Abstract
Objective
To determine whether diffusion-weighted imaging (DWI) can help differentiate peri-ictal signal abnormality from limbic encephalitis (LE) among patients with medial temporal lobe T2-hyperintensity.
Methods
We retrospectively identified patients with peri-ictal medial temporal lobe T2-hyperintensity using a Mayo Clinic database, and reviewed their DWI to look for unique diffusion restriction patterns. We then identified patients with medial temporal lobe T2-hyperintensity and LE, and reviewed their DWI to see if these patterns were ever present. Presence of diffusion restriction patterns was confirmed by a blinded neuro-radiologist.
Results
We identified 10 patients without LE who had peri-ictal unilateral medial temporal lobe T2-hyperintensity, ipsilateral to focal seizure onset. Nine of 10 (90%) had at least one of two diffusion restriction patterns potentially unique to seizure activity; four had gyriform hippocampal diffusion restriction (“Pattern 1”), three had diffuse hippocampal diffusion restriction that spared the most medial temporal lobe structures (“Pattern 2”), and two had both diffusion restriction patterns. The median patient age was 62 years (range 2–76 years) and 3/9 (33%) were female. In comparison, among patients with medial temporal lobe T2-hyperintensity and LE, only 5/57 (9%) had one of the diffusion restriction patterns (“Pattern 2”) identified (P < 0.0001); all five had seizures reported.
Conclusions
In patients with medial temporal lobe T2-hyperintensity and one of the diffusion restriction patterns described herein, the signal abnormality may be a peri-ictal phenomenon rather than indicative of LE and should prompt investigation for seizure. Even in patients with LE, these patterns should raise concern for seizure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Limbic encephalitis (LE) is an immune-mediated neurologic disease that typically presents with memory deficits, psychiatric symptoms, and/or seizures [1]. Common etiologies include herpes simplex virus encephalitis (HSV-E) and autoimmune limbic encephalitis (ALE) [2]. On brain magnetic resonance imaging (MRI) medial temporal lobe T2-hyperintensity is classically seen in LE but is not pathognomonic of this disease [1, 3, 4]. Peri-ictal signal abnormality can have a similar radiographic appearance and cause diagnostic confusion [5, 6]. Medial temporal lobe diffusion restriction has been reported in patients with a recent seizure, indicating that diffusion-weighted imaging (DWI) may help distinguish between peri-ictal signal abnormality and LE [7,8,9]. Among patients with medial temporal lobe T2-hyperintensity, we thus sought to identify patients with recent seizure activity as well as patients with LE to look for diffusion restriction patterns that could help differentiate between these two entities.
Methods
We searched radiology reports from 1999 to 2019 in the Mayo Clinic Rochester Electronic Medical Record (EMR) using the Advanced Cohort Explorer search engine to identify patients with medial temporal lobe T2-hyperintensity attributed to recent seizure. An established non-encephalitic cause of seizure was required for inclusion. The following search terms were queried in brain MRI reports: ‘limbic AND seizure’, ‘seizure-related’, ‘seizure related’, ‘post-ictal’, ‘postictal’, ‘status epilepticus’, ‘mesial temporal T2-hyperintensity’, ‘mesial temporal T2 hyperintensity’, ‘medial temporal T2-hyperintensity’, ‘medial temporal T2 hyperintensity’, ‘limbic encephalitis’, or ‘herpes encephalitis’, which returned 3022 unique patients. We then proceeded as per the Flow Diagram. Patients with a clear cause of medial temporal lobe T2-hyperintensity other than peri-ictal signal abnormality (e.g. stroke, tumor) were excluded. Remaining patients were queried to identify those with an electroencephalogram (EEG) performed within 24 h of MRI, which was used as a surrogate marker of high suspicion for seizure. Of these patients, radiology reports were again reviewed to identify those with medial temporal lobe T2-hyperintensity deemed to be of likely peri-ictal origin (e.g. seizure listed in MRI indication, resolution of abnormality on short-term repeat MRI); mention of a seizure by the reporting radiologist, however, was not required for inclusion. The EMR of these patients were then reviewed to identify those with medial temporal lobe T2-hyperintensity that was definitely peri-ictal in origin. This was defined as (1) clinical and/or electrographic seizure documented within 48 h of MRI; (2) non-LE etiology of seizure identified; and (3) no other cause of medial temporal lobe T2-hyperintensity. Although EEG performed within 24 h of MRI was a criterion for inclusion, clinical and/or electrographic seizure documentation was required within 48 h for medial temporal lobe T2-hyperintensity to be classified as definitely peri-ictal in origin. This decision was made to increase the study sample size while ensuring a close temporal relationship between brain MRI and seizure activity. The DWI sequences of patients meeting these criteria were then reviewed to identify diffusion restriction patterns potentially unique to recent seizure activity.

