018) Dural and pial pain-sensitive structures in humans: new inputs from awake craniotomies. Brain J Neurol 141(4):1040–1048. https://doi.org/10.1093/brain/awy005
Arbab MA, Wiklund L, Svendgaard NA (1986) Origin and distribution of cerebral vascular innervation from superior cervical, trigeminal and spinal ganglia investigated with retrograde and anterograde WGA-HRP tracing in the rat. Neuroscience 19(3):695–708
Liu-Chen LY, Han DH, Moskowitz MA (1983) Pia arachnoid contains substance P originating from trigeminal neurons. Neuroscience 9(4):803–808
Goadsby PJ, Edvinsson L, Ekman R (1988) Release of vasoactive peptides in the extracerebral circulation of humans and the cat during activation of the trigeminovascular system. Ann Neurol 23(2):193–196. https://doi.org/10.1002/ana.410230214
Goadsby PJ, Knight YE, Hoskin KL, Butler P (1997) Stimulation of an intracranial trigeminally-innervated structure selectively increases cerebral blood flow. Brain Res 751(2):247–252
Macfarlane R, Moskowitz MA (1995) The innervation of pial blood vessels and their role in cerebrovascular regulation. In: Caplan LR (ed) Brain ischemia. Springer, Berlin, pp 247–259
Sakas DE, Moskowitz MA, Wei EP, Kontos HA, Kano M, Ogilvy CS (1989) Trigeminovascular fibers increase blood flow in cortical gray matter by axon reflex-like mechanisms during acute severe hypertension or seizures. Proc Natl Acad Sci USA 86(4):1401–1405
Lambert GA, Bogduk N, Goadsby PJ, Duckworth JW, Lance JW (1984) Decreased carotid arterial resistance in cats in response to trigeminal stimulation. J Neurosurg 61(2):307–315. https://doi.org/10.3171/jns.1984.61.2.0307
Mai J, Paxinos G (2011) The human nervous system, 3rd edn. Accessed 23 Sept 2019. https://www.elsevier.com/books/the-human-nervous-system/mai/978-0-12-374236-0
Penfield W, McNaughton F (1940) Dural headache and innervation of the dura mater. Arch Neurol Psychiatry 44(1):43–75. https://doi.org/10.1001/archneurpsyc.1940.02280070051003
Lee S-H, Shin K-J, Koh K-S, Song W-C (2017) Visualization of the tentorial innervation of human dura mater. J Anat 231(5):683–689. https://doi.org/10.1111/joa.12659
Edvinsson L, McCulloch J, Uddman R (1981) Substance P: immunohistochemical localization and effect upon cat pial arteries in vitro and in situ. J Physiol 318:251–258
Liu-Chen LY, Mayberg MR, Moskowitz MA (1983) Immunohistochemical evidence for a substance P-containing trigeminovascular pathway to pial arteries in cats. Brain Res 268(1):162–166
Liu-Chen LY, Liszczak TM, King JC, Moskowitz MA (1986) Immunoelectron microscopic study of substance P-containing fibers in feline cerebral arteries. Brain Res 369(1–2):12–20
Lee S-H, Hwang S-J, Koh K-S, Song W-C, Han S-D (2017) Macroscopic innervation of the dura mater covering the middle cranial fossa in humans correlated to neurovascular headache. Front Neuroanat 11:127. https://doi.org/10.3389/fnana.2017.00127
Kemp WJ, Tubbs RS, Cohen-Gadol AA (2012) The innervation of the cranial dura mater: neurosurgical case correlates and a review of the literature. World Neurosurg 78(5):505–510. https://doi.org/10.1016/j.wneu.2011.10.045
Liu GT (2005) Walsh and Hoyt’s clinical neuro-ophthalmology, vol 1, 6th edn. Lippincott Williams and Wilkins. https://collections.lib.utah.edu/ark:/87278/s6rj4hsw
Burstein R, Blake P, Schain A, Perry C (2017) Extracranial origin of headache. Curr Opin Neurol 30(3):263–271. https://doi.org/10.1097/WCO.0000000000000437
Noseda R, Burstein R (2013) Migraine pathophysiology: anatomy of the trigeminovascular pathway and associated neurological symptoms, cortical spreading depression, sensitization, and modulation of pain. Pain 154(Suppl 1):S44–S53. https://doi.org/10.1016/j.pain.2013.07.021
Ramachandran R (2018) Neurogenic inflammation and its role in migraine. Semin Immunopathol 40(3):301–314. https://doi.org/10.1007/s00281-018-0676-y
Donaldson IM (2000) The functions of the proprioceptors of the eye muscles. Philos Trans R Soc Lond B Biol Sci 355(1404):1685–1754. https://doi.org/10.1098/rstb.2000.0732
Augustine JR (2017) Human neuroanatomy. Wiley, New York
Borsook D, DaSilva AFM, Ploghaus A, Becerra L (2003) Specific and somatotopic functional magnetic resonance imaging activation in the trigeminal ganglion by brush and noxious heat. J Neurosci Off J Soc Neurosci 23(21):7897–7903
Peker S, Kurtkaya O, Uzün I, Pamir MN (2006) Microanatomy of the central myelin-peripheral myelin transition zone of the trigeminal nerve. Neurosurgery 59(2):354–359. https://doi.org/10.1227/01.NEU.0000223501.27220.69 (discussion 354–359)
Young R (1977) “Pain in the Trigeminal Region”, fiber spectrum of the trigeminal sensory root of frog by cat and man determined electron microscopy. Elsevier/North-Holland Biomedical Press, New York
