Abstract
In this study, we examined autism spectrum disorder (ASD) symptoms in adults with attention-deficit hyperactivity disorder (ADHD) in comparison with normal controls using the Autism Diagnostic Observation Schedule, Second Edition (ADOS-2). Sixty-three adults with ADHD (mean age, 35.3 years; 38 men) and 31 controls (mean age, 38.7 years; 17 men) completed Module 4 of the ADOS-2, Autism Spectrum Quotient, Conners’ Adult ADHD Rating Scale, and Wechsler Adult Intelligence Scale, Third Edition. Adults with ADHD were not clinically diagnosed with ASD, and their ADHD diagnosis was based on the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition criteria. Between-group comparisons on the scoring patterns revealed significant ASD symptoms present in adults with ADHD, which was congruent with our previous study. Further, item level and correlation analyses showed the possibility that ASD symptoms in adult ADHD comprised of two distinct mechanisms, one qualitatively similar to ASD and the other arising from ADHD characteristics, highlighting the complex nature of ADHD-ASD symptom overlaps.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the heat of ever-growing interests in adult attention-deficit/hyperactivity disorder (ADHD) among researchers, one of the unsolved questions is how ADHD is related to autism spectrum disorder (ASD). While the former is characterized by inattention, hyperactivity, and impulsivity, the latter presents deficits in social reciprocal communication and restricted and repetitive behaviors or interests [1]. Despite the long history of being considered as etiologically different and mutually exclusive disorders, literature now suggests that ADHD and ASD frequently co-occur [2,3,4,5], and their overlaps may not be limited to symptoms but extend to shared genetic underpinnings [3, 6,7,8]. In studies with children, up to 50% of individuals with ADHD were found to exhibit some form of ASD symptoms [3,4,5, 9, 10], and importantly, the co-existing ASD symptoms affect various clinical and functional outcomes of children with ADHD [11, 12]. For example, Cooper et al. [11] found that higher levels of autistic traits were associated with more severe ADHD symptoms, more psychiatric comorbidities, lower intelligence quotient (IQ), and greater motor and language problems in children with ADHD. Given the complex social pressure in adult life and the effect of developmental sequel, it is possible that adults with ADHD may be more affected by ASD symptoms compared to children, and their consequences can be profound. It is, therefore, crucial to evaluate the detailed profile and extent of ASD symptoms exhibited by adults with ADHD. However, only a handful of studies have examined such status in adults, and none used objective clinical measures. This led to our previous study, which examined ASD symptoms in adults with ADHD using the Autism Diagnostic Observation Schedule, Second Edition (ADOS-2) [13].
The ADOS-2 is a standardized, well-validated measure for assessing ASD symptoms and diagnosis [14]. Its uniqueness lies in the objectivity, thoroughness, and reliability of the assessment, in which a well-trained administrator performs a direct observation of an individual in a series of semi-structured plays and interviews. Adults with fluent speech receive Module 4, consisting of 15 activities that are rated using 32 items based on specific evaluation criteria. The scores of the specific items are converted into domain scores and then to a final algorithm score, which determines ASD or non-ASD classification. Both the original and recently revised Module 4 algorithms have good sensitivity (original, 89.6; revised, 90.5) and specificity (original, 72.2; revised: 82.2) [15]. While the original algorithm produces two domain scores: Communication and Reciprocal Social Interaction (RSI), the revised algorithm yields three domain scores: Language and Communication (LC), RSI, and Restricted and Repetitive Behaviors (RRB). The sum of LC and RSI is called Social Affect (SA), and together with RRB, it matches the two domain ASD criteria of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) [1].
In our preliminary study, we examined ASD symptoms in a clinical sample of adults with ADHD using the ADOS-2 for the first time [13]. Though the participants showed no apparent ASD symptoms in childhood and therefore were not clinically diagnosed with ASD, 23.3% met the ASD cut-off on the ADOS-2. While the domain level analysis showed significant scoring on SA than RRB items, indicating the evident challenges in communication and reciprocal social interaction in adults with ADHD, item-level analysis revealed that (1) the quantitative rather than qualitative aspect of social communication is compromised in individuals with ADHD and (2) emotion processing and affective interaction may also be impaired in ADHD. These results were in line with previous studies, indicating that individuals with ADHD tend to be one-sided in their communication and lack relational reciprocity [9, 16,17,18,19]. Difficulty in affect recognition [20] and lack of emotional empathy [21] are also identified in individuals with ADHD. While our study successfully revealed the profile of more objective ASD symptoms present in adults with ADHD, only those with ADHD were involved, and as many as 11 items were endorsed by more than 30% of the sample. A comparative study was necessary to obtain a better picture of ASD symptoms in adults with ADHD. Therefore, we conducted this consecutive study in which the scoring patterns on the ADOS-2 were compared between adults with ADHD and healthy controls.
