Abstract
Introduction
Superior semicircular canal dehiscence syndrome (SCDS) is a clinical syndrome that can cause instability, vertigo, fullness, tinnitus, autophony, hearing loss (HL), Tullio phenomenon, or Hennebert’s sign. Historically, surgery has been the primary treatment reported in the literature, although some medical treatments may also be proposed. This study aims to comprehensively characterize SCDS in a large series of patients from clinical, auditory, and vestibular perspectives, and explore medical alternatives to conventional surgical treatments by comparing their results and evolution.
Methods
A retrospective observational study was designed in a tertiary care center. Audiovestibular tests evaluated included pure-tone audiometry (PTA), VEMPs, video head impulse test (vHIT), and CT imaging. Improvement was assessed over a follow-up period of up to 6 months for seven cardinal symptoms to verify the efficacy of the proposed treatments.
Results
71 subjects with SCDS and a mean age of 51.20 ± 12.22 years were included in the study. The most common symptom found in our sample was instability in 31 patients (43.66%), followed by aural fullness or tinnitus in 29 subjects (40.85%). 36 patients (43.66%) received medical treatment, with 28 of them (77.78%) showing symptom reduction. Surgical repair was indicated in five patients, with all showing symptom improvement. Statistically significant improvement (p < 0.05) was observed, particularly with surgical treatment and acetazolamide, in both symptoms and objective tests such as pure-tone audiometry and VEMPs.
Conclusion
SCDS shows significant similarities with other otic capsule dehiscences. It is essential to perform VEMPs and CT scans to complete the diagnosis, which is usually accompanied by clearly recognizable clinical criteria. Surgery for SCDS is effective, safe, and without complications. However, in cases where symptoms are mild to moderate, addressing this condition with medical treatment using diuretics such as acetazolamide has shown promising results.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Superior semicircular canal dehiscence syndrome (SCDS) is a clinical syndrome that is part of the known third mobile window syndromes and is associated with a defect in the continuity of the otic capsule, specifically in the bone covering this canal, as reported by Minor et al. in 1998 [1]. Changes in intracranial pressure cause movements in the dehiscence and changes in the fluid dynamics in the inner ear, resulting in the typical symptoms of the third mobile window, making it a recognized clinical condition that manifests in a wide variety of vestibular and auditory symptoms [2]. The third mobile window is located in various anatomical locations of the otic capsule, with SCDS being the most common. In fact, since 1998, more than 700 cases of SCDS have been reported [3]. Some symptoms of SCDS include instability, vertigo attacks, or increased sensation of instability induced by sound stimuli (Tullio phenomenon) or pressure changes (Hennebert’s sign), greater sensitivity to loud sounds, fullness, tinnitus, autophony (hearing one’s own footsteps or voice), or hearing loss (HL) [4,5,6].
Vestibular evoked myogenic potentials (VEMPs) have shown high value and utility in diagnosing SCDS [7], with reduced thresholds on the affected side and increased amplitudes [8, 9]. The 4000 Hz ocular VEMP threshold is the most sensitive and specific diagnostic test for SCDS [10]. Similarly, high-resolution computed tomography (CT) scans of the temporal bone, especially with thin slices, have very high sensitivity and specificity [11], aligning with the diagnostic criteria proposed by Ward et al. [12], making them essential diagnostic tools. Along with VEMPs, they are the reference diagnostic studies today [1, 11, 13].
Until now, surgery has been the primary therapeutic approach in the literature and the only one considered curative, performing canal occlusion via a middle fossa approach [1, 2] or even transmastoid. However, are there any other measures beyond surgical treatment to improve the quality of life for SCDS patients who do not want to undergo surgery? Given that the prevalence of this syndrome has been increasing in recent years [3], there is a need not only for greater awareness of this pathology and all alterations of the otic capsule in general but also to offer minimally invasive surgeries or even replace it with conservative treatments with much less risk to the patient [1, 14].
The main objectives of this study are to initially become one of the largest series of patients in the literature to perform a comprehensive characterization from clinical, auditory, and vestibular perspectives of SCDS, and secondly, to propose medical alternatives to conventional surgical approaches, comparing the results and evolution of these patients once treatment is carried out.
Materials and methods
Study design
A retrospective observational study was designed in a tertiary care center.
