Abstract
Background and Objective
We conducted a systematic review and meta-analysis to compare the accuracy of the interferon-gamma release assays (IGRAs) and the tuberculin skin test (TST) for the diagnosis of Mycobacterium tuberculosis infection.
Methods
We systematically searched PubMed, Embase, Cochrane library, and Web of Science databases for relevant published studies in recent decades and calculated pooled estimated sensitivities, specificities, DOR, and SROC curve of the QFT-IT, T-SPOT and TST. Random-effects models were used to assess estimates from studies with significant heterogeneity. Moreover, area under the curve was used to evaluate the accuracy of the tests.
Results
Overall, 9 studies for QFT-IT, 12 studies for T-SPOT, and 16 studies for TST involving 3586 participants were included in this analysis. We found that sensitivities of the QFT-IT, T-SPOT, and TST were respectively 0.842 (95 % CI 0.811–0.870), 0.840 (95 % CI 0.814–0.864), and 0.665 (CI 0.635–0.693); specificities were respectively 0.745 (95 % CI 0.715–0.775), 0.658 (95 % CI 0.621–0.693), and 0.633 (CI 0.605–0.661); positive likelihood ratios were respectively 3.652 (95 % CI 2.180–6.117), 2.196 (95 % CI 1.727–2.794), and 1.825 (95 % CI 1.351–2.464); negative likelihood ratios were respectively 0.212 (95 % CI 0.109–0.414), 0.246 (95 % CI 0.161–0.377), and 0.556 (95 % CI 0.385–0.804); the SROC curves were 19.205, 10.397, and 3.810.
Conclusions
The two IGRAs showed better performance than TST for the diagnosis of the tuberculosis. However, neither of them showed stability in the diagnosis of TB.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tuberculosis (TB) remains a worldwide infectious disease attracting extensive attention, whose morbidity rate under persistent elevation displayed inevitable hazards. In 2013, approximately 9.0 million people developed into TB with 1.5 million deaths, 360,000 of whom were HIV positive. The proportion of cured TB patients every year is almost constant with subtle declinations, for instance, only estimated 37 million people were cured between 2000 and 2013 ascribed to advanced and effective methods of diagnosis in conjunction with treatments (WHO) [1]. Meanwhile, tuberculosis’s early diagnosis remains an intricate problem. Nowadays, tuberculin skin test (TST) is one of the most widely applied methods in major countries considering its lower cost and convenience. However, this recommended diagnostic approach has several deficiencies, such as its low specificity, cross-reactions with bacille Calmette-Gue´rin (BCG) vaccination, and infection of nontuberculous mycobacteria (NTM) [2]. Ample evidences have demonstrated that Interferon (IFN)-γ release assays (IGRAs) seems to be an alternative for the diagnosis of the TB. IGRAs applies proteins that are more unique and specific for Mycobacterium tuberculosis than the purified protein derivative (PPD) which is encoded by genes located in the region of difference 1 (RD1) among the whole M. tuberculosis genome. These genes are not found in the BCG substrains or the most environmental mycobacteria (apart from Mycobacterium kansasii, Mycobacterium szulgai, Mycobacterium marinum, and Mycobacterium flavescens) [3]. Several studies have depicted the relative accuracy of IGRAs, but the majority of them were just verified in one aspect, respectively, such as children’s tuberculosis [4], active tuberculosis [5], and latent tuberculosis [6], which had restricted a comprehensive application of IGRAs.
Therefore, aiming to access the exact diagnostic value of IGRAs, we performed this meta-analysis to investigate the accuracy of IGRAs and determine whether IGRAs has the probability to replace the conventional diagnostic approaches.
Methods
Search Strategy
A systematic search was performed in the PubMed, Embase, Cochrane Library, and Web of Science databases in recent decades up to May 2015. The following terms were used to search for relevant investigations: “Tuberculosis/diagnosis” and “T-SPOT” or “QFT-IT” and “specificity”.
Inclusion and Exclusion Criteria
Researches retrieved from the databases were first scanned through titles with abstracts, and then full-text studies were further reviewed for eligibility. The studies will be included if TB patients have been diagnosed etiologically or sufficient information such as false or true positives and negatives have been provided to construct the analysis.
The studies will be excluded by the following reasons: case reports, editorials, or animal studies; systematic review and meta-analysis; sensitivity and specificity were not reported or could not be calculated; full text were not available or published in English. Two investigators participated in the search of available references individually and they reached the consensus on each eligible study.
