Abstract
A man and a woman were found dead in the same car with a burned coal briquette. The cause of death of the woman was assigned to acute carbon monoxide (CO) poisoning without difficulty based on typical findings associated with this condition, including elevation of carboxyhaemoglobin (COHb). However, the man had an unremarkable elevation of COHb and a higher rectal temperature compared to that of the woman. Postmortem computed tomography (PMCT) revealed ambiguous low-density areas in the bilateral globi pallidi. Further analysis by postmortem magnetic resonance (PMMR) imaging showed these lesions more clearly; the lesions appeared as marked high signal intensity areas on both the T2-weighted images and the fluid-attenuated inversion recovery sequences. A subsequent autopsy revealed signs of pneumonia, dehydration, starvation, and hypothermia, suggesting that the man died from prolonged CO poisoning. Both globi pallidi contained grossly ambiguous lesions, and a detailed neuropathologic investigation revealed these lesions to be coagulative necrotic areas; this finding was compatible with a diagnosis of prolonged CO poisoning. This case report shows that postmortem imaging, especially PMMR, is useful for detecting necrotic lesions associated with prolonged CO poisoning. This report further exemplifies the utility of PMMR for detecting brain lesions, which may be difficult to detect by macroscopic analysis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Carbon monoxide (CO) poisoning is life-threatening and can be acute, prolonged, or delayed. Acute CO poisoning can cause hypoxia and death. After blood carboxyhaemoglobin (COHb) levels decrease, prolonged neurological impairment, including impaired consciousness, or prolonged cardiac sequelae may occur, which can cause death. Even if the victim recovers from acute and prolonged disturbances, some delayed neurological sequelae may occur. Such sequelae include impaired consciousness with a lucid interval of approximately 2–4 weeks [1], which may be fatal.
A number of studies have reported lesions in several regions of the brain in some cases of prolonged or delayed CO poisoning. Lapresle and Fardeau [2] described 22 different types of CO poisoning autopsy cases and various CO poisoning-related brain lesions, including: coagulative necrosis in the globus pallidus, hippocampi, cerebral cortex, cerebral white matter, and cerebellum, as well as demyelination in cerebral and cerebellar white matter. These lesions have also been detected using imaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI) in living patients with CO poisoning [1, 3,4,5,6,7]. Bilateral globi pallidi lesions are the most recognized type of lesion associated with CO poisoning. Detection of such changes may be important in diagnosing fatalities due to prolonged or delayed death associated with CO poisoning because, in such cases, the COHb level in the cadavers is within the normal range and other findings of acute CO poisoning may have subsided.
Recently, postmortem imaging, including postmortem computed tomography (PMCT) and postmortem magnetic resonance imaging (PMMR), has been introduced to the field of forensic pathology [8,9,10,11,12,13]. Postmortem imaging of the head region has been performed in previous studies to report on the detection of various findings in cases of trauma-related intra- or extracranial injury, including haemorrhage, fracture, oedema, and herniation [14,15,16], arteriovenous malformation-related intracerebral haemorrhage [17], cerebral infarction [11, 14], ischaemic white matter changes [18], and air embolism [19, 20]. However, the findings of postmortem imaging of brain lesions related to CO poisoning have not been reported previously.
In this article, we report a case of death caused by prolonged CO poisoning where PMCT and PMMR revealed necrotic lesions in the bilateral globi pallidi, findings that were confirmed by autopsy.
Case report
A man in his fifties was found dead in a car on the driver’s seat in the parking space of his house on a cold winter day (day 0). His common law wife was found in the back seat, and a burnt coal briquette was also found near her body, placed in the luggage storage space. Neither individual received cardiopulmonary resuscitation as their death was obvious. The man’s rectal temperature soon after discovery was 20.5 °C, while that of the woman was 9.1 °C. The ambient temperature was 11.0 °C.
The subjects had purchased coal briquettes 3 days prior to the discovery of their bodies, which was the last time they were seen alive (day − 3). Suicide was suspected based on the prevailing situation. The subjects had no significant medical history. The man had undergone a cranial CT scan for minor head trauma 6 days prior to discovery, and this scan revealed no significant findings other than calcification in the bilateral globi pallidi (Fig. 1a). PMCT of the man and the woman was performed 2 days after their discovery; PMMR of the man was performed 6 days after discovery; and autopsy of both of their bodies was performed 11 days after discovery. The cadavers were stored in a refrigerator prior to the autopsy.