Patient selection
Clinical data from subjects with clinical suspicion of SCDS were collected from 2017 to 2024. A detailed review of medical records and audiovestibular tests allowed the classification of patients with SCDS. All included patients agreed to participate in the study, which was designed and conducted following the ethical guidelines of the 1975 Declaration of Helsinki.
Audiovestibular tests
All patients underwent a physical examination based on otoscopy and otoneurological examination with videonystagmography glasses (VideoFrenzel Interacoustics VF505m, Denmark). The presence or absence of spontaneous nystagmus, gaze-evoked nystagmus, position-induced nystagmus, and eye movements induced by sound or pressure stimuli were evaluated. Audiovestibular tests evaluated included pure-tone audiometry (PTA AC40, Interacustics, Denmark), VEMPs (Eclipse, Interacoustics, Denmark), video head impulse test (vHIT GN Otometrics, Denmark), and CT imaging of the petrous bones of both ears. All these tests were performed at diagnosis. VEMPs, PTA, and vHIT were also repeated during follow-up after the surgical procedure.
Audiometric tests
Audiometric findings are reported in terms of pure-tone thresholds from 0.25 to 6 kHz, expressed in hearing level in decibels (dB HL). Results were qualitatively graded based on severity according to the criteria of the Bureau International d’Audiophonologie (BIAP).
Vestibular tests
The vHIT (GN Otometrics, Denmark) analyzed the gain of the vestibulo-ocular reflex and the occurrence of refixation saccades obtained for head impulses in the plane of each of the three semicircular canals (SCC) of the affected and unaffected ear. The average gain is considered normal for each of the evaluated canals when it is above the lower limit for the patient’s age according to the system used. If the gain is lower than expected, it will be considered abnormal if there are refixation saccades (overt or covert). A test is considered normal when all three SCCs are normal and abnormal when at least one SCC is abnormal [1]. In our case, a gain equal to or greater than 0.8 was considered normal for the studied canal. For vestibular evoked myogenic potentials (VEMP), both cervical (cVEMP) and ocular (oVEMP) tests, normal vestibular function is defined as the presence of a vestibular evoked myogenic potential in both ears. It will be analyzed by the interaural asymmetry ratio (RAIA (%)) for air-conducted stimulation at 0.5 kHz, 1 kHz, and 4 kHz. The intensity of the acoustic stimulus used will be 97 dB HL, normalized, and 100 averages were presented at a rate of 5.1/s. cVEMP was recorded with patients sitting upright. The obtained signals were rectified by the contraction value of the SCM (sternocleidomastoid muscle). oVEMP was recorded with the patient sitting upright with the head facing forward and instructed to look at a fixed point on the wall with an upward tilt of 35°. Normality was defined as a RAIA below 50% in the studied stimulus [11, 13].
Computed tomography
Helical thin section computed tomography scans of the temporal bones were performed with 0.4 mm sections obtained. A positive diagnosis of SCD (semicircular canal dehiscence) was considered when the dehiscence was visible in three consecutive images. In cases of doubt about possible dehiscence, or in the presence of discordant findings between audiovestibular tests or otoneurological findings and the clinical presentation, the possibility of performing a higher resolution image such as Cone Beam CT was done.
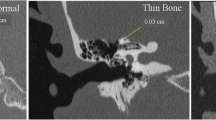
In Fig. 1, the typical audiovestibular and imaging findings of a patient diagnosed with SCDS are shown.
Typical presentation form of SCDS. In image A, we observe right tonal audiometry with moderate mixed hearing loss (43 dB), with an air-bone gap, especially in low frequencies (250 to 750 Hz). In images B and D, vestibular tests showing a right side with a hypofunctioning deficit, as measured by vHIT (Figure B) and VEMPs (Figure D), observing the specific frequency at 4 kHz with an RAIA of 85% due to increased amplitude on the pathological side. Finally, in image C, a coronal projection CT scan of the petrous bone shows the superior semicircular canal with an absence of bony coverage
Medical treatment
The proposed treatment algorithm was based on the progression of symptoms and the impact on quality of life they caused. Specifically, as occurred in most patients, when they were asymptomatic or had a mild limitation of their symptoms, no treatment beyond active surveillance was considered. However, if the symptoms resulted in an inability to carry out daily activities, the initial approach was to introduce acetazolamide for 3 months. If there was improvement with this drug, it was continued for 3 more months. In cases of poor tolerance, adverse effects, or poor response, the alternative was vasodilator treatment for the same period. If there was no response to this medication either, surgery was then considered.