Methods
Participants
Participants were recruited from outpatients and inpatients at Showa University Karasuyama Hospital, central Tokyo, Japan, between October 2018 and December 2020. Only those fulfilling the DSM-5 criteria [1] for ADHD and with a full-scale IQ of over 85 on the Japanese version of the Wechsler Adult Intelligence Scale-Third Edition (WAIS-III) [22] were included. The exclusion criteria included age < 17 years and the presence of psychiatric disorders other than ADHD according to the DSM-5 criteria. The control group of neurotypical individuals was recruited from volunteer relatives or acquaintances of healthcare workers. In addition to the abovementioned exclusion criteria, those diagnosed with psychiatric disorders according to the DSM-5 criteria and those having first-order relatives with neurodevelopmental disorders were not included as controls. The final sample comprised 63 adults (56 from our previous study [13]) with ADHD and 31 neurotypical individuals.
Procedure
The detailed diagnostic process is explained elsewhere [13]. All participants were individually administered a comprehensive clinical assessment battery, which included (1) two sets of thorough interviews with participants and their caretakers on their developmental and medical history by psychiatrists, (2) the Japanese version of Conners’ Adult ADHD Diagnostic Interview for DSM-IV (CAADID) [23, 24], and (3) reviews of the maternity record books and the elementary school (6–12 years old) reports. The final diagnosis of ADHD and comorbid mental disorders, if any, was determined by a consensus between psychiatrists and psychologists who were involved in this process based on the DSM-5 criteria. ASD diagnosis was a good clinical estimate (GCE), as neither the ADOS-2 nor the Autism Diagnostic Interview-Revised (ADI-R) [25] was employed in the assessment process. However, all psychiatrists and psychologists involved in this study are experts in neurodevelopmental disorders, and the clinical assessment was thorough with multiple resources. Individuals with a GCE of ASD diagnosis were excluded.
Control group participants underwent a 30-min interview with a psychiatrist to confirm the absence of mental disorders in themselves and any neurodevelopmental disorder in their first-order relatives.
The above diagnostic process was followed by the administration of the following assessments to all participants: (1) the ADOS-2 Module 4; (2) the Japanese version of the Conners’ Adult ADHD Rating Scale-Self-Report: Long Version (CAARS-S:L-J), in which subjective ADHD symptoms were evaluated [26, 27]; (3) the autism-spectrum quotient (AQ) for measuring subjective ASD symptoms [28, 29], in which 33 is considered as the suitable cut-off [29]; and (4) the Japanese version of WAIS-III [22] to evaluate the IQ profile.
Autism diagnostic observation schedule, second edition
The ADOS-2 Module 4 (standard and all optional activities) was administered by trained and certified psychiatrists, and both the original (cut-off of 7) and revised algorithms (clinical cut-off of 8 and research cut-off of 10 [15]) were scored. Details of the administration and scoring processes are explained in the previous study, and our administration has a good inter-rater reliability [13].
Statistical analysis
All statistical analyses were performed using SPSS version 25.0 (IBM Corp., Armonk, NY, USA). The ADHD and control groups were compared on demographic (age, sex, and years of education) and clinical measurements (AQ, CAARS scores, and WAIS-III), as well as on the domain scores and proportions of individuals meeting the ASD cut-offs of the original and revised ADOS-2 algorithms. Independent samples t tests and chi-square tests were used for continuous and categorical variables, respectively. For detailed item-level analysis, the proportion of participants with each score (0, 1, 2, 3, 7, and 8) on every ADOS-2 item was calculated for the two groups. Additionally, the proportion of non-zero scorers (those who scored 1, 2, 3, or 8 for B3) on each ADOS-2 item was compared between the ADHD and control groups using chi-square tests. To assess the relationship between each measurement, Pearson product-moment correlation coefficients were calculated between AQ, CAARS, WAIS-III, and ADOS-2 (revised algorithm) scores for the groups. Further, we created a “typical ADHD” group by excluding participants who met the ASD cut-off (clinical cut-off of the revised algorithm) from the ADHD group and compared the characteristics between this group and the control group by performing the abovementioned analyses. The significance level was set at 0.05, except for the correlations, for which 0.01 was set to account for the possibility of a type I error.
