Abstract
An increasing interest in the assessment of neuropsychological performance variability in people with first-episode psychosis (FEP) has emerged. However, its association with clinical and functional outcomes requires further study. Furthermore, FEP neuropsychological subgroups have not been characterized by clinical insight or metacognition and social cognition domains. The aim of this exploratory study was to identify specific groups of patients with FEP based on neuropsychological variables and to compare their sociodemographic, clinical, metacognition and social cognition profiles. A sample of 149 FEP was recruited from adult mental health services. Neuropsychological performance was assessed by a neuropsychological battery (WAIS-III; TMT; WSCT; Stroop Test; TAVEC). The assessment also included sociodemographic characteristics, clinical, functional, metacognition and social cognition variables. Two distinct neuropsychological profiles emerged: one neuropsychological impaired cluster (N = 56) and one relatively intact cluster (N = 93). Significant differences were found between both profiles in terms of sociodemographic characteristics (age and level of education) (p = 0.001), clinical symptoms (negative, positive, disorganized, excitement and anxiety) (p = 0.041–0.001), clinical insight (p = 0.038–0.017), global functioning (p = 0.014), as well as in social cognition domains (emotional processing and theory of mind) (p = 0.001; p = 0.002). No significant differences were found in metacognitive variables (cognitive insight and ‘jumping to conclusions’ bias). Relationship between neurocognitive impairment, social cognition and metacognition deficits are discussed. Early identifying of neuropsychological profiles in FEP, characterized by significant differences in clinical and social cognition variables, could provide insight into the prognosis and guide the implementation of tailored early-intervention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Schizophrenia is a chronic disorder that concurs with delusions, hallucinations, disorganized speech, disorganized/catatonic behavior or negative symptoms of sufficient severity to disrupt everyday functioning [1], resulting in a life expectancy of 15–20 years shorter that of the general population [2]. Poor insight into mental disorder is also a prevalent feature in those patients [3], which has been linked with positive and negative symptoms, as well as depression in patients with first-episode psychosis (FEP) [4].
Recently, however, the study of deficits in social cognition [5, 6] and metacognition [7] in FEP has attracted increasing interest [8]. Social cognition refers to perception, interpretation, and information processing for adaptive social interactions [9]. Metacognition refers to the spectrum of mental processes involving the reflection upon others’ and one’s own thinking, and the synthesis of these phenomena into an integrated sense of self and others [10]. Unlike social cognition, the domains of metacognition are not as well identified and confusion is often created by the variety of definitions we find in the literature [8]. Both deficits in social cognition and metacognition have been related to functional outcome [9, 11].
At the same time, FEP is highly heterogeneous in symptom presentation, severity of clinical course, and functional impact [12, 13]. Recent research has also suggested that subjects with FEP experience different substantial deficits in social cognition and metacognition [5]. Identifying factors predicting outcome overtime in the early stages of psychosis is especially necessary given that recovery rates in FEP appear to be stable two years after illness onset [14].
Some of this variability may be partly attributable to heterogeneity in neuropsychological functioning in FEP [15,16,17]. Cognitive impairments, including attention, verbal memory, processing speed, working memory and executive functioning [18], have been consistently accepted as core features of the disorder that play a role as vulnerability indicators, and linked with both clinical and functional outcomes over time [11, 19, 20]. However, recent meta-analysis have shown only small to medium effect sizes for the association between neuropsychological performance and functional outcomes, as well as a limited role of neuropsychological deficits to explain lack of insight for people with schizophrenia [21]. Regarding the relationship between neurocognitive impairment, social cognition and metacognition deficits, there is still debate about if these are influenced by neuropsychological deficits [15, 22, 23], or, conversely, they are not [24,25,26].
It has been increasingly recognized that neuropsychological impairment severity varies among patients with FEP, ranging from broad deficits to performance at levels similar to healthy samples [27]. Cluster analysis provides an opportunity to classify individuals with FEP based on their neuropsychological profiles and guide early treatment while preventing functional decline. However, the vast majority of current neuropsychological clustering work has occurred in chronic populations, with relatively few cognitive heterogeneity studies in FEP. Published FEP studies to date have reported two [28], three [16, 29,30,31], and four [17] clusters neuropsychological structures. In all cases, relatively intact, results within the typical limits in all scores on standardized tests, and impaired groups were identified. One recent study [28] has found two distinct subgroups: the first cluster characterized by moderate neuropsychological impairments in processing speed, verbal learning, working memory and verbal fluency and the second one with relatively intact neurocognition.
Research aimed at identifying specific groups of patients with FEP based on neuropsychological variables requires extension and replication, notably to aid in improving the characterization of the clusters. Previous studies have associated neuropsychological clusters with distinct clinical characteristics [28]. Cluster groups with neuropsychological deficits are more likely to present severe clinical symptoms and lower global functioning than those without neuropsychological deficits [17, 29]. In contrast, other authors have found no significant differences in symptom severity [30] and global functioning [16] across clusters. To our knowledge, FEP cognitive clusters have not been characterized by clinical insight or their metacognitive or social cognitive skills. Given the significant impact of these variables on clinical severity and psychosocial functioning, understanding whether different neuropsychological profiles present differences in clinical, metacognitive and social cognitive variables may help identify which persons are at a bigger risk of chronic illness and may help develop treatments tailored to their illness profile.
In light of these issues, the present study sought to identify neuropsychological profiles in a group of FEP patients using cluster analysis, and to further investigate the relationships with sociodemographic, clinical and global functioning features, and metacognition and social cognition variables. Based on previous findings, we hypothesized that at least two profiles would emerge, one relatively neuropsychological intact and one generally impaired cluster. We also predicted significant differences between clusters on sociodemographic and clinical characteristics, clinical symptoms, clinical insight and global functioning, as well as on metacognition and social cognition skills between the clusters.
Material and methods
The design of the study was performed based on the baseline data of two multi-centric clinical trials, registered under NCT02340559 and NCT04429412 [32].