We next evaluated whether any identified peri-ictal diffusion restriction patterns were observed in patients with LE. Among our patients with medial temporal lobe T2-hyperintensity we identified 31 patients with HSV-E defined by cerebrospinal fluid (CSF) HSV polymerase chain reaction positivity, and 26 patients with ALE defined by the positivity of a neural antibody in serum and/or CSF (e.g. LGI1, CASPR2, AMPAR, GABA(B)R, ANNA-1) via testing at the Mayo Clinic as previously described [10, 11]. In this LE comparison cohort, brain MRI at presentation and up to 30 days thereafter was reviewed to look for diffusion restriction patterns we had identified in our non-LE seizure cohort.
In all suspected cases, the presence of any diffusion restriction pattern was confirmed by a board-certified radiologist with a certificate of added qualification in neuroradiology who was blinded to the diagnosis (G.B.L).
Results
We identified 10 patients with peri-ictal medial temporal lobe T2-hyperintensity. In all patients, the finding was unilateral, with evidence of ipsilateral focal-onset seizures. Nine of 10 patients (90%) had medial temporal lobe diffusion restriction; four had gyriform hippocampal diffusion restriction (“Pattern 1”, Fig. 1), three had diffuse hippocampal diffusion restriction that spared the most medial temporal lobe structures (“Pattern 2”, Fig. 2), and two had both gyriform and diffuse hippocampal diffusion restriction patterns (Table 1). The median patient age was 62 years (range 2–76 years) and 3/9 (33%) were female. All patients had prolonged and/or recurrent seizures concerning for status epilepticus (SE). Repeat brain MRI was performed greater than one month after the acute presentation in 6/9 (66%) in patients with diffusion restriction. All six demonstrated hippocampal atrophy, in addition to the resolution of diffusion restriction as would be expected with signal abnormality related to acute seizure activity.