Ethics
This study was evaluated and approved by the Medical Ethics Committee of Showa University School of Medicine, and the protocols were carried out in accordance with the Declaration of Helsinki. Written informed consent was obtained from all included participants after information was provided about the study.
Results
Demographic and clinical characteristics
A total of 94 participants, 63 adults with ADHD (mean [M] age, 35.3 years, standard deviation [SD] = 10.9 years, 38 men) and 31 controls (M age, 38.7 years, SD = 6.7 years, 17 men) were analyzed and compared. Their demographics and clinical characteristics are shown in Table 1. The background demographics showed no group differences, in which both groups had relatively high average years of education and slight male preponderance. The average full, verbal, and performance IQs were all above 100 and showed no group differences. The ADHD group scored significantly higher than the control group not only on all CAARS scales but also on AQ total score. The typical ADHD group comprised of 48 individuals (M age, 35.3 years, SD = 11.1 years, 29 men), and its comparison against controls took the same pattern as that of the ADHD group, with no background differences but significantly higher AQ and CAARS scores than the control group (Table 2).
ADOS-2 domain and total scores
Table 3 shows the final scores of the original and revised ADOS-2 algorithms and the percentage of individuals who met the ASD cut-off. While no one in the control group met the ASD cut-off, a total of 11 (17.5%), 15 (23.8%), and 11 (17.5%) individuals met the ASD cut-off by the original, revised clinical, and revised research algorithms, respectively, in the ADHD group. All average domain and total scores of the original and revised algorithms were significantly higher in the ADHD group than those in controls.
Comparison between adults with ADHD and controls on ADOS-2 scoring
Table 4 compares the proportion of non-zero scorers on each ADOS-2 item between the ADHD and control groups. Eleven items differentiated the two groups, in which adults with ADHD scored significantly higher than controls on A2 (Speech Abnormalities Associated With Autism), A8 (Conversation), B1 (Unusual Eye Contact), B2 (Facial Expressions Directed to Examiner), B3 (Language Production and Linked Nonverbal Communication), B4 (Shared Enjoyment in Interaction), B7 (Insight Into Typical Social Situations and Relationships), B8 (Responsibility), B9 (Quality of Social Overtures), B11 (Quality of Social Response), and B12 (Amount of Reciprocal Social Communication). No item in C (Imagination), D (Stereotyped Behaviors and Restricted Interests), and E (Other Abnormal Behaviors) domains showed a significant group difference.
Table 5 compares the proportion of non-zero scorers on each ADOS-2 item between the typical ADHD and control groups. Only six items differentiated the two groups: A2, B2, B3, B4, B8, and B12. A8, B1, B7, B9, and B11 no longer showed significant differences between the typical ADHD and control groups.
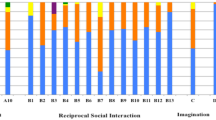
The detailed scoring patterns on each ADOS-2 item by the groups are depicted in Fig. 1a (A: Language and Communication), Fig. 1b (B: Reciprocal Social Interaction), and Fig. 1c (C: Imagination, D: Stereotyped Behaviors and Restricted Interests, E: Other Abnormal Behaviors). Fifty-two (82.5%) individuals in the ADHD group scored on at least two of A, B, and D domains, which are relatively important for the algorithm.
Percentage of scores on each ADOS-2 item for the ADHD, typical ADHD, and control groups of a Language and Communication (A); b Reciprocal Social Interaction (B); and c Imagination (C), Stereotyped Behaviors and Restricted Interests (D), and Other Abnormal Behaviors (E) domains. t-ADHD typical ADHD, commun: communication; behav: behaviors. *See Tables 4, 5 for full item names
Relationship between the ADOS-2 and other clinical measures
Tables 6, 7, and 8 show the correlations between AQ, CAARS, WAIS-III, and ADOS-2 scores in the ADHD, control, and typical ADHD groups, respectively. While ADOS-2 scores did not correlate with AQ, CAARS, or WAIS-III in the ADHD and control groups, ADOS-2 RSI, SA, and total scores positively correlated with CAARS Impulsivity/Emotional Lability score in the typical ADHD group. ADOS-2 intra-scale correlations also differed between the groups, in which RRB scores were correlated with LC, RSI, and SA scores only in the ADHD group.