Participants
A total of 149 participants with FEP were recruited by staff members of the following Spanish mental health centers: Fundación Jiménez Díaz (Madrid), Servicio Andaluz de Salud (Jaén, Málaga and Granada), Centro de Salud Mental de Corporació Sanitària i Universitària Parc Taulí (Sabadell), Hospital del Mar, Consultas externas del Hospital de Santa Creu i Sant Pau (Barcelona), Centro de Higiene Mental Les Corts (Barcelona), Hospital Universitari Institut Pere Mata (Reus), Institut d´Assistència Sanitària Girona, Hospital Clínico de Valencia, and Parc Sanitari Sant Joan de Déu (PSSJD).
The Ethics Committee of each participating center approved this project and was carried out in accordance with the Declaration of Helsinki in its recent review.
Inclusion criteria were as follows: (1) a diagnosis of schizophrenia, unspecified psychotic disorder, delusional disorder, schizoaffective disorder, brief psychotic disorder, or schizophreniform disorder (DSM-IV-TR criteria); (2) less than 5 years from the onset of psychotic symptomatology; (3) PANSS scores of ≥ 3 in items Delusions, Grandiosity, or Suspiciousness, in the last year; (4) age between 16 and 50 years.
Exclusion criteria included: (1) traumatic brain injury, dementia, or intellectual disability (premorbid IQ ≤ 70); (3) substance dependence (DSM-IV-TR criteria); (4) PANSS scores of ≥ 5 in Hostility and Uncooperativeness items, and ≥ 6 in Suspiciousness.
All selected individuals were informed of the study objectives and methodology by their psychiatrist and signed the required informed consent form. In the case of participants under the age of 18, in addition to their own consent, the consent of their parents, guardians or legal representatives was also obtained.
Assessment measures
Sociodemographic questionnaire: Data on age, sex, marital status and education level were collected using a questionnaire created ad hoc. Diagnosis and treatment were collected from the clinical history of the participants.
Clinical measures: Positive and Negative Syndrome Scale (PANSS) Spanish version [33, 34] was used to measure clinical and general symptoms. We used the seven-factor solution [35]. This solution separates anxiety and depression into two different factors and includes a motor factor. The Spanish version of the Scale Unawareness of Mental Disorder (SUMD) [36, 37] was used to measure unawareness of the mental disorder. A score ranging from 1 to 5 was computed for the unawareness of symptoms, and misattribution of symptoms dimensions. A global insight score was calculated by adding up the first three dimension scores (unawareness of illness, unawareness of the need for treatment and unawareness of the social consequences).
Global functioning: The Global Assessment of Functioning (GAF) [38] was used to measure global functioning on a scale of 0–100.
Metacognition: A Spanish version of the Beck Cognitive Insight Scale (BCIS) [39, 40] was used to evaluate cognitive insight yielding a self-reflectiveness subscale and a self-certainty subscale, as well as a Composite Index score. Jumping to Conclusions (JTC) reasoning bias was measured by means of the The Beads Task [41, 42]. We used three trials with different conditions: a probabilistic trial with an 85/15 ratio, a second probabilistic trial with a 60/40 ratio, and a final trial with an affective condition in a 60/40 ratio. Our outcome variable was the draws to decision in the three probabilistic conditions. JTC was considered when participants took a decision after extracting one or two balls [43].
Social cognition: Attributional style was assessed by the Internal, Personal and Situational Attributions Questionnaire (IPSAQ) [44, 45]. We used two indexes: personalizing bias and externalizing bias. Personalizing bias refers to a tendency to blame others rather than circumstances for negative events. Externalizing bias refers to a tendency to attribute the causes of negative events to others or circumstances rather than to oneself. The Faces Test [46, 47] was used to measure emotion processing. A Spanish version of the Hinting Task [48, 49] was used to measure Theory of Mind (ToM).
Neurocognition. The following scales were used for the variables in the cluster analysis. The scores used in the analysis were demographically-adjusted (t distribution) according to the Spanish validation of the instruments:
The WAIS-III [50] Vocabulary and Digits subtests were used to measure premorbid estimated intelligence quotient (IQ) and verbal fluency and working memory, respectively.
The Trail Making Test [51, 52] was used to assess information processing speed and visuomotor attention (TMT-A), as well as cognitive flexibility and inhibitory control (TMT-B).
The Stroop Test [53, 54] was used to measure selective attention, processing speed, and resistance to interference.
The Wisconsin Card Sorting Test (WSCT) [55, 56] was used to assess executive functioning in clinical settings. The analysis variables were total errors, perseverative errors and non-perseverative errors.
The Complutense Verbal Learning Test (TAVEC) [57] is the Spanish version of the California Verbal Learning Test (CVLT) [58]. This was used to assess the different verbal memory processes, such as immediate memory (RIAT), short-term memory and long-term memory.
Finally, part of the sample was assessed with the Continuous Performance Test (CPT-II for Windows) [59]. The other subset was assessed with the MATRICS CPT [60,61,62]. To obtain a homogeneous measure of attention, we created the composite variable “Attention” by adding the D-prime scores of both measures.
Statistical analysis
The cluster analysis was performed with the K-means iterate and classify conglomerate method. The optimal number of clusters was determined by the average silhouette width (Dunn index calculated with Euclidian distances) for 1–10 clusters, with 1000 replications for each number of clusters. Hence, the algorithm proposes the optimal number of clusters by considering the number of subgroups that exhibit the highest average silhouette width. The Dunn index, on the other hand, is a measure of the ratio between compactness within clusters and the separation between clusters [63, 64]. The number of clusters tested was based on the number of subgroups of the average silhouette width and previous literature. Cluster assignment was carried out by 1000 replications of a tenfold cross-validation [65]. For each step of the cross-validation process, data were split into 10 parts and one of the parts functioned as a test set while a clustering model was performed on the rest training set. A comparison of the two selected clusters with categorical variables was made using the Chi-square test, and the comparisons of the two groups with continuous variables were made using Student’s t-test. We did not perform multiple comparison corrections due mainly to the exploratory nature of this study [66]. The effect size was calculated with Cohen’s d,
Analyses were carried out using the IBM SPSS Statistical software package (version 26).