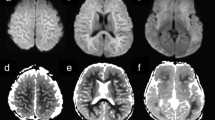
Gyriform hippocampal diffusion restriction in patients with recent seizure (“Pattern 1”). Axial T2-weighted FLAIR imaging shows left medial temporal lobe T2-hyperintensity in a patient with recent seizure activity (A1, A2, red arrows). In the same patient, DWI shows gyriform left hippocampal diffusion hyperintensity (A3, A4, red arrows). A similar diffusion restriction pattern is seen in a second patient (B1, B2) and third patient (C1, C2) with seizure activity; apparent diffusion coefficient (ADC) map of the third patient shows representative corresponding hypointensity in keeping with true diffusion restriction (C3, C4, red arrows)
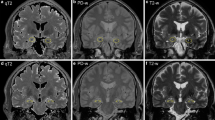
Diffuse hippocampal diffusion restriction in patients with recent seizure (“Pattern 2”). Axial T2-weighted FLAIR imaging shows left medial temporal lobe hyperintensity in a patient with recent seizure activity (A1, A2, red arrows). In the same patient, DWI shows diffuse left hippocampal diffusion hyperintensity (A3, A4, red arrows) with sparing of the most medial temporal lobe structures (A3, blue arrow). A similar diffusion restriction pattern is seen in a second patient (B1, B2) and third patient (C1, C2) with seizure activity; ADC map of the third patient shows representative corresponding hypointensity in keeping with true diffusion restriction (C3, C4, red arrows)
In comparison, only 5/57 patients with LE (9%) had one these diffusion restriction patterns (P < 0.0001, Fisher’s Exact test). Of the 31 with HSV-E, four (13%) had “Pattern 2” diffusion restriction; one presented with confusion and developed tonic–clonic SE less than 24 h after MRI, one presented with confusion and was in focal-onset SE on EEG less than 24 h after MRI, and two had a high clinical concern for focal-onset SE (one presented with confusion followed by right facial twitching and automatisms less than 48 h after MRI, the other presented with left head deviation and shaking movements followed by a persistent decreased level of consciousness less than 48 h before MRI). All four of these patients had repeat brain MRI performed within one week of acute presentation, and progressive diffusion restriction outside the medial temporal lobe involving typical regions of HSV-E (i.e. anterior temporal lobe, insular cortex and/or cingulate cortex) was observed in all [2]. One of four patients had a repeat brain MRI that was performed greater than one month after acute presentation, which showed resolution of diffusion restriction and temporo-insular encephalomalacia. Of the 26 with antibody-positive ALE (17 LGI1, 5 ANNA-1, 3 CASPR2, 1 AMPAR), only one (4%) with LGI1-antibody encephalitis had “Pattern 2” diffusion restriction. This patient presented with confusion and initial EEG showed only slowing, but repeat EEG for persistent cognitive difficulties 2 weeks later showed focal electrographic seizures. Repeat brain MRI performed within one week of the acute presentation showed additional subtle diffusion restriction of the caudate, which has been previously described in LGI1-antibody encephalitis [12]. Repeat brain MRI performed greater than one month after acute presentation showed resolution of diffusion restriction, as well as hippocampal atrophy and caudate encephalomalacia.
Discussion
Among patients with medial temporal lobe T2-hyperintensity, we describe two patterns of diffusion restriction that can help differentiate peri-ictal signal abnormality from LE. Although LE may be investigated as a substrate of seizure activity in this setting, other etiologies should be considered if supportive evidence of LE is lacking.
Medial temporal lobe diffusion restriction has been reported previously with recent seizure activity, and presumably reflects transient seizure-induced vasogenic and cytotoxic edema [5, 7]. However, diffusion restriction in this region has also been described in infectious and autoimmune encephalitis [13, 14], indicating that the simple presence of diffusion restriction cannot distinguish seizure-related change from LE. Furthermore, patients with LE are at high risk for seizures, making it difficult to determine whether diffusion restriction reported in the previous series was related to LE or recent seizure activity [13]. For this reason, a systematic investigation of diffusion restriction patterns that can help differentiate between these two entities is needed.
There are several limitations to this retrospective study. Due to the stringency of criteria that needed to be met before classifying medial temporal lobe T2-hyperintensity as peri-ictal in origin, the number of patients who were ultimately included in this cohort was small. These highly selective criteria, however, served to minimize the possibility of factors other than recent seizure contributing to the medial temporal lobe T2-hyperintensity observed. In particular, the requirement of an established cause of seizure other than LE minimized the likelihood of antibody-negative ALE in this cohort, permitting identification of diffusion restriction patterns attributable to recent seizure activity. Furthermore, because our search strategy was designed to identify patients with a high likelihood of peri-ictal signal abnormality, clinical sensitivity and specificity of the diffusion restriction patterns we identified for recent seizure activity in all patients with medial temporal lobe T2-hyperintensity could not be determined. Nonetheless, the diffusion restriction patterns we identified were reproducibly seen in patients with peri-ictal medial temporal lobe T2-hyperintensity and were also associated with seizures in patients with medial temporal lobe T2-hyperintensity and LE.