Emmons WF, Rhoton AL (1968) Functional subdivision of the trigeminal sensory root. Surg Forum 19:440–441
Emmons WF, Rhoton AL (1971) Subdivision of the trigeminal sensory root. Experimental study in the monkey. J Neurosurg 35(5):585–591. https://doi.org/10.3171/jns.1971.35.5.0585
Gudmundsson K, Rhoton AL, Rushton JG (1971) Detailed anatomy of the intracranial portion of the trigeminal nerve. J Neurosurg 35(5):592–600. https://doi.org/10.3171/jns.1971.35.5.0592
Jannetta PJ (1967) Gross (mesoscopic) description of the human trigeminal nerve and ganglion. J Neurosurg 26(suppl 1):109–111. https://doi.org/10.3171/jns.1967.26.1part2.0109
Terrier L-M, Amelot A, François P, Destrieux C, Zemmoura I, Velut S (2018) Therapeutic failure in trigeminal neuralgia: from a clarification of trigeminal nerve somatotopy to a targeted partial sensory rhizotomy. World Neurosurg 117:e138–e145. https://doi.org/10.1016/j.wneu.2018.05.211
Alper J, Shrivastava RK, Balchandani P (2017) Is there a magnetic resonance imaging-discernible cause for trigeminal neuralgia? a structured review. World Neurosurg 98:89–97. https://doi.org/10.1016/j.wneu.2016.10.104
Pelletier VA, Poulos DA, Lende RA (1974) Functional localization in the trigeminal root. J Neurosurg 40(4):504–513. https://doi.org/10.3171/jns.1974.40.4.0504
Olszewski J (1950) On the anatomical and functional organization of the spinal trigeminal nucleus. J Comp Neurol 92(3):401–413
Taber E (1961) The cytoarchitecture of the brain stem of the cat. I. Brain stem nuclei of cat. J Comp Neurol 116:27–69
Aström KE (1953) On the central course of afferent fibres in the trigeminal, facial, glossopharyngeal, and vagal nerves and their nuclei in the mouse. Acta Physiol Scand 29(S106):209–320. https://doi.org/10.1111/apha.1953.29.s106.209
Arbab MA-R, Delgado T, Wiklund L, Svendgaard NAA (1988) Brain stem terminations of the trigeminal and upper spinal ganglia innervation of the cerebrovascular system: WGA-HRP transganglionic study. J Cereb Blood Flow Metab 8(1):54–63. https://doi.org/10.1038/jcbfm.1988.8
Eisenman J, Landgren S, Novin D (1963) Functional organization in the main sensory trigeminal nucleus and in the rostral subdivision of the nucleus of the spinal trigeminal tract in the cat. Acta Physiol Scand Suppl 214:1–44
Capra NF, Dessem D (1992) Central connections of trigeminal primary afferent neurons: topographical and functional considerations. Crit Rev Oral Biol Med 4(1):1–52. https://doi.org/10.1177/10454411920040010101
Henssen DJHA, Mollink J, Kurt E et al (2018) Ex vivo visualization of the trigeminal pathways in the human brainstem using 11.7 T diffusion MRI combined with microscopy polarized light imaging. Brain Struct Funct. https://doi.org/10.1007/s00429-018-1767-1
Henssen DJHA, Kurt E, Kozicz T, van Dongen R, Bartels RHMA, van Cappellen van Walsum AM (2016) New insights in trigeminal anatomy: a double orofacial tract for nociceptive input. Front Neuroanat 10:53. https://doi.org/10.3389/fnana.2016.00053
Afshar F, Dykes E (1984) Computer-generated three-dimensional visualization of the trigeminal nuclear complex. Surg Neurol 22(2):189–196
Peker S, Sirin A (2017) Primary trigeminal neuralgia and the role of pars oralis of the spinal trigeminal nucleus. Med Hypotheses 100:15–18. https://doi.org/10.1016/j.mehy.2017.01.008
Crosby EC (1962) Correlative anatomy of the nervous system. Macmillan, New York. Accessed 28 Aug 2019. http://archive.org/details/correlativeanato00cros
Graham SH, Sharp FR, Dillon W (1988) Intraoral sensation in patients with brainstem lesions: role of the rostral spinal trigeminal nuclei in pons. Neurology 38(10):1529–1533. https://doi.org/10.1212/wnl.38.10.1529
Jacquin MF, Semba K, Rhoades RW, Egger MD (1982) Trigeminal primary afferents project bilaterally to dorsal horn and ipsilaterally to cerebellum, reticular formation, and cuneate, solitary, supratrigeminal and vagal nuclei. Brain Res 246(2):285–291
Hayashi H, Sumino R, Sessle BJ (1984) Functional organization of trigeminal subnucleus interpolaris: nociceptive and innocuous afferent inputs, projections to thalamus, cerebellum, and spinal cord, and descending modulation from periaqueductal gray. J Neurophysiol 51(5):890–905. https://doi.org/10.1152/jn.1984.51.5.890
Ohya A, Tsuruoka M, Imai E et al (1993) Thalamic- and cerebellar-projecting interpolaris neuron responses to afferent inputs. Brain Res Bull 32(6):615–621
Bartsch T, Goadsby PJ (2002) Stimulation of the greater occipital nerve induces increased central excitability of dural afferent input. Brain J Neurol 125(Pt 7):1496–1509. https://doi.org/10.1093/brain/awf166