Discussion
This is a consecutive study to our first attempt in evaluating ASD symptoms in clinical adults with ADHD using the ADOS-2. Herein, we compared the scoring patterns on the ADOS-2 between adults with ADHD who were not clinically diagnosed with ASD and typically developing controls. The overall analysis clearly indicated that adults with ADHD have significant ASD symptoms, particularly impairment in reciprocal social interaction. Item-level and correlational analyses revealed impairment in both qualitative and quantitative aspects of social communication; however, ASD symptoms in ADHD may not be a uniform construct.
Consistent with our preliminary study, approximately 20% of adults with ADHD without apparent childhood ASD symptoms met the ASD cut-off on the ADOS-2 as opposed to none among the controls. Moreover, all domain and final algorithm scores were significantly higher in individuals with ADHD than the controls. These results clearly indicate that adults with ADHD have symptoms, similar to ASD, of both impairment in social communication and restricted and repetitive behaviors and interests. It is possible that, as some researchers postulate, ADHD and ASD are overarching disorders [30, 31], and since the manifestation of symptoms change along with development [32], one might exhibit ASD symptoms later in adulthood despite the absence of overt ASD symptoms in childhood. It also adds support to the shared biological underpinnings between the two disorders as reported by previous researchers [3, 6,7,8]. Additionally, another possibility is that ADHD symptoms and their related issues, such as being rejected by others [33,34,35,36,37,38] or being involved in bullying [39], can give rise to ASD-like symptoms later in adulthood as they had been deprived of the appropriate opportunity and experience of developing healthy social interaction skills.
The item-level analysis between the ADHD and control groups suggested that adults with ADHD have difficulties not only in quantitative but also qualitative aspects of social communication. Most RSI items and some LC items were significantly more frequently endorsed by the ADHD group than the control group. These included A2 (Speech Abnormalities Associated With Autism), A8 (Conversation), B1 (Unusual Eye Contact), B2 (Facial Expressions Directed to Examiner), B3 (Language Production and Linked Nonverbal Communication), B7 (Insight Into Typical Social Situations and Relationships), B9 (Quality of Social Overtures), B11 (Quality of Social Response), and B12 (Amount of Reciprocal Social Communication). While the same reasons described in our previous study were behind the high scoring on A2, A8, and B12 (they tended to be loud and communicate in a one-sided manner such as expressing their own interest without paying much attention to the examiner), the current study also revealed that in comparison to controls, they were also impaired in the appropriate use of eye contact and its coordination in social communication (B1, B2, B3, B9, and B11). Further, these impairments are also reflected in the high B7 scoring. The vicious circle of peer rejection, reduced interaction, and poor social skills [40,41,42] likely damages the quality and understanding of social relationships. Theory of mind deficits [43, 44] may also contribute to the difficulties in social communication and grasping the nature of typical relationships.
Interestingly however, when we excluded those with overt ASD symptoms (i.e., those who met ASD cut-off on ADOS-2) and compared “typical ADHD” against controls, most of the qualitative items were no longer endorsed more frequently in the ADHD than control group. Such items include B1 (Unusual Eye Contact), B9 (Quality of Social Overtures), and B11 (Quality of Social Response). The quantitative item; B12 (Amount of Reciprocal Social Communication), affect-related items; B2 (Facial Expressions Directed to Examiner) and B4 (Shared Enjoyment in Interaction), and the sense of responsibility (B8) remained significantly different than controls in endorsement frequency. This possibly suggests that ASD symptoms present in ADHD are not on a quantitative continuum to ASD but rather a mixture of distinct mechanisms. One is qualitatively the same as those with ASD, in which the integration of different forms of social communication is significantly impaired. The other is the resulting symptom resemblance of ADHD to ASD, in which inattentive or impulsive communication wrecks reciprocal interaction, rather more quantitatively (one-sided communication style). The correlation patterns support this point as only the “typical ADHD” group showed CAARS-ADOS-2 correlations, especially between impulsivity and reciprocal social interaction, whereas the overall ADHD group did not. Further, non-identical SA-RRB relationships in these two groups may describe the heterogeneity of ASD symptoms in adults with ADHD. It is possible that the entanglement of these mechanisms may become more prominent in adulthood than in childhood as they mutually potentiate along development and experiences. Our study highlights the importance of careful and detailed assessment of ASD symptoms and co-occurrence in ADHD as they may not be homogenous in nature. Recent neuroimaging studies have identified several overlapping and distinctive cortical and subcortical structural changes between ADHD and ASD [32, 45, 46]. In particular, O’Dwyer et al. [47] studied the neural structural correlates of ASD-like symptoms in ADHD, in which the volumes of caudate nucleus and globus pallidus were found to play a critical role. Although the results remain inconsistent and are subject to longitudinal evaluation, it is crucial to examine these biological markers in combination with clinical symptoms in future studies.