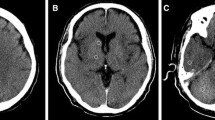
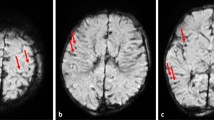
Brain imaging findings of the man. a An antemortem computed tomography (CT) scan taken 6 days before the discovery of his body revealing no lesions in the globi pallidi; calcification is observed in the region. b A postmortem computed tomography (PMCT) image obtained 2 days after the discovery of the body showing a low-density area in the bilateral globi pallidi (red arrows). c A T2-weighted image (T2WI) obtained by postmortem magnetic resonance (PMMR) imaging performed 6 days after the discovery of the body demonstrates high signal intensities in the bilateral globi pallidi (red arrows). d A diffusion-weighted image (DWI) obtained by PMMR performed 6 days after the discovery of the body shows high signal intensities in the bilateral globi pallidi (red arrows). e An apparent diffusion coefficient (ADC) map of a PMMR imaging scan obtained 6 days after the discovery of the body reveals no abnormality in the bilateral globi pallidi compared with the other regions of the brain. The ADC values decreased diffusely. f A T1-weighted image (T1WI) obtained by PMMR performed 6 days after the discovery of the body shows obscure low-signal intensity areas in the bilateral globi pallidi (red arrows). The basal ganglia have diffuse high signal intensity (yellow arrows), which is a normal finding in PMMR scans
PMCT was performed using a 64-row multidetector CT scanner (Supria Grande; Hitachi Ltd., Tokyo, Japan) with the following settings: 120-kV tube voltage, 250-mA tube current, 0.75 s per rotation, and 0.625-mm collimation.
PMMR for the man was performed using a 1.5 T scanner (Intera Achieva; Philips Healthcare, Best, the Netherlands). The total PMMR scanning time of the brain, neck, and thoracic regions was 7 h, including 1.5-h brain scanning time; this amount of imaging acquisition time was used to improve image quality. The man’s rectal temperatures measured before and after scanning were 3.5 °C and 5.5 °C, respectively. A 16-channel head/neck coil was used for brain scanning. Brain scans were performed under the following conditions: T2-weighted imaging (T2WI), fast spin echo (echo time (TE) 100 ms, repetition time (TR) 4500 ms, and 5-mm slice thickness); T1-weighted imaging (T1WI), spin echo (TE 12 ms, TR 600 ms, and 5-mm slice thickness); fluid-attenuated inversion recovery (FLAIR) (TE 140 ms, TR 11,000 ms, inversion time (TI) 1700 ms [21], and 5 mm slice thickness); and diffusion-weighted imaging (DWI) (b 1000 s/mm2, TE 75 ms, TR 3000 ms, and 5-mm slice thickness). The images were evaluated by two board-certified radiologists. Image analysis software (SYNAPSE VINCENT 3D image analysis system; Fujifilm Medical, Tokyo, Japan) was used for the analysis.
The PMCT imaging of the woman did not reveal any abnormalities in any part of her body. The woman was diagnosed with acute CO poisoning due to typical autopsy findings, such as > 70% COHb levels in blood from the heart and the cherry-pink colour of hypostasis, organs, muscles, and blood. No other significant lesions were detected in the whole body, either macroscopically or microscopically.
The PMCT imaging of the man revealed segmental consolidation in the lower lobe of the left lung, and obscure low-density areas in the bilateral globi pallidi, with relatively ambiguous contrast to the surrounding areas (Fig. 1b). These lesions were depicted with clearer contrast as high signal intensity areas on T2WI and FLAIR images (Fig. 1c). They were also detected with high signal intensity on DWI (Fig. 1d), although the apparent diffusion coefficient (ADC) values were not lower than those of the other brain tissue (Fig. 1e). On T1WI, the basal ganglia, namely, the caudate nucleus, putamen, and globus pallidus, showed diffuse high signal intensity, which is a frequent normal finding on PMMR images [9, 22]; the lesions showed comparatively low signal intensity with obscure contrast in these images (Fig. 1f). No other abnormalities were observed in the brain.