Darian-Smith I, Phillips G, Ryan RD (1963) Functional organization in the trigeminal main sensory and rostral spinal nuclei of the cat. J Physiol 168:129–146. https://doi.org/10.1113/jphysiol.1963.sp007182
Déjerine J (1914) Sémiologie Des Affections Du Système Nerveux. Masson. Librairies de l’Académie de médecine
Das A, Shinde PD, Kesavadas C, Nair M (2011) Teaching neuroimages: onion-skin pattern facial sensory loss. Neurology 77(8):e45-46. https://doi.org/10.1212/WNL.0b013e31822affc6
Freeman L, Wu OC, Sweet J, Cohen M, Smith GA, Miller JP (2019) Facial sensory restoration after trigeminal sensory rhizotomy by collateral sprouting from the occipital nerves. Neurosurgery. https://doi.org/10.1093/neuros/nyz306
Ko MW, Prasad S (2019) Headache, facial pain, and disorders of facial sensation. In: Liu GT, Volpe NJ, Galetta SL (eds) Liu, Volpe, and Galetta’s neuro-ophthalmology, 3rd edn. Elsevier, New York, pp 661–684. https://doi.org/10.1016/B978-0-323-34044-1.00019-5
Davis KD, Dostrovsky JO (1988) Responses of feline trigeminal spinal tract nucleus neurons to stimulation of the middle meningeal artery and sagittal sinus. J Neurophysiol 59(2):648–666. https://doi.org/10.1152/jn.1988.59.2.648
Marfurt CF, Del Toro DR (1987) Corneal sensory pathway in the rat: a horseradish peroxidase tracing study. J Comp Neurol 261(3):450–459. https://doi.org/10.1002/cne.902610309
Pozo MA, Cervero F (1993) Neurons in the rat spinal trigeminal complex driven by corneal nociceptors: receptive-field properties and effects of noxious stimulation of the cornea. J Neurophysiol 70(6):2370–2378. https://doi.org/10.1152/jn.1993.70.6.2370
Marfurt CF, Echtenkamp SF (1988) Central projections and trigeminal ganglion location of corneal afferent neurons in the monkey. Macaca fascicularis J Comp Neurol 272(3):370–382. https://doi.org/10.1002/cne.902720307
Arbab MA, Delgado T, Wiklund L, Svendgaard NA (1988) Brain stem terminations of the trigeminal and upper spinal ganglia innervation of the cerebrovascular system: WGA-HRP transganglionic study. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 8(1):54–63. https://doi.org/10.1038/jcbfm.1988.8
Piovesan EJ, Kowacs PA, Tatsui CE, Lange MC, Ribas LC, Werneck LC (2001) Referred pain after painful stimulation of the greater occipital nerve in humans: evidence of convergence of cervical afferences on trigeminal nuclei. Cephalalgia Int J Headache 21(2):107–109. https://doi.org/10.1046/j.1468-2982.2001.00166.x
Angus-Leppan H, Lambert GA, Boers P, Zagami AS, Olausson B (1992) Craniovascular nociceptive pathways relay in the upper cervical spinal cord in the cat. Neurosci Lett 137(2):203–206. https://doi.org/10.1016/0304-3940(92)90404-u
Goadsby PJ, Charbit AR, Andreou AP, Akerman S, Holland PR (2009) Neurobiology of migraine. Neuroscience 161(2):327–341. https://doi.org/10.1016/j.neuroscience.2009.03.019
Rausell E, Jones EG (1991) Chemically distinct compartments of the thalamic VPM nucleus in monkeys relay principal and spinal trigeminal pathways to different layers of the somatosensory cortex. J Neurosci Off J Soc Neurosci 11(1):226–237
Wallenberg A (1905) Sekundäre Bahnen aus den frontalen sensiblen Trigeminuskerne des Kaninchens. Anat Anz:145–155.
Woodburne RT (1936) A phylogenetic consideration of the primary and secondary centers and connections of the trigeminal complex in a series of vertebrates. J Comp Neurol 65(1):403–501. https://doi.org/10.1002/cne.900650113
Fleming GWTH (1937) The dorsal trigeminal tract and the centre median nucleus of luys. J Nerv Ment Disord 85:505 (Papez JW, Rundles W (1938) J Ment Sci 84(349):415–415. https://doi.org/10.1192/bjp.84.349.415-b)
Walker AE (1939) The origin, course and terminations of the secondary pathways of the trigeminal nerve in primates. J Comp Neurol 71(1):59–89. https://doi.org/10.1002/cne.900710105
Carpenter MB (1957) The dorsal trigeminal tract in the rhesus monkey. J Anat 91(Pt 1):82-90.3
Torvik A (1957) The ascending fibers from the main trigeminal sensory nucleus. An experimental study in the cat. Am J Anat 100(1):1–15. https://doi.org/10.1002/aja.1001000102
Smith JB, Watson GDR, Alloway KD, Schwarz C, Chakrabarti S (2015) Corticofugal projection patterns of whisker sensorimotor cortex to the sensory trigeminal nuclei. Front Neural Circuits 9:53. https://doi.org/10.3389/fncir.2015.00053
Matsushita M, Ikeda M, Okado N (1982) The cells of origin of the trigeminothalamic, trigeminospinal and trigeminocerebellar projections in the cat. Neuroscience 7(6):1439–1454
Juergen K (2004) The Human Nervous System (Hardback) par George Paxinos, 2edn. New Hardback, Mai
Brodal P (2016) The central nervous system, 5th edn. Oxford University Press, p 640
