Abstract
The aim of the present study was to evaluate the thyroarytenoid muscle response during bilateral thyroid surgery using vagal nerve stimulation. 195 patients (390 nerves at risk) underwent a total thyroidectomy. The recurrent laryngeal nerve’s function was checked by analyzing the amplitude and the latency of the thyroarytenoid muscle’s responses after a vagal nerve’s stimulation (0.5 and 1 mA) using the NIM3 Medtronic system. All patients were submitted to preoperative and postoperative laryngoscopy. 20 patients get no thyroarytenoid muscle response to the vagal nerve stimulation, and 14 postoperative recurrent laryngeal nerve palsies were confirmed (3.8 %). Two palsies were present after 6 months (0.51 %). All the patients with muscle’s response have normal mobility vocal fold. The test sensitivity was 100 % and the test specificity was 98 %. Physiologically, the mean latencies of the muscular potentials for the right RLN were, respectively, 3.89 and 3.83 ms (p > 0.05) for the stimulation at 0.5 and 1 mA. The mean latencies for the left RLN were, respectively, 6.25 and 6.22 ms for the stimulation at 0.5 and 1 mA (p > 0.05). The difference of the latencies between the right and the left nerve was 2.30 ms (1.75–3.25 ms) with a stimulation of 0.5 or 1 mA (p < 0.05). Thyroarytenoid muscle’s response via a vagal nerve stimulation showed a functional asymmetry of the laryngeal adduction with a faster right response. Surgically, this method can predict accurately an immediate postoperative vocal folds function in patients undergoing a bilateral thyroid surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
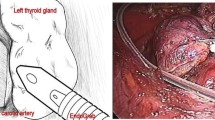
Thyroid gland surgery is the most frequently performed endocrine procedure worldwide. The postoperative dysfunction of the recurrent laryngeal nerve (RLN) is one of the most serious complications in thyroid surgery, sometimes irreversible and the leading cause of life-quality decrease [1]. The gold standard to protect the nerve is the visual identification of the recurrent laryngeal nerve during thyroid surgery [2, 3]. As early as 1938, Lahey reported a routine exposure of the RLN in thyroid surgery [4]. Identification of the recurrent laryngeal nerve with neurostimulation during surgery can also provide physiological data. Intra-operative neuromonitoring has been advocated as a mean to localize and identify the recurrent laryngeal nerve and to predict the vocal folds function [5, 6]. Various methods have been used to facilitate the intraoperative monitoring of the RLN: the first method was the direct visualization of the vocal folds movements intraoperatively, by using laryngeal mask anesthesia [7]. Other methods rely on RLN stimulation with an observation of posterior cricoarytenoid muscle contraction or palpation [8]. Presently, neuro monitoring was the most frequent implemented method (Figs. 1, 2).
Different types of electrodes for the RLN monitoring are used [9]: intralaryngeal surface electrodes attached to a conventional endotracheal tube [5, 10–12], monopolar needle electrodes placed on the vocal cords by direct laryngoscopy [13], or bipolar needle electrodes inserted intraoperatively through the cricothyroid ligament [14–17]. Two different types of stimulation can be performed: the direct stimulation of the RLN and the indirect stimulation of the RLN via the ipsilateral vagal nerve [10, 14]. Intra-operative direct neuromonitoring is frequently used; however, selective laryngeal response by vagal nerve stimulation is poorly studied [6].
The aim of this study was to evaluate the physiological and pathological data of thyroarytenoid muscle response via a vagal nerve stimulation during bilateral thyroid surgery correlated to laryngoscopy.
Methods
A prospective study was conducted from January 2012 to January 2015. One hundred and ninety-five consecutive patients underwent bilateral thyroid surgery for various thyroid diseases. Exclusion criteria included previous thyroid surgery and unilateral procedures. Patients were informed about the use of the monitoring system to assess the function of the recurrent laryngeal nerve during operation.
All patients were submitted to preoperative and postoperative direct laryngoscopy. Postoperative laryngoscopy was performed 2 days and 1 month after the surgical procedure, then 6 months later in the absence of recovery. The dysfunction was considered as permanent over 12 months postoperatively.