The autopsy of the man confirmed a body length of 173 cm and a body weight of 66.5 kg. The hypostasis, organs, muscles, and blood of the cadaver did not have a cherry-pink colour. No significant injury was detected. The left lung weighed 600 g, and the lower lobe of the left lung contained a hard lesion, which was histologically proven to be acute pneumonia. Microscopic pneumonia was also shown in the lower lobe of the right lung. Blackish to brownish spots (Wischnewski spots, which are frequently detected in hypothermia-related deaths [23]) were found in the gastric mucosa. Except for these findings, there were no other notable findings in any of the organs.
Macroscopically, obscure lesions of a slightly browner colour (compared to the surrounding tissue) were found in the internal segment of the bilateral globi pallidi of the brain; there was no haemorrhage, softening, or cavitation in this area (Fig. 2a, b). Histologically, demyelination of the neurons was observed (Fig. 2c, d). Coagulative necrosis was observed in some areas (Fig. 2e, f), although no inflammatory changes, including lipid-laden macrophage infiltration or haemorrhage, were found. Mineralization of blood vessel walls and the proliferation and swelling of endothelial cells were also observed. Macroscopic and microscopic evaluations did not reveal any other brain lesions.
Macroscopic and microscopic findings of the brain lesions in the man’s globus pallidus. a Coronal section of the brain at the level of the lentiform nucleus. b Image of the right lentiform nucleus at high magnification; a slightly browner lesion (blue arrow) compared with the surrounding tissue can be detected in the internal segment of the globus pallidus. The area shows no haemorrhage, softening, or cavitation. c Histological specimen showing the lesion (blue arrows); the specimen was stained using haematoxylin-eosin staining and shows areas with lower eosinophilicity without haemorrhage compared to the surrounding tissue. d Histological specimen showing the lesion (blue arrows); the specimen was stained using the Klüver-Barrera method and reveals demyelination. (e, f) Magnified image of a haematoxylin-eosin specimen showing several neuronal cells with coagulative necrosis
The COHb level of the heart blood was 1.0%. The results of the serological examinations of his heart blood and urine were as follows, with the reference ranges of living adults given in parentheses: heart blood, C-reactive protein, 45.2 mg/dL (< 0.3 mg/dL); procalcitonin, 0.94 ng/mL (< 0.05 ng/dL); blood urea nitrogen, 97.5 mg/dL (9–21 mg/dL); creatinine, 7.43 mg/dL (0.6–1.2 mg/dL); and total ketone bodies, 2181 μmol/L (< 130 μmol/L); and urine, total ketone bodies, 1506 μmol/L (negative). Alcohol and acetone levels were analysed by gas chromatography using a flame-ionization detector and revealed the following: ethanol and acetone levels in femoral blood, below the limit of detection and 0.01 mg/mL, respectively, and in urine, 0.06 mg/dL and 0.01 mg/mL, respectively. Screening for basic drugs in the heart blood and urine was performed using liquid chromatography-tandem mass spectrometry and revealed the presence of diphenhydramine and propranolol.
Discussion
In Japan, suicide using coal briquettes is common, and the situation in the present case suggests that the subjects may have committed suicide using coal briquettes. Although the typical findings associated with death from acute CO poisoning were detected in the woman’s body, no such findings could be detected in the man’s body; his COHb level was within the normal range. While the rectal temperature of the woman was close to the ambient temperature, the rectal temperature of the man soon after discovery was higher than that of the woman. This suggests that the man may have died hours or days later than the woman and may have experienced prolonged CO poisoning. Findings from autopsy of the man’s body support this possibility. Pneumonia was proven macroscopically and microscopically and may be explained as hypostatic pneumonia due to impaired consciousness caused by prolonged CO poisoning. Dehydration and starvation suggested from the examination of the blood urea nitrogen, creatinine, ketone bodies, and acetone may have also resulted from the impaired consciousness. Hypothermia, as suggested by Wischnewski spots, may have been the result of neurological sequelae. Thus, our assumptions regarding these cases are as follows: after the man and woman were exposed to CO from the coal briquette burning, 3 days before discovery and the day when they were last seen alive, the woman died immediately from acute CO poisoning. However, the man survived and prolonged CO poisoning caused him to suffer from secondary pneumonia, probable dehydration and starvation, and possible hypothermia. He eventually died approximately half a day before discovery, approximately 2 days after poisoning (Table 1). Bilateral globi pallidi lesions detected by postmortem imaging and histological examinations are also considered products of prolonged CO poisoning and help confirm the cause of death in this case.