Surgical treatment
In cases of poor response to medical treatment, with persistent incapacitating symptoms interfering with patients with defined SCDS, some patients underwent a surgical procedure, either through a middle fossa approach for access and obliteration of the superior semicircular canal (Fig. 2A) or a transmastoid approach for the same purpose (Fig. 2B).
In the first row, for the middle fossa approach, the first step was to perform an anterior temporal craniotomy. The dura mater was dissected until the arcuate eminence (AE) was located, which was reduced using a diamond burr [15]. Subsequently, the dehiscence of the superior semicircular canal was controlled and obliterated with autologous fascia and bone wax, followed by reinforcement of the tegmen with fascia, muscle, and synthetic fibrin glue, as described by Cheng et al. [15, 16] and Jan et al. [17]. Finally, the temporal lobe was repositioned, metal plates were placed to close the temporal craniotomy, and sutures were applied in layers. Conversely, in patients with the transmastoid approach, the procedure consisted of performing a standard mastoidectomy at the level of the labyrinthine vestibule. Next, in a posterolateral location to the horizontal semicircular canal, using a diamond burr, the dehiscent area of the superior semicircular canal was located and obliterated in the same manner as previously described (Fig. 2B). AE: Arcuate eminence; SSCD: Semicircular Superior Canal Dehiscence
There was also a considerable number of patients with bilateral involvement. In these cases, the decision to treat one ear or the other, if surgery was required, was based on the results of audiovestibular tests, primarily VEMPs due to their high sensitivity in these pathologies, in conjunction with otoneurological findings during the examination.
Follow-up
A clinical and audiovestibular function evaluation was performed both preoperatively and postoperatively. The evolution was measured using a dichotomous variable: persistence or disappearance of nine symptoms: hearing loss, aural fullness or tinnitus, autophony, instability, vertigo, falls, and the Hennebert or Tullio phenomenon. Audiometric improvement was evaluated by considering the difference between pure-tone thresholds, observing the most frequent pattern of hearing loss qualitatively. Vestibular improvement was evaluated through VEMPs and vHIT, both of which were assessed as normal/abnormal according to previously described criteria. The evolution of the different variables was recorded at the first consultation and at six months, considering prolonged follow-up if the response was not favorable.
However, it is important to emphasize the difficulty, in these patients, of achieving normalization of the audiovestibular parameters measured in the complementary tests. Therefore, to conduct this study, the improvement of the main previously described symptoms was determined exhaustively as a tool to represent an improvement in quality of life once the chosen treatment was implemented.
Statistical analysis
Descriptive statistical methods, including arithmetic means, standard deviations, and ranges, were used for each group before and after treatment.
Initially, a statistical analysis using multivariate logistic regression was carried out to assess the association between the presence of seven cardinal symptoms at two different times (Pretreatment and six months). To evaluate the relationship between the pre-posttreatment period with PTA (Pure Tone Average) and vestibular tests based on VEMPs and vHIT, controlling for different treatments, sex, and patient age, a repeated measures ANOVA statistical analysis was used. As with symptom improvement, the statistical significance of the coefficients was evaluated using the associated p-value, considering a p-value < 0.05 as statistically significant.
The suitability of the data distribution was checked using the Shapiro-Wilk test to assess the normality of the distribution. For the statistical analysis, a p-value < 0.05 was considered a measure of statistical significance, and the Stata 16.1 program was used.
Results
Population
Seventy-one patients were included. 49 (69.01%) participants were women, and 22 (30.99%) were men. Demographic results are summarized in Table 1.
Symptoms
The most common symptom found in our sample (Fig. 3) was instability in 31 patients (43.66%), followed by a sensation of hearing loss in 29 patients (40.85%), fullness or tinnitus in 20 (28.17%), vertigo attacks in 17 patients (23.94%), autophony in 14 (19.72%), Tullio or Hennebert phenomena in 10 patients (14.08%), and finally, falls in 6 of them (8.45%). The representation of symptoms at the time of diagnosis, prior to surgical treatment, is shown in Fig. 3.
Of the patients with hearing loss (n = 22), the most common pattern in the affected ear was mild mixed hearing loss in 54.54% (n = 12), with an average PTA of 26.38 dB for air conduction and 16.60 dB for bone conduction. The average air-bone gap was 9.78 dB.