Technique of intra-operative neuromonitoring
The patient is placed in the supine position with the neck extended, under general anesthesia and with endotracheal intubation. No long-acting muscle relaxants were used during the intubation and no muscle relaxants of any kind were used at any other time during the procedure. A transverse incision was made in the line of a naturel skin crease, approximately 3 cm above the suprasternal notch.
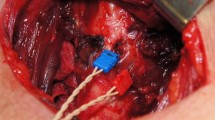
After completion of total thyroidectomy, the cricothyroid ligament is identified and a double-needle electrode is placed through the ligament on each side within the thyroarytenoid muscle. Two neutral electrodes were inserted into the upper skin. These electrodes were connected to the preamplifier of the NIM III Medtronic system (Medtronic Xomed Inc., Jacksonville, FL, USA). The stimulation current of the recurrent laryngeal nerve via the ipsilateral vagal nerve was set to 0.5 and 1 mA, and the response threshold was set to 100 µV.
The functioning of the recurrent laryngeal nerve was checked by stimulation of the vagal nerve at the thyroid cartilage level before wound closure, to analyze the amplitude and latency from the responses of the thyroarytenoid muscle. The detection of activity of the thyroarytenoid muscle was auditory and visual on the screen monitor and the direct motion of the bipolar electrode within the muscle. The clinical usefulness to the method was to predict intraoperatively potential patient with RLN palsy to close survey severe voice changes or above all airway compromise notably for at risk patients (obesity, etc.) during the immediate postoperative period.
Statistical analysis
Parameters were tested using the Student test. A p value of less than 0.05 was considered to be significant. Absence or alterations in the normal neuromonitoring signal were defined as a positive test result, possibly heralding recurrent laryngeal nerve injury. Sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were calculated. The sensitivity is the probability that the test is positive if there is paralysis. The specificity is the probability that the test is negative if there is no paralysis. The PPV is the probability that paralysis is present if the test is positive. The NPV is the probability that there is no paralysis if the test is negative.
Results
The study group consisted of 195 patients with a total of 390 nerves at risk. Patients ranged in age from 14 to 88 years, with a mean age of 53.4 years, and there was a female predominance of 4.7:1. Indications for surgery included multinodular goiter (69 %), Graves’ disease (19 %), thyroid carcinoma (10 %) and hyperthyroidism to the cordarone (2 %). Patients with hyperthyroidism: 58 patients (30 %), euthyroidism: 135 patients (69 %), and with hypothyroidism: 1 patient (0.5 %) (Table 1). The weight of the specimen was 18–360 g (average: 62 g). No complications were attributable to the use of intra-operative neuromonitoring. We found no cardiac or pulmonary interaction during vagal nerve stimulation. Ten hematomas required additional drainage, without advent palsy.
Out of 195 right thyroarytenoid muscles analyzed, 14 electromyographic responses were absent (7.2 %). Out of 195 left thyroarytenoid muscles analyzed, 6 electromyographic responses were absent (3.1 %). Out of 20 patients without response (positive test), 14 palsies were confirmed postoperatively by fibroscopy at 2 days (3.8 %), therefore the PPV was 70 %. Ten vocal fold palsies were observed 1 month after surgery and 2 palsies (0.51 %) were observed 6 months and then 12 months after surgery. So 0.51 % palsy was permanent (12 months). Out of 370 nerves with response (negative test), 370 vocal cords normally moved bilaterally. The NPV was 100 %. The sensitivity was 100 % and the specificity was 98.4 % (Table 2). No bilateral palsy was noticed postoperatively.
