Abstract
Age estimation based on aspartic acid racemization (AAR) in dentine is one of the most precise methods in adult age. Caries induces protein degradation and may have an impact on the kinetics of AAR in dentine. We systematically examined standardized prepared dentine samples from caries-affected teeth to clarify the question, if caries-affected teeth should not be used for age estimation based on AAR at all, or if the analysis of dentine samples from such teeth may be useful after removal of the caries-affected tissue according to clinical standards. Our results suggest that caries may lead to an extensive protein degradation even in macroscopically healthy-appearing dentine samples from caries-affected teeth and may significantly affect the precision of age estimation. To ensure the quality of age estimation based on AAR in forensic practice, we recommend using dentine samples from healthy teeth. If only caries-affected teeth are available, dentine samples from at least two teeth from the same individual should be analyzed as it seems unlikely that caries-induced protein degradation occurred with identical kinetics in two different teeth. In any case, results of the analysis of caries-affected teeth must be interpreted with caution.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Estimation of age is an important task in forensic practice, regarding identification of unidentified bodies as well as age estimation of living persons of unknown age. One of the most precise methods (in adult age) is based on aspartic acid racemization (AAR) [1,2,3].
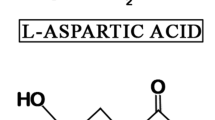
The molecular basis of AAR has extensively been described [4,5,6]. In permanent and long-living proteins, an accumulation of D-aspartic acid residues may occur with increasing age. If the relationship of D-aspartic acid content and age is close enough, it can be used for age estimation [7, 8]. An age-dependent accumulation of D-aspartic acid has been observed. e. g., in dentine [9, 10], enamel [11,12,13], tooth cementum [14], erythrocytes [15,16,17], bone [18, 19], intervertebral discs [20, 21], lens [22,23,24], sclera [25], elastic cartilage from epiglottis [26], and rib [27], as well as in purified proteins, e. g., osteocalcin from bone [28] and elastin from lungs [29], artery walls [30], human skin [31], and ligamenta flava [32].
In forensic practice, dentine is the tissue of choice for age estimation based on AAR, as it is a bradytrophic tissue exhibiting a very close relationship of D-aspartic acid content and age [2, 33]. For reasons of quality assurance, the analysis of healthy intact teeth is strongly recommended [34]. However, completely unaffected teeth are often unavailable in forensic case work. The question arises if caries-affected teeth should not be used for age estimation based on AAR at all, or if the analysis of dentine samples from such teeth may be useful after removal of the caries-affected tissue.
Experimental evidence for a relevant influence of caries on AAR already comes from data presented by Tiemeyer [13] and Griffin et al. [12]. Tiemeyer described elevated AAR values in enamel from teeth with carious lesions, even if the samples had been taken from caries-unaffected areas. Griffin et al. [12] analyzed acid-extracted fractions of enamel; samples from carious teeth exhibited a higher deviation of estimated from real ages than healthy teeth. Tiemeyer [13] as well as Griffin et al. [12] recommended further investigation of the influence of caries on the precision of the method.
The precise implications of the effects of caries on AAR in dentine have not yet been examined in detail. Therefore, we examined the influence of caries-induced protein degradation on AAR by analysis of standardized prepared dentine samples from caries-affected teeth. Under consideration of the results of these analyses we discuss, if caries-affected teeth are at least suitable after removal of the caries-affected dentinal tissue according to clinical standards, or if such teeth are not suitable for forensic age estimation based on AAR in dentine at all.
Materials and methods
AAR in standardized prepared dentine samples from caries-affected teeth
To investigate the relationship between the severity of a carious lesion (as detectable by morphological methods according to clinical standards) and the risk of false results of age estimation, AAR was determined in standardized prepared and categorized samples.
Ninety-nine standardized prepared root dentine samples (cube) from 25 wisdom teeth with carious lesions (23 individuals, with known ages between 21 and 79 years) were analyzed. To check the influence of the preparation method, three sound wisdom teeth (from 3 individuals, with known ages between 17 and 35 years) were analyzed in addition.
The samples were taken from vital teeth free of fillings and root fillings. All teeth were extracted by dentists due to carious or periodontal diseases, or for orthodontic and other medical reasons. Patients consented to the analysis of their extracted teeth for scientific purposes. Each extracted carious tooth was X-rayed to assess the severity of the carious lesions. For classification, the International Caries Detection Assessment System (ICDAS) was used (www.icdas.org). Only teeth with a score of 5 and 6 were selected for further analysis.