Results
Sample characteristics
A total of 149 patients with FEP were included in the analysis (68% males, age range: 16–47 years, Mage = 27.8, SDage = 7.32). According to their marital status, 128 patients referred being single (84.2%). Most of sample was employed, with 61 participants (41%), 27 were students (18.1%), 2 were homemakers (1.3%), 14 were retired (9.4%), and 45 were on temporary leave due to disability, or inactive (30.2%). Regarding clinical characteristics, the most prevalent diagnosis was schizophrenia in 66 participants (44.3%), followed by 39 with unspecified psychotic disorder (26.2%), 12 with schizoaffective disorder (8.1%), 9 with delusional disorder (6%), 14 with brief psychotic disorder (9.4%), and 9 with schizophreniform disorder (6%).
Cluster solution
The tested clusters were 2, 3 and 4. The four-cluster solution was excluded from further analysis based on a comprehensive assessment of both the sample distribution (Cluster 1, N = 57; Cluster 2, N = 44; Cluster 3, N = 5; Cluster 4, N = 4) and the average silhouette width (0.169) (see Fig. 2). Table 1 summarizes the scores of the two cluster solutions in the neuropsychological variables. Significant differences were found between all neuropsychological variables evaluated in the two cluster solutions (p < 0.001). However, the average silhouette width shows a better adjustment to two-cluster solution (0.31) (Fig. 2). This suggests that the two-cluster solution performed better than the three-cluster one. After assessment of the values for each number of clusters, the two-group clusters were selected for further analyses. Furthermore, the two-cluster solution showed both greater homogeneous sample size distribution and neuropsychological characterization in the patients’ subgroups. Equal-size clusters and homogeneity within-group are commonly argued in the statistics literature for determining the optimal number of clusters [67, 68]. Table 1 and Fig. 1 contain a summary the characteristics of participants in the two-cluster solution in relation to neuropsychological variables. The variable “Attention” was excluded from further analyses as it did not show significant differences between the groups in any of the cluster’s solutions (p = 0.784).
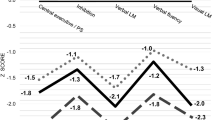
The analysis of the two-group cluster showed two profiles. Figure 3 each profile in relation to neuropsychological variables.
Means Z-scores in the neuropsychological variables for each neurocognitive clusters. For detailed statistics, see Table 1. Note: IQ Intelligence quotient, RIAT 1 Immediate recall (first trial), RIAT 5 Immediate recall (fifth trial), TAVEC Verbal Learning Test Spain-Complutense, TMT Trail Making Test, WAIS-III Wechsler Adult Intelligence Scale, WCST Wisconsin Card Sorting Test. ªHigher scores indicate worse functioning. bHigher scores indicate better functioning
Profile 1 was characterized by the following: low premorbid IQ (WAIS-III Vocabulary), severe impairment in processing speed (TMT-A) and in neurocognitive flexibility and divided attention (TMT-B), medium–low performance in executive functions when those were assessed by WSCT, severe to moderate impairment in verbal memory (TAVEC), and moderate deficits in working memory measured by the digits subscale of the WAIS–III. Based on these results and previous research, Profile 1 was labeled as “Impaired”.
In contrast, Profile 2 emerged as the relatively neuropsychological intact cluster (results within the typical limits in all scores on standardized tests), outperforming the other cluster on all neuropsychological measures. This profile was named “Relatively intact”.
Socio-demographic and clinical characteristics by cluster
Table 2 details the sociodemographic and clinical characteristics between profiles. When comparing profiles, subjects with relatively intact neuropsychological performance Profile 2) had significantly higher age and level of education.
Clinical and functional performance by cluster
Profiles also showed significant differences in all clinical and functional domains evaluated, except for the motor and depression factors of the Emsley PANSS (Table 3).
Specifically, those in the “Impaired” profile had significantly higher scores in the PANSS total and in the negative positive, disorganized and anxiety symptoms subscales than their counterparts. The magnitudes of the significant differences found in this group of variables were moderate for the total scale and disorganized symptoms of the PANSS, and small for the rest of the subscales.
Similarly, as seen in Table 3, the “Impaired” profile achieved higher scores in each SUMD measure (global insight score, unawareness of symptoms and misattribution of symptoms), indicating a worse clinical insight. The magnitude of the differences was small in all the dimensions.
Finally, small significant differences were found in global functioning (total GAF score, Table 3). FEP patients in the “Impaired” profile had worse functioning compared to patients in the “Relatively intact” profile.
Social cognition and metacognition by clusters
Table 4 summarizes the scores of each profile in the metacognitive and social cognitive variables. Regarding social cognition, clusters showed significant differences. Specifically, the “Impaired” profile displayed worse emotional processing and greater impairment in ToM, compared to the other group. After calculating the effect sizes associated with these differences, great and moderate differences were found in emotional processing and in theory of mind, respectively.
As seen in Table 4, clusters did not differ either in attributional style or in any metacognition variables.
Discussion
This study showed two profiles characterized by impairment (Profile 1) and relatively intact (Profile 2) neuropsychological performance, which was in line with our first hypothesis. The second hypothesis was partially supported, with significant group differences observed for age and level of education, clinical symptoms (negative, positive, disorganized, excitement and anxiety), clinical insight, and global functioning, as well as in the domains of social cognition, but not for any metacognition domains between clusters. To the best of our knowledge, this is the first study investigating clinical insight, metacognition and social cognition profiles in neuropsychological subgroups of FEP patients.
Our findings are aligned with other authors [28] demonstrating two well defined neuropsychological profiles, supporting the presence of neuropsychological heterogeneity in FEP. These groups of patients differed significantly across all neurocognitive domains. Thus, our results suggest that some groups of patients with a FEP are nearer to healthy groups, which is concurrent with previous research [29, 31]. However, it should be noted that neuropsychological performance similar to that of general samples is not necessarily synonymous with no cognitive impairment, since there is no confirmation of them not having declined from premorbid levels (pre-onset) because there is no previous neuropsychological assessment available.
Nevertheless, three-cluster solution demonstrating three neuropsychological subgroups is also consistent with previous literature on FEP and the notion of a continuum of cognitive functioning [16, 29,30,31]. Of note, the subgroup showing intermediate scores across three outperformed the most impaired group in all subdomains except verbal memory processes. To our knowledge, such an exception has not been found in previous studies exploring neuropsychological profiles in FEP. Findings regarding the intermediate cluster should be further investigated in future studies.