The mean of the latencies on the right side was 3.89 ms (min: 1.75 ms, max: 6 ms) and 3.83 ms (min: 2 ms, max: 5.75 ms), respectively for the stimulation of 0.5 and 1 mA (Table 3). The difference of the latencies on the right side was not significant for stimulation of 0.5 mA and stimulation of 1 mA (p = 0.7). The mean of the latencies on the left side were 6.25 ms (min: 3.75 ms, max: 9 ms) and 6.22 ms (min: 3.75 ms, max: 10 ms), respectively, for the stimulation of 0.5 and 1 mA. The difference of the latencies on the left side was not significant for stimulation of 0.5 mA and stimulation of 1 mA (p = 0.49). The difference of the latencies between the right and the left was 2.30 ms with stimulation 0.5 and 1 mA. The difference of the latencies with stimulation 0.5 mA was significant between the right and the left (p < 0.001). The difference of the latencies with stimulation 1 mA was significant between the right and the left (p < 0.001).
The mean of the amplitudes on the right side was 993.96 µV (min: 100 µV, max: 6249 µV) and 1158.34 µV (min: 100 µV, max: 6997 µV), respectively, for the stimulation of 0.5 and 1 mA (Table 4).
The difference of the amplitudes on the right was not significant for stimulation of 0.5 mA and stimulation of 1 mA (p = 0.16). The mean of the amplitudes on the left side was 1005.21 µV (min: 100 µV, max: 6120 µV) and 1236.26 µV (min: 117 µV, max: 6981 µV), respectively, for the stimulation of 0.5 and 1 mA.
The difference of the amplitudes on the right was not significant for stimulation of 0.5 mA and stimulation of 1 mA (p = 0.06). The difference of the amplitudes with stimulation 0.5 mA was not significant between the right and the left nerves (p = 0.88). The difference of the amplitudes with stimulation 1 mA was not significant between the right and the left nerves (p = 0.2).
Discussion
Iatrogenic injury to the RLN during thyroid and parathyroid surgery is the main origin of vocal fold paralysis throughout the world. Unilateral vocal fold palsy can be associated with severe voice changes, dysphagia and difficulties with aspiration, which could result in pulmonary complications. Bilateral vocal fold paralysis results in loss of airway and then tracheotomy [1].
Visual identification of the RLN during thyroid operations has been considered as the gold standard of recurrent laryngeal nerve treatment by many studies [2, 3]. In 1938, Lahey first dissected the recurrent laryngeal nerve and thanks to that the number of injuries to the RLN decreased [4]. With RLN visual identification, Lahey observed 1.6 % of paralysis. Calo et al. observed without neuromonitoring 2.62 % unilateral RLN paralysis (2.01 % transient and 0.6 % permanent) and 0.2 % bilateral palsy [5].
Mechanisms of RLN injury include transection, clamping, stretching, electro-thermal injury, ligature entrapment or ischemia. Chiang et al. showed that excessive stretching of the RLN at the region of Berry’s ligament plays a major role in the occurrence of RLN palsy during total lobectomy. They observed 15 nerves injuries (9 %), of which 12 were caused by apparent overstretching at the region of Berry’s ligament. So excessive traction on the thyroid may cause a stretch injury of the RLN, especially when the nerve coursed through the Berry’s ligament [10].
Intraoperative neuromonitoring is used as a means to localize and identify the RLN as well as to predict postoperative vocal fold dysfunction [5, 6]. In 1984, Woltering et al. first demonstrated reliability of intraoperative neuromonitoring in the thyroid surgery [18]. Various methods of monitoring are used [8, 9]. Direct stimulation of the RLN and muscular detection by the surface electrodes is the most frequent method [5, 10–12]. The results of postoperative transient and permanent paralysis are, respectively, 6 and 1 % for Chiang et al. [10], 2.2 and 0.3 % for Genther et al. [11], and 1.44 and 0.57 % for Calo et al. [5] (Table 5). A systematic review, including 27 articles and 25000 patients, reported mean incidences of 9.8 % for temporary vocal fold paresis and 2.3 % for permanent vocal fold paresis after thyroid surgery, with reported values of vocal fold paresis (temporary or permanent) ranging from 2.3 to 26 %. The incidence of paralysis varies considerably in the literature probably because different tools of postoperative examination were used: indirect laryngoscopy (mirror), fiber-optic laryngoscopy and video-stroboscopy [19].