The samples were processed and analyzed as in the procedure described by Ritz-Timme [35] as follows:
Preparation of root dentine samples
All teeth were cleaned with water and stored dry at −20 °C. Root dentine samples were prepared using a dental drill under constant cooling. The roots were separated from the crown at the enamel-dentine junction. The lower third of the root was removed. The remaining root dentine was freed from cementum and pulp tissue. The precision of the preparation was checked using an UV lightener at a wavelength of 366 nm; at this wavelength, dentine is fluorescent, while cementum is not.
Root dentine samples were separated into four “cubes” of about 3 mm height each (see Fig. 1). The prepared cube samples were classified with regard to the extent of the carious lesions visible:
-
category 0: no carious lesions visible in the concerned cube,
-
category a: carious lesions cover 30% or more of the cube,
-
category b: carious lesions cover less than 30% of the cube.
Washing procedure
The prepared cubes were rinsed in distilled water and washed in 15% sodium chloride solution for 1 h, in ethanol-diethylether (3:1) for 15 min, and in 2% sodium lauryl sulfate (SDS) for 1 hour. The samples were then lyophilized and stored at −20 °C until further processing.
Pulverization of dentine samples
Pulverization of dentine was performed by a hydraulic press (P/O/Weber, Remshalden) at 20 kN. Four milligrams of the pulverized dentine was used for the following steps.
Hydrolysis, derivatization, and chromatographic analysis
After pulverization of dentine, all samples were hydrolyzed in 1 ml 6 N hydrochloric acid for 6 h at 100 °C and dried in a vacuum desiccator.
For derivatization, 1 ml isopropanol and 1 μl sulfuric acid were added to each sample, and the samples were heated at 110 °C for 1 h. Isopropanol was removed by a nitrogen stream. After adding 1 ml 4 N ammonia solution and 1 ml dichlormethane, the samples were centrifuged and the resulting two phases were separated and dried again. One milligram dichlormethane and 50 μl trifluoroacetic acid (TFAA) were added; the samples were heated for 15 min at 60 °C and dried using a nitrogen stream.
Separation and quantification of D- and L-aspartic acid were performed on a chiral capillary column in a gas chromatograph (GC: Shimadzu GC-2014, column: Chirasil-L-Val, Varian). A defined sample of aspartic acid with known D/L ratio (Merck, Darmstadt) was used as a standard for quality assurance. Each sample was analyzed at least twice. D/L ratio was calculated by using the arithmetic mean. The extent of AAR was calculated as ln ((1 + D/L)/(1 − D/L)) [11].
Age estimation
D/L values were used to estimate the individuals’ ages using the regression equation established by Ritz-Timme et al. [35] (ln ((1 + D/L)/(1 − D/L)) = 0.00165 t + 0.00716). Deviations of estimated ages from real ages were determined.
Results
AAR in samples from sound wisdom teeth
Age estimation based on AAR in the prepared cubes from the sound teeth resulted in deviations between estimated and real ages between − 1.87 and 3.69 years.
AAR in samples from caries-affected teeth
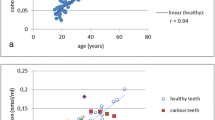
Figures 2, 3, and 4 depict the deviations of estimated from true ages in the prepared cubes from caries-affected teeth under consideration of the observed degree of caries affection.
In all categories (of visible carious lesions), precise age estimates as well as remarkably large deviations of estimated from real ages were observed:
-
Deviations of less than +/− 3 years were observed in 33 (out of 99) analyzed cubes from caries-affected teeth.
-
62 (out of 99) cubes exhibited deviations of less than +/− 6 years.
-
The analyses of 15 (out of 99) cubes resulted in deviations of more than 10 years.
-
Overall, deviations from − 12.67 to 20.39 years were observed.
-
In c. 92% of the cases with deviations of more than +/− 6 years, false high AAR values were observed.
There was no relationship between the severity of the carious lesions in the prepared cubes and the precision of age estimation; in each defined category of affection, very precise as well as very imprecise results of age estimation were observed:
-
Samples in category “0” (no caries lesions visible in the concerned cube) exhibited deviations of 0.02 years at best and 20.39 years at worst (see Fig. 2).
-
Samples of category “a” (carious lesion covered 30% or more of the cube) exhibited deviations of estimated from true age of 0.02 years at best and 14.33 years at worst (see Fig. 3).
-
In samples of category “b” (carious lesion covered less than 30%), deviations between − 1.10 years at best and 15.08 years at worst were observed (see Fig. 4).
Discussion
Tiemeyer [13] and Griffin et al. [12] already concluded from their data on AAR in enamel of caries-affected teeth that caries may have an impact on the precision of age estimation based on AAR. However, the precise implications of the effects of caries on AAR in dentine remained unclear.