FEP patients in the “Relatively intact” profile were significantly older and with more years of education than those in the “Impaired” profile. These findings are substantiated by previous work in both FEP [16, 17, 31, 69] and chronic samples [70, 71], suggesting that reduced educational success is related to poorer cognitive function [72]. Although age at onset variable was not included, we could hypothesize that Profile 1 (youngest cluster) would have had an earlier age at onset, which has been widely associated with greater impairment [73].
Neuropsychological profiles showed also distinct clinical characteristics. Our findings of more severe clinical symptomatology, specifically negative symptoms, in the group with severely impaired cognition is in line with previous research in FEP [17, 31, 74]. The lack of differences in the depression levels is not surprising, as previous studies have suggested that greater awareness of having a mental disorder might lead to depression [4, 75, 76]. Although this is the first study to characterize FEP neuropsychological profiles by insight into their mental disorder, the results are consistent with earlier reports [4] indicating relationships between poor insight and several domains of neurocognition. This is relevant as poor insight is associated with treatment compliance and recovery. In the same line, disparity in the global functioning were also found across profiles. Other studies also suggest that global functioning may be related to neuropsychological cluster membership [16, 31, 70].
These cross-sectional findings could be explained by the widely acknowledged abnormal neurodevelopmental hypothesis, based on some schizophrenias have a developmental origin- suggesting that multiple genetic and environmental factors can disrupt normal brain development, and at a certain point, the person may be affected by the external and internal factors that trigger the onset of the disease [77]. In this way, biological alterations and other characteristics of the illness would be present in the patient well before the emergence of the psychotic symptoms. For instance, it well documented that a large number of individuals affected by the spectrum of psychotic disorders manifest lower intelligence or academic adjustment prior to initiation of the diseases. This, in turn, could lead to poor neuropsychological performance [78], aligning with our emerging Profile 1 characterized by impairment. In addition, these abnormalities could act as vulnerability markers for the illness leading potential trajectories. Individuals with abnormal neurodevelopmental may face higher risk of developing more negative symptoms a poorer functional outcome [79], in line with our findings.
One of the main findings of our study is that neuropsychological profiles were related with some variables of social cognition. Specifically, patients with higher impaired in neuropsychological functioning (Profile 1) obtained significantly lower scores on emotional processing and ToM measures. Previous research have also found distinct social cognition profiles regarding to impairment in psychosis [5, 80]. Our findings are in line with Lysaker [81], who observed in a sample of individuals with a schizophrenia spectrum disorder, that better performance in social cognition was associated with higher educational level, a variable broadly linked to neuropsychological performance. In FEP samples, Ayesa-Arriola [69] has also evidenced the relationship between ToM deficits and neurocognitive function, and its possible link with premorbid social [23].
However, no significant differences between profiles could be demonstrated in attributional style (social cognition domain) and in metacognitive variables. The separation of attributional style and social cognition variables (like theory of mind and emotion perception) giving support to prior works [5, 82] and are consistent with conceptual differences between these two constructs [82]. It must also be added that previous research has found profiles of patients encompassing metacognitive variables and attributional style [5]. However, it is worth noting that differences with a trend towards statistical significance were found in jumping to conclusions bias between the clusters. Again, these results would be in line with previous proposal [82] suggesting the existence of a social cognition factor involving emotion perception, ToM and JTC.
Thus, in general, the results of our study support previous research suggesting that social cognition and metacognition may act through different pathways [5, 81, 83]. In addition, we consider that the results presented here help to elucidate the debate about whether social cognition and metacognition deficits are influenced by neuropsychological deficits. However, taking into account the differences with tendency towards statistical significance in some variables, replicated longitudinal studies are required.
The results have important implications for clinical practice with FEP. Identifying neuropsychological profiles at the early stages of psychosis may provide insights into how to direct early treatment. Early interventions as cognitive remediation [84], exercise intervention [85, 86] or metacognitive training for psychosis (MCT) [87], as well integrative therapy combining these treatments could be beneficial in promoting recovery and preventing or reducing the impact of the illness. MCT, for example, has demonstrated recently its efficacy in neurocognitive deficits in FEP [88]. Furthermore, understanding the social cognition and metacognition profiles that present neuropsychological subgroups has the potential to aid in the tailoring of therapeutic interventions to better address the heterogeneity of psychotic illness, as well as gaining more knowledge about cognition-enhancing interventions. For example, individuals with general social cognition impairments could benefit more by interventions that aim to compensate or remediate these deficits.
Some limitations should be considered. First, although we followed the recommended guidelines for reporting on cluster analysis [27], cluster analyses have the limitation that the determination of the number of clusters can be arbitrary, as it depends on the methods used. Future studies should replicate the present results with other methods such as the Partition Around Medoids (PAM) algorithm [89]. Second, although our utilization of scores demographically-adjusted (t distribution) offered a reasonable estimate of neurocognitive impairment or intactness in the emerging clusters, incorporating a demographically matched healthy control group in future analyses would be advantageous. Third, the cross-sectional design causality does not allow for any claims regarding the potential trajectories of patients’ cognitive performance and their relationship with prospective clinical, metacognition and social cognition variables. Furthermore, we considered neuropsychological variables after psychosis appears but it is possible that neuropsychological deficits were present before the onset of the illness. Finally, the only measure of functioning was the GAF. Although it is widely used in research, it fails to cover all nuances of functional outcome, as it is a general measure. Future research should include longitudinal design and test the presence of these neuropsychological profiles in other samples as high risk for psychosis, as well as including more measures of metacognition, as the Spanish version of the Cognitive Bias Questionnaire (CBQ) [90] which evaluates a wide range of cognitive biases. It would also be interesting to test the effect of other sociodemographic and contextual variables (i.e., parent’s education level, economic level or presence of trauma history) over neurocognition performance in future research. A more exhaustive assessment is needed to explore the association of these neuropsychological profiles to more clinical characteristics as premorbid adjustment social, duration of untreated psychosis or age at onset. Finally, while the groups here did not significantly differ on medication dose, the complex relationship between cognitive functioning and antipsychotic medications warrants additional exploration. Investigating this issue to elucidate whether neuropsychological performance could be influenced by dose or type of antipsychotic medication, as noted in other studies [29], would provide further insights.