In our study, using fiber-optic laryngoscopy, we reported mean incidences of temporary vocal fold paresis of 3.1 % and permanent vocal fold paresis of 0.51 % over 12 months postoperatively.
Dralle et al. [6] have defined the impact of the monitoring on the vocal fold paresis on the basis of six published articles: they observed that RLN palsies were lower with intraoperative nerve monitoring (IONM) than with visual nerve identification only: 1.1–3.5 % transient paralysis and 0–0.9 % permanent paralysis with IONM, versus 2.6–4.2 % transient paralysis and 0.89–2.5 % permanent paralysis without IONM.
In the current study, the bipolar trans-cricothyroid ligament electrode was used: Tschopp and Gottardo reported consistently muscular responses in 100 % of the stimulated recurrent laryngeal nerve if made with the bipolar transligament electrode and only 76 % had reliable muscular potentials with the intralaryngeal surface electrode [9]. Chiang et al., using the intralaryngeal surface electrode, reported five patients (0.04 %) encountered equipment failure that was caused by the malposition of electrodes at the start of operation [10].
In an experimental study on pigs, Schneider et al. showed that the continuos real-time monitoring of the RLN by vagal nerve stimulation appears technically feasible and safe. Indeed the cardiac and pulmonal parameters registered during the experimentation did not show any changes [20]. Ulmer et al. presented the results of 19 patients after the implementation of a novel real-time nerve monitoring system, based on a new vagal nerve cuff electrode. This technique was feasible, safe, reproducible and easy to perform [21].
Indirect stimulation of the RLN via the vagal nerve stimulation was considered significantly better predictor of postoperative RLN dysfunction than direct recurrent laryngeal nerve stimulation, because the entire course of the recurrent laryngeal nerve was analyzed [10, 14]. Chiang et al. [10] showed that the monitoring of a direct RLN stimulation is not able to register injury if the stimulation electrode is placed at the distal part of the nerve, below the injury. Thomusch et al. [14] showed that the accuracy (p < 0.05) was significantly improved with the indirect method relative to the direct method. IONM can afford the surgeon real-time information regarding neurophysiologic function of the recurrent laryngeal nerve.
In the present study, out of 390 recurrent laryngeal nerves at risk, 14 nerves exhibited temporary vocal fold paresis on flexible fiberoptic laryngoscopy performed. For these 14 nerves, postdissection muscular responses were absent (100 % true-positive findings). Out of 390 RLN at risk, 376 presented postoperative function (96.4 %). Out of these 376 nerves, 370 obtained postdissection responses (94.9 % true-negative findings) and 6 failed to obtain a response (1.5 % false-positive findings). In total, 20 of 390 (5.1 %) recurrent laryngeal nerves failed to get a response, and 370 of 390 (94.9 %) got response.
The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) of the test were 100, 98.4, 70 and 100 %, respectively. The misplacement of the transligamentary needle electrode can lead to a reduction of the signal amplitude or the absence of response. So, if intraoperative RLN response is absent, the surgeon is able to check needle electrode position [21]. In the study of Genther et al., using an endotracheal tube with surface electrodes, 674 patients with 1000 recurrent laryngeal nerves were analyzed. The sensitivity, specificity, PPV, NPV, and accuracy for postdissection amplitude of less than 200 μV regarding prediction of vocal fold paresis were, 95.5, 99.2, 72.4, 99.9, and 99.1 %, respectively [11].
Caragacianu et al. aimed to provide electrophysiological reference range values for threshold stimulation and amplitude of evoked action potential in the setting of recurrent laryngeal nerve preservation and normal postoperative vocal cord function [12]. For group 1 (normal pre and postoperative laryngeal exams), at a suprathreshold stimulation current 1 mA, mean final amplitude was 1179 μV (min: 152 μV, max: 3843 μV); for group 2 (abnormal postoperative vocal cord function), mean final amplitude was 97.5 μV.
In our study, we found similar mean amplitude (1158.34 µV on the right and 1236.26 µV on the left) and the mean amplitude for RLN palsy was below 100 µV (Table 5).