Brain lesions resulting from CO poisoning have been reported from the autopsy results of dead subjects as well as from imaging results of subjects who were still living. Lapresle and Fardeau [2] described 22 autopsy cases, including various types of CO poisoning. Acute, prolonged, and delayed cases of CO poisoning were described in the report. One person who died 10 h after CO poisoning had no brain lesions. However, brain lesions in the bilateral globi pallidi and cerebral white matter were revealed in another person who died 24 h after CO poisoning. Brain lesions in the globus pallidus detected within 6 days of CO poisoning were characterized as coagulative necrosis with or without haemorrhage. After 6 days, the lesions were reported to infiltrate lipid-laden macrophages. The present case also showed similar coagulative necrosis, and the lack of inflammation or haemorrhage may suggest that the necrosis in this case was in a relatively early stage.
Several mechanisms have been proposed to explain these lesions. One of the most widely accepted mechanisms to explain CO poisoning-related brain lesions is the process of hypoxia and ischaemia [1, 3, 7, 24,25,26]. The affinity of the binding of CO to haemoglobin is approximately 200 times greater than that of oxygen [1, 3, 7, 24,25,26,27], and CO binding competes with oxygen binding. In addition, oxyhaemoglobin binding CO has difficulty in releasing oxygen in the peripheral tissues because the oxyhaemoglobin dissociation curve shifts to the left [1, 3, 7, 24,25,26]. In addition, hypoxia in cardiac tissue causes circulation impairment. It has also been suggested that cardiac dysfunction can be worsened by the binding of CO to myoglobin in cardiac muscle [1, 24, 28]. All of these hypoxic-ischaemic effects result in the exacerbation of tissue damage, especially in low-perfusion areas such as the globus pallidus [3, 7, 25, 29]. It is also said that CO can selectively affect the globus pallidus, as this region can bind easily to CO because of its iron-rich component [30]. Numerous other hypotheses, including the direct inhibition of cytochrome c oxidase in mitochondria [1, 2, 24, 31], or neutrophil activation from the direct effects on platelets [32], may be associated with specific lesions to which CO can easily bind.
Although lesions in the bilateral globi pallidi are thought to be characteristic to CO poisoning [2], they are not specific [1, 33]. For example, impairments of the bilateral globi pallidi resembling CO poisoning-related lesions have been reported in CT or MRI in living patients after other hypoxic-ischaemic situations such as drowning [34], hanging [35], cyanide toxicity [36], methanol toxicity [37], illicit drug toxicity [38,39,40,41,42,43], antidepressant overdose [44, 45], altitude sickness [46], and other diseases [47,48,49,50]. Similar lesions may also be caused by genetic metabolic disorders such as methylmalonic acidaemia [51]. Macroscopic and histological findings of these lesions have rarely been reported, and the difference with those of CO poisoning-related lesions remains unknown. Due to a lack of specificity, the interpretation of the findings of this study requires care. However, because most bilateral globi pallidi lesions were reported in CO poisoning cases, detection of these lesions in the context of evidence from detailed scene investigations, other autopsy findings, and toxicological examinations may be useful in diagnosing prolonged CO poisoning. In the present case, we were able to exclude some differential diagnoses of bilateral globi pallidi lesions with autopsy and laboratory examination, including drug toxicity, although some toxic agents could not be detected because of metabolism or excretion as the carbon monoxide level was within the normal range in this case. We excluded several situation-specific causes such as drowning, hanging, and altitude sickness. Genetic metabolic disorders were excluded based on the man’s medical history and the lack of significant findings of the premortem CT scan taken 6 days before his body was discovered. It is unlikely that the lesions developed for the first time at his age. Although, to the best of our knowledge, such a phenomenon has never been reported, there is a remote possibility that pneumonia, dehydration and starvation, or hypothermia caused the lesions.