Conclusions
The results of this initial exploratory study reported evidence of one neuropsychological impaired profile (“Impaired”) and one relatively intact profile (“Relatively intact”) with the corresponding differences in sociodemographic characteristics as well as clinical and social cognition variables. This research encourages early interventions to adapt to impairment profiles as there seems to be heterogeneity in neuropsychological functioning in FEP.
Data availability
Due to confidentiality issues, access to data will only be granted on request (S.O.; susana.ochoa@sjd.es).
References
Nevarez-Flores AG, Sanderson K, Breslin M, Carr VJ, Morgan VA, Neil AL (2019) Systematic review of global functioning and quality of life in people with psychotic disorders. Epidemiol Psychiatr Sci. https://doi.org/10.1017/S2045796018000549
Laursen TM, Nordentoft M, Mortensen PB (2014) Excess early mortality in schizophrenia. Annu Rev Clin Psychol. https://doi.org/10.1146/annurev-clinpsy-032813-153657
Lincoln TM, Lüllmann E, Rief W (2007) Correlates and long-term consequences of poor insight in patients with schizophrenia. A systematic review. Schizophr Bull 33:1324–1342. https://doi.org/10.1093/schbul/sbm002
Pousa E, Brébion G, López-Carrilero R, Ruiz AI, Grasa E, Barajas A et al (2022) Clinical insight in first-episode psychosis: clinical, neurocognitive and metacognitive predictors. Schizophr Res 248:158–167. https://doi.org/10.1016/j.schres.2022.08.007
Ferrer-Quintero M, Fernández D, López-Carrilero R, Birulés I, Barajas A, Lorente-Rovira E et al (2022) Persons with first episode psychosis have distinct profiles of social cognition and metacognition. Npj Schizophr 8:3. https://doi.org/10.1038/s41537-021-00199-4
Healey KM, Bartholomeusz CF, Penn DL (2016) Deficits in social cognition in first episode psychosis: a review of the literature. Clin Psychol Rev 50:108–137. https://doi.org/10.1016/j.cpr.2016.10.001
MacBeth A, Gumley A, Schwannauer M, Carcione A, McLeod HJ, Dimaggio G (2016) Metacognition in first episode psychosis: item level analysis of associations with symptoms and engagement. Clin Psychol Psychother. https://doi.org/10.1002/cpp.1959
Moritz S, Lysaker PH (2018) Metacognition – What did James H. Flavell really say and the implications for the conceptualization and design of metacognitive interventions. Schizophr Res 201:20–6. https://doi.org/10.1016/J.SCHRES.2018.06.001
Green MF, Horan WP, Lee J (2019) Nonsocial and social cognition in schizophrenia: current evidence and future directions. World Psychiatry 18:146–161. https://doi.org/10.1002/wps.20624
Lysaker PH, Vohs J, Minor KS, Irarrázaval L, Leonhardt B, Hamm JA et al (2015) Metacognitive deficits in schizophrenia: presence and associations with psychosocial outcomes. J Nerv Ment Dis. https://doi.org/10.1097/NMD.0000000000000323
Lysaker PH, Pattison ML, Leonhardt BL, Phelps S, Vohs JL (2018) Insight in schizophrenia spectrum disorders: relationship with behavior, mood and perceived quality of life, underlying causes and emerging treatments. World Psychiatry. https://doi.org/10.1002/wps.20508
Jaracz K, Górna K, Kiejda J, Grabowska-Fudala B, Jaracz J, Suwalska A et al (2015) Psychosocial functioning in relation to symptomatic remission: a longitudinal study of first episode schizophrenia. Eur Psychiatry 30:907–913. https://doi.org/10.1016/j.eurpsy.2015.08.001
Santesteban-Echarri O, Paino M, Rice S, González-Blanch C, McGorry P, Gleeson J et al (2017) Predictors of functional recovery in first-episode psychosis: a systematic review and meta-analysis of longitudinal studies. Clin Psychol Rev. https://doi.org/10.1016/j.cpr.2017.09.007
Lally J, Ajnakina O, Stubbs B, Cullinane M, Murphy KC, Gaughran F et al (2017) Remission and recovery from first-episode psychosis in adults: systematic review and meta-analysis of long-term outcome studies. Br J Psychiatry J Ment Sci 211:350–358. https://doi.org/10.1192/bjp.bp.117.201475
Ayesa-Arriola R, Setién-Suero E, Neergaard KD, Ferro A, Fatjó-Vilas M, Ríos-Lago M et al (2016) Evidence for trait related theory of mind impairment in first episode psychosis patients and its relationship with processing speed: a 3 year follow-up study. Front Psychol. https://doi.org/10.3389/fpsyg.2016.00592
Oomen PP, Begemann MJH, Brand BA, de Haan L, Veling W, Koops S et al (2021) Longitudinal clinical and functional outcome in distinct cognitive subgroups of first-episode psychosis: a cluster analysis. Psychol Med. https://doi.org/10.1017/s0033291721004153
Reser MP, Allott KA, Killackey E, Farhall J, Cotton SM (2015) Exploring cognitive heterogeneity in first-episode psychosis: what cluster analysis can reveal. Psychiatry Res 229:819–827. https://doi.org/10.1016/j.psychres.2015.07.084
Carrión RE, Walder DJ, Auther AM, McLaughlin D, Zyla HO, Adelsheim S et al (2018) From the psychosis prodrome to the first-episode of psychosis: no evidence of a cognitive decline. J Psychiatr Res. https://doi.org/10.1016/j.jpsychires.2017.10.014
Johansson M, Hjärthag F, Helldin L (2020) Cognitive markers related to long-term remission status in Schizophrenia Spectrum Disorders. Psychiatry Res 289:113035. https://doi.org/10.1016/J.PSYCHRES.2020.113035
Lindgren M, Holm M, Kieseppä T, Suvisaari J (2020) Neurocognition and social cognition predicting 1-year outcomes in first-episode psychosis. Front Psychiatry 11:603933. https://doi.org/10.3389/fpsyt.2020.603933