Welker WI (1973) Principles of organization of the ventrobasal complex in mammals. Brain Behav Evol 7(4):253–336. https://doi.org/10.1159/000124417
Kaas JH, Nelson RJ, Sur M, Dykes RW, Merzenich MM (1984) The somatotopic organization of the ventroposterior thalamus of the squirrel monkey, Saimiri sciureus. J Comp Neurol 226(1):111–140. https://doi.org/10.1002/cne.902260109
Lenz FA, Seike M, Lin YC et al (1993) Neurons in the area of human thalamic nucleus ventralis caudalis respond to painful heat stimuli. Brain Res 623(2):235–240. https://doi.org/10.1016/0006-8993(93)91433-s
Malick A, Strassman RM, Burstein R (2000) Trigeminohypothalamic and reticulohypothalamic tract neurons in the upper cervical spinal cord and caudal medulla of the rat. J Neurophysiol 84(4):2078–2112. https://doi.org/10.1152/jn.2000.84.4.2078
Edvinsson JCA, Viganò A, Alekseeva A et al (2020) The fifth cranial nerve in headaches. J Headache Pain 21(1):65. https://doi.org/10.1186/s10194-020-01134-1
Patel NM, M Das J (2021) Neuroanatomy, spinal trigeminal nucleus. In: StatPearls. StatPearls Publishing. Accessed 17 Jul 2021. http://www.ncbi.nlm.nih.gov/books/NBK539729/
Craig AD, Dostrovsky JO (1997) Anaesthesia: biologic foundations. In: Yaksh TL (ed) Processing of nociceptive information at supraspinal levels. Lippincott-Raven, Philadelphia, pp 625–642
Noseda R, Burstein R (2011) Advances in understanding the mechanisms of migraine-type photophobia. Curr Opin Neurol 24(3):197–202. https://doi.org/10.1097/WCO.0b013e3283466c8e
Noseda R, Jakubowski M, Kainz V, Borsook D, Burstein R (2011) Cortical projections of functionally identified thalamic trigeminovascular neurons: implications for migraine headache and its associated symptoms. J Neurosci Off J Soc Neurosci 31(40):14204–14217. https://doi.org/10.1523/JNEUROSCI.3285-11.2011
Noseda R, Kainz V, Jakubowski M et al (2010) A neural mechanism for exacerbation of headache by light. Nat Neurosci 13(2):239–245. https://doi.org/10.1038/nn.2475
Jantsch HHF, Kemppainen P, Ringler R, Handwerker HO, Forster C (2005) Cortical representation of experimental tooth pain in humans. Pain 118(3):390–399. https://doi.org/10.1016/j.pain.2005.09.017
Nash PG, Macefield VG, Klineberg IJ, Gustin SM, Murray GM, Henderson LA (2010) Bilateral activation of the trigeminothalamic tract by acute orofacial cutaneous and muscle pain in humans. Pain 151(2):384–393. https://doi.org/10.1016/j.pain.2010.07.027
Nash PG, Macefield VG, Klineberg IJ, Murray GM, Henderson LA (2009) Differential activation of the human trigeminal nuclear complex by noxious and non-noxious orofacial stimulation. Hum Brain Mapp 30(11):3772–3782. https://doi.org/10.1002/hbm.20805
Servos P, Engel SA, Gati J, Menon R (1999) fMRI evidence for an inverted face representation in human somatosensory cortex. NeuroReport 10(7):1393–1395
Nguyen BT, Tran TD, Hoshiyama M, Inui K, Kakigi R (2004) Face representation in the human primary somatosensory cortex. Neurosci Res 50(2):227–232. https://doi.org/10.1016/j.neures.2004.07.004
Moulton EA, Pendse G, Morris S, Aiello-Lammens M, Becerra L, Borsook D (2009) Segmentally arranged somatotopy within the face representation of human primary somatosensory cortex. Hum Brain Mapp 30(3):757–765. https://doi.org/10.1002/hbm.20541
Ramachandran VS, Hirstein W (1998) The perception of phantom limbs. The DO Hebb lecture. Brain J Neurol 121(Pt 9):1603–1630. https://doi.org/10.1093/brain/121.9.1603
Ramachandran VS, Brang D, McGeoch PD (2010) Dynamic reorganization of referred sensations by movements of phantom limbs. NeuroReport 21(10):727–730. https://doi.org/10.1097/WNR.0b013e32833be9ab
Uysal H, Özkan Ö, Barçın E, Şenol U, Tombak K, Özkan Ö (2016) Referred facial sensation on the hand after full face transplantation. Neurology 86(9):836–839. https://doi.org/10.1212/WNL.0000000000002409
Disbrow E, Roberts T, Krubitzer L (2000) Somatotopic organization of cortical fields in the lateral sulcus of Homo sapiens: evidence for SII and PV. J Comp Neurol 418(1):1–21. https://doi.org/10.1002/(sici)1096-9861(20000228)418:1%3c1::aid-cne1%3e3.0.co;2-p
Eickhoff SB, Schleicher A, Zilles K, Amunts K (2006) The human parietal operculum. I. Cytoarchitectonic mapping of subdivisions. Cereb Cortex 16(2):254–267. https://doi.org/10.1093/cercor/bhi105
Sanchez Panchuelo RM, Besle J, Schluppeck D, Humberstone M, Francis S (2018) Somatotopy in the human somatosensory system. Front Hum Neurosci. https://doi.org/10.3389/fnhum.2018.00235
Schulte LH, Sprenger C, May A (2016) Physiological brainstem mechanisms of trigeminal nociception: an fMRI study at 3T. Neuroimage 124(Pt A):518–525. https://doi.org/10.1016/j.neuroimage.2015.09.023