The differences of latency between the right and the left may be explained by morphological and functional asymmetry of the human RLN. We showed previously that the mean lengths of the left and right recurrent laryngeal nerves were, respectively, 136.6 and 75.0 mm with a mean difference of 61.6 mm and the distal and proximal total intraperineural area ratio demonstrated differences between the left and right recurrent laryngeal nerve [22]. The left recurrent laryngeal nerve presents a slight delay about 2.30 ms in the conduction velocity when compared with the right nerve. In our study, the left recurrent laryngeal nerve demonstrated a slight delay about 2.30 ms in the conduction velocity when compared with the right. Continuous vagal intraoperative neural monitoring can provide real-time recurrent laryngeal nerve evaluation during surgical maneuvers in identifying real-time adverse concordant amplitude and latency changes. Prompt modification of the associated surgical maneuver could prevent recurrent laryngeal nerve paralysis [23]. A recent report about the amplitude and latency values for the vagus nerve, RLN and EBSLN (external branch of the superior laryngeal nerve) in a prospective series with normal pre and postoperative vocal cord function, reported a significant divergent latencies of the left and right vagus: so the mean left vagal latency was 8.14 ms compared to a mean right vagal latency of 5.47 ms [24].
A keen interest has been shown recently for the superior laryngeal nerve in thyroid surgeries, because the EBSLN innervates the cricothyroid muscle: cricothyroid muscle paralysis, due to EBSLN injury during thyroid surgery, can lead to a change in voice and reduce ability to produce high frequency tones. So IONM may be also a helpful tool in the identification and preservation of the EBSLN in thyroid surgeries [25].
Conclusion
Using vagal stimulation, IONM of the thyroarytenoid muscle can predict immediate postoperative vocal fold function in patients undergoing bilateral thyroid surgery and be aware of potential airway compromise. If we can get the muscular response, we can be sure that the vocal cord isn’t paralyzed. Using vagal nerve stimulation, the sensitivity and NPV were 100 %.
Physiologically, the thyroarytenoid muscle’s response showed functional asymmetry of laryngeal adduction with a faster vago-recurrent right response of 2.30 ms.
References
Munch S, deKryger L (2001) A piece of my mind. Moral wounds: complicated complications. JAMA 285:1131–1132
Jatzko GR, Lisborg PH, Müller MG, Wette VM (1994) Recurrent nerve palsy after thyroid operations: principal nerve identification and a literature review. Surgery 115:139–144
Wagner HE, Seiler C (1994) Recurrent laryngeal nerve palsy after thyroid gland surgery. Br J Surg 81:226–228
Lahey FH (1938) Routine dissection and demonstration of the recurrent laryngeal nerve in subtotal thyroidectomy. Surg Gynecol Obstet 66:775–777
Calò PG, Pisano G, Medas F, Pittau MR, Gordini L, Demontis R, Nicolosi A (2014) Identification alone versus intraoperative neuromonitoring of the recurrent laryngeal nerve during thyroid surgery: experience of 2034 consecutive patients. J Otolaryngol Head Neck Surg 43:16
Dralle H, Sekulla C, Lorenz K, Brauckhoo M, Machens A (2008) Intraoperative monitoring of the recurrent laryngeal nerve in thyroid surgery. World J Surg 32:1358–1366
Hillermann CL, Tarpey J, Phillips DE (2003) Laryngeal nerve identification during thyroid surgery: feasibility of a novel approach. Can J Anaesth 50:189–192
Randolph GW, Kobler JB, Wilkins J (2004) Recurrent laryngeal nerve identification and assessment during thyroid surgery: laryngeal palpation. World J Surg 28:755–760