Subotnik KL, Ventura J, Hellemann GS, Zito MF, Agee ER, Nuechterlein KH (2020) Relationship of poor insight to neurocognition, social cognition, and psychiatric symptoms in schizophrenia: a meta-analysis. Schizophr Res 220:164–171. https://doi.org/10.1016/j.schres.2020.03.038
Davies G, Greenwood K (2020) A meta-analytic review of the relationship between neurocognition, metacognition and functional outcome in schizophrenia. J Ment Health 29:496–505. https://doi.org/10.1080/09638237.2018.1521930
Ferraro L, La Cascia C, La Barbera D, Sanchez-Gutierrez T, Tripoli G, Seminerio F et al (2021) The relationship of symptom dimensions with premorbid adjustment and cognitive characteristics at first episode psychosis: findings from the EU-GEI study. Schizophr Res 236:69–79. https://doi.org/10.1016/j.schres.2021.08.008
Chan KKS (2016) Associations of symptoms, neurocognition, and metacognition with insight in schizophrenia spectrum disorders. Compr Psychiatry 65:63–69. https://doi.org/10.1016/j.comppsych.2015.09.009
Mehta UM, Thirthalli J, Subbakrishna DK, Gangadhar BN, Eack SM, Keshavan MS (2013) Social and neuro-cognition as distinct cognitive factors in schizophrenia: a systematic review. Schizophr Res 148:3–11. https://doi.org/10.1016/j.schres.2013.05.009
Bozikas VP, Giannakou M, Kosmidis MH, Kargopoulos P, Kioseoglou G, Liolios D et al (2011) Insights into theory of mind in schizophrenia: the impact of cognitive impairment. Schizophr Res 130:130–136. https://doi.org/10.1016/j.schres.2011.04.025
Carruthers SP, Van Rheenen TE, Gurvich C, Sumner PJ, Rossell SL (2019) Characterising the structure of cognitive heterogeneity in schizophrenia spectrum disorders. A systematic review and narrative synthesis. Neurosci Biobehav Rev 107:252–78. https://doi.org/10.1016/J.NEUBIOREV.2019.09.006
Amoretti S, Rabelo-da-Ponte FD, Rosa AR, Mezquida G, Sánchez-Torres AM, Fraguas D et al (2021) Cognitive clusters in first-episode psychosis. Schizophr Res 237:31–39. https://doi.org/10.1016/j.schres.2021.08.021
Sauvé G, Malla A, Joober R, Brodeur MB, Lepage M (2018) Comparing cognitive clusters across first- and multiple-episode of psychosis. Psychiatry Res 269:707–718. https://doi.org/10.1016/j.psychres.2018.08.119
Tan EJ, Rossell SL, Subotnik KL, Ventura J, Nuechterlein KH (2022) Cognitive heterogeneity in first-episode psychosis and its relationship with premorbid developmental adjustment. Psychol Med. https://doi.org/10.1017/S0033291721000738
Uren J, Cotton SM, Killackey E, Saling MM, Allott K (2017) Cognitive clusters in first-episode psychosis: Overlap with healthy controls and relationship to concurrent and prospective symptoms and functioning. Neuropsychology. https://doi.org/10.1037/neu0000367
Ochoa S, Lopez-Carrilero R, Luisa Barrigon M, Pousa E, Grasa E, González F et al (2020) S34 effectiveness of individual metacognitive training (MCT+) in first-episode psychosis. Schizophr Bull 46:S44–S45. https://doi.org/10.1093/schbul/sbaa031.100
Kay SR, Fiszbein A, Opler LA (1987) The Positive and Negative Syndrome Scale (PANSS) for Schizophrenia. Schizophr Bull 13:261–276. https://doi.org/10.1093/schbul/13.2.261
Peralta V, Cuesta M (1994) Psychometric properties of the Positive and Negative Syndrome Scale (PANSS) in Schizophrenia. Psychiatry Res 53:31–40. https://doi.org/10.1016/0165-1781(94)90093-0
Emsley R, Rabinowitz J, Torreman M (2003) The factor structure for the Positive and Negative Syndrome Scale (PANSS) in recent-onset psychosis. Schizophr Res 61:47–57. https://doi.org/10.1016/S0920-9964(02)00302-X
Amador XF, Strauss DH, Yale SA, Flaum MM, Endicott J, Gorman JM (1993) Assessment of insight in psychosis. Am J Psychiatry 150:873–873. https://doi.org/10.1176/ajp.150.6.873
Ruiz AI, Pousa E, LastNameDuñó R, Crosas JM, Cuppa S, Garcia-Ribera C (2010) Adaptación al español de la escala de valoración de la no conciencia de trastorno mental (SUMD). Actas Esp Psiquiatr 36:111–119
Endicott J, Spitzer RL, Fleiss JL, Cohen J (1976) The global assessment scale. A procedure for measuring overall severity of psychiatric disturbance. Arch Gen Psychiatry 33:766–71
Gutiérrez-Zotes JA, Valero J, Cortés MJ, Labad A, Ochoa S, Ahuir M et al (2012) Adaptación española de la Escala de Insight Cognitivo de Beck (EICB) en esquizofrénicos. Actas Esp Psiquiatr 40:2–9
Beck A, Baruch E, Balter JM, Steer RA, Warman DM (2004) A new instrument for measuring insight: the beck cognitive insight scale. Schizophr Res 68:319–329. https://doi.org/10.1016/S0920-9964(03)00189-0
Garety PA, Hemsley DR, Wessely S (1991) Reasoning in deluded schizophrenic and paranoid patients: biases in performance on a probabilistic inference task. J Nerv Ment Dis 179:194
Dudley REJ, John CH, Young AW, Over DE (1997) The effect of self-referent material on the reasoning of people with delusions. Br J Clin Psychol 36:575–84. https://doi.org/10.1111/j.2044-8260.1997.tb01262.x
Brett-Jones J, Garety P, Hemsley D (1987) Measuring delusional experiences: a method and its application. Br J Clin Psychol 26:257–265. https://doi.org/10.1111/j.2044-8260.1987.tb01359.x
Díez-Alegría C, Vázquez-Valverde C (2006) Teorías cognitivas de las creencias delirantes. Apunt Psicol 24:51–82
Kinderman P, Bentall RP (1996) A new measure of causal locus: the internal, personal and situational attributions questionnaire. Person Individ Differ 20:261–264. https://doi.org/10.1016/0191-8869(95)00186-7