Mehnert J, Schulte L, Timmann D, May A (2017) Activity and connectivity of the cerebellum in trigeminal nociception. Neuroimage 150:112–118. https://doi.org/10.1016/j.neuroimage.2017.02.023
Stoodley CJ, Schmahmann JD (2010) Evidence for topographic organization in the cerebellum of motor control versus cognitive and affective processing. Cortex J Devoted Study Nerv Syst Behav 46(7):831–844. https://doi.org/10.1016/j.cortex.2009.11.008
Stoodley CJ, Valera EM, Schmahmann JD (2012) Functional topography of the cerebellum for motor and cognitive tasks: an fMRI study. Neuroimage 59(2):1560–1570. https://doi.org/10.1016/j.neuroimage.2011.08.065
Strata P, Scelfo B, Sacchetti B (2011) Involvement of cerebellum in emotional behavior. Physiol Res 60(Suppl 1):S39-48
Silverman JD, Kruger L (1985) Projections of the rat trigeminal sensory nuclear complex demonstrated by multiple fluorescent dye retrograde transport. Brain Res 361(1–2):383–388
Steindler DA (1985) Trigeminocerebellar, trigeminotectal, and trigeminothalamic projections: a double retrograde axonal tracing study in the mouse. J Comp Neurol 237(2):155–175. https://doi.org/10.1002/cne.902370203
Gobel S, Purvis MB (1972) Anatomical studies of the organization of the spinal V nucleus: the deep bundles and the spinal V tract. Brain Res 48:27–44. https://doi.org/10.1016/0006-8993(72)90169-2
Contreras RJ, Beckstead RM, Norgren R (1982) The central projections of the trigeminal, facial, glossopharyngeal and vagus nerves: an autoradiographic study in the rat. J Auton Nerv Syst 6(3):303–322. https://doi.org/10.1016/0165-1838(82)90003-0
Rustioni A, Baan JW, Verdonk-Karlsen S (1972) Afferents from the facial, vago-glossopharyngeal and second cervical nerves to the substantia gelatinosa of the rat. Brain Res 37(1):137–140. https://doi.org/10.1016/0006-8993(72)90355-1
Henssen DJHA, Derks B, van Doorn M et al (2019) Vagus nerve stimulation for primary headache disorders: an anatomical review to explain a clinical phenomenon. Cephalalgia 39(9):1180–1194. https://doi.org/10.1177/0333102419833076
Nomura S, Mizuno N (1984) Central distribution of primary afferent fibers in the Arnold’s nerve (the auricular branch of the vagus nerve): a transganglionic HRP study in the cat. Brain Res 292(2):199–205. https://doi.org/10.1016/0006-8993(84)90756-x
Ganchrow D (1978) Intratrigeminal and thalamic projections of nucleus caudalis in the squirrel monkey (Saimiri sciureus): a degeneration and autoradiographic study. J Comp Neurol 178(2):281–312. https://doi.org/10.1002/cne.901780206
McGee S (2012) Chapter 61—Examination of the reflexes. In: McGee S (ed) Evidence-based physical diagnosis (3rd ed). W.B. Saunders, Philadelphia, pp 581–592. doi:https://doi.org/10.1016/B978-1-4377-2207-9.00061-6
Schaller B, Probst R, Strebel S, Gratzl O (1999) Trigeminocardiac reflex during surgery in the cerebellopontine angle. J Neurosurg 90(2):215–220. https://doi.org/10.3171/jns.1999.90.2.0215
Meuwly C, Chowdhury T, Sandu N et al (2017) Definition and diagnosis of the trigeminocardiac reflex: a grounded theory approach for an update. Front Neurol 8:533. https://doi.org/10.3389/fneur.2017.00533
Chowdhury T, Mendelowith D, Golanov E et al (2015) Trigeminocardiac reflex: the current clinical and physiological knowledge. J Neurosurg Anesthesiol 27(2):136–147. https://doi.org/10.1097/ANA.0000000000000065
Leon-Ariza DS, Leon-Ariza JS, Nangiana J, Vargas Grau G, Leon-Sarmiento FE, Quiñones-Hinojosa A (2018) Evidences in neurological surgery and a cutting edge classification of the trigeminocardiac reflex: a systematic review. World Neurosurg 117:4–10. https://doi.org/10.1016/j.wneu.2018.05.208
Kumada M, Dampney RA, Reis DJ (1977) The trigeminal depressor response: a novel vasodepressor response originating from the trigeminal system. Brain Res 119(2):305–326
Sugrue A, DeSimone CV, Gaba P, El-Harasis MA, Deshmukh AJ, Asviravtham SJ (2018) Grinding to a halt: stimulation of the trigeminal cardiac reflex from severe bruxism. Hear Case Rep 4(8):329–331. https://doi.org/10.1016/j.hrcr.2017.06.013
Lapi D, Scuri R, Colantuoni A (2016) Trigeminal cardiac reflex and cerebral blood flow regulation. Front Neurosci 10:470. https://doi.org/10.3389/fnins.2016.00470
May A, Bahra A, Büchel C, Frackowiak RS, Goadsby PJ (1998) Hypothalamic activation in cluster headache attacks. Lancet Lond Engl 352(9124):275–278. https://doi.org/10.1016/S0140-6736(98)02470-2
May A (2005) Cluster headache: pathogenesis, diagnosis, and management. Lancet Lond Engl 366(9488):843–855. https://doi.org/10.1016/S0140-6736(05)67217-0
Leone M, Bussone G (2009) Pathophysiology of trigeminal autonomic cephalalgias. Lancet Neurol 8(8):755–764. https://doi.org/10.1016/S1474-4422(09)70133-4