It has been reported that lesions in the bilateral globi pallidi of living patients with CO poisoning can be detected using CT and MRI. In the subacute phase, CT scans can show lesions with ambiguous low densities [6, 7], while MRI scans provide clearer high signal intensity areas on T2WI, FLAIR, and DWI sequences, accompanied by ADC reduction [1, 3,4,5]. It has been reported that MRI scans obtained within hours of CO exposure may reveal such lesions in living patients [4, 5]. Although MRI detection of these lesions is well described in the literature, to the best of our knowledge, the use of postmortem imaging to detect such lesions has never been reported. In the present case, we were able to detect brain lesions using PMCT and PMMR imaging. In particular, the T2WI and FLAIR sequences of PMMR scans revealed high signal intensity areas similar to those observed in living subjects. On the other hand, the lesions presented as high signal intensity areas on DWI, although ADC reduction was not observed. There may be two reasons for this observation: first, it was reported that postmortem changes may lower whole-brain ADC values [52]; second, the ADC values of the lesions may have been at normal levels prior to death, similar to those observed in the lesions of some living patients [4]. Although lesions in this case could be observed macroscopically, microscopically, on PMCT, and on PMMR imaging, better contrast was obtained with the PMMR imaging, especially on the T2WI and FLAIR sequences. Although it is preferable to study a higher number of cases, this report raises the possibility that postmortem imaging, especially PMMR imaging, can be useful in detecting these lesions, which sometimes have macroscopically ambiguous contrast.
Conclusions
We present a case of death caused by suspected prolonged CO poisoning, with bilateral globi pallidi lesions. This is the first study to report that PMCT and PMMR imaging can be used to detect CO poisoning-related brain lesions. Postmortem imaging, especially PMMR, should be considered useful for detecting CO poisoning-related brain lesions.
Data availability
Not applicable.
References
Weaver LK (2009) Clinical practice. Carbon monoxide poisoning. N Engl J Med 360:1217–1225. https://doi.org/10.1056/NEJMcp0808891
Lapresle J, Fardeau M (1967) The central nervous system and carbon monoxide poisoning. II. Anatomical study of brain lesions following intoxication with carbon monoxide (22 cases). Prog Brain Res 24:31–74. https://doi.org/10.1016/s0079-6123(08)60181-8
Beppu T (2014) The role of MR imaging in assessment of brain damage from carbon monoxide poisoning: a review of the literature. AJNR Am J Neuroradiol 35:625–631. https://doi.org/10.3174/ajnr.A3489
Jeon SB, Sohn CH, Seo DW, Oh BJ, Lim KS, Kang DW, Kim WY (2018) Acute brain lesions on magnetic resonance imaging and delayed neurological sequelae in carbon monoxide poisoning. JAMA Neurol 75:436–443. https://doi.org/10.1001/jamaneurol.2017.4618
Moon JM, Chun BJ, Baek BH, Hong YJ (2018) Initial diffusion-weighted MRI and long-term neurologic outcomes in charcoal-burning carbon monoxide poisoning. Clin Toxicol (Phila) 56:161–169. https://doi.org/10.1080/15563650.2017.1352098
Choi IS, Kim SK, Choi YC, Lee SS, Lee MS (1993) Evaluation of outcome after acute carbon monoxide poisoning by brain CT. J Korean Med Sci 8:78–83. https://doi.org/10.3346/jkms.1993.8.1.78
Lo CP, Chen SY, Lee KW, Chen WL, Chen CY, Hsueh CJ, Huang GS (2007) Brain injury after acute carbon monoxide poisoning: early and late complications. Am J Roentgenol 189:W205–W211. https://doi.org/10.2214/AJR.07.2425