Huerta-Ramos E, Ferrer-Quintero M, Gómez-Benito J, González-Higueras F, Cuadras D, Del Rey-Mejías AL et al (2021) Translation and validation of Baron Cohen’s face test in a general population from Spain. Actas Esp Psiquiatr 49:106–113
Baron-Cohen S, Wheelwright S, Jolliffe T (1997) Is there a “Language of the Eyes”? Evidence from normal adults, and adults with Autism or Asperger Syndrome. Vis Cogn 4:311–31. https://doi.org/10.1080/713756761
Corcoran R, Mercer G, Frith CD (1995) Schizophrenia, symptomatology and social inference: investigating “theory of mind” in people with schizophrenia. Schizophr Res 17:5–13. https://doi.org/10.1016/0920-9964(95)00024-G
Gil D, Fernández-Modamio M, Bengochea R, Arrieta M (2012) Adaptación al español de la prueba de teoría de la mente Hinting Task. Rev Psiquiatr Salud Ment 5:79–88. https://doi.org/10.1016/j.rpsm.2011.11.004
Wechsler D (1999) WAIS-III: Escala de inteligencia de Wechsler para adultos-III. TEA Ediciones
Sánchez-Cubillo I, Periáñez JA, Adrover-Roig D, Rodríguez-Sánchez JM, Ríos-Lago M, Tirapu J et al (2009) Construct validity of the Trail Making Test: role of task-switching, working memory, inhibition/interference control, and visuomotor abilities. J Int Neuropsychol Soc JINS 15:438–450. https://doi.org/10.1017/S1355617709090626
Spreen O, Strauss E (1998) A compendium of neuropsychological tests: Administration, norms, and commentary, 2nd edn. Oxford University Press, New York
Golden CJ (2001) Test de Colores y Palabras Stroop. TEA Ediciones
Stroop JR (1935) Studies of interference in serial verbal reactions. J Exp Psychol 18:643–662. https://doi.org/10.1037/h0054651
Grant DA, Berg E (1948) A behavioral analysis of degree of reinforcement and ease of shifting to new responses in a Weigl-type card-sorting problem. J Exp Psychol 38:404–411. https://doi.org/10.1037/h0059831
Tien AY, Spevack TV, Jones DW, Pearlson GD, Schlaepfer TE, Strauss ME (1996) Computerized Wisconsin Card Sorting Test: comparison with manual administration. Kaohsiung J Med Sci 12:479–485
Benedet MJ, Alejandre MA (1998) TAVEC. Test de Aprendizaje verbal EspañaComplutense. TEA Ediciones
Delis DC, Freeland J, Kramer JH, Kaplan E (1988) Integrating clinical assessment with cognitive neuroscience: construct validation of the California Verbal Learning Test. J Consult Clin Psychol 56:123–130. https://doi.org/10.1037/0022-006X.56.1.123
Conners CK, Epstein JN, Angold A, Klaric J (2003) Continuous performance test performance in a normative epidemiological sample. J Abnorm Child Psychol 31:555–562. https://doi.org/10.1023/A:1025457300409
Nuechterlein KH, Green MF, Kern RS, Baade LE, Barch DM, Cohen JD et al (2008) The MATRICS consensus cognitive battery, Part 1: test selection, reliability, and validity. Am J Psychiatry 165:203–213. https://doi.org/10.1176/appi.ajp.2007.07010042
Kern RS, Nuechterlein KH, Green MF, Baade LE, Fenton WS, Gold JM et al (2008) The MATRICS consensus cognitive battery, Part 2: Co-norming and standardization. Am J Psychiatry 165:214–220. https://doi.org/10.1176/appi.ajp.2007.07010043
Green MF, Horan WP (2010) Social cognition in schizophrenia. Curr Dir Psychol Sci 19:243–248. https://doi.org/10.1177/0963721410377600
Hassani M, Seidl T (2017) Using internal evaluation measures to validate the quality of diverse stream clustering algorithms. Vietnam J Comput Sci 4:171–183. https://doi.org/10.1007/s40595-016-0086-9
Tomasini C, Borges EN, Machado K, Emmendorfer L (2024) A study on the relationship between internal and external validity indices applied to partitioning and density-based clustering algorithms p. 89–98
Murty GS, Kumar DVV, Naresh T (2012) Learning number of clusters in unlabeled dataset using rotation estimation 2
Bender R, Lange S (2001) Adjusting for multiple testing—when and how? J Clin Epidemiol 54:343–349. https://doi.org/10.1016/S0895-4356(00)00314-0
Landau S, Chis Ster I (2010) Cluster analysis: overview. Int Encycl Educ https://doi.org/10.1016/B978-0-08-044894-7.01315-4
Norusis MJ (2010) Chapter 16: Cluster analysis. PASW Statistics 18 Statistical Procedures Companion (pp. 361–391). Upper Saddle River, NJ: Prentice Hall. n.d
Ayesa-Arriola R, Setién-Suero E, Neergaard KD, Belzunces ÀA, Contreras F, van Haren NEM et al (2018) Premorbid IQ subgroups in first episode non affective psychosis patients: long-term sex differences in function and neurocognition. Schizophr Res 197:370–377. https://doi.org/10.1016/J.SCHRES.2017.12.006
Lewandowski KE, Sperry SH, Cohen BM, Öngür D (2014) Cognitive variability in psychotic disorders: a cross-diagnostic cluster analysis. Psychol Med 44:3239–3248. https://doi.org/10.1017/S0033291714000774
Van Rheenen TE, Lewandowski KE, Tan EJ, Ospina LH, Ongur D, Neill E et al (2017) Characterizing cognitive heterogeneity on the schizophrenia-bipolar disorder spectrum. Psychol Med 47:1848–1864. https://doi.org/10.1017/S0033291717000307
Lee J, Rizzo S, Altshuler L, Glahn DC, Miklowitz DJ, Sugar CA et al (2017) Deconstructing bipolar disorder and schizophrenia: a cross-diagnostic cluster analysis of cognitive phenotypes. J Affect Disord 209:71–79. https://doi.org/10.1016/j.jad.2016.11.030
De la Serna E, Puig O, Mezquida G, Moreno-Izco L, Merchan-Naranjo J, Amoretti S et al (2023) Relationship between cognition and age at onset of first-episode psychosis: comparative study between adolescents, young adults, and adults. Eur Child Adolesc Psychiatry 32:639–649. https://doi.org/10.1007/s00787-021-01901-8