Data Extraction
The following data were collected: first author, publishing year, numbers of cases and controls, country of origin, individuals’ characteristics, age, percent of male, percent of BCG vaccinated, percent of HIV-positive, TST reagent, cutoff for TST, percent with TST, T-SPOT, QFT-IT results, and percent of indeterminacy.
Statistical Analysis
To decrease heterogeneity, data extraction was separated by T-SPOT, QFT-IT, and TST. Indeterminate results were rejected from these studies. We used Statistical analysis I 2 and P value to describe the heterogeneity, and use correlation coefficient to determine its threshold effect. Pooled sensitivity and specificity of each assay and their 95 % confidence interval (CI) were calculated using random effects [7]. The meta-analysis was performed using MetaDiSc and Review Manager version 5.3.
Quality Assessment
The risk of bias table, which consists of seven domains covering (1) random sequence generation, (2) allocation concealment, (3) blinding of participants and personnel, (4) blinding of outcome assessment, (5) incomplete outcome data, (6) selective reporting, and (7) other bias, was used to access the inclusive studies’ risk of bias considering a total of five aspects, including the selection bias, the performance bias, the detection bias, the attrition bias, and the reporting bias. The risk of bias was finally judged as “low,” “high,” or “unclear” according to the answers of the signaling questions. The “unclear” category was used only when insufficient data were reported [8].
Results
Characteristics of the Studies
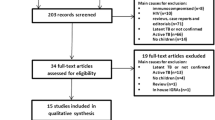
The search and selection process is described in Fig. 1. A total of 961 studies were found through retrieval, except for 393 duplications. In the remaining 568 studies, 529 did not conduct diagnostic tests or have the associative sensitivity or specificity, and 21 were meta-analysis and systematic reviews. Consequently, after further excluding 2 studies not published in English, a total of 16 studies were available as full texts for the final analysis [2, 3, 9–22]. Furthermore, among the 16 studies, 4 made comparison between QFT-IT and TST including 1855 participants, 7 compared T-SPOT and TST including 1731 participants, and the other 5 studies distinguished TST and T-SPOT from QFT-IT. All of these studies were representative in which a total of 3586 participants took part in the present analysis (Tables 1, 2). The risk of bias is shown in Fig. 2, with the incomplete outcome data locating in the high risk of bias and other bias under relatively low risk.
Sensitivity and Specificity of Interferon-γ Release Assays
We identified 9 studies correlated with QFT-IT, and the pooled sensitivity and specificity in diagnosis were 0.840 (95 % CI 0.814–0.864) and 0.658 (95 % CI 0.621–0.693), respectively (Fig. 3). The positive likelihood ratio, negative likelihood ratio, and pooled DOR of QFT-IT for the diagnosis of TB were 3.652 (95 % CI 2.180–6.117), 0.212 (95 % CI 0.109–0.414), and 10.397 (95 % CI 5.527–19.560) (Fig. 4), respectively. The pooled sensitivity, specificity, positive likelihood ratio, negative likelihood ratio, and pooled DOR of the T-SPOT were 0.842 (95 % CI 0.811–0.870), 0.745 (95 % CI 0.715–0.775), (Fig. 5), 2.196 (95 % CI 1.727–2.794), 0.246 (95 % CI 0.161–0.377), and 19.205 (95 % CI 7.049–52.326) (Fig. 6).
Sensitivity and Specificity of the Tuberculin Skin Test
Sixteen studies were identified to describe the tuberculin skin test. The meta-analytic estimate for sensitivity and specificity were 0.665 (CI 0.635–0.693) and 0.633 (CI 0.605–0.661) (Fig. 7). In addition, the positive likelihood ratio, negative likelihood ratio, and pooled DOR of the TST were 1.825 (95 % CI 1.351–2.464), 0.556 (95 % CI 0.385–0.804), and 3.810 (95 % CI 1.837–7.902) in Fig. 8.
SROC Curve of Interferon-γ Release Assays and the Tuberculin Skin Test
Area under the curve (AUC) was adopted to measure the accuracy of the tests. SROC curve of Interferon-γ release assays and the tuberculin skin test are shown in Figs. 6, 7, and 8. The AUC of the QFT and T-SPOT were 0.8818 and 0.9006, which were significantly higher than 0.7301 which was the AUC of the TST. From the SROC curve, we came to the assumption that all of the three methods might have a threshold effect. The analysis of diagnostic threshold of the QFT, T-SPOT and TST are conveyed in Figs. 6, 7, and 8. Their spearman correlation coefficients were, respectively, −0.375, −0.227, and −0.063, which proved the hypothesis that all the three methods had a threshold effect.