We performed vHIT in 53 patients (74.64%), showing normal results in 34 of them (68%) and reduced gain in the superior semicircular canal in 16 of them (32%). The mean gain for hypofunctioning results in the affected semicircular canals was 0.64. Regarding VEMPs, which were performed in 100% of the cohort patients, the average VEMP threshold before treatment was 68 dB Normal Hearing Level (NHL). 73.23% (n = 52) of the ears affected by SCDS had significantly higher VEMP amplitudes than the unaffected ears, especially under vibratory stimulation in ocular records at 4000 Hz, averaging 5.23 µV.
Through otoneurological exploration with videonystagmography glasses, it was observed that 36 of the patients (50.70%) had some pathological nystagmus. 17 of these patients (47.22%) presented vertical torsional nystagmus with hyperextension. 11 patients (30.55%) had upbeat vertical nystagmus also with hyperextension, 6 (16.66%) had horizontal nystagmus, and 2 (5.55%) had downbeat nystagmus.
SCDS treatment
Overall, 36 patients (50.70%) received medical treatment at some point. Of these patients, 19 (26.76%) received what is called vasodilator treatment, consisting of calcium antagonists, topiramate, corticosteroids, or betahistine. 17 (23.94%) received Acetazolamide 250 mg/24 h. Three of these patients had to discontinue treatment, one due to an allergic reaction and two due to intolerable adverse effects such as headaches, paresthesias, and tremors. The rest of the patients, 29 (40.85%), did not receive any treatment and were only subjected to active surveillance. Finally, 6 of them (8.45%) underwent surgical intervention, 5 via middle fossa, and the remaining patient through a transmastoid approach. All this is summarized in Fig. 4.
Treatment response
The response to the various medical and surgical treatments proposed is summarized in Table 2. It briefly reports the evolution of symptoms prior to diagnosis, as well as the evolution through audiovestibular tests, with the corresponding p-values.
Once the treatment was completed, an improved response was also observed in the results of the audiovestibular tests. Although the average gains of the superior semicircular canals with a history of dehiscence remained hypofunctional, there was an observed improvement (0.73). The same occurred with the VEMPs, with a threshold after treatment that was 76 dB Normal Hearing Level (NHL), and amplitudes in the oVEMPs averaging 4.44 µV. Finally, the average PTA was 17.75 dB for air conduction and 13.50 dB for bone conduction, resulting in an air-bone gap of 4.25 dB.
Thus, as summarized in Fig. 5A, at 6 months, once the treatment of choice was completed, it was observed that patients with instability had reduced to 7 patients (9.86%), followed by those with a sensation of hearing loss, totaling 10 patients (14.08%). Fullness or tinnitus affected 13 patients (18.31%), and vertigo attacks were present in 4 patients (5.63%). Autophony was observed in 10 patients (14.08%), Tullio or Hennebert phenomena in 3 patients (4.23%), and finally, falls only occurred in one patient (1.41%). Figure 5B expresses the evolution of audiovestibular tests at different evaluated moments. However, the most important interpretation of these data is that the perceived improvement with treatment translated into a more satisfactory quality of life.
As shown previously, in terms of absolute value reduction, all symptoms showed improvement. However, the statistical significance value was distributed for each studied variable as observed in Table 2.
Discussion
SCDS has been diagnosed more frequently than expected since its first description in 1998 by Minor [4]. In fact, in our series, we have diagnosed 71 patients over the last six years. Going back to the retrospective study, we could find more cases of SCDS that may have gone unnoticed due to ignorance of the disease or low-sensitivity complementary tests that may have been performed. As with all different otic capsule dehiscences, the more these diseases are studied and understood, the greater the diagnostic capability will be, and thus more cases will appear, and a greater understanding and characterization of these pathologies will be achieved [3].
The two theories in the development of SCDS, congenital and acquired, are currently accepted although the definitive cause is still unknown [3]. Minor et al., in their study, conclude that the etiology of SCDS is mainly congenital. However, this does not mean there are no acquired cases [18], as demonstrated by Chari in 2021 through sequential CT imaging of the same patient with and without the dehiscence. Other studies report that patients under ten years old showed a high prevalence of SCDS in CT scans, which decreased in the second decade [19,20,21], supporting the congenital theory. Furthermore, it is common to observe other embryological developmental alterations of adjacent structures, frequently seeing defects in the tegmen or geniculate ganglion dehiscence along with a superior semicircular canal dehiscence [22, 23].