Engen MJ, Simonsen C, Melle I, Færden A, Lyngstad SH, Haatveit B et al (2019) Cognitive functioning in patients with first-episode psychosis stratified by level of negative symptoms: a 1-year follow-up study. Psychiatry Res 281:112554. https://doi.org/10.1016/j.psychres.2019.112554
McEVOY JP, Johnson J, Perkins D, Lieberman JA, Hamer RM, Keefe RSE et al (2006) Insight in first-episode psychosis. Psychol Med 36:1385–1393. https://doi.org/10.1017/S0033291706007793
Saravanan B, Jacob KS, Johnson S, Prince M, Bhugra D, David AS (2010) Outcome of first-episode schizophrenia in India: longitudinal study of effect of insight and psychopathology. Br J Psychiatry 196:454–459. https://doi.org/10.1192/bjp.bp.109.068577
Murray RM, Bhavsar V, Tripoli G, Howes O (2017) 30 Years on: how the neurodevelopmental hypothesis of schizophrenia morphed into the developmental risk factor model of psychosis. Schizophr Bull 43:1190–1196. https://doi.org/10.1093/schbul/sbx121
Pino O, Guilera G, Gómez-Benito J, Najas-García A, Rufián S, Rojo E (2014) Neurodevelopment or neurodegeneration: review of theories of schizophrenia. Actas Esp Psiquiatr 42:185–195
Picchioni MM, Dazzan P (2009) Clinical significance of neurological abnormalities in psychosis. Adv Psychiatr Treat 15:419–427. https://doi.org/10.1192/apt.bp.107.004408
Vaskinn A, Sundet K, Haatveit B (2022) Social cognitive heterogeneity in schizophrenia: a cluster analysis. Schizophr Res Cogn 30:100264. https://doi.org/10.1016/j.scog.2022.100264
Lysaker PH, Gumley A, Luedtke B, Buck KD, Ringer JM, Olesek K et al (2013) Social cognition and metacognition in schizophrenia: evidence of their independence and linkage with outcomes. Acta Psychiatr Scand 127:239–247. https://doi.org/10.1111/ACPS.12012
Buck BE, Healey KM, Gagen EC, Roberts DL, Penn DL (2016) Social cognition in schizophrenia: factor structure, clinical and functional correlates. J Ment Health 25:330–337. https://doi.org/10.3109/09638237.2015.1124397
Lysaker PH, Cheli S, Dimaggio G, Buck B, Bonfils KA, Huling K et al (2021) Metacognition, social cognition, and mentalizing in psychosis: are these distinct constructs when it comes to subjective experience or are we just splitting hairs? BMC Psychiatry 21:329. https://doi.org/10.1186/s12888-021-03338-4
Vita A, Barlati S, Ceraso A, Nibbio G, Ariu C, Deste G et al (2021) Effectiveness, core elements, and moderators of response of cognitive remediation for schizophrenia. JAMA Psychiat 78:848. https://doi.org/10.1001/jamapsychiatry.2021.0620
Firth J, Carney R, French P, Elliott R, Cotter J, Yung AR (2018) Long-term maintenance and effects of exercise in early psychosis. Early Interv Psychiatry 12:578–585. https://doi.org/10.1111/eip.12365
Firth J, Cotter J, Elliott R, French P, Yung AR (2015) A systematic review and meta-analysis of exercise interventions in schizophrenia patients. Psychol Med 45:1343–1361. https://doi.org/10.1017/S0033291714003110
Moritz S, Kerstan A, Veckenstedt R, Randjbar S, Vitzthum F, Schmidt C et al (2011) Further evidence for the efficacy of a metacognitive group training in schizophrenia. Behav Res Ther 49:151–157. https://doi.org/10.1016/j.brat.2010.11.010
Ruiz-Delgado I, Moreno-Küstner B, García-Medina M, Barrigón ML, Gonzalez-Higueras F, López-Carrilero R et al (2022) Is metacognitive training effective for improving neurocognitive function in patients with a recent onset of psychosis? Psychiatry Res 318:114941. https://doi.org/10.1016/j.psychres.2022.114941
Schubert E, Rousseeuw PJ (2019) Faster K-medoids clustering: improving the PAM, CLARA, and CLARANS algorithms. Lect Notes Comput Sci Subser Lect Notes Artif Intell Lect Notes Bioinforma 11807 LNCS:171–87. https://doi.org/10.1007/978-3-030-32047-8_16/FIGURES/6
Corral L, Labad J, Ochoa S, Cabezas A, Muntané G, Valero J et al (2021) Cognitive Biases Questionnaire for Psychosis (CBQp): Spanish Validation and Relationship With Cognitive Insight in Psychotic Patients. Front Psychiatry. https://doi.org/10.3389/fpsyt.2020.596625
Acknowledgements
The authors are grateful to the patients for their generosity of time and effort. Furthermore, the authors would like to thank all the members of the Spanish Metacognition Study Group for their valuable advice in this work.
Funding
This work was supported by the Instituto de Salud Carlos III, Spanish Government (PI11/01347, PI14/00044, PI18/ 00212), the Fondo Europeo de Desarrollo Regional (FEDER), Health Department of Catalonia, PERIS call (SLT006/17/00231), Progress and Health Foundation of the Andalusia, (PI-0634/2011 and PI-0193/2014), and Obra Social La Caixa (RecerCaixa call 2013). They were not involved in the study design, collection, analysis and interpretation of the data, in the writing of the manuscript, or the decision to submit it for publication.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
All individuals were given an informative sheet, and all of them signed an informed consent file for participation in this study. The Ethics Committee of each participating center approved this project. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Espinosa, V., Bagaeva, A., López-Carrilero, R. et al. Neuropsychological profiles in first-episodes psychosis and their relationship with clinical, metacognition and social cognition variables. Eur Arch Psychiatry Clin Neurosci (2024). https://doi.org/10.1007/s00406-024-01813-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00406-024-01813-z