In contrast to our previous work, we did not find as much evidence as we did before, for the difficulties with affective interaction in adults with ADHD. The only emotion-related items that were scored significantly more likely by the ADHD than control groups were B2 (Facial Expressions Directed to Examiner) and B4 (Shared Enjoyment in Interaction). Other items such as A10 (Emphatic or Emotional Gestures), B5 (Communication of Own Affect), and B6 (Comments on Others’' Emotions/Empathy), which were frequently endorsed by adults with ADHD in the preliminary work, did not show any group differences in the scoring patterns. Given the frequent scoring by controls on B5, B6, and B10, cultural factors may have skewed these results. In Japan, it is often considered impolite to ask personal questions to examiners or others at the first meeting, and the Japanese have fewer and more covert expressions of emotions and body language than the Western culture [48, 49]. It is possible that these factors masked the group differences, including A6 (Asks for Information), and further cross-cultural studies are necessary to evaluate affect-related difficulties in ADHD. Further, given the relatively lower percentage of adults with ADHD scoring positive on all D domain (Stereotyped Behaviors and Restricted Interests) items, A3 (Immediate Echolalia), and A4 (Stereotyped/Idiosyncratic Use of Words or Phrases), these particularities in behavior and language may play a critical role in differentiating between ADHD and ASD. However, the absence of group differences on these items may be due to the small number of positive cases. Since RRB may not be fully observed during the limited time in the ADOS-2 [15], the combined use of other RRB evaluations is ideal in exploring these possible distinctive symptoms in ADHD and ASD.
The current study has some limitations. First, since the exclusion of ASD was based on the clinical diagnosis without the ADOS-2 and ADI-R, adults with ASD may have been included amiss. However, this effect should be small as we conducted a thorough examination of developmental history from multiple resources. Second, we did not use medication-naïve individuals, and psychotropic medications could have altered the ADOS-2 performance. A further study on a larger non-medicated ADHD sample is currently planned. Finally, there are reports on a rather limited specificity of the ADOS-2 Module 4 [50]. Although we believe that the ADOS-2 was appropriate in our study to identify the details of ASD symptoms, further evaluations of validity, especially on items that may have cultural effects, are warranted.
In conclusion, the present comparative study using the ADOS-2 demonstrated that adults with ADHD have significant ASD symptoms compared with neurotypical controls. While both qualitative and quantitative aspects of social communication were found impaired, similar to in those with ASD, the results highlighted the heterogeneous nature of ASD symptoms present in adult ADHD.
Availability of data and material
Research data will not be shared as the participants did not consent for open data sharing.
Code availability
Not applicable.
References
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatric Association, Washington, DC
Simonoff E, Pickles A, Charman T, Chandler S, Loucas T, Baird G (2008) Psychiatric disorders in children with autism spectrum disorders: prevalence, comorbidity, and associated factors in a population-derived sample. J Am Acad Child Adolesc Psychiatry 47:921–929. https://doi.org/10.1097/chi.0b013e318179964f
Ronald A, Simonoff E, Kuntsi J, Asherson P, Plomin R (2008) Evidence for overlapping genetic influences on autistic and ADHD behaviours in a community twin sample. J Child Psychol Psychiatry 49:535–542. https://doi.org/10.1111/j.1469-7610.2007.01857.x
Ames C, White S (2011) Brief report: are ADHD traits dissociable from the autistic profile? Links between cognition and behavior. J Autism Dev Disord 41:357–363. https://doi.org/10.1007/s10803-010-1049-0