Understanding the natural history, symptoms, and signs of SCDS is essential for diagnosis. SCDS is very similar to other otic capsule dehiscences and other entities [3]. Various labyrinthine dehiscences could cause symptoms similar to those of SCDS [24], with very similar results in complementary tests from an audiovestibular perspective. Minor et al. reported in 2005 that symptoms appeared in 25% of patients after head trauma or Valsalva maneuver [7].
Therefore, there is a great variability in SCDS symptoms [25], and not all patients have all possible manifestations, as described in our cohort. In our series, the three most common symptoms of SCDS are instability, fullness, and hearing loss, but they are not specific enough. The more specific symptoms are autophony, Tullio phenomenon, and Hennebert’s sign, although we only found them in less than 30% of cases. There is also enormous variation among groups; for example, Powell classifies symptoms in descending order of frequency as autophony, aural fullness, instability, vertigo attacks, tinnitus, Tullio phenomenon, Hennebert’s sign, and hearing loss was less frequent [2]. Merchant and Rosowski proposed that a dehiscence in the otic capsule of the inner ear could cause conductive hearing loss due to a third window syndrome [26]. Sluydts recently published a study and reported a patient in whom SCDS was masked by otosclerosis [27]. Moreover, all these symptoms could overlap with other otoneurological entities and challenge the diagnosis [7]. For instance, Johanis describes a symptomatic patient with two otoneurological diseases in the medical history, SCDS and endolymphatic hydrops [25]. We can observe that, with very slight differences, the trend in our series from a symptomatic perspective is consistent with what is described in the literature.
We must be aware that, if SCDS is suspected, the VEMP threshold test should be performed, and a high-resolution CT scan of the temporal bone should be requested. Although VEMP testing was considered the gold standard for SCDS, it is not always able to detect it [28]. Currently, the only widely proven effective treatment is surgery. The most common surgical approach is the middle fossa approach [16]. However, transmastoid approaches have also been used, as in one of our patients [2], and even round window approaches [3]. In fact, round window reinforcement has been proposed as a minimally invasive technique to control SCDS symptoms, but it is associated with variable results and high failure rates [14]. Middle fossa or transmastoid approaches are often used to rescue these patients [14]. In fact, the six patients who underwent surgical intervention had previously used various unsuccessful medical treatments, leading to clearly limited quality of life, which made them decide on the surgery. On the other hand, the selection of the surgical approach should be based on the patient’s anatomy [7, 29] and the surgeon’s experience. While the middle cranial fossa approach has advantages in exposing the superior semicircular canal and ensuring proper canal occlusion, the transmastoid approach could be an option in cases where SCDS is located near the petrous sinus or the posterior edge of the incus.
No complications occurred in our series of surgically treated patients. Previous studies show that it is an effective and safe technique [7]. The only notable symptom is vertigo during the 3–7 days after surgery, as occurred in our cases, usually accompanied by torsional nystagmus with hyperextension. Both findings progressively decreased within the first 3–4 weeks, and the otoneurological examination showed no findings then. Meanwhile, findings such as vHIT gain or VEMP amplitudes and thresholds became almost normal at the six-month follow-up, where the surgical approach showed the best results in symptom reduction and from a statistical significance perspective, as shown in Table 2. A plausible hypothesis for this event could be that there is a new alteration in fluid dynamics once the dehiscence is obliterated. This, combined with inflammation and irritability from surgical manipulation, might explain the vestibular symptoms that tend to diminish over time and with the administration of vestibular sedatives, preferably at low doses to avoid compromising vestibular compensation phenomena.
Apart from surgical treatment, one of the novelties of our work is that, until now, there are no effective medical treatments described for SCDS. Based on Minor’s experience [7], only half of the patients diagnosed with SCDS accepted surgery, and the other half were content with having a diagnosis that explains their symptoms. When a patient rejects surgery, as occurred in our cases, we have two options. The first is to control the symptoms expectantly, as we did with 29 of our patients, and the second option is empirical medical treatment, as we did with 36 patients. Within this second group, the most commonly used medications were diuretics, such as Acetazolamide, calcium channel blockers, such as nimodipine, betahistine, or topiramate. The main diuretic used was Acetazolamide, commonly used in Meniere’s disease (MD) or endolymphatic hydrops, to reduce the amount of endolymph in the inner ear and eliminate associated symptoms [30,31,32]. In both cases, the use of these treatments in the study was empirical since the efficacy of this drug for this condition had not been demonstrated at that time for SCDS. The plausible hypothesis is that, considering that an alteration in the continuity of the otic capsule can cause changes in fluid dynamics, responsible for fluctuating audiovestibular symptoms (Gurkov et al. [33]), the diuretic or vasodilator effect can reduce the pressure gradient and, consequently, alleviate cochleovestibular symptoms. In fact, in our cohort, both with diuretics and vasodilators, a reduction in various symptoms was observed, particularly with acetazolamide, with statistical significance in several clinical and even vestibular variables studied, such as VEMPs.