Discussion
This meta-analysis aimed to inquire the accuracy of IGRAs, which was witnessed as an accurate diagnostic method for latent and active TB, considering its unique preponderance. The summary results, as derived from 16 studies, indicated that both the sensitivity and the specificity of the IGRAs were significantly higher than those of the conventional TST. Moreover, the DOR of the TST was signally lower than the IGRAs, which meant the IGRAs had distinctly higher diagnostic value than TST. Besides, the reported TST sensitivity fluctuated between 0.258 and 1.000, with its specificity ranging from 0.336 to 0.919, while the sensitivity of the T-SPOT ranged from 0. 557 to 0.929, with its specificity were from 0.493 to 1.000. Hence, these results accounted for the better accuracy of IGRAs, although the outcomes were a little bit unsteady.
Through sub-analysis, we found that young adults, latent tuberculosis, TB patients with immunodeficiency, and TB patients with concurrent HIV infection may explain their stability [23–26] of data.
Because of the discontinuation of BCG mass vaccination in countries with a low incidence of TB, there has been an increase in NTM infection [27, 28], from which TST fails to distinguish NTM infection. However, IGRAs has a propensity for discriminating cross-reactivity induced by nontuberculous mycobacteria from bacille Calmette–Gue´rin vaccination, which demonstrates its superior capability of reducing over-diagnosis of TB and guiding clinical management.
As a fact that about one-third people in the world have been infected by M. tuberculosis in which estimated 2 billion people under latent tuberculosis infection (LTBI) have a 10 % lifetime risk of developing active TB [29], there is an urgent need to develop the golden standard of early LTBI’s diagnosis to solve the problems separating the LTBI from the ATB. Although increasing studies has demonstrated that the IGRAs promoted diagnosing LTBI because of its better specificity [20], there is no doubt that the false-positive rate is still high, which may lead to abundant unnecessary treatments resulting in drug resistance. So more researches are needed to be conducted to evaluate the value of the diagnosis of IGRAs.
The risk of tuberculosis (TB) in patients with an immunocompromised medical condition is greater than that in the general population [30]. Several studies conducted in South Korean populations have manifested that IGRAs had a predominant diagnostic sensitivity in active TB patients who were immunosuppressed [31–33]. The sensitivity of the T-SPOT was 0.720 (95 % CI 0.542–0.862), which was far higher than the TST (0.423), while the specificity of the T-SPOT was 0.423 and the specificity of the TST was 0.918. T-SPOT’s low specificity could make it insufficient to rule out TB disease, and the TST’s low sensitivity made it fail to rule in TB disease. So when referring to the immunocompromised TB patients, IGRAs might replace T-SPOT or TST for a definite diagnosis.
Besides, it is difficult to detect tuberculosis infections in HIV-infected patients, since the decreased number of CD4+ and CD8+ cells in their immune system brings about immune escape [34]. In this study, the sensitivity of the T-SPOT was 0.413 (95 % CI 0.354–0.488), which might be inaccurate due to different microenvironments in patients with other latent diseases or diverse received treatments. This was also far lower than the pooled sensitivity of 72 % in low/middle-income countries in a meta-analysis [35]. Another limitation of IGRA testing among HIV-infected patients was the rate of indeterminate results. Previous reports from the UK described indeterminate T-SPOT results in 2–7.4 % of HIV-infected patients [36–38]. However, in this study, it had not been found. As to TST, it failed to be a diagnostic method for the TB patients infected by HIV taken its sensitivity of 12.9 % into consideration.
In conclusion, IGRAs showed a superior capability than the TST to be a diagnostic approach for the tuberculosis because of its far higher sensitivity and specificity contributing to tell the TB patients apart. Also, its higher DOR and accuracy made it a valid alternative to the TST. However, neither the IGRAs nor the TST revealed ideal stability, which led to their restricted use. Furthermore, both of their costs and trauma should be considered. So we finally concluded that without a better precise diagnosis, the IGRAs could be a priority option to detect TB patients.