Blokker BM, Wagensveld IM, Weustink AC, Oosterhuis JW, Hunink MG (2016) Non-invasive or minimally invasive autopsy compared to conventional autopsy of suspected natural deaths in adults: a systematic review. Eur Radiol 26:1159–1179. https://doi.org/10.1007/s00330-015-3908-8
Ruder TD, Thali MJ, Hatch GM (2014) Essentials of forensic post-mortem MR imaging in adults. Br J Radiol 87:20130567. https://doi.org/10.1259/bjr.20130567
Eriksson A, Gustafsson T, Höistad M, Hultcrantz M, Jacobson S, Mejare I, Persson A (2017) Diagnostic accuracy of postmortem imaging vs autopsy-a systematic review. Eur J Radiol 89:249–269. https://doi.org/10.1016/j.ejrad.2016.08.003
Roberts IS, Benamore RE, Benbow EW, Lee SH, Harris JN, Jackson A, Mallett S, Patankar T, Peebles C, Roobottom C, Traill ZC (2012) Post-mortem imaging as an alternative to autopsy in the diagnosis of adult deaths: a validation study. Lancet 379:136–142. https://doi.org/10.1016/S0140-6736(11)61483-9
Leth PM (2009) Computerized tomography used as a routine procedure at postmortem investigations. Am J Forensic Med Pathol 30:219–222. https://doi.org/10.1097/PAF.0b013e318187e0af
Bolliger SA, Thali MJ, Ross S, Buck U, Naether S, Vock P (2008) Virtual autopsy using imaging: bridging radiologic and forensic sciences. A review of the Virtopsy and similar projects. Eur Radiol 18:273–282. https://doi.org/10.1007/s00330-007-0737-4
Yen K, Lövblad KO, Scheurer E, Ozdoba C, Thali MJ, Aghayev E, Jackowski C, Anon J, Frickey N, Zwygart K, Weis J, Dirnhofer R (2007) Post-mortem forensic neuroimaging: correlation of MSCT and MRI findings with autopsy results. Forensic Sci Int 173:21–35. https://doi.org/10.1016/j.forsciint.2007.01.027
Añon J, Remonda L, Spreng A, Scheurer E, Schroth G, Boesch C, Thali M, Dirnhofer R, Yen K (2008) Traumatic extra-axial hemorrhage: correlation of postmortem MSCT, MRI, and forensic-pathological findings. J Magn Reson Imaging 28:823–836. https://doi.org/10.1002/jmri.21495
Aghayev E, Yen K, Sonnenschein M, Ozdoba C, Thali M, Jackowski C, Dirnhofer R (2004) Virtopsy post-mortem multi-slice computed tomography (MSCT) and magnetic resonance imaging (MRI) demonstrating descending tonsillar herniation: comparison to clinical studies. Neuroradiology 46:559–564
Franckenberg S, Schulze C, Bolliger SA, Gascho D, Thali MJ, Flach PM (2015) Postmortem angiography in computed tomography and magnetic resonance imaging in a case of fatal hemorrhage due to an arterio-venous malformation in the brain. Leg Med (Tokyo) 17:180–183. https://doi.org/10.1016/j.legalmed.2014.11.006
Weustink AC, Hunink MG, van Dijke CF, Renken NS, Krestin GP, Oosterhuis JW (2009) Minimally invasive autopsy: an alternative to conventional autopsy? Radiology 250:897–904. https://doi.org/10.1148/radiol.2503080421
Oliver J, Lyons TJ, Harle R (1999) The role of computed tomography in the diagnosis of arterial gas embolism in fatal diving accidents in Tasmania. Australas Radiol 43:37–40. https://doi.org/10.1046/j.1440-1673.1999.00615.x
Iwama T, Andoh H, Murase S, Miwa Y, Ohkuma A (1994) Diffuse cerebral air embolism following trauma: striking postmortem CT findings. Neuroradiology 36:33–34. https://doi.org/10.1007/BF00599191
Abe K, Kobayashi T, Shiotani S, Saito H, Kaga K, Tashiro K, Someya S, Hayakawa H, Homma K (2015) Optimization of inversion time for postmortem fluid-attenuated inversion recovery (FLAIR) MR imaging at 1.5T: temperature-based suppression of cerebrospinal fluid. Magn Reson Med Sci 14:251–255. https://doi.org/10.2463/mrms.2014-0086
Wagensveld IM, Blokker BM, Wielopolski PA, Renken NS, Krestin GP, Hunink MG, Oosterhuis JW, Weustink AC (2017) Total-body CT and MR features of postmortem change in in-hospital deaths. PLoS One 12:e0185115. https://doi.org/10.1371/journal.pone.0185115