In summary, as expected, our work statistically significantly demonstrates that surgical treatment provides superior improvement compared to other empirical treatments. This improvement is observed not only in symptomatic relief but also in results measured through audiovestibular tests, as seen in our study. However, it is worth noting that although there is a smaller absolute value gain compared to surgery, considering that patients undergoing surgical procedures had a more deteriorated audiovestibular condition, acetazolamide, and even vasodilator treatments in variables like instability, also contribute to clinical improvement, even optimizing results measured in VHIT, VEMPs, and pure-tone audiometry.
Study limitations
While these findings should be approached with caution due to the limited sample size, they offer optimism and present a real alternative to surgery, especially suitable for cases with moderate deterioration or for patients hesitant about undergoing surgery. Another limitation is possibly the evolution time. It would be interesting to extend the follow-up time, considering the difficulty in patients with conditions of multiple pathologies, where the administration of a diuretic or a vasodilator could pose a risk of causing hypotension or hydroelectrolytic alterations, which is why it was only extended to six months. Although the measurement of various cardinal symptoms has been used as a tool to represent improvement once treatment has been initiated, we believe it would be interesting, in future studies, to use standardized quality of life questionnaires such as the Glasgow Benefit Inventory or the Dizziness Handicap Inventory.
Conclusions
Suspicion of SCDS by symptoms and signs is key to diagnosis. SCDS has a great similarity with other otic capsule dehiscences and other entities. The three most common symptoms of SCDS are instability, fullness, and hearing loss, but they are not specific enough. We must be aware that if SCDS is suspected, the VEMP threshold test should be performed, and a high-resolution CT scan of the temporal bone should be requested. To monitor follow-up and know the evolution of the disease, we can analyze vestibular function through tests like VEMPs and vHIT, and perform audiological tests like pure-tone audiometry. Although surgery is effective and safe, with almost no complications, medical treatment with Acetazolamide has shown to be effective in some cases, but more research is needed to establish clear criteria for its indication.
Data availability
Data pertaining to this study can be shared upon request to the corresponding author.
References
Halmagyi GM, Chen L, MacDougall HG, Weber KP, McGarvie LA, Curthoys IS (2017) The video head impulse test. Front Neurol. ;8(JUN)
Powell HRF, Khalil SS, Saeed SR (2016) Outcomes of transmastoid surgery for Superior Semicircular Canal Dehiscence Syndrome. Otol Neurotol 37(7):e228–e233
Ward BK, Carey JP, Minor LB (2017) Superior canal dehiscence syndrome: lessons from the first 20 years. Front Neurol 8(APR):1–10
Minor LB, Solomon D, Zinreich JS, Zee DS (1998) Sound- and/or pressure-induced vertigo due to bone dehiscence of the superior semicircular canal. Arch Otolaryngol - Head Neck Surg 124(3):249–258
Dlugaiczyk J, Burgess AM, Goonetilleke SC, Sokolic L, Curthoys IS Superior Canal Dehiscence Syndrome:Relating Clinical Findings With
Lin K, Lahey R, Beckley R, Bojrab D, Wilkerson B, Johnson E Validating the Utility of High Frequency Ocular Vestibular, Zhou G, Gopen Q, Poe DS et al (2007) Clinical and diagnostic characterization of canal dehiscence syndrome: A great otologic mimicker. Otol Neurotol [Internet]. Oct [cited 2021 Apr 3];28(7):920–6. https://pubmed.ncbi.nlm.nih.gov/17704722/