Jannetta PJ (1967) Arterial compression of the trigeminal nerve at the pons in patients with trigeminal neuralgia. J Neurosurg 26(Suppl 1):159–162. https://doi.org/10.3171/jns.1967.26.1part2.0159
Headache Classification Committee of the International Headache Society (IHS) (2018) The international classification of headache disorders, 3rd edn. Cephalalgia Int J Headache 38(1):1–211. doi:https://doi.org/10.1177/0333102417738202
Cruccu G, Finnerup NB, Jensen TS et al (2016) Trigeminal neuralgia: new classification and diagnostic grading for practice and research. Neurology 87(2):220–228. https://doi.org/10.1212/WNL.0000000000002840
Cruccu G, Di Stefano G, Truini A (2020) Trigeminal neuralgia. N Engl J Med 383(8):754–762. https://doi.org/10.1056/NEJMra1914484
Liu J, Wu G, Xiang H et al (2020) Long-term retrospective analysis of microvascular decompression in patients with recurrent trigeminal neuralgia. Front Neurol. https://doi.org/10.3389/fneur.2020.584224
Barker FG, Jannetta PJ, Bissonette DJ, Larkins MV, Jho HD (1996) The long-term outcome of microvascular decompression for trigeminal neuralgia. N Engl J Med 334(17):1077–1083. https://doi.org/10.1056/NEJM199604253341701
Jannetta PJ (1980) Neurovascular compression in cranial nerve and systemic disease. Ann Surg 192(4):518–525
Lopez BC, Hamlyn PJ, Zakrzewska JM (2004) Systematic review of ablative neurosurgical techniques for the treatment of trigeminal neuralgia. Neurosurgery 54(4):973–982 (discussion 982–983)
Lopez BC, Hamlyn PJ, Zakrzewska JM (2004) Stereotactic radiosurgery for primary trigeminal neuralgia: state of the evidence and recommendations for future reports. J Neurol Neurosurg Psychiatry 75(7):1019–1024
Cheng JS, Lim DA, Chang EF, Barbaro NM (2014) A review of percutaneous treatments for trigeminal neuralgia. Neurosurgery 10(Suppl 1):25–33. https://doi.org/10.1227/NEU.00000000000001687 (discussion 33)
Broggi G, Ferroli P, Franzini A, Servello D, Dones I (2000) Microvascular decompression for trigeminal neuralgia: comments on a series of 250 cases, including 10 patients with multiple sclerosis. J Neurol Neurosurg Psychiatry 68(1):59–64
Keravel Y, Gaston A, Ciampi de Andrade D, Mencattini G, Le Guérinel C (2009) Traitement de la névralgie trigéminale par la compression par ballon. Neurochirurgie 55(2):197–202. https://doi.org/10.1016/j.neuchi.2009.01.008
Wang C, Dou Z, Yan M et al (2021) The comparison of efficacy and complications of coblation and radiofrequency thermocoagulation for V2/V3 idiopathic trigeminal neuralgia: a retrospective cohort study of 292 cases. BMC Anesthesiol 21:6. https://doi.org/10.1186/s12871-020-01224-2
Udupi BP, Chouhan RS, Dash HH, Bithal PK, Prabhakar H (2012) Comparative evaluation of percutaneous retrogasserian glycerol rhizolysis and radiofrequency thermocoagulation techniques in the management of trigeminal neuralgia. Neurosurgery 70(2):407–412. https://doi.org/10.1227/NEU.0b013e318233a85f (discussion 412–413)
Holste K, Chan AY, Rolston JD, Englot DJ (2020) Pain outcomes following microvascular decompression for drug-resistant trigeminal neuralgia: a systematic review and meta-analysis. Neurosurgery 86(2):182–190. https://doi.org/10.1093/neuros/nyz075
Jones MR, Urits I, Ehrhardt KP et al (2019) A comprehensive review of trigeminal neuralgia. Curr Pain Headache Rep 23(10):74. https://doi.org/10.1007/s11916-019-0810-0
Dandy WE (1929) An operation for the cure of tic douloureux: partial section of the sensory root at the pons. Arch Surg 18(2):687–734. https://doi.org/10.1001/archsurg.1929.04420030081005
Fontaine D, Vandersteen C, Magis D, Lanteri-Minet M (2015) Neuromodulation in cluster headache. Adv Tech Stand Neurosurg 42:3–21. https://doi.org/10.1007/978-3-319-09066-5_1
Hoffmann J, May A (2019) Neuromodulation for the treatment of primary headache syndromes. Expert Rev Neurother 19(3):261–268. https://doi.org/10.1080/14737175.2019.1585243
Hoffmann J, May A (2018) Diagnosis, pathophysiology, and management of cluster headache. Lancet Neurol 17(1):75–83. https://doi.org/10.1016/S1474-4422(17)30405-2
Rozen TD, Fishman RS (2012) Cluster headache in the United States of America: demographics, clinical characteristics, triggers, suicidality, and personal burden. Headache 52(1):99–113. https://doi.org/10.1111/j.1526-4610.2011.02028.x
Miller S, Matharu M (2017) Non-invasive neuromodulation in primary headaches. Curr Pain Headache Rep 21(3):14. https://doi.org/10.1007/s11916-017-0608-x
Antony AB, Mazzola AJ, Dhaliwal GS, Hunter CW (2019) Neurostimulation for the treatment of chronic head and facial pain: a literature review. Pain Physician 22(5):447–477