The results of the analysis of dentine samples from caries-affected teeth (Figs. 2, 3, and 4) confirm the assumption of a relevant impact of caries on the quality of age estimation based on AAR. Deviations between estimated and real ages of up to 20.39 years were observed. Deviations in these dimensions have not been observed in age estimation based on the analysis of healthy dentine in large sample series by our group using the method employed here [10, 34], and also other experienced groups did not report such high deviations (e.g., Ohtani et al. [36, 37]).
Most deviations of estimated from real ages were caused by remarkably high AAR values resulting in falsely high age estimates (see Figs. 2, 3, and 4). This is in line with the assumption that caries-induced protein degradation generates small fragments with lower steric hints, promoting a faster accumulation of D-aspartic acid [4, 5].
However, there were also samples with relatively low AAR values resulting in falsely low age estimates. In these cases, dentine samples may already have lost small protein fragments with high AAR rates due to further degradation and elimination.
The observed deviations of estimated from real ages in samples from caries-affected teeth prove that caries may indeed influence the precision of age estimation by AAR in dentine considerably. The question arises, whether precise age estimation based on AAR is even possible in caries-affected teeth, if macroscopically healthy dentine is prepared from these affected teeth according to clinical standards. According to our results, this is not possible. Cubes labeled as “category 0” (no caries visible) exhibited very precise as well as very imprecise results of age estimation (Fig. 2). Our findings indicate that caries may lead to extensive changes on a molecular level (protein degradation) even in regions that appear “healthy” in the morphological examination according to clinical standards. On the other hand, cubes with visible carious lesions also presented very precise as well as very imprecise results of age estimation. There was no relationship between the severity of a carious lesion (as detectable by morphological methods) and the risk of false results of age estimation based on AAR.
Considering these results, the analysis of caries-affected teeth should be avoided in forensic practice. Nevertheless, in some cases, only carious teeth are available. In these cases, at least two teeth from the same individual should be analyzed. Since carious lesions are the result of complex biological processes, it seems highly unlikely that caries produces the same extent of protein degradation in two affected teeth from the same individual. If the analysis of two carious teeth from the same individual yields roughly the same results, we would consider these results as being plausible, although they should still be interpreted with caution.
References
Meissner C, Ritz-Timme S (2010) Molecular pathology and age estimation. Forensic Sci Int 203:34–43
Ritz-Timme S, Cattaneo C, Collins MJ, Waite ER, Schütz HW, Kaatsch HJ, Borrman HIM (2000) Age estimation: the state of the art in relation to the specific demands of forensic practise. Int J Legal Med 113:129–136
Ohtani S (1995) Estimation of age from the teeth of unidentified corpses using the amino acid racemization with reference to actual cases. Am J Forensic Med Pathol 16:238–242
Collins MJ, Waite ER, van Duin ACT (1999) Predicting protein decomposition: the case of aspartic-acid racemization kinetics. Phil Trans R Soc Lond B 354:51–64
Robinson NE, Robinson AB (2004) Molecular clocks. Deamidation of asparaginyl and glutaminyl residues in peptides and proteins. Althouse Press, Cave Junction
Stephenson RC, Clarke S (1989) Succinimide formation from aspartyl and asparaginyl peptides as a model for the spontaneous degradation of proteins. J Biol Chem 11:6164–6170
Truscott RJW (2011) Macromolecular deterioration as the ultimate constraint on human lifespan. Ageing Res Rev 10:397–403
Truscott RJW, Schey KL, Friedrich MG (2016) Old proteins in man: a field in its infancy. Trends Biochem Sci 41(8):654–664
Helfman PM, Bada JL (1976) Aspartic acid racemization in dentine as a measure of ageing. Nature 262:279–281
Ritz S, Schütz HW, Peper C (1993) Postmortem estimation of age at death based on aspartic acid racemization in dentin: its applicability for root dentin. Int J Legal Med 105:289–293
Helfman PM, Bada JL (1975) Aspartic acid racemization in tooth enamel from living humans. Proc Nat Acad Sci USA 72:2891–2894
Griffin RC, Moody H, Penkman KEH, Collins MJ (2008) The application of amino acid racemization in the acid soluble fraction of enamel to the estimation of the age of human teeth. For Sci Int 175(1):11–16