Tschopp KP, Gottardo C (2002) Comparison of various methods of electromyographic monitoring of the recurrent laryngeal nerve in thyroid surgery. Ann Otol Rhinol Laryngol 111:811–816
Chiang FY, Lu IC, Kuo WR, Lee KW, Chang NC, Wu CW (2008) The mechanism of recurrent laryngeal nerve injury during thyroid surgery–the application of intraoperative neuromonitoring. Surgery 143:743–749
Genther DJ, Kandil EH, Noureldine SI, Tufano RP (2014) Correlation of final evoked potential amplitudes on intraoperative electromyography of the recurrent laryngeal nerve with immediate postoperative vocal fold function after thyroid and parathyroid surgery. JAMA Otolaryngol Head Neck Surg 140:124–128
Caragacianu D, Kamani D, Randolph GW (2013) Intraoperative monitoring: normative range associated with normal postoperative glottic function. Laryngoscope 123:3026–3031
Lipton RJ, McCaffrey TV, Litchy WJ (1988) Intraoperative electrophysiologic monitoring of laryngeal muscle during thyroid surgery. Laryngoscope 98:1292–1296
Thomusch O, Sekulla C, Machens A, Neumann HJ, Timmermann W, Dralle H (2004) Validity of intra-operative neuromonitoring signals in thyroid surgery. Arch Surg 389:499–503
Alon EE, Hinni ML (2009) Transcricothyroid electromyographic monitoring of the recurrent laryngeal nerve. Laryngoscope 119:1918–1921
Petro ML, Schweinfurth JM, Petro AB (2006) Transcricothyroid, intraoperative monitoring of the vagus nerve. Arch Otolaryngol Head Neck Surg 132:624–628
Hermann M, Hellebart C, Freissmuth M (2004) Neuromonitoring in thyroid surgery. Ann Surg 240:9–17
Woltering EA, Dumond D, Ferrara J, Farrar WB, James AG (1984) A method for intraoperative identification of the recurrent laryngeal nerve. Am J Surg 148:438–440
Jeannon JP, Orabi AA, Bruch GA, Abdalsalam HA, Simo R (2009) Diagnosis of recurrent laryngeal nerve palsy after thyroidectomy: a systematic review. Int J Clin Pract 63:624–629
Schneider R, Przybyl J, Pliquett U, Hermann M, Wehner M, Pietsch UC et al (2010) A new vagal anchor electrode for real-time monitoring the recurrent laryngeal nerve. Am J Surg 199:507–514
Ulmer C, Koch KP, Seimer A, Molnar V, Meyding-Lamadé U, Thon KP, Lamadé W (2008) Real-time monitoring of the recurrent laryngeal nerve: an observational clinical trial. Surgery 143:359–365
Prades JM, Dubois MD, Dumollard JM, Tordella L, Rigail J, Timoshenko AP, Peoc’h M (2012) Morphological and functional asymmetry of the human recurrent laryngeal nerve. Surg Radiol Anat 34:903–908
Phelan E, Schneider R, Lorenz K, Dralle H, Kamani D, Potenza A, Sritharan N, Shin J, Randolph GW (2014) Continuous vagal IONM prevents recurrent laryngeal nerve paralysis by revealing initial EMG changes of impending neuropraxic injury: a prospective, multicenter study. Laryngoscope 124:1498–1505
Sritharan N, Chase M, Kamani D, Randolph M, Randolph GW (2015) The vagus nerve, recurrent laryngeal nerve, and external branch of the superior laryngeal nerve have unique latencies allowing for intraoperative documentation of intact neural function during thyroid surgery. Laryngoscope 125:E84–E89
Darr EA, Tufano RP, Ozdemir S, Kamani D, Hurwitz S, Randolph G (2014) Superior Laryngeal nerve quantitative intraoperative monitoring is possible in all thyroid surgeries. Laryngoscope 124:1035–1041
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial interests to declare in relation to the content of this article.
Ethical standards
The authors declare that the study comply with the current ethical laws of the state of France.
Rights and permissions
About this article
Cite this article
Farizon, B., Gavid, M., Karkas, A. et al. Intraoperative monitoring of the recurrent laryngeal nerve by vagal nerve stimulation in thyroid surgery. Eur Arch Otorhinolaryngol 274, 421–426 (2017). https://doi.org/10.1007/s00405-016-4191-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-016-4191-2