Minor LB (2005) Clinical manifestations of superior semicircular canal dehiscence. Laryngoscope 115(10 I):1717–1727
Colebatch JG, Rosengren SM, Welgampola MS. Vestibular-evoked myogenic potentials. In: Handbook of Clinical Neurology [Internet]., Elsevier BV (2016) [cited 2021 Apr 3]. pp. 133–55. https://pubmed.ncbi.nlm.nih.gov/27638068/
Rosengren SM, Aw ST, Halmagyi GM, McAngus Todd NP, Colebatch JG Ocular vestibular evoked myogenic potentials in superior canal dehiscence. J Neurol Neurosurg Psychiatry [Internet]. 2008 May [cited 2021 Apr 3];79(5):559–66. https://pubmed.ncbi.nlm.nih.gov/17766428/
Mukherjee P, Chiarovano E, Cheng K, Manzari L, McGarvie LA, MacDougall HG (2021) Video-head impulse test in superior canal dehiscence. Acta Otolaryngol [Internet]. [cited 2021 Apr 3]; https://pubmed.ncbi.nlm.nih.gov/33641579/
Gürkov R, Jerin C, Flatz W, Maxwell R (2018) Superior canal dehiscence syndrome: diagnosis with vestibular evoked myogenic potentials and fremitus nystagmus. German version. HNO 66(5):390–395
Ward BK, van de Berg R, van Rompaey V, Bisdorff A, Hullar TE, Welgampola MS et al Superior semicircular canal dehiscence syndrome: Diagnostic criteria consensus document of the committee for the classification of vestibular disorders of the bárány society. J Vestib Res [Internet]. 2021 Jan 29 [cited 2021 Apr 3];1–10. https://pubmed.ncbi.nlm.nih.gov/33522990/
Zuniga MG, Janky KL, Nguyen KD, Welgampola MS, Carey JP Ocular versus cervical VEMPs in the diagnosis of superior semicircular canal dehiscence syndrome. Otol Neurotol [Internet]. 2013 Jan [cited 2021 Apr 3];34(1):121–6. https://pubmed.ncbi.nlm.nih.gov/23183641/
Chemtob RA, Noij KS, Qureshi AA, Klokker M, Nakajima HH, Lee DJ (2019) Superior Canal Dehiscence surgery outcomes following failed round window surgery. Otol Neurotol 40(4):535–542
Cheng YS, Kozin ED, Remenschneider AK, Nakajima HH, Lee DJ (2016) Characteristics of wax occlusion in the surgical repair of superior canal dehiscence in human temporal bone specimens. Otol Neurotol 37(1):83–88
Cheng YS, Kozin ED, Lee DJ (2016) Endoscopic-Assisted Repair of Superior Canal Dehiscence [Internet]. Vol. 49, Otolaryngologic Clinics of North America. W.B. Saunders; [cited 2021 Apr 3]. pp. 1189–204. https://pubmed.ncbi.nlm.nih.gov/27565386/
Jan TA, Cheng YS, Landegger LD, Lin BM, Srikanth P, Niesten MEF et al Relationship between Surgically Treated Superior Canal Dehiscence Syndrome and Body Mass Index. Otolaryngol - Head Neck Surg (United States) [Internet]. 2017 Apr 1 [cited 2021 Apr 3];156(4):722–7. https://pubmed.ncbi.nlm.nih.gov/28116976/
Chari DA, Juliano AF, Jung DH Radiologically-proven New Development of Superior Semicircular Canal Dehiscence Associated With Development of Superior Semicircular Canal Dehiscence Syndrome. Otol Neurotol [Internet]. 2021 Feb 1 [cited 2021 Apr 3];42(2):285–9. https://pubmed.ncbi.nlm.nih.gov/33273305/
Chen EY, Paladin A, Phillips G, Raske M, Vega L, Peterson D et al (2009) Semicircular canal dehiscence in the pediatric population. Int J Pediatr Otorhinolaryngol 73(2):321–327
Meiklejohn DA, Corrales CE, Boldt BM, Sharon JD, Yeom KW, Carey JP et al (2015) Pediatric Semicircular Canal Dehiscence: Radiographic and Histologic Prevalence, with Clinical Correlation [Internet]. Vol. 36, Otology and Neurotology. Lippincott Williams and Wilkins; [cited 2021 Apr 3]. pp. 1383–9. https://pubmed.ncbi.nlm.nih.gov/26164444/
Saxby AJ, Gowdy C, Fandiño M, Chadha NK, Kozak FK, Sargent MA et al Radiological prevalence of superior and posterior semicircular canal dehiscence in children. Int J Pediatr Otorhinolaryngol [Internet]. 2015 Mar 1 [cited 2021 Apr 3];79(3):411–8. https://pubmed.ncbi.nlm.nih.gov/25636666/