Tiemeyer H (2002) Razemisierung von Asparaginsäure in Schmelzproteinen: Forensische Nutzbarkeit zur biochemischen Lebensaltersschatzung und grundlagenwissenschaftliche Aspekte. Dissertation, University of Kiel
Ohtani S, Sugimoto H, Sugeno H, Yamamoto S, Yamamoto K (1995) Racemization of asparagin acid in human cementum with age. Archs oral Biol 40(2):91–94
Barber JR, Clarke S (1983) Membrane protein carboxyl methylation increases with human erythrocyte age. J Biol Chem 258:1189–1196
Brunauer L, Clarke S (1986) Age-dependent accumulation of protein residues which can be Hydolized to D-aspartic acid in human erythrocytes. J Biol Chem 261:12538–12543
Lowenson J, Clarke S (1988) Does the chemical instability of aspartyl and asparaginyl residues in proteins contribute to erythrocyte aging? Blood Cells 14:103–117
Pfeiffer H, Mörnstad H, Teivens A (1995) Estimation of chronologic age using the aspartic acid racemization method. II. On human cortical bone. Int J Legal Med 108:24–26
Ritz S, Turzynski A, Schütz HW (1994) Estimation of age at death based on aspartic acid racemization in noncollagenous bone proteins. Forensic Sci Int 69:149–159
Maroudas A, Stockwell RA, Nachemson A, Urban J (1975) Factors involved in nutrition of the human lumbar intervertebral disc: cellularity and diffusion of glucose in vitro. J Anat 120:113–130
Ritz S, Schütz HW (1993) Aspartic acid racemization in intervertebral discs as an aid to postmortem estimation of age at death. J Forensic Sci 38:633–640
Garner WH, Spector A (1978) Racemization in human lens: evidence of rapid insolubilization of specific polypeptides in cataract formation. Proc Natl Acad Sci U S A 75:3618–3620
Helfman PM, Bada JL (1977) Aspartic acid racemization in the human lens during ageing and in cataract formation. Nature 268:71–73
Masters PM, Bada JL, Zigler JS Jr (1978) Aspartic acid racemization in heavy molecular weight crystallins and water-insoluble protein from normal human lenses and cataracts. Proc Natl Acad Sci U S A 75:1204–1208
Klumb K, Matzenauer C, Reckert A, Lehmann K, Ritz-Timme S (2016) Age estimation based on aspartic acid racemization in human sclera. Int J Legal Med 130:207–211
Matzenauer C, Reckert A, Ritz-Timme S (2014) Estimation of age at death based on aspartic acid racemization in elastic cartilage of the epiglottis. Int J Legal Med 128:995–1000
Pfeiffer H, Mörnstad H, Teivens A (1995) Estimation of chronologic age using the aspartic acid racemization method. I. On human rib cartilage. Int J Legal Med 108:19–23
Ritz S, Turzynski A, Schütz HW, Hollmann A, Rochholz G (1996) Identification of osteocalcin as a permanent aging constituent of the bone matrix: basis for an accurate age at death determination. For Sci Int 770:13–26
Shapiro SD, Endicott SK, Province MA, Pierce JA, Campbell EJ (1991) Marked longevity of human lung parenchymal elastic fibers deduced from prevalence of D-aspartate and nuclear weapons-related radiocarbon. J Clin Invest 87:1828–1834
Powell JT, Vine N, Crossman M (1992) On the accumulation of D-aspartate in elastin and other proteins of the ageing aorta. Atherosclerosis 97:201–208
Ritz-Timme S, Laumeier I, Collins MJ (2003) Aspartic acid racemization: evidence for marked longevity of elastin in human skin. Brit J Derm 149:951–959
Ritz-Timme S, Laumeier I, Collins MJ (2003) Age estimation on aspartic acid racemization in elastin from the yellow ligaments. Int J Legal Med 117:96–101
Ohtani S, Yamamoto T (2005) Strategy for the estimation of chronological age using the aspartic acid racemization method with special reference to coefficient of correlation between D/L ratios and ages. J Forensic Sci 50:1–7
Ritz-Timme S, Rochholz G, Schütz HW, Collins MJ, Waite ER, Cattaneo C, Kaatsch HJ (2000) Quality assurance in age estimation based on aspartic acid racemisation. Int J Legal Med 114:83–86
Ritz-Timme S (1999) Lebensaltersbestimmung aufgrund des Razemisierungs-grades von Asparaginsäure. Grundlagen, Methodik, Möglichkeiten, Grenzen, Anwendungsbereiche. In: Berg S, Brinkmann B (eds) Arbeitsmethoden der medizinischen und naturwissenschaftlichen Kriminalistik, 23rd edn. Lübeck, Schmidt-Römhild, pp 1–84
Ohtani S (1995) Estimation of age from dentin by using the racemization reaction of aspartic acid. Am J Forensic Med Pathol 16:158–161
Ohtani S, Yamamoto K (1991) Age estimation using the racemization of amino acid in human dentin. J Forensic Sci 36:792–800
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Sirin, N., Matzenauer, C., Reckert, A. et al. Age estimation based on aspartic acid racemization in dentine: what about caries-affected teeth?. Int J Legal Med 132, 623–628 (2018). https://doi.org/10.1007/s00414-017-1667-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-017-1667-6