Wischnewsky S (1895) Ein neues Kennzeichen des Todes durch Erfrieren. Bote Gerichtl Med 3:12–20
Prockop LD, Chichkova RI (2007) Carbon monoxide intoxication: an updated review. J Neurol Sci 262:122–130. https://doi.org/10.1016/j.jns.2007.06.037
Bleecker ML (2015) Carbon monoxide intoxication. Handb Clin Neurol 131:191–203. https://doi.org/10.1016/B978-0-444-62627-1.00024-X
Ernst A, Zibrak JD (1998) Carbon monoxide poisoning. N Engl J Med 339:1603–1608. https://doi.org/10.1056/NEJM199811263392206
Rodkey FL, O'Neal JD, Collison HA (1969) Oxygen and carbon monoxide equilibria of human adult hemoglobin at atmospheric and elevated pressure. Blood 33:57–65
Wittenberg BA, Wittenberg JB (1987) Myoglobin-mediated oxygen delivery to mitochondria of isolated cardiac myocytes. Proc Natl Acad Sci U S A 84:7503–7507. https://doi.org/10.1073/pnas.84.21.7503
Okeda R, Funata N, Takano T, Miyazaki Y, Higashino F, Yokoyama K, Manabe M (1981) The pathogenesis of carbon monoxide encephalopathy in the acute phase--physiological and morphological correlation. Acta Neuropathol 54:1–10. https://doi.org/10.1007/BF00691327
Kawanami T, Kato T, Kurita K, Sasaki H (1998) The pallidoreticular pattern of brain damage on MRI in a patient with carbon monoxide poisoning. J Neurol Neurosurg Psychiatry 64:282. https://doi.org/10.1136/jnnp.64.2.282
Alonso JR, Cardellach F, López S, Casademont J, Miró O (2003) Carbon monoxide specifically inhibits cytochrome c oxidase of human mitochondrial respiratory chain. Pharmacol Toxicol 93:142–146. https://doi.org/10.1034/j.1600-0773.2003.930306.x
Thom SR, Bhopale VM, Han ST, Clark JM, Hardy KR (2006) Intravascular neutrophil activation due to carbon monoxide poisoning. Am J Respir Crit Care Med 174:1239–1248. https://doi.org/10.1164/rccm.200604-557OC
Yarid NA, Harruff RC (2015) Globus pallidus necrosis unrelated to carbon monoxide poisoning: retrospective analysis of 27 cases of basal ganglia necrosis. J Forensic Sci 60:1484–1487. https://doi.org/10.1111/1556-4029.12838
Murray RR, Kapila A, Blanco E, Kagan-Hallet KS (1984) Cerebral computed tomography in drowning victims. AJNR Am J Neuroradiol 5:177–179
Bianco F, Floris R (1987) Computed tomography abnormalities in hanging. Neuroradiology 29:297–298. https://doi.org/10.1007/BF00451772
Mohan A, Lee T, Sachdev P (2014) Surviving acute cyanide poisoning: a longitudinal neuropsychological investigation with interval MRI. BMJ Case Rep 19:2014. https://doi.org/10.1136/bcr-2013-203025
Karayel F, Turan AA, Sav A, Pakis I, Akyildiz EU, Ersoy G (2010) Methanol intoxication: pathological changes of central nervous system (17 cases). Am J Forensic Med Pathol 31:34–36. https://doi.org/10.1097/PAF.0b013e3181c160d9
Renard D, Brunel H, Gaillard N (2009) Bilateral haemorrhagic infarction of the globus pallidus after cocaine and alcohol intoxication. Acta Neurol Belg 109:159–161
Daras MD, Orrego JJ, Akfirat GL, Samkoff LM, Koppel BS (2001) Bilateral symmetrical basal ganglia infarction after intravenous use of cocaine and heroin. Clin Imaging 25:12–14. https://doi.org/10.1016/s0899-7071(00)00232-1
De Smet K, De Maeseneer M, Talebian YA, Stadnik T, De Mey J (2011) Bilateral globus pallidus infarcts in ecstasy use. JBR-BTR 94:93. https://doi.org/10.5334/jbr-btr.512
De Roock S, Hantson P, Laterre PF, Duprez T (2007) Extensive pallidal and white matter injury following cocaine overdose. Intensive Care Med 33:2030–2031. https://doi.org/10.1007/s00134-007-0773-1
Miller JM, Vorel SR, Tranguch AJ, Kenny ET, Mazzoni P, van Gorp WG, Kleber HD (2006) Anhedonia after a selective bilateral lesion of the globus pallidus. Am J Psychiatry 163:786–788. https://doi.org/10.1176/ajp.2006.163.5.786