Weiner RL, Reed KL (1999) Peripheral neurostimulation for control of intractable occipital neuralgia. Neuromodulation J Int Neuromodulation Soc 2(3):217–221. https://doi.org/10.1046/j.1525-1403.1999.00217.x
Magis D, Bruno M-A, Fumal A et al (2011) Central modulation in cluster headache patients treated with occipital nerve stimulation: an FDG-PET study. BMC Neurol 11:25. https://doi.org/10.1186/1471-2377-11-25
Ansarinia M, Rezai A, Tepper SJ et al (2010) Electrical stimulation of sphenopalatine ganglion for acute treatment of cluster headaches. Headache 50(7):1164–1174. https://doi.org/10.1111/j.1526-4610.2010.01661.x
Goadsby PJ, Sahai-Srivastava S, Kezirian EJ et al (2019) Safety and efficacy of sphenopalatine ganglion stimulation for chronic cluster headache: a double-blind, randomised controlled trial. Lancet Neurol 18(12):1081–1090. https://doi.org/10.1016/S1474-4422(19)30322-9
Schoenen J, Jensen RH, Lantéri-Minet M et al (2013) Stimulation of the sphenopalatine ganglion (SPG) for cluster headache treatment. Pathway CH-1: a randomized, sham-controlled study. Cephalalgia Int J Headache 33(10):816–830. https://doi.org/10.1177/0333102412473667
Jürgens TP, Barloese M, May A et al (2017) Long-term effectiveness of sphenopalatine ganglion stimulation for cluster headache. Cephalalgia 37(5):423–434. https://doi.org/10.1177/0333102416649092
Leone M, Franzini A, Bussone G (2001) Stereotactic stimulation of posterior hypothalamic gray matter in a patient with intractable cluster headache. N Engl J Med 345(19):1428–1429. https://doi.org/10.1056/NEJM200111083451915
Fontaine D, Lanteri-Minet M, Ouchchane L et al (2010) Anatomical location of effective deep brain stimulation electrodes in chronic cluster headache. Brain J Neurol 133(Pt 4):1214–1223. https://doi.org/10.1093/brain/awq041
Nowacki A, Schober M, Nader L et al (2020) Deep brain stimulation for chronic cluster headache: meta-analysis of individual patient data. Ann Neurol. https://doi.org/10.1002/ana.25887
Schoenen J, Di Clemente L, Vandenheede M et al (2005) Hypothalamic stimulation in chronic cluster headache: a pilot study of efficacy and mode of action. Brain J Neurol 128(Pt 4):940–947. https://doi.org/10.1093/brain/awh411
Leone M, Franzini A, Broggi G, Bussone G (2006) Hypothalamic stimulation for intractable cluster headache: long-term experience. Neurology 67(1):150–152. https://doi.org/10.1212/01.wnl.0000223319.56699.8a
Monsalve GA (2012) Motor cortex stimulation for facial chronic neuropathic pain: A review of the literature. Surg Neurol Int 3(Suppl 4):S290–S311. https://doi.org/10.4103/2152-7806.103023
Fontaine D, Hamani C, Lozano A (2009) Efficacy and safety of motor cortex stimulation for chronic neuropathic pain: critical review of the literature. J Neurosurg 110(2):251–256. https://doi.org/10.3171/2008.6.17602
Graff-Radford SB (2009) Facial pain. Neurologist 15(4):171–177. https://doi.org/10.1097/NRL.0b013e31819827d8
Henderson JM, Lad SP (2006) Motor cortex stimulation and neuropathic facial pain. Neurosurg Focus 21(6):E6. https://doi.org/10.3171/foc.2006.21.6.9
Nuti C, Peyron R, Garcia-Larrea L et al (2005) Motor cortex stimulation for refractory neuropathic pain: four year outcome and predictors of efficacy. Pain 118(1–2):43–52. https://doi.org/10.1016/j.pain.2005.07.020
Rasche D, Ruppolt M, Stippich C, Unterberg A, Tronnier VM (2006) Motor cortex stimulation for long-term relief of chronic neuropathic pain: a 10 year experience. Pain 121(1–2):43–52. https://doi.org/10.1016/j.pain.2005.12.006
Raslan AM, Nasseri M, Bahgat D, Abdu E, Burchiel KJ (2011) Motor cortex stimulation for trigeminal neuropathic or deafferentation pain: an institutional case series experience. Stereotact Funct Neurosurg 89(2):83–88. https://doi.org/10.1159/000323338
Mercier C, Léonard G (2011) Interactions between pain and the motor cortex: insights from research on phantom limb pain and complex regional pain syndrome. Physiother Can Physiother Can 63(3):305–314. https://doi.org/10.3138/ptc.2010-08p
Meyerson B (2005) Motor cortex stimulation–effective for neuropathic pain but the mode of action remains illusive. Pain 118(1–2):6–7. https://doi.org/10.1016/j.pain.2005.07.019
Kishima H, Saitoh Y, Osaki Y et al (2007) Motor cortex stimulation in patients with deafferentation pain: activation of the posterior insula and thalamus. J Neurosurg 107(1):43–48. https://doi.org/10.3171/JNS-07/07/0043
Slavin KV, Nersesyan H, Colpan ME, Munawar N (2007) Current algorithm for the surgical treatment of facial pain. Head Face Med 3:30. https://doi.org/10.1186/1746-160X-3-30
Silberstein SD, Yuan H, Najib U et al (2020) Non-invasive vagus nerve stimulation for primary headache: a clinical update. Cephalalgia Int J Headache 40(12):1370–1384. https://doi.org/10.1177/0333102420941864