Recognition of these diffusion restriction patterns in patients with medial temporal lobe T2-hyperintensity should prompt consideration of seizure rather than LE as the cause of the signal abnormality. Even in patients with LE, the presence of these patterns should raise concern for seizure.
Availability of data and material
Anonymized data is available for all study patients and will be shared by request from any qualified investigator.
References
Graus F, Titulaer MJ, Balu R, Benseler S, Bien CG, Cellucci T et al (2016) A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol 15(4):391–404
Chow FC, Glaser CA, Sheriff H, Xia D, Messenger S, Whitley R et al (2015) Use of clinical and neuroimaging characteristics to distinguish temporal lobe herpes simplex encephalitis from its mimics. Clin Infect Dis 60(9):1377–1383
Budhram A, Leung A, Nicolle MW, Burneo JG (2019) Diagnosing autoimmune limbic encephalitis. CMAJ 191(19):E529–E534
Vogrig A, Joubert B, Ducray F, Thomas L, Izquierdo C, Decaestecker K et al (2018) Glioblastoma as differential diagnosis of autoimmune encephalitis. J Neurol 265(3):669–677
Kim SE, Lee BI, Shin KJ, Ha SY, Park J, Park KM et al (2017) Characteristics of seizure-induced signal changes on MRI in patients with first seizures. Seizure 48:62–68
Macher S, Zimprich F, De Simoni D, Hoftberger R, Rommer PS (2018) Management of autoimmune encephalitis: an observational monocentric study of 38 patients. Front Immunol 9:2708
Kim JA, Chung JI, Yoon PH, Kim DI, Chung TS, Kim EJ et al (2001) Transient MR signal changes in patients with generalized tonicoclonic seizure or status epilepticus: periictal diffusion-weighted imaging. AJNR 22(6):1149–1160
Chatzikonstantinou A, Gass A, Forster A, Hennerici MG, Szabo K (2011) Features of acute DWI abnormalities related to status epilepticus. Epilepsy Res 97(1–2):45–51
Szabo K, Poepel A, Pohlmann-Eden B, Hirsch J, Back T, Sedlaczek O et al (2005) Diffusion-weighted and perfusion MRI demonstrates parenchymal changes in complex partial status epilepticus. Brain 128(Pt 6):1369–1376
Zalewski NL, Lennon VA, Lachance DH, Klein CJ, Pittock SJ, McKeon A (2016) P/Q- and N-type calcium-channel antibodies: oncological, neurological, and serological accompaniments. Muscle Nerve 54(2):220–227
Dubey D, Pittock SJ, Kelly CR, McKeon A, Lopez-Chiriboga AS, Lennon VA et al (2018) Autoimmune encephalitis epidemiology and a comparison to infectious encephalitis. Ann Neurol 83(1):166–177
López Chiriboga AS, Siegel JL, Tatum WO, Shih JJ, Flanagan EP (2017) Striking basal ganglia imaging abnormalities in LGI1 ab faciobrachial dystonic seizures. Neurol (R) Neuroimmunol Neuroinflamm 4(3):e336
Kotsenas AL, Watson RE, Pittock SJ, Britton JW, Hoye SL, Quek AM et al (2014) MRI findings in autoimmune voltage-gated potassium channel complex encephalitis with seizures: one potential etiology for mesial temporal sclerosis. AJNR 35(1):84–89
Sener RN (2001) Herpes simplex encephalitis: diffusion MR imaging findings. Compute Med Imaging Gr 25(5):391–397
Funding
There is no funding to report for this research.