Leyfer OT, Folstein SE, Bacalman S, Davis NO, Dinh E, Morgan J, Tager-Flusberg H, Lainhart JE (2006) Comorbid psychiatric disorders in children with autism: interview development and rates of disorders. J Autism Dev Disord 36:849–861. https://doi.org/10.1007/s10803-006-0123-0
Reiersen AM, Constantino JN, Grimmer M, Martin NG, Todd RD (2008) Evidence for shared genetic influences on self-reported ADHD and autistic symptoms in young adult Australian twins. Twin Res Hum Genet 11:579–585. https://doi.org/10.1007/s10803-010-1135-3
Rommelse NN, Franke B, Geurts HM, Hartman CA, Buitelaar JK (2010) Shared heritability of attention-deficit/hyperactivity disorder and autism spectrum disorder. Eur Child Adolesc Psychiatry 19:281–195. https://doi.org/10.1007/s00787-010-0092-x
Mulligan A, Anney RJ, O’Regan M, Chen W, Butler L, Fitzgerald M, Buitelaar J, Steinhausen HC, Rothenberger A, Minderaa R, Nijmeijer J, Hoekstra PJ, Oades RD, Roeyers H, Buschgens C, Christiansen H, Franke B, Gabriels I, Hartman C, Kuntsi J, Marco R, Meidad S, Mueller U, Psychogiou L, Rommelse N, Thompson M, Uebel H, Banaschewski T, Ebstein R, Eisenberg J, Manor I, Miranda A, Mulas F, Sergeant J, Sonuga-Barke E, Asherson P, Faraone SV, Gill M (2009) Autism symptoms in attention-deficit/hyperactivity disorder: a familial trait which correlates with conduct, oppositional defiant, language and motor disorders. J Autism Dev Disord 39:197–209. https://doi.org/10.1007/s10803-008-0621-3
Reiersen AM, Constantino JN, Volk HE, Todd RD (2007) Autistic traits in a population-based ADHD twin sample. J Child Psychol Psychiatry 48:464–472. https://doi.org/10.1111/j.1469-7610.2006.01720.x
Grzadzinski R, Di Martino A, Brady E, Mairena MA, O’Neale M, Petkova E, Lord C, Castellanos FX (2011) Examining autistic traits in children with ADHD: does the autism spectrum extend to ADHD? J Autism Dev Disord 41:1178–1191. https://doi.org/10.1007/s10803-010-1135-3
Cooper M, Martin J, Langley K, Hamshere M, Thapar A (2014) Autistic traits in children with ADHD index clinical and cognitive problems. Eur Child Adolesc Psychiatry 23:23–34. https://doi.org/10.1007/s00787-013-0398-6
Mattard-Labrecque C, Ben Amor L, Couture MM (2013) Children with autism and attention difficulties: a pilot study of the association between sensory, motor, and adaptive behaviors. J Can Acad Child Adolesc Psychiatry 22:139–146
Hayashi W, Hanawa Y, Yuriko I, Aoyagi K, Saga N, Nakamura D, Iwanami A (2021) ASD symptoms in adults with ADHD: a preliminary study with the ADOS-2. Eur Arch Psychiarty Clin Neurosci. https://doi.org/10.1007/s00406-021-01250-2
Lord C, Rutter M, DiLavore P, Risi S, Gotham K, Bishop S (2012) Autism Diagnostic Observation Schedule, Second Edition (ADOS-2) Manual (Part I): Modules 1–4. Western Psychological Services, Torrance, CA
Hus V, Lord C (2014) The autism diagnostic observation schedule, module 4: revised algorithm and standardized severity scores. J Autism Dev Disord 44:1996–2012. https://doi.org/10.1007/s10803-014-2080-3
Clark T, Feehan C, Tinline C, Vostanis P (1999) Autistic symptoms in children with attention deficit-hyperactivity disorder. Eur Child Adolesc Psychiatry 8:50–55. https://doi.org/10.1007/s007870050083
Biederman J, Faraone SV, Mick E, Williamson S, Wilens TE, Spencer TJ, Weber W, Jetton J, Kraus I, Pert J, Zallen B (1999) Clinical correlates of ADHD in females: findings from a large group of girls ascertained from pediatric and psychiatric referral sources. J Am Acad Child Adolesc Psychiatry 38:966–975. https://doi.org/10.1097/00004583-199908000-00012
Greene RW, Biederman J, Faraone SV, Ouellette CA, Penn C, Griffin SM (1996) Toward a new psychometric definition of social disability in children with attention-deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 35:571–578. https://doi.org/10.1097/00004583-199605000-00011
Hoza B, Mrug S, Gerdes AC, Hinshaw SP, Bukowski WM, Gold JA, Kraemer HC, Pelham WE Jr, Wigal T, Arnold LE (2005) What aspects of peer relationships are impaired in children with attention-deficit/hyperactivity disorder? J Consult Clin Psychol 73:411–423. https://doi.org/10.1037/0022-006x.73.3.411