Suryanarayanan R, Lesser TH Honeycomb tegmen: Multiple tegmen defects associated with superior semicircular canal dehiscence. J Laryngol Otol [Internet]. 2010 May [cited 2021 Apr 3];124(5):560–3. https://pubmed.ncbi.nlm.nih.gov/19852870/
Isaacson B, Vrabec JT (2007) The radiographic prevalence of geniculate ganglion dehiscence in normal and congenitally thin temporal bones [Internet]. Vol. 28, Otology and Neurotology. Otol Neurotol; [cited 2021 Apr 3]. pp. 107–10. https://pubmed.ncbi.nlm.nih.gov/17031323/
Krombach GA, DiMartino E, Schmitz-Rode T, Prescher A, Haage P, Kinzel S et al Posterior semicircular canal dehiscence: A morphologic cause of vertigo similar to superior semicircular canal dehiscence. Eur Radiol [Internet]. 2003 Jun 1 [cited 2021 Apr 3];13(6):1444–50. https://pubmed.ncbi.nlm.nih.gov/12764665/
Johanis M, De Jong R, Miao T, Hwang L, Lum M, Kaur T et al (2021) Concurrent superior semicircular canal dehiscence and endolymphatic hydrops: A novel case series. Int J Surg Case Rep [Internet]. ;78:382–6. https://doi.org/10.1016/j.ijscr.2020.12.074
Merchant SN, Rosowski JJ Conductive Hearing Loss Caused by Third-Window Lesions of the Inner Ear. Otol Neurotol [Internet]. 2008 Apr [cited 2021 Apr 3];29(3):282–9. https://journals.lww.com/00129492-200804000-00005
Sluydts M, Bernaerts A, De Foer B, Zarowski A, van Dinther J, Vanspauwen R et al (2020) The sensitivity of the cVEMP test in detecting a superior semicircular canal dehiscence and the influence of a coexisting incudal lysis: a case report. J Int Adv Otol 16(1):123–126
M Geraldine Zuniga 1 Angela Schell, Benjamin G Engst JPC. [Internet]. 2021;Online ahe
Lookabaugh S, Kelly HR, Carter MS, Niesten MEF, McKenna MJ, Curtin H et al (2015) Radiologic classification radiologic classificationof superior canal dehiscence: implications for surgical repair. Otol Neurotol [Internet]. 36(1):118–125 Jan 20 [cited 2021 Apr 3];
Brookes GB, Booth JB (1984) Oral acetazolamide in Menière’s disease. J Laryngology Otology [Internet] 98(11):1087–1095 Nov 1 [cited 2023 Nov 18];
Varga G, Ribari O (1958) Action of acetazolamide in Ménière’s disease. J Laryngology Otology [Internet] 72(11):920–925 Nov 1 [cited 2023 Nov 18];
Muftic MK Acetazolamide in Ménière’s disease. AMA Archives of Otolaryngology [Internet]. 1957 Jun 1 [cited 2023 Nov 18];65(6):575–9
Gürkov R, Flatz W, Louza J, Strupp M, Krause E (2011) In vivo visualization of endolyphatic hydrops in patients with Meniere’s disease: correlation with audiovestibular function. Eur Arch Otorhinolaryngol 268(12):1743–1748. https://doi.org/10.1007/s00405-011-1573-3
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Institutional review board statement
This study protocol was reviewed in November 2023 and approved by Ethics committee of Clínica Universidad de Navarra, approval number (CEI 2023.163). Even more, this study was designed and performed in accordance with the ethical guidelines of the 1975 Declaration of Helsinki. Written informed consent was obtained from all to participate in the study. Even though, written informed consent was obtained from the patient for publication of the details of their medical case and any accompanying images.
Informed consent
Informed consent was obtained from all subjects involved in the study.
Conflict of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lorente-Piera, J., Prieto-Matos, C., Fernández, N.P. et al. Insights from therapeutic strategies in superior canal dehiscence syndrome: is there anything beyond surgical treatment?. Eur Arch Otorhinolaryngol (2024). https://doi.org/10.1007/s00405-024-08896-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00405-024-08896-3