Author information
Authors and Affiliations
Contributions
Dr. AB conceptualized and designed the study; drafted manuscript; analyzed and interpreted the data; composed the figures. Dr. JB interpreted the data; revised the manuscript for intellectual content. Dr. GL interpreted the data; revised the manuscript for intellectual content. Dr. DD interpreted the data; revised the manuscript for intellectual content. Dr. AZ interpreted the data; revised the manuscript for intellectual content. Dr. EF interpreted the data; revised the manuscript for intellectual content. Dr. AM interpreted the data; revised the manuscript for intellectual content. Dr. SP interpreted the data; revised the manuscript for intellectual content. Dr. SB interpreted the data; revised the manuscript for intellectual content. Dr. NZ conceptualized and designed the study; analyzed and interpreted the data; revised the manuscript for intellectual content; supervised the study.
Corresponding author
Ethics declarations
Conflicts of interest
Dr. Adrian Budhram has no disclosures to report. Dr. Jeff Britton is a consultant for UCB pharmaceuticals, investigational drug discussion, Rozanolixizumab; a co-investigator, unpaid, for GW Pharma in a double-blind, randomized, placebo-controlled study to investigate the efficacy and safety of cannabidiol (GWP42003-P, CBD) as add-on therapy in patients with tuberous sclerosis complex who experience inadequately-controlled seizures; a co-investigator, unpaid, for Grifols Pharmaceuticals in a Randomized Double-Blind Placebo Controlled Study of IVIG in Patients with Voltage-Gated Potassium Channel Complex Antibody Associated Autoimmune Epilepsy. Dr. Greta Liebo has no disclosures to report. Dr. Divyanshu Dubey has a patent pending for Kelch-like protein 11 as a marker of neurological autoimmunity and has received research support from Grifols, Translational Research Innovation and Test Development Office and, Center for Clinical and Translational Science. Dr. Dubey has consulted for UCB and Astellas. All compensation for consulting activities is paid directly to Mayo Clinic. Dr. Anastasia Zekeridou has a patent pending on PDE10A-IgG as a biomarker of neurological autoimmunity. Dr. Eoin Flanagan is a site principal investigator in a randomized placebo-controlled clinical trial of Inebilizumab (A CD19 inhibitor) in neuromyelitis optica spectrum disorders funded by MedImmune/Viela Bio. He receives no personal compensation and just receives reimbursement for the research activities related to the trial. Dr. Andrew McKeon reports grants from Alexion, grants from Grifols, grants from Euroimmun, outside the submitted work; in addition, Dr. McKeon has a patent Septin-5-IgG pending, a patent PDE10A-IgG pending, a patent MAP1B-IgG pending, and a patent GFAP-IgG pending. Dr. Sean Pittock reports grants, personal fees and non-financial support from Alexion Pharmaceuticals, Inc.; grants from Grifols, Autoimmune Encephalitis Alliance; grants, personal fees, non-financial support and other from MedImmune, Inc.; Dr. Pittock has a patent # 9,891,219 (Application#12-573942) “Methods for Treating Neuromyelitis Optica (NMO) by Administration of Eculizumab to an individual that is Aquaporin-4 (AQP4)-IgG Autoantibody positive”. Dr Pittock also has patents pending for the following IgGs as biomarkers of autoimmune neurological disorders (septin-5, Kelch-like protein 11, GFAP, PDE10A and MAP1B). Dr. Sherri Braksick has no disclosures to report. Dr. Nicholas Zalewski has no disclosures to report.
Ethics approval
The Mayo Clinic Institutional Review Board (IRB) approved the study (IRB number 08‐006647).
Consent to participate
As patients were identified retrospectively informed consent was not specifically obtained for this submission, but the patients did agree to use of their medical records for research purposes and no patient-identifying images have been included.
Consent to publication
The authors consent to the publication of this manuscript. The manuscript and figures have not been published or copyrighted elsewhere.
Rights and permissions
About this article
Cite this article
Budhram, A., Britton, J.W., Liebo, G.B. et al. Use of diffusion-weighted imaging to distinguish seizure-related change from limbic encephalitis. J Neurol 267, 3337–3342 (2020). https://doi.org/10.1007/s00415-020-10007-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-10007-1