Miller M, Hanford RB, Fassbender C, Duke M, Schweitzer JB (2011) Affect recognition in adults with ADHD. J Atten Disord 15:452–460. https://doi.org/10.1177/1087054710368636
Groen Y, den Heijer AE, Fuermaier ABM, Althaus M, Tucha O (2018) Reduced emotional empathy in adults with subclinical ADHD: evidence from the empathy and systemizing quotient. Atten Defic Hyperact Disord 10:141–150. https://doi.org/10.1007/s12402-017-0236-7
Wechsler D (1997) Wechsler Adult Intelligence Scale, 3rd edn. The Psychological Corporation, San Antonio
Epstein JN, Johnson DE, Conners CK (2001) Conners’ adult ADHD diagnostic interview for DSM-IV (CAADID): technical manual. Multi-Health Systems, Toronto
Nakamura K (2012) CAADID. Japanese edition manual. Tokyo, KanekoShobo
Lord C, Rutter M, Le Couteur A (1994) Autism diagnostic interview-revised: a revised version of a diagnostic interview for caregivers of individuals with possible pervasive developmental disorders. J Autism Dev Disord 24:659–685. https://doi.org/10.1007/bf02172145
Conners CK, Erhardt D, Sparrow E (1999) Conners’ adult ADHD rating scales (CAARS)-technical manual. Multi-Health Systems, North Tonawanda, New York
Nakamura K (2012) CAARS. Japanese edition manual. Tokyo, KanekoShobo
Baron-Cohen S, Wheelwright S, Skinner R, Martin J, Clubley E (2001) The autism-spectrum quotient (AQ): evidence from Asperger syndrome/high-functioning autism, males and females, scientists and mathematicians. J Autism Dev Disord 31:5–17. https://doi.org/10.1023/a:1005653411471
Wakabayashi A, Tojo Y, Baron-Cohen S, Wheelwright S (2004) The Autism-Spectrum Quotient (AQ) Japanese version: evidence from high-functioning clinical group and normal adults. Shinrigaku Kenkyu 75:78–84. https://doi.org/10.4992/jjpsy.75.78
Rommelse NN, Geurts HM, Franke B, Buitelaar JK, Hartman CA (2011) A review on cognitive and brain endophenotypes that may be common in autism spectrum disorder and attention-deficit/hyperactivity disorder and facilitate the search for pleiotropic genes. Neurosci Biobehav Rev 35:1363–1396. https://doi.org/10.1016/j.neubiorev.2011.02.015
Van der Meer JM, Oerlemans AM, van Steijn DJ, Lappenschaar MG, de Sonneville LM, Buitelaar JK, Rommelse NN (2012) Are autism spectrum disorder and attention-deficit/hyperactivity disorder different manifestations of one overarching disorder? Cognitive and symptom evidence from a clinical and population-based sample. J Am Acad Child Adolesc Psychiatry 51:1160-1172.e3. https://doi.org/10.1016/j.jaac.2012.08.024
Rommelse NN, Buitellar JK, Hartman CA (2017) Structural brain imaging correlates of ASD and ADHD across the lifespan: a hypothesis-generating review on developmental ASD-ADHD subtypes. J Neural Transm (Vienna) 124:259–271. https://doi.org/10.1007/s00702-016-1651-1
Nijmeijer JS, Minderaa RB, Buitelaar JK, Mulligan A, Hartman CA, Hoekstra PJ (2008) Attention-deficit/hyperactivity disorder and social dysfunctioning. Clin Psychol Rev 28:692–708. https://doi.org/10.1016/j.cpr.2007.10.003
Hoza B (2007) Peer functioning in children with ADHD. J Pediatr Psychol 32:655–663. https://doi.org/10.1093/jpepsy/jsm024
Flicek M (1992) Social status of boys with both academic problems and attention-deficit hyperactivity disorder. J Abnorm Child Psychol 20:353–366. https://doi.org/10.1007/bf00918981
De Boo GM, Prins PJM (2007) Social incompetence in children with ADHD: possible moderators and mediators in social-skills training. Clin Psychol Rev 27:78–97. https://doi.org/10.1016/j.cpr.2006.03.006
Ronk MJ, Hund AM, Landau S (2011) Assessment of social competence of boys with attention-deficit/hyperactivity disorder: problematic peer entry, host responses, and evaluations. J Aborm Child Pyschol 39:829–840. https://doi.org/10.1007/s10802-011-9497-3
Bagwell CL, Molina BS, Pelham WE Jr, Hoza B (2001) Attention-deficit hyperactivity disorder and problems in peer relations: predictions from childhood to adolescence. J Am Acad Child Adolesc Psychiatry 40:1285–1292. https://doi.org/10.1097/00004583-200111000-00008