Abbreviations
- TB:
-
Tuberculosis
- TST:
-
Tuberculin skin test
- BCG:
-
Bacille Calmette–Gue´rin
- IGRAs:
-
Interferon (IFN)-γ release assays
- QFT-IT:
-
Quantiferon-TB gold in-tube test
References
Organization WH (2014) Global tuberculosis report 2014. http://www.who.int/tb/publications/global_report/en/
Detjen AK, Keil T, Roll S et al (2007) Interferon-gamma release assays improve the diagnosis of tuberculosis and nontuberculous mycobacterial disease in children in a country with a low incidence of tuberculosis. Clin Infect Dis 45(3):322–328. doi:10.1086/519266
Dilektasli AG, Erdem E, Durukan E et al (2010) Is the T-cell-based interferon-gamma releasing assay feasible for diagnosis of latent tuberculosis infection in an intermediate tuberculosis-burden country? Jpn J Infect Dis 63(6):433–436
Sun L, Xiao J, Miao Q et al (2011) Interferon gamma release assay in diagnosis of pediatric tuberculosis: a meta-analysis. FEMS Immunol Med Microbiol 63(2):165–173. doi:10.1111/j.1574-695X.2011.00838.x
Sester M, Sotgiu G, Lange C et al (2011) Interferon-gamma release assays for the diagnosis of active tuberculosis: a systematic review and meta-analysis. Eur Respir J 37(1):100–111. doi:10.1183/09031936.00114810
Pai M, Zwerling A, Menzies D (2008) Systematic review: T-cell-based assays for the diagnosis of latent tuberculosis infection. An update (Structured abstract). Ann Intern Med 149(3):177–184
Deville WL, Buntinx F, Bouter LM et al (2002) Conducting systematic reviews of diagnostic studies: didactic guidelines. BMC Med Res Methodol 2:9
Ng SC, Hirai HW, Tsoi KK et al (2014) Systematic review with meta-analysis: accuracy of interferon-gamma releasing assay and anti-saccharomyces cerevisiae antibody in differentiating intestinal tuberculosis from Crohn’s disease in Asians. J Gastroenterol Hepatol 29(9):1664–1670. doi:10.1111/jgh.12645
Chung JH, Han CH (2010) Clinical utility of QuantiFERON-TB Gold in tube interferon-gamma assay and tuberculin skin test in patients with tuberculous pleural effusions. Diagn Microbiol Infect Dis 71(3):263–266
Kabeer BSA, Raman B, Thomas A et al (2010) Role of QuantiFERON-TB gold, interferon gamma inducible protein-10 and tuberculin skin test in active tuberculosis diagnosis. PLoS One. doi:10.1371/journal.pone.0009051
Lee JE, Kim HJ, Lee SW (2011) The clinical utility of tuberculin skin test and interferon-γ release assay in the diagnosis of active tuberculosis among young adults: a prospective observational study. BMC Infect Dis. doi:10.1186/1471-2334-11-96
Syed Ahamed Kabeer B, Raman B, Thomas A et al (2010) Role of QuantiFERON-TB gold, interferon gamma inducible protein-10 and tuberculin skin test in active tuberculosis diagnosis. PLoS One. doi:10.1371/journal.pone.0009051
Goletti D, Stefania C, Butera O et al (2008) Accuracy of immunodiagnostic tests for active tuberculosis using single and combined results: a multicenter TBNET-study. PLoS One. doi:10.1371/journal.pone.0003417
Jung JY, Lim JE, Lee HJ et al (2012) Questionable role of interferon-gamma assays for smear-negative pulmonary TB in immunocompromised patients. J Infect 64(2):188–196. doi:10.1016/j.jinf.2011.09.008
Kang YA, Lee HW, Hwang SS et al (2007) Usefulness of whole-blood interferon-gamma assay and interferon-gamma enzyme-linked immunospot assay in the diagnosis of active pulmonary tuberculosis. Chest 132(3):959–965. doi:10.1378/chest.06-2805
Lee JY, Choi HJ, Park IN et al (2006) Comparison of two commercial interferon-gamma assays for diagnosing Mycobacterium tuberculosis infection. Eur Respir J 28(1):24–30. doi:10.1183/09031936.06.00016906
Ball PM, Pernollet M, Bouillet L et al (2010) Usefulness of an in vitro tuberculosis interferon-gamma release assay (T-SPOT.TB) in the first-line check-up of uveitis patients. Ann Med 42(7):546–554. doi:10.3109/07853890.2010.518155
Fei B, Wu Z, Min K et al (2014) Interferon-gamma release assay in the diagnosis of laryngeal tuberculosis. Acta Otolaryngol 134(3):314–317. doi:10.3109/00016489.2013.850174
Feng Y, Diao N, Shao L et al (2012) Interferon-gamma release assay performance in pulmonary and extrapulmonary tuberculosis. PLoS One. doi:10.1371/journal.pone.0032652