Kim DW, Im HJ, Oh J (2013) Selective injury of the globus pallidus and hippocampus in methamphetamine-induced encephalopathy. Clin Neuroradiol 23:51–53. https://doi.org/10.1007/s00062-011-0115-0
Lin YH, Huang E, Chien YL (2012) Bilateral pallidal necrosis and cardiac toxicity in a patient with venlafaxine and bupropion overdose. Am J Psychiatry 169:993–994. https://doi.org/10.1176/appi.ajp.2012.11111708
Szolics M, Chaudhry M, Ljubisavljevic M, Corr P, Samir HA, van Gorkom KN (2012) Neuroimaging findings in a case of fluoxetine overdose. J Neuroradiol 39:254–257. https://doi.org/10.1016/j.neurad.2011.10.006
Jeong JH, Kwon JC, Chin J, Yoon SJ, Na DL (2002) Globus pallidus lesions associated with high mountain climbing. J Korean Med Sci 17:861–863. https://doi.org/10.3346/jkms.2002.17.6.861
Rabitsch W, Brugger SA, Pirker W, Baumgartner C, Reiter E, Keil F, Prayer D, Hoecker P, Lechner K, Kalhs P, Greinix HT (1997) Symmetrical necrosis of globus pallidus with severe gait disturbance in a patient with myelodysplastic syndrome given allogeneic marrow transplantation. Ann Hematol 75:235–237. https://doi.org/10.1007/s002770050349
Lou M, Jing CH, Selim MH, Caplan LR, Ding MP (2009) Delayed substantia nigra damage and leukoencephalopathy after hypoxic-ischemic injury. J Neurol Sci 277:147–149. https://doi.org/10.1016/j.jns.2008.09.032
Abou Hassan OK, Karnib M, El-Khoury R, Nemer G, Ahdab-Barmada M, BouKhalil P (2016) Linezolid toxicity and mitochondrial susceptibility: a novel neurological complication in a Lebanese patient. Front Pharmacol 7:325. https://doi.org/10.3389/fphar.2016.00325
Finelli PF (1984) Bilateral hemorrhagic infarction of the pallidum. J Comput Assist Tomogr 8:125–127. https://doi.org/10.1097/00004728-198402000-00025
Michel SJ, Given CA 2nd, Robertson WC Jr (2004) Imaging of the brain, including diffusion-weighted imaging in methylmalonic acidemia. Pediatr Radiol 34:580–582. https://doi.org/10.1007/s00247-004-1155-2
Schmidt TM, Fischer R, Acar S, Lorenzen M, Heinemann A, Wedegärtner U, Adam G, Yamamura J (2012) DWI of the brain: postmortal DWI of the brain in comparison with in vivo data. Forensic Sci Int 220:180–183. https://doi.org/10.1016/j.forsciint.2012.02.022
Acknowledgments
The authors would like to thank Hiroko Abe, Yoshikazu Yamagishi, Katsura Otsuka, Kazuhiro Kobayashi, Yuriko Odo, and Miyuki Miura for their technical support. The authors also would like to thank Editage (www.editage.com) for English language editing.
Funding
This work was supported by JSPS KAKENHI Grant Numbers JP16H06242 and JP26870102.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception of this case report. Data collection and analysis were performed by Shigeki Tsuneya, Yohsuke Makino, Masatoshi Kojima, Maiko Yoshida, Takashi Kishimoto, Hiroki Mukai, and Shinya Hattori. The first draft of the manuscript was written by Shigeki Tsuneya, and all authors commented on previous versions of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval/consent to participate/consent for publication
The ethics committee of the University of Tokyo approved this study and permitted waiver of consent from next of kin.
Code availability
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tsuneya, S., Makino, Y., Chiba, F. et al. Postmortem magnetic resonance imaging revealed bilateral globi pallidi lesions in a death associated with prolonged carbon monoxide poisoning: a case report. Int J Legal Med 135, 921–928 (2021). https://doi.org/10.1007/s00414-021-02506-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-021-02506-1