Chou WJ, Liu TL, Yang P, Yen CF, Hu HF (2018) Bullying victimization and perpetration and their correlates in adolescents clinically diagnosed with ADHD. J Atten Disord 22:25–34. https://doi.org/10.1177/1087054714558874
Mrug S, Brooke SG, Molina B, Hoza AC, Gerdes SP, Hinshaw L, Hechtman L, Eugene A (2012) Peer rejection and friendships in children with attention deficit/hyperactivity disorder: contributions to long-term outcomes. J Abnorm Child Psychol 40:1013–1026. https://doi.org/10.1007/s10802-012-9610-2
Mikami AY, Hinshaw SP (2003) Buffers of peer rejection among girls with and without ADHD: the role of Popularity with adults and goal-directed solitary play. J Abnorm Child Psychol 31:381–397. https://doi.org/10.1023/a:1023839517978
Kok FM, Groen Y, Fuermaier AB, Tucha O (2016) Problematic peer functioning in girls with ADHD: a systematic literature review. PLoS ONE 11:e0165119. https://doi.org/10.1371/journal.pone.0165119
Maoz H, Gvirts HZ, Sheffer M, Bloch Y (2019) Theory of mind and empathy in children with ADHD. J Atten Disord 23:1331–1338. https://doi.org/10.1177/1087054717710766
Buitelaar JK, van der Wees M, Swaab-Barneveld H, van der Gaag RJ (1999) Theory of mind and emotion-recognition functioning in autistic spectrum disorders and in psychiatric control and normal children. Dev Psychopathol 11:39–58. https://doi.org/10.1017/s0954579499001947
Hoogman M, van Rooij D, Klein M et al (2020) Consortium neuroscience of attention deficit/hyperactiveity disorder and autism spectrum disorder: the ENIGMA adventure. Hum Brain Mapp. https://doi.org/10.1002/hbm.25029
Boedhoe P, van Rooij D, Hoogman M et al (2020) Subcortical brain volume, regional cortical thickness and cortical surface area across attention-deficit/hyperactivity disorder (ADHD), autism spectrum disorder (ASD), and obsessive-compulsive disorder (OCD). Am J Psychiatry 177:834–843. https://doi.org/10.1176/appi.ajp.2020.19030331
O’Dwyer L, Tanner C, van Dongen EV, Greven CU, Bralten J, Zwiers MP et al (2016) Decreased left caudate volume is associated with increased severity of autistic-like symptoms in a cohort of ADHD patients and their unaffected siblings. PLoS ONE 11:e0165620. https://doi.org/10.1371/journal.pone.0165620
IES (2021). Cultural Atlas, Japanese culture at: https://culturalatlas.sbs.com.au/japanese-culture/japanese-culture-communication#japanese-culture. Accessed 4 July 2021
Taylor HM (1975). Americans and Japanese Nonverbal Communication. Linguistic Communications. Japanese Linguistics 3 at https://files.eric.ed.gov/fulltext/ED119489.pdf. Accessed 4 July 2021
Pugliese CE, Kenworthy L, Hus V, Wallace GL, Yerys BE, Maddox BB, White SW, Popal H, Armour AC, Miller J, Herrington JD, Schultz RT, Martin A, Anthony LG (2015) Replication and comparison of the newly proposed ADOS-2, Module 4 algorithm in ASD without ID: a multi-site study. J Autism Dev Disord 45:3919–3931. https://doi.org/10.1007/s10803-015-2586-3
Acknowledgments
We would like to thank Dr. Atsushi Harano for giving us helpful advice on psychological testing. Moreover, we would like to express our thanks to Professor Miho Kuroda, at the Department of Psychology of Teikyo University, for teaching us ADOS-2 administration. We thank Editage for English language editing.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
This study was evaluated and approved by the Medical Ethics Committee of Showa University School of Medicine, and the protocols were carried out in accordance with the Declaration of Helsinki.
Consent to participate
Written informed consent was obtained from all participants included in the study after information was provided about the study.
Consent for publication
Not applicable.
Rights and permissions
About this article
Cite this article
Hayashi, W., Hanawa, Y., Saga, N. et al. ASD symptoms in adults with ADHD: a comparative study using ADOS-2. Eur Arch Psychiatry Clin Neurosci 272, 1481–1494 (2022). https://doi.org/10.1007/s00406-021-01362-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00406-021-01362-9