Simsek H, Alpar S, Ucar N et al (2010) Comparison of tuberculin skin testing and T-SPOT.TB for diagnosis of latent and active tuberculosis. Jpn J Infect Dis 63(2):99–102
Yu Y, Zhao X, Wang W et al (2013) Diagnostic performance of interferon-gamma releasing assay in HIV-infected patients in China. PLoS One 8(8):e70957. doi:10.1371/journal.pone.0070957
Zhong L, Zhou XL, Li J et al (2015) The T-SPOT.TB test for diagnosis of breast tuberculosis. Labmedicine. doi:10.1309/lmfbrto5mwv3ayim
Arenas Miras MDM, Hidalgo-Tenorio C, Jimenez-Gamiz P et al (2014) Diagnosis of latent tuberculosis in patients with systemic lupus erythematosus: T.SPOT.TB versus tuberculin skin test. BioMed Res Int 2014:291031. doi:10.1155/2014/291031
Cai R, Chen J, Guan L et al (2014) Relationship between T-SPOT.TB responses and numbers of circulating CD4+ T-cells in HIV infected patients with active tuberculosis. Biosci Trends 8(3):163–168
Wang X, Wu Y, Wang M et al (2014) The sensitivity of T-SPOT.TB assay in diagnosis of pediatric tuberculosis. Fetal Pediatr Pathol. doi:10.3109/15513815.2013.878010
Chen J, Sun J, Zhang R et al (2011) T-SPOT.TB in the diagnosis of active tuberculosis among HIV-infected patients with advanced immunodeficiency. AIDS Res Hum Retrovir 27(3):289–294. doi:10.1089/aid.2010.0062
Romanus V (1995) Mycobacterial infections in Sweden. Scand J Infect Dis Suppl 98:15–16
Vu TT, Daniel SJ, Quach C (2005) Nontuberculous mycobacteria in children: a changing pattern. J Otolaryngol 34(Suppl 1):S40–S44
Markowitz N, Hansen NI, Hopewell PC et al (1997) Incidence of tuberculosis in the United States among HIV-infected persons. The pulmonary complications of HIV infection study group. Ann Intern Med 126(2):123–132
Kobashi Y, Mouri K, Obase Y et al (2007) Clinical evaluation of QuantiFERON TB-2G test for immunocompromised patients. Eur Respir J 30(5):945–950. doi:10.1183/09031936.00040007
Kim SH, Choi SJ, Kim HB et al (2007) Diagnostic usefulness of a T-cell based assay for extrapulmonary tuberculosis. Arch Intern Med 167(20):2255–2259. doi:10.1001/archinte.167.20.2255
Kim SH, Song KH, Choi SJ et al (2009) Diagnostic usefulness of a T-cell-based assay for extrapulmonary tuberculosis in immunocompromised patients. Am J Med 122(2):189–195. doi:10.1016/j.amjmed.2008.07.028
Lee JY, Choi HJ, Park IN et al (2006) Comparison of two commercial interferon-gamma assays for diagnosing Mycobacterium tuberculosis infection. Eur Respir J 28(1):24–30. doi:10.1183/09031936.06.00016906
Rangaka MX, Wilkinson KA, Seldon R et al (2007) Effect of HIV-1 infection on T-Cell-based and skin test detection of tuberculosis infection. Am J Respir Crit Care Med 175(5):514–520. doi:10.1164/rccm.200610-1439OC
Cattamanchi A, Smith R, Steingart KR et al (2011) Interferon-gamma release assays for the diagnosis of latent tuberculosis infection in HIV-infected individuals: a systematic review and meta-analysis. JAIDS J Acquir Immune Defic Syndr 56(3):230–238. doi:10.1097/QAI.0b013e31820b07ab
Clark SA, Martin SL, Pozniak A et al (2007) Tuberculosis antigen-specific immune responses can be detected using enzyme-linked immunospot technology in human immunodeficiency virus (HIV)-1 patients with advanced disease. Clin Exp Immunol 150(2):238–244. doi:10.1111/j.1365-2249.2007.03477.x
Kall MM, Coyne KM, Garrett NJ et al (2012) Latent and subclinical tuberculosis in HIV infected patients: a cross-sectional study. BMC Infect Dis 12:107. doi:10.1186/1471-2334-12-107
Dheda K, Lalvani A, Miller RF et al (2005) Performance of a T-cell-based diagnostic test for tuberculosis infection in HIV-infected individuals is independent of CD4 cell count. AIDS (London, England) 19(17):2038–2041
Acknowledgments
We thank Li-mei Zhu M.D. for her discussion and help.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Additional information
Peng Lu and Xiu Chen have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Lu, P., Chen, X., Zhu, Lm. et al. Interferon-Gamma Release Assays for the Diagnosis of Tuberculosis: A Systematic Review and Meta-analysis. Lung 194, 447–458 (2016). https://doi.org/10.1007/s00408-016-9872-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-016-9872-5