Yuan H, Silberstein SD (2016) Vagus nerve and vagus nerve stimulation, a comprehensive review: part I. Headache 56(1):71–78. https://doi.org/10.1111/head.12647
Yuan H, Silberstein SD (2016) Vagus nerve and vagus nerve stimulation, a comprehensive review: Part II. Headache 56(2):259–266. https://doi.org/10.1111/head.12650
Yuan H, Silberstein SD (2016) Vagus nerve and vagus nerve stimulation, a comprehensive review: Part III. Headache 56(3):479–490. https://doi.org/10.1111/head.12649
Yuan H, Silberstein SD (2017) Vagus nerve stimulation and headache. Headache 57(Suppl 1):29–33. https://doi.org/10.1111/head.12721
Silberstein SD, Calhoun AH, Lipton RB et al (2016) Chronic migraine headache prevention with noninvasive vagus nerve stimulation: the EVENT study. Neurology 87(5):529–538. https://doi.org/10.1212/WNL.0000000000002918
Goadsby PJ, de Coo IF, Silver N et al (2018) Non-invasive vagus nerve stimulation for the acute treatment of episodic and chronic cluster headache: a randomized, double-blind, sham-controlled ACT2 study. Cephalalgia Int J Headache 38(5):959–969. https://doi.org/10.1177/0333102417744362
Gaul C, Diener H-C, Silver N et al (2016) Non-invasive vagus nerve stimulation for PREVention and Acute treatment of chronic cluster headache (PREVA): a randomised controlled study. Cephalalgia Int J Headache 36(6):534–546. https://doi.org/10.1177/0333102415607070
Tassorelli C, Grazzi L, de Tommaso M et al (2018) Noninvasive vagus nerve stimulation as acute therapy for migraine: the randomized PRESTO study. Neurology 91(4):e364–e373. https://doi.org/10.1212/WNL.0000000000005857
Diener H-C, Goadsby PJ, Ashina M et al (2019) Non-invasive vagus nerve stimulation (nVNS) for the preventive treatment of episodic migraine: the multicentre, double-blind, randomised, sham-controlled PREMIUM trial. Cephalalgia Int J Headache 39(12):1475–1487. https://doi.org/10.1177/0333102419876920
Silberstein SD, Mechtler LL, Kudrow DB et al (2016) Non-invasive vagus nerve stimulation for the acute treatment of cluster headache: findings from the randomized, double-blind, sham-controlled ACT1 study. Headache 56(8):1317–1332. https://doi.org/10.1111/head.12896
Marin J, Giffin N, Consiglio E, McClure C, Liebler E, Davies B (2018) Non-invasive vagus nerve stimulation for treatment of cluster headache: early UK clinical experience. J Headache Pain 19(1):114. https://doi.org/10.1186/s10194-018-0936-1
Magis D, D’Ostilio K, Thibaut A et al (2017) Cerebral metabolism before and after external trigeminal nerve stimulation in episodic migraine. Cephalalgia Int J Headache 37(9):881–891. https://doi.org/10.1177/0333102416656118
Chou DE, Shnayderman Yugrakh M, Winegarner D, Rowe V, Kuruvilla D, Schoenen J (2019) Acute migraine therapy with external trigeminal neurostimulation (ACME): a randomized controlled trial. Cephalalgia Int J Headache 39(1):3–14. https://doi.org/10.1177/0333102418811573
Schoenen J, Vandersmissen B, Jeangette S et al (2013) Migraine prevention with a supraorbital transcutaneous stimulator: a randomized controlled trial. Neurology 80(8):697–704. https://doi.org/10.1212/WNL.0b013e3182825055
Lan L, Zhang X, Li X, Rong X, Peng Y (2017) The efficacy of transcranial magnetic stimulation on migraine: a meta-analysis of randomized controlled trials. J Headache Pain 18(1):86. https://doi.org/10.1186/s10194-017-0792-4
Andreou AP, Holland PR, Akerman S, Summ O, Fredrick J, Goadsby PJ (2016) Transcranial magnetic stimulation and potential cortical and trigeminothalamic mechanisms in migraine. Brain J Neurol 139(Pt 7):2002–2014. https://doi.org/10.1093/brain/aww118
Lipton RB, Dodick DW, Silberstein SD et al (2010) Single-pulse transcranial magnetic stimulation for acute treatment of migraine with aura: a randomised, double-blind, parallel-group, sham-controlled trial. Lancet Neurol 9(4):373–380. https://doi.org/10.1016/S1474-4422(10)70054-5
Gangitano M, Valero-Cabré A, Tormos JM, Mottaghy FM, Romero JR, Pascual-Leone A (2002) Modulation of input-output curves by low and high frequency repetitive transcranial magnetic stimulation of the motor cortex. Clin Neurophysiol Off J Int Fed Clin Neurophysiol 113(8):1249–1257. https://doi.org/10.1016/s1388-2457(02)00109-8
Acknowledgements
We thank the French Academy of Medicine, the French Society of Neurosurgery, and the Foundation «Les Gueules Cassées» for their financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Terrier, LM., Hadjikhani, N. & Destrieux, C. The trigeminal pathways. J Neurol 269, 3443–3460 (2022). https://doi.org/10.1007/s00415-022-11002-